Abstract
Introduction
According to the World Health Organization, 1 lung cancer is the most common cancer (2.09 million cases) and the most common cause of cancer death (1.76 million deaths). The 5-year survival rate of patients with different stage of lung cancer (stage I, 67%; stage III, 23%) 2 suggested the benefits of early diagnosis for improving the prognosis. Low-dose computed tomography (CT) for lung cancer screening has been recommended for the high-risk population in Canada, the EU, Japan, and the United States. 3 In addition, a number of serum tumor-related markers have been explored for early detection of lung cancer. Cancer antigen 125 (CA125), carcinoembryonic antigen (CEA), neuron-specific enolase (NSE) and others have been investigated for lung cancer screening in several clinical trials. 4 Multiple techniques based on the analysis of exhaled breath condensate, volatile organic compounds, or specific genomic features, have also been developed for the early diagnosis of lung cancer. 5
Circulating tumor cells (CTCs) are cells that detach from a primary solid tumor and enter into the bloodstream, which become an emerging target to provide pathological information for diagnosis and prognosis in a variety of cancers. 6 The CELLSEARCH® Circulating Tumor Cell Kit, an in vitro diagnostic device approved by the US Food and Drug Administration (USFDA), is used to assess the prognosis of metastatic breast or metastatic colorectal cancer. However, to date no clinical data clearly support the utility of this medical device for the disease management of lung cancer. On the other hand, recent studies showed that the sensitivity and specificity of CTCs identified by the chromosome enumeration probe 8 (CEP8) are 83.3% and 98.6% in patients with lung cancer, respectively.7,8 This unique CEP8+ CTCs has been examined in a series of studies, which showed a significant association with diagnosis and/or prognosis of lung cancer with promising sensitivity and specificity.9-11 According to the potential application of the CEP8+ CTCs on lung cancer management, this technique was utilized in this study.
The primary objective of this study is to investigate the potential of CTCs as a biomarker for early detection of lung cancer. The association between the identification of CTCs and lung cancer was examined. The diagnosis performance between CTCs and selected serum tumor-related markers were analyzed from patients with a solitary pulmonary nodule in outpatients of our hospital, Zhuhai People’s Hospital (Zhuhai hospital affiliated with Jinan University), China.
Materials and Methods
Patients
Inpatients in Zhuhai People’s Hospital (Zhuhai hospital affiliated with Jinan University), China from September 2020 to February 2021 were enrolled in this prospective, evaluator-blinded study. The inclusion criterion was patients who had a solitary pulmonary nodule 12 first identified by chest CT imaging. Exclusion criteria were patients who (1) received radiotherapy, chemotherapy, or immunotherapy recently; (2) had cancer history; (3) were not able/willing to receive sampling (blood, sputum, bronchoalveolar lavage fluid [BALF] or lung biopsy); (4) had no clear diagnosis. This study was approved by the institutional review board of Zhuhai People’s Hospital (IRB number: LW-[2020] no. 17) and registered (clinical trial registration number: 20200731105109530). All patients gave signed informed to allow the analyses of their medical records and samples. All samples used in this study were collected at the same timepoint after the informed consent was signed.
Detection of CTCs and Tumor-Related Markers
A CEP8 CTC detection kit (Cyttel®, Cyttel Bio), which is developed based on CD45-FISH technique,7,8 was used for identification of CTCs, and the experimental procedure was according to the user manual. Briefly, CTCs were first enriched from BALF by depletion of CD45+ cells. The remaining cells were fixed on a slide and hybridized with CEP8 probe. The identification criteria for CTCs (DAPI+/CD45−/CEP8+, the number of CEP8 signal dots ≥ 3) have been described previously.7,8
The serum levels of 3 tumor-related markers, CA125 (Siemens Healthcare Diagnostics Inc.), CEA (Siemens Healthcare Diagnostics Inc.), and NSE (Roche Diagnostics GmbH), were measured in the peripheral blood samples using electrochemiluminescence with an analyzer (Cabas6000-e601, Roche, Swiss).
