Abstract
Introduction
Colorectal cancer (CRC) is one of the most common malignant tumors. According to the Global Cancer Surveillance Report 2020 released by the International Agency for Research on Cancer, CRC was ranked third and second in the world in terms of incidence and mortality, respectively, and accounted for about one-tenth of all malignant tumor-related deaths.1,2 Due to factors, such as atypical early clinical symptoms and incomplete screening, at the time of diagnosis, most CRC patients are at the middle or late stage of disease, accompanied by metastasis, thereby also called a “silent disease.” 3 Therefore, due to the molecular diversity of CRC, the personalized targeted therapies should be developed to improve the patients’ quality of life.4,5 For this purpose, novel biomarkers are needed to be explored to develop diagnostics and targeted therapeutics of CRC.
MARCH (membrane-associated RING-CH) family proteins are a key component of the ubiquitination cascade, which can strictly control the efficiency and specificity of ubiquitination. MARCH2 is a member of the MARCH family and is located at exon 7, zone 2, region 13, on the short arm of chromosome 19. It is widely expressed in various human tissues and is mainly located in the Golgi apparatus and endoplasmic reticulum membrane of the cells. 6 Its main functions in cells include specific binding to amino acids, ubiquitin-protein ligase activity, and selective non-covalent binding with zinc ions.7,8 Studies have shown that MARCH2 can directly bind to the PDZ domain of CFTR through its PDZ domain-binding motif, promote the ubiquitination and degradation of CFTR, and negatively regulate autophagy, thereby influencing tumor occurrence, development, proliferation, and metastasis. 8 The over-expression of MARCH2 can promote cell proliferation, while the knockdown of MARCH2 can promote cell apoptosis and G2/M phase cell cycle arrest in CRC. 9 However, little is known about the function of MARCH2 in the invasion and migration of CRC.
Considering the correlations of MARCH2 with the interstitial invasive depth and lymph node metastasis of CRC, 9 it was hypothesized that MARCH2 could promote the invasion and migration of CRC. Epithelial–mesenchymal transition (EMT) is a major factor, which leads to cancer cell metastasis. 10 This study focused on the role of MARCH2 in the progression of CRC as well as in the invasion and migration of CRC through EMT.
Materials and Methods
Ethics statement: The ethical approval for this study was obtained from the Clinical Trials Ethics Committee of the Affiliated Hospital of Southwest on December 24, 2021. All the patients signed an informed consent form before participating in the clinical study.
Tissue samples and immunohistochemistry: CRC tissue chips were purchased from Shanghai Outdo Biotech Co., Ltd (http://superchip.bioon.com.cn/). A total of 180 tissue samples, including 100 CRC tissues and 80 corresponding adjacent tissues, were obtained from the patients. All the clinicopathological and follow-up data were also obtained. The tissue chip was baked overnight at 60 °C, and then dewaxed and hydrated. Antigen repair was performed with sodium citrate buffer (pH 6.0), and the chip was incubated with 3% hydrogen peroxide for 10 min to block endogenous peroxidase. The chips were incubated with anti-MARCH2 antibody (1:50; ab220292; Abcam; Cambridge; UK) or anti-E Cadherin antibody (1: 500; ab40772; Cambridge; UK) overnight at 4 °C. The next day, the tissue chip was incubated with the respective secondary antibodies (Beyotime; Shanghai; China) at room temperature for 30 min. The color was generated using DAB (Zsbio; Beijing; China), and the tissue sections were counterstained with hematoxylin. The IHC score was assessed independently by 2 experienced pathologists using a semi-quantitative method. The tissue sections were scored based on staining intensity (0 [no staining], 1 [weak staining], 2 [moderate staining], and 3 [intense staining]) and percentage of positive tumor cells using the rating system (0 [≤5%], 1 [6%-25%], 2 [26%-50%], 3 [51%-75%], and 4 [≥76%]). The final score was the sum of the above 2 scores: ≤2 scores were regarded as negative (−), 3-4 scores were regarded as weakly positive (+), 5 score was regarded as positive (++), and 6 score was regarded as strongly positive (+++).
Cell culture: Human CRC cell lines, including Lovo (CC0509), SW480 (CC0505), and SW620 (CC0503), were purchased from CELLCOOK (Guangzhou; China), HT29 cells were obtained from the Pathology Laboratory of Southern Medical University. All the cells were cultured in DMEM (Gibco; MA; USA), containing 10% fetal bovine serum (FBS, Biological industries; Kibbutz Beit Haemek; Israel), and maintained in a humidified incubator at 37 °C in the presence of 5% CO2.