The cut-off value of a CTC positive result was determined as CTC count ≥2, which has been published previously. 11 The upper limits of normality were 35 U/mL for CA125, 5 ng/mL for CEA, and 16.3 ng/mL for NSE.
Statistics
The association between CTCs and lung cancer was tested by the Fisher’s exact test. A
Results
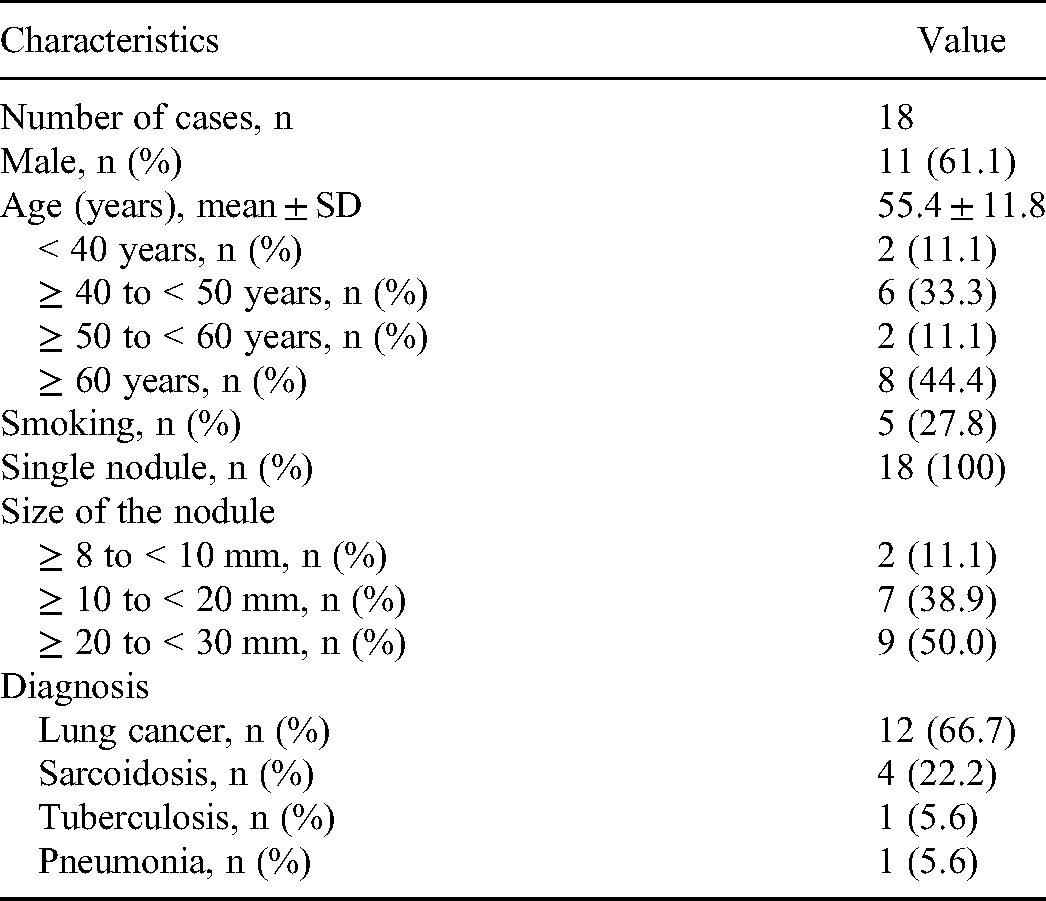
As shown in Figure 1, a total of 24 patients were enrolled and screened for the eligibility of this study. Twenty-four patients had a first identified solitary pulmonary nodule. Among them, 0 recently received radiotherapy, chemotherapy or immunotherapy; 3 had a cancer history; 0 had no sampling/biopsy; 3 had no clear diagnosis. Finally, 18 patients were analyzed in this study; 61.1% (11/18) of them were males. The mean age was 55.4 ± 11.8 years; 27.8% (5/18) of them had smoking history or were current smoking. The nodules were 11.1% (2/18) in a diameter with ≥8 to <10 mm, 38.9% (7/18) in a diameter with ≥10 to <20 mm, and 50.0% (9/18) in a diameter with ≥20 to <30 mm. The confirmed diagnosis of lung cancer, sarcoidosis, tuberculosis, and pneumonia was 66.7% (12/18), 22.2% (4/18), 5.6% (1/18), and 5.6% (1/18), respectively (Table 1). Information on the nodule size and CTC findings for each patient was shown in Table 2.