Cell transfection: The siRNA MARCH2-1 (target sequence: CTGGCACCCTCGACTTTAT) and siRNA MARCH2-2 (target sequence: GCAAGCTTCTTGCGCTTCT) were designed by Guangzhou RiboBio Co. Ltd, China, to downregulate MARCH2, and the negative control (NC) siRNA was used as control. MARCH2 or NC siRNAs were transfected into SW480 cells using Lipofectamine 2000 (Invitrogen; California; USA) following the manufacturer's instructions.
Quantitative real-time PCR (qRT-PCR): Total RNA was extracted using an RNA Extraction Kit (TIANGEN; Beijing; China) and then reverse transcribed into cDNA using the PrimeScript RT Reagent Kit (TaKaRa; Dalian; China). All the primers used were designed and synthesized by Sango Biotech Co., Ltd. The primers used in this study are listed in Table 1.
Primer Sequences Used in This Study.
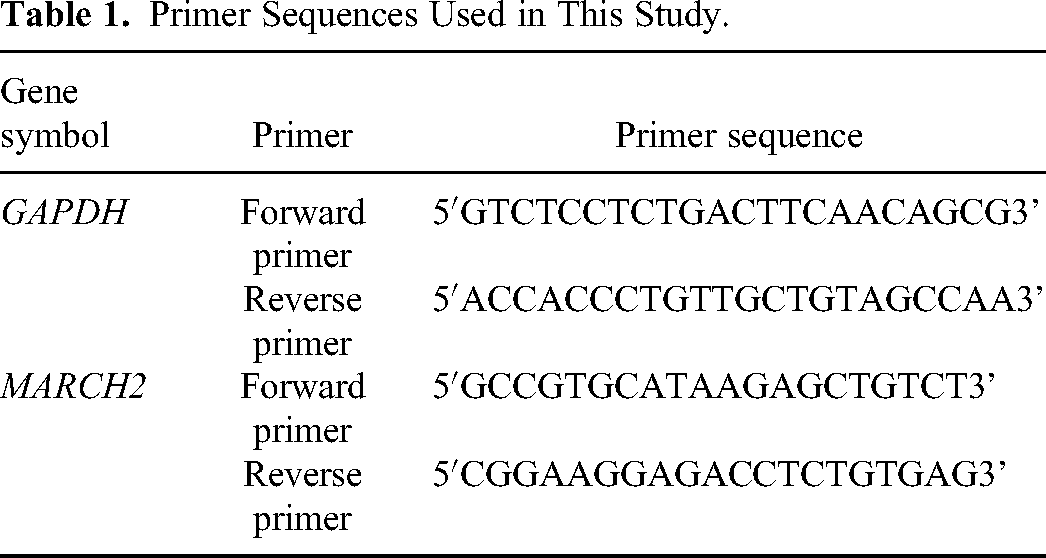
GAPDH was used as an internal reference. The 10 μL qRT-PCR reaction included 5.0 μL SYBR Premix Ex TaqII, 0.4 μL each PCR primer, 0.4 μL cDNA, and 3.8 μL RNase Free dH2O (TaKaRa; Dalian; China). The qRT-PCR and data collection were performed using Roche LightCycler 480. Each qRT-PCR experiment for a different passage of cells was performed in triplicates.
Western blotting: The total proteins were extracted from the cells using cell lysis buffer, containing protease inhibitors (Beyotime; Shanghai; China). Protein concentrations were determined using a Bicinchoninic acid Protein Assay Kit (Solarbio; Beijing; China). Equal amounts of proteins (50 μg) were separated using SDS-PAGE and transferred to a PVDF membrane. The membranes were blocked with 5% skimmed milk for 1 h and then incubated with different primary antibodies overnight on a shaking incubator at 4 °C. The primary antibodies used in this study were as follows: anti-MARCH2 antibody (1: 100; ab220192), anti-E Cadherin antibody (1: 10000; ab40772), anti-Vimentin antibody (1: 2000; ab92547) (all from Abcam; Cambridge; UK), and β-Actin (4D3) monoclonal antibody (1: 10000; BS6007M; Bioworld; Nanjing). The membranes were incubated with the respective secondary antibodies (Beyotime; Shanghai; China) on a shaking incubator at room temperature for 1 h. The membranes were then washed, and proteins were visualized using an ECL kit (Beyotime; Shanghai; China) using UVP ChemStudio PLUS (Analytik Jena AG; Jena; Germany). Densitometric analysis was performed using ImageJ software by measuring the grayscale intensity of the target bands derived from scanned films to quantify the protein bands. Each western blotting experiment for a different passage of cells was performed in triplicates.