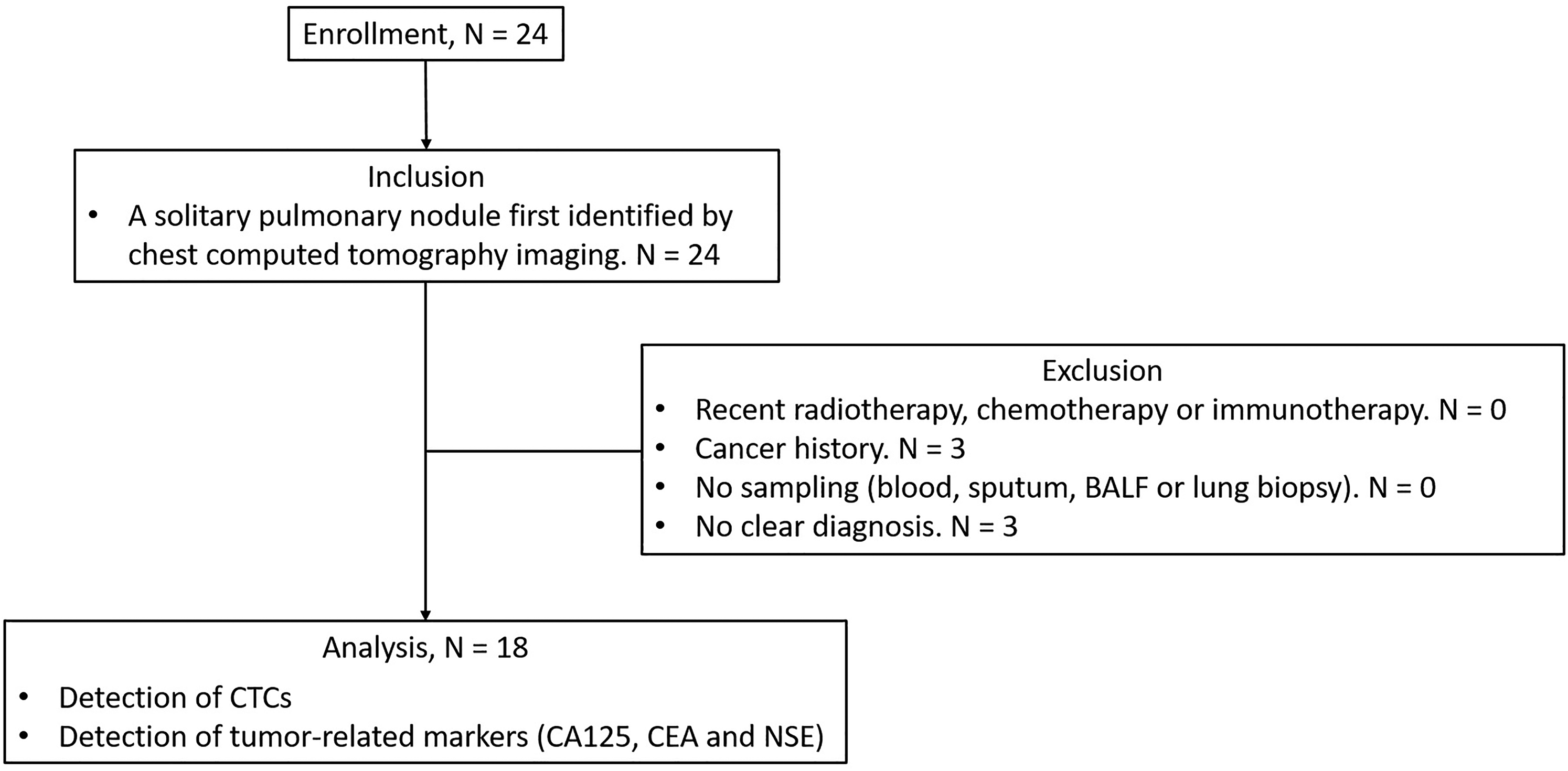
Study flow diagram. Twenty-four patients were enrolled in this study. Six of them were excluded due to cancer history (n = 3) and no clear diagnosis (n = 3). Finally, data from 18 patients were analyzed in this study.