Transwell invasion assay: Invasion assays were performed in a 24-well Transwell chamber (Corning-Costar; USA). After 36 h of transfection, the SW480 cells were cultured in a serum-free medium for 12 h. Matrigel (Corning-Costar; USA) and serum-free medium were mixed at a ratio of 1:8, and 80 μL of the mixture was added into the top chamber of each well. The plate was placed in an incubator at 37 °C for about 1 to 2 h until the mixture solidified. The cells were resuspended in a serum-free medium, and the cell concentration was adjusted to 2 × 105 cells/mL. A 100 μL cell suspension was added to each top chamber, and a 500 μL culture medium, containing 10% serum, was added to the bottom chamber. After incubating at 37 °C for 48 h, the cells and Matrigel remaining in the top chambers were gently wiped using cotton swabs. The cells that had passed through the chambers were fixed with 4% paraformaldehyde for 30 min and stained with 0.1% crystal violet aqueous solution for 30 min. The cells that had passed through the chamber were observed and photographed under an inverted microscope. Each experiment for a different passage of cells was performed in triplicates.
Scratch assay: The well-grown cells were selected and seeded into a 6-well plate. The SW480 cells were transfected for 48 h until they reached 100% cell confluency, covering just the bottom of a 6-well plate. Horizontal lines with approximately the same width were drawn on the bottom of the 6-well plate with a 10 μL pipette tip using a sterilized ruler. The suspended cells were washed away, and an appropriate medium was added prior to further incubation. Photographs were taken at the same magnification (10×) at 0, 24, and 48 h time-points after scratching under an inverted microscope, and the scratch area was measured using ImageJ software. The average width of the scratch was calculated by dividing it by the corresponding length. Each experiment for a different passage of cells was performed in triplicates.
Statistical analyses: All the statistical analyses were performed using SPSS Statistics 17. The rank-sum test was used to analyze the differences in the MARCH2 expression levels between CRC and adjacent tissues. A chi-square test was used to analyze the correlations among the high and low MARCH2 protein expression groups and clinicopathological parameters of CRC. The CRC patient survival data were analyzed using Kaplan–Meier survival curve analysis, and the log-rank method was used to evaluate the statistical significance of differences in prognosis. Spearman correlation analysis was used to analyze the correlations between MARCH2 expression and E-cadherin expression. One-way ANOVA was used to analyze the difference between the MARCH2 gene silencing and control groups. A P-value of <.05 was considered statistically significant.
Results
MARCH2 was an Important Progression Factor of CRC
First, the expression of MARCH2 was detected in tumor tissues (n = 99) and adjacent normal tissues (n = 78). The results showed that the MARCH2 expression was mainly located in the cytoplasm, and its expression level in CRC tissues was significantly higher than that in the adjacent normal tissues (Table 2) (Z = −3.279, P < .001). Survival analysis showed that the 5-year survival rate of CRC patients was 48.5% (48/99) with an average survival time of 54.3 months. In the high MARCH2 expression group, the 5-year survival rate of CRC patients was 41.9% (31/74) with an average survival time of 50.1 months. In the low MARCH2 expression group, the 5-year survival rate of CRC patients was 68.0% (17/25) with the average survival time of 66.6 months. According to the Kaplan–Meier survival curve (Figure 1A), the survival time of the high MARCH2 expression group was significantly lower than that of the low expression group (χ2 = 4.417, P = .036, log-rank test). These results demonstrate that MARCH2 plays a critical role in CRC progression.