Baseline Characteristics.
Information on the Nodule Size and CTC Findings for Each Patient.

CEP8+: chromosome enumeration probe 8-positive; CTC: circulating tumor cell.
Considering the small sample size of this study, the Fisher’s exact test was utilized to examine the significance of the association between the detection of CTCs and lung cancer. As shown in Table 3, the
Association between the identification of CTCs and lung cancer.
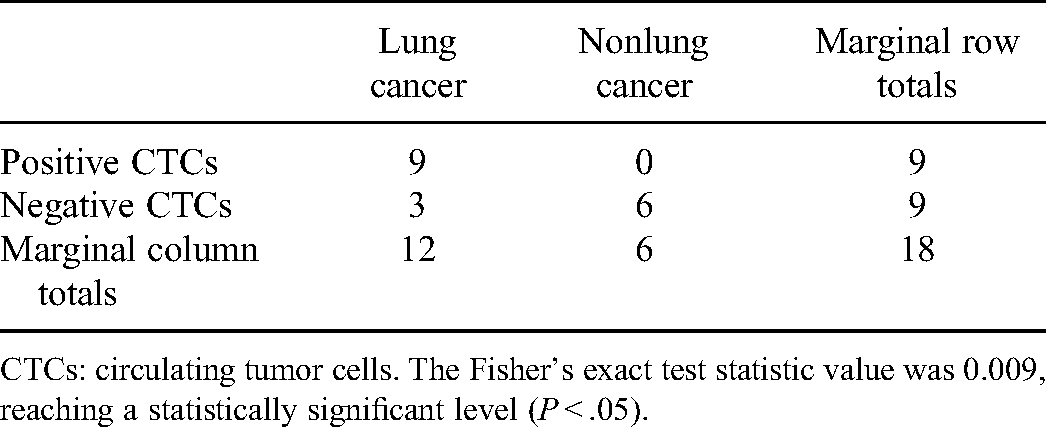
CTCs: circulating tumor cells. The Fisher’s exact test statistic value was 0.009, reaching a statistically significant level (
Comparison of the Diagnostic Performance for Lung Cancer Between the CTCs and Examined Tumor-Related Markers.
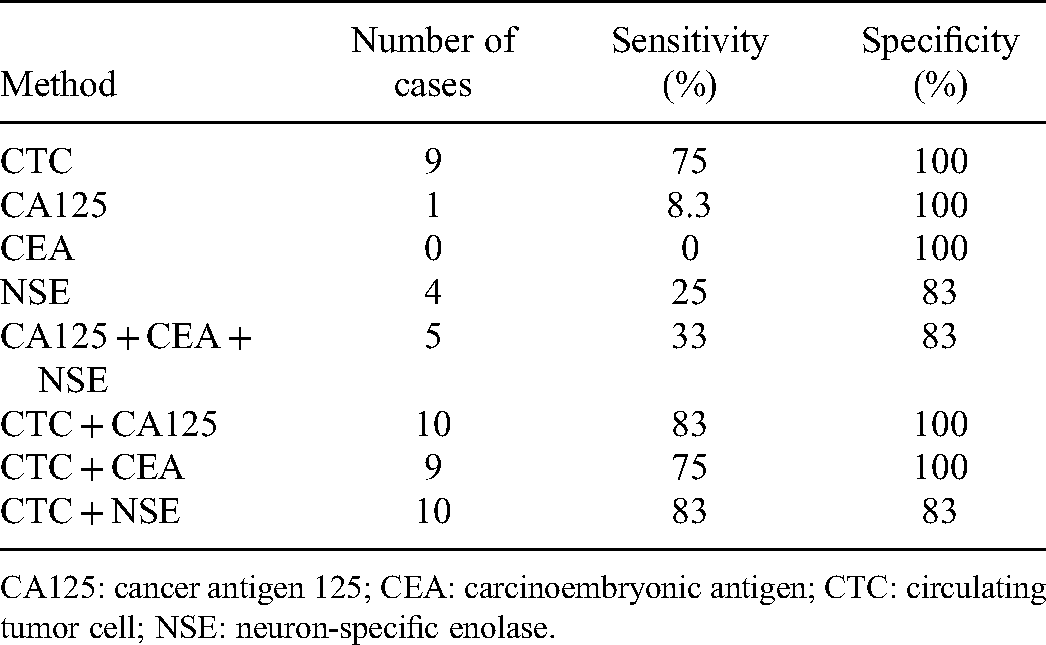
CA125: cancer antigen 125; CEA: carcinoembryonic antigen; CTC: circulating tumor cell; NSE: neuron-specific enolase.