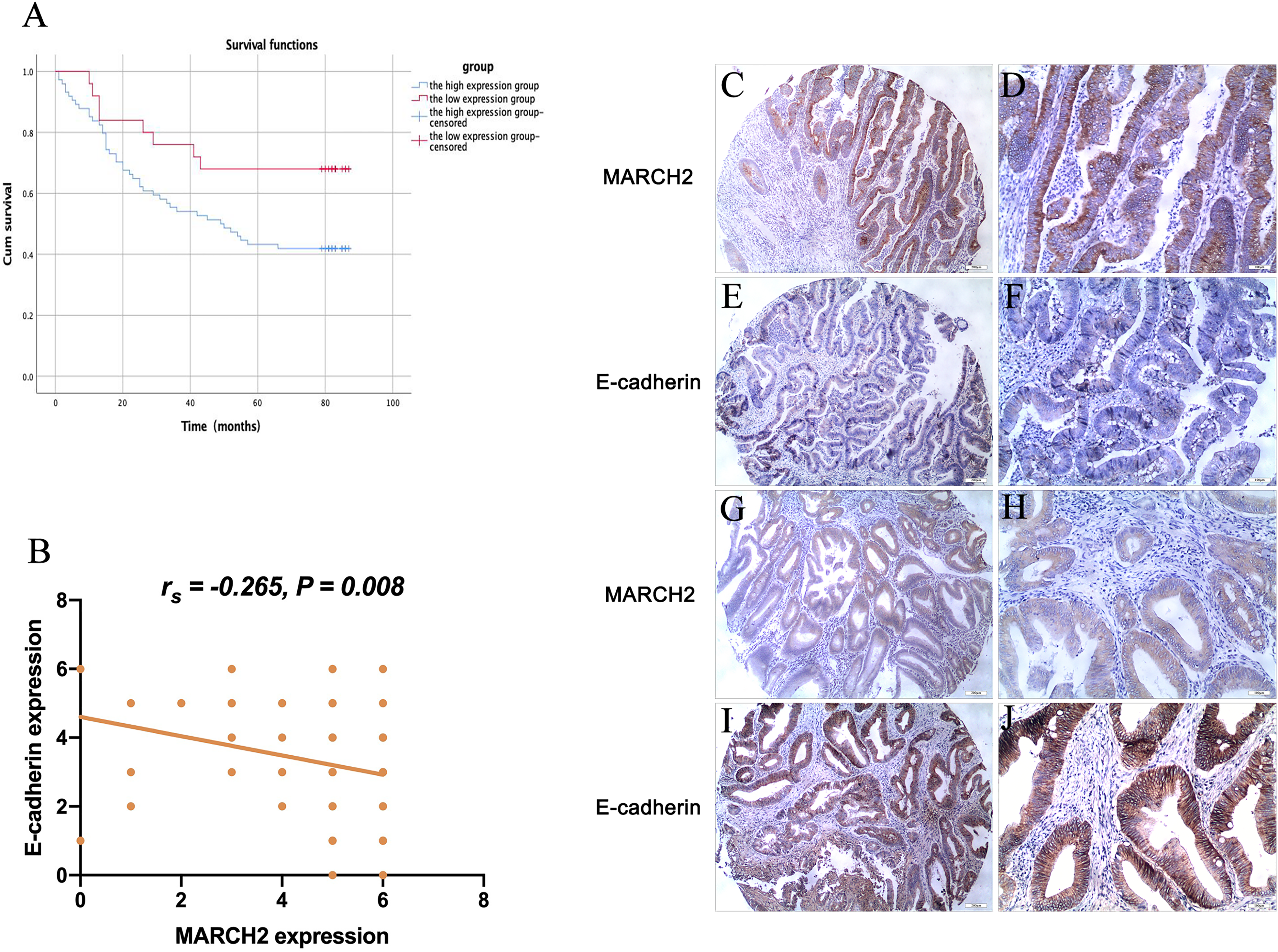
(A) Kaplan–Meier survival analysis of the correlation between MARCH2 protein expression level and survival time of the CRC patients (χ2 = 4.417, P = .036, log-rank test). (B) Expression level of MARCH2 protein in CRC tissues was negatively correlated with E-cadherin (r = −0.265, P = .008, Spearman correlation analysis). (C-J) Representative images of MARCH2 and E-cadherin immunohistochemical-stained CRC tissues. (C-F) Same patient tissues, in which C and D show high expression level of MARCH2, and E and F show low expression of E-cadherin. (G-J) Same patient tissues, in which G and H show a low expression of MARCH2, and I and J show a high expression of E-cadherin. (C, E, G, I) Bars correspond to 200 μm (40×). (D, F, H, J) Bars correspond to 100 μm (100×).