Discussion
For the management of pulmonary nodules, CT follow-up is recommended by both Fleischner and British Thoracic Society guidelines; the frequency of CT follow-up is depending on the size, number, and density of the nodules. 13 However, the neoplasms are needed to be distinguished from other lesions by further examinations and are growing during the follow-up period. CTCs were considered as a potential biomarker to provide preliminary pathological information (eg, tumor or nontumor) for early stage of cancers. Recently, the method for detection of CTCs in lung cancers has been improved by several aspects, including isolation devices for CTC capture, 14 three-dimensional imaging for translocation of specific DNA fragments,15,16 and others. Further analysis of DNA translocation could be achieved using CTCs from transient cell culture. 17
Previous studies have shown that the number of CEP8+ CTCs was significantly higher in patients with lung cancer than with benign lung lesions and was similar across the disease stage of lung cancer.11,18 In this study, this CEP8+ CTCs detection was applied to patients with a solitary pulmonary nodule first identified by CT imaging. We found that detection of CEP8+ CTCs was significantly associated with the diagnosis of lung cancer (
On the other hand, we noted that the diagnosis performance of examined serum tumor-related markers is not as great as previously reported. CA125 is a sensitive but not specific marker for a number of benign and malignant conditions; 19 the sensitivity and specificity for lung cancer in published reports were 28.9% to 52.6% and > 90%, respectively.20-22 The sensitivity of CEA and NSE in the literature was 40.7% to 62.1% and 19.1% to 40.8%.20,21,23 In this study, the sensitivity of each serum tumor-related marker was 0% to 25%. Moreover, the diagnosis performance of CA125/CEA/NSE combination (sensitivity: 61.9%-79.5%; specificity: 72.7%-78.4%) previously reported was better than that in this study.20,21 One possible reason may be the differences in the enrolled population (eg, disease stage). In this study, patients with solitary pulmonary nodule first found by CT were enrolled. This time point was earlier than most of published studies, which retrospectively enrolled patients with definitive diagnosis of lung cancer for analysis of serum tumor-related markers. Therefore, CEP8+ CTCs may potentially be a useful biomarker for lung cancer detection at an early stage of the disease, compared to the examined serum tumor-related markers.
There are some limitations in this study. First, this single-center study had a small sample size. Second, the reference value of CEP8+ CTCs for lung cancer diagnosis is not well established. Further studies are needed to confirm the diagnosis performance of CEP8+ CTCs for lung cancer.
In conclusion, this study showed that CEP8+ CTCs may be served as an adjunctive technique for early detection of lung cancer in patients with solitary pulmonary nodule and may be helpful for the disease management.
Footnotes
Abbreviations
Authors’ Note
This study was approved by the institutional review board of Zhuhai People’s Hospital (IRB number: LW-[2020] no. 17) and registered (clinical trial registration number: 20200731105109530). Written informed consents were obtained from all patients.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