Immunohistochemical Staining Detection of MARCH2 Expression in Colorectal Cancer (CRC) Tissues (n = 99) and Para-Carcinoma Tissues (n = 78).

The scoring system relates to staining intensity and percentage of positive tumor cells. For details, refer to the methods section.
*** P < .001.
MARCH2 was Strongly Correlated With Tumor Size, Pathological Grade, and Lymph Node Metastasis of CRC
In order to better understand the function of MARCH2 in CRC patients, the patients’ clinicopathological characteristics were compared with the MARCH2 expression (Table 3). The results showed that the expression of MARCH2 was not correlated with age, gender, distant metastasis, and the presence or absence of invasion at the entire thickness of the intestinal wall (P > .05), however, the expression level of MARCH2 was significantly correlated with tumor size (P = .008), pathological grade (P = .031), and lymph node metastasis (P = .001).
Correlation Between MARCH2 Protein Expression and Clinicopathological Characteristics of CRC.

*P < .05, **P < .01, ***P < .001.
MARCH2 was Negatively Correlated With EMT-Related Protein E-Cadherin in CRC
EMT is a crucial biological process for acquiring the migration and invasion ability of epithelial malignant tumor cells. Down-regulation or deletion of E-cadherin expression is a marker of EMT occurrence and an important regulatory factor of EMT. The expression levels of E-cadherin were further detected in cancer tissues. The results showed that patients with higher MARCH2 expression had lower E-cadherin expression, and MARCH2 was negatively correlated with E-cadherin (r = −0.265, P = .008) (Figure 1B to J). It was speculated that MARCH2 could promote the invasion and migration of CRC by activating EMT.
MARCH2 Silencing in CRC Cells Resulted in Enhanced Invasion and Migration of Cells
The expression levels of MARCH2 in CRC cell lines (Lovo, SW480, HT29, and SW620) were further explored. The results showed that the HT29 and SW480 cells had higher expression of MARCH2 mRNA (Figure 2A). As compared to the HT29 lines, the SW480 lines were easier to transfect, less differentiated, and easier to metastasize. Therefore, the SW480 cell line was used for the knockdown of MARCH2 with siRNA. After transfecting the SW480 cells with siRNA MARCH2-1 and siRNA MARCH2-2, the mRNA and protein expression levels of MARCH2 (Figure 2B to D) significantly decreased. Transwell assay results showed that the number of siRNA MARCH2-1/-2 penetrating cells in the experimental group was significantly lower than that of the control group (Figure 2E and F). Migration assay results showed that after 24 and 48 h, the migration distance of siRNA MARCH2-1/-2 cells in the experimental group was significantly smaller than that of the control group (Figure 2G and H). These results showed that MARCH2 might promote CRC progression by affecting cell invasion and migration.
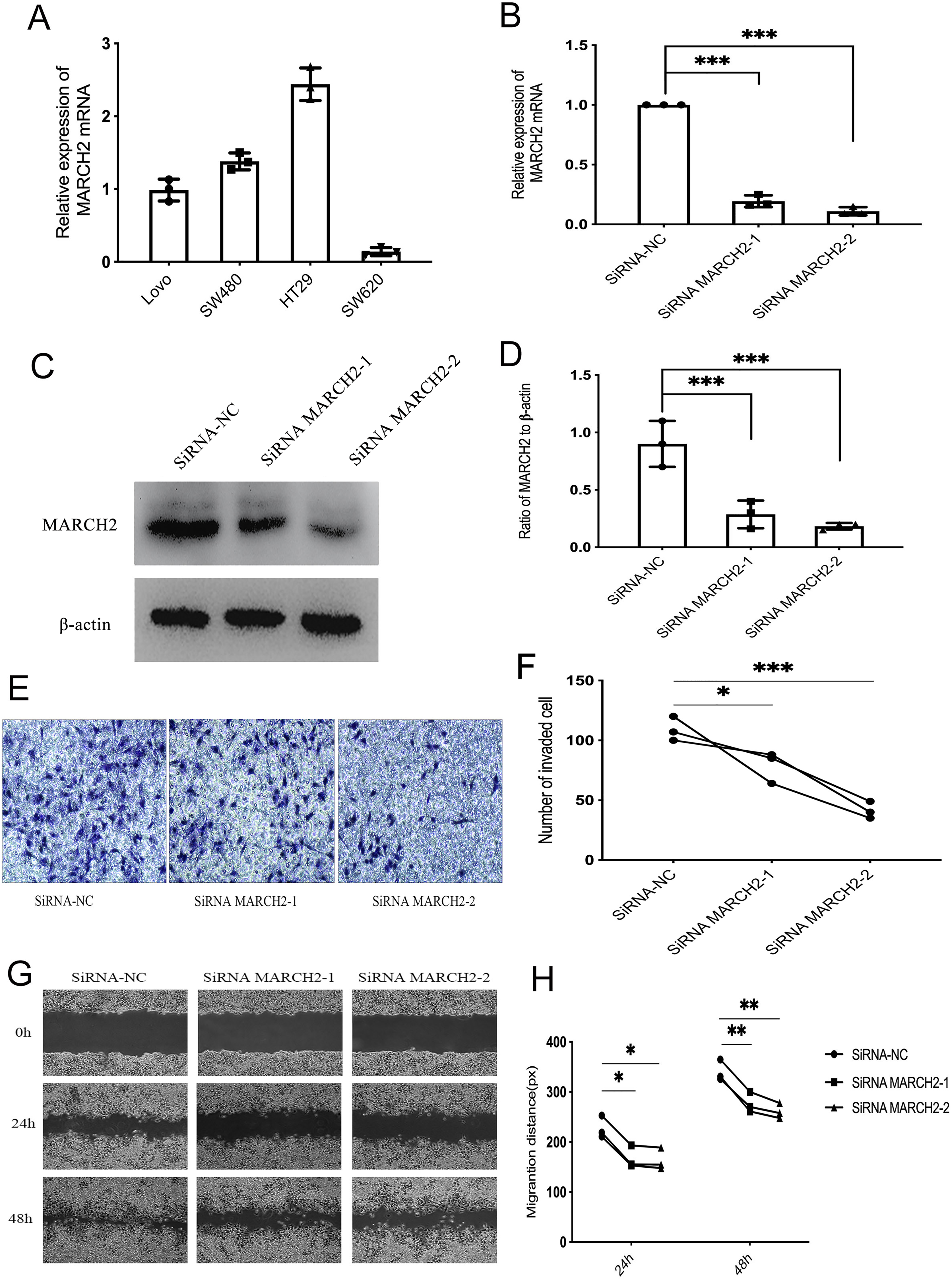
MARCH2 silencing in the CRC cells resulted in an enhanced invasion and migration. (A) Expression level of MARCH2 in the CRC cell lines was analyzed using qRT-PCR. (B) Expression levels of MARCH2 in the SW480 cells transfected with siRNA MARCH2-1/-2 and negative control were analyzed using qRT-PCR. (C) Representative images of Western blotting for MARCH2 protein expression levels in SW480 cells transfected with siRNA MARCH2-1/-2 and negative control. (D) Gray level analysis showing the protein level of MARCH2 (β-actin acted as internal control). (E, F) Transwell assay was performed to determine the invasion of SW480 cells. (E) Representative images. (G, H) Scratch assay was performed to determine the migration of SW480 cells. (G) Representative images.
MARCH2 Could Affect E-Cadherin and Vimentin in SW480
In order to further study the pathway of MARCH2, affecting the invasion and migration ability of CRC, the expression levels of EMT-related markers in SW480 were examined. The results showed that as compared to the NC group, the mRNA and protein expression levels of E-cadherin were upregulated in the experimental group siRNA MARCH2-1/-2, while those of Vimentin were downregulated (Figure 3). Altogether, these results suggest a role of silencing MARCH2 in preventing the transition of EMT.
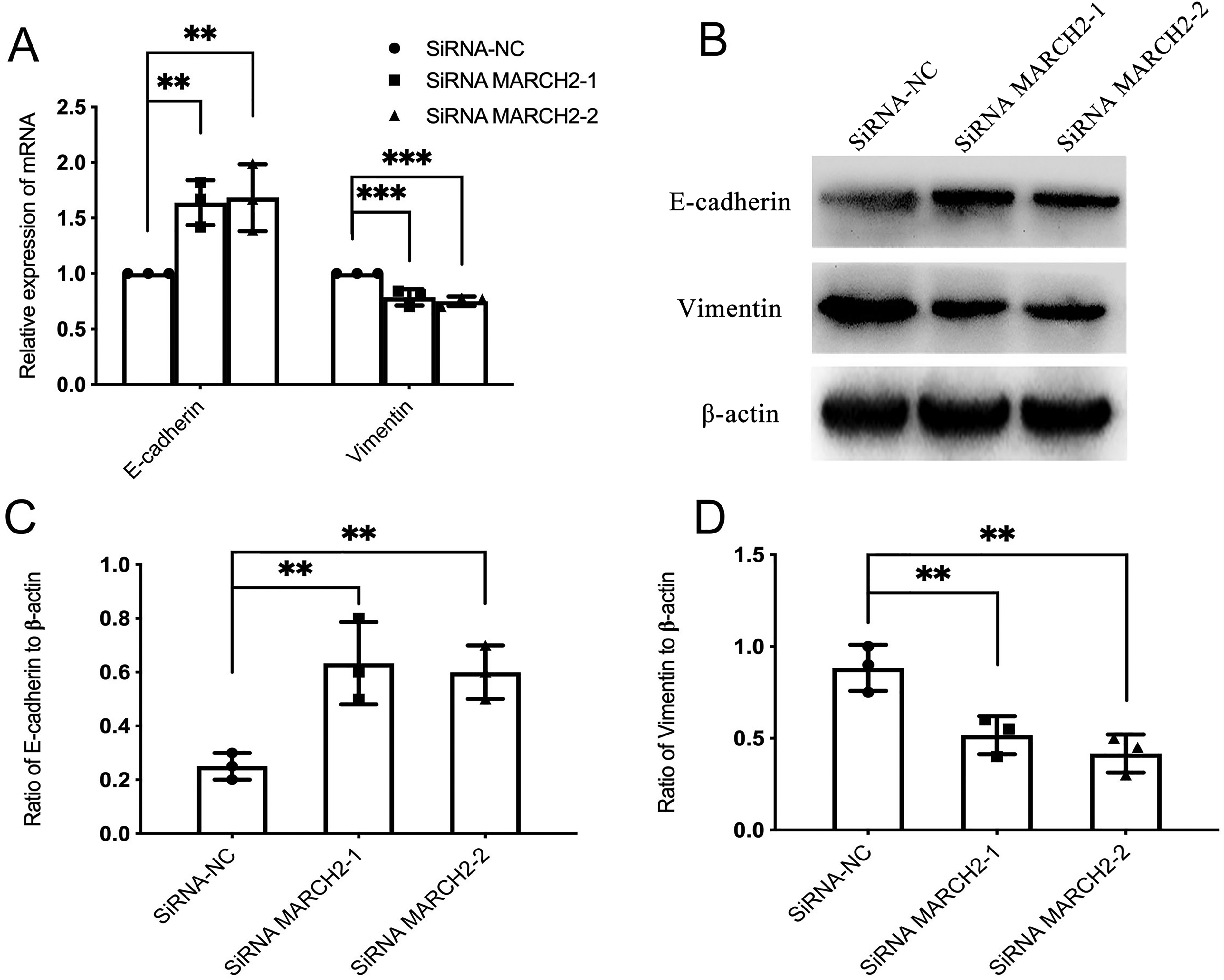
MARCH2 affected the E-cadherin and Vimentin expression levels in SW480. (A) Expression levels of EMT-related markers in the SW480 cells transfected with siRNA MARCH2-1/-2 and negative control were analyzed using qRT-PCR. (B-D) Protein expression levels of E-cadherin and Vimentin in SW480 cells transfected with siRNA MARCH2-1/-2 and negative control were determined using Western blotting. (B) Representative images of Western blotting. (C, D) Gray level analysis, showing the protein expression levels of E-cadherin and Vimentin (β-actin acted as internal control).
Discussion
The current study investigated the role of MARCH2 in CRC in the tissues and cell cultures. The main findings were as follows: MARCH2 was highly expressed in CRC, inversely correlated with survival, and correlated with tumor size, pathological grade, and lymph node metastasis. These data suggested that the high MARCH2 expression could contribute to CRC progression and promote aggressive behavior. In order to determine the role of MARCH2 in promoting its invasiveness and metastatic ability in CRC, the expression levels of E-cadherin expression were further examined. The results indicated that MARCH2 was negatively correlated with E-cadherin. It was speculated that MARCH2 could regulate the occurrence of EMT, which was verified using subsequent cell-based experiments. Moreover, MARCH2 silencing was also related to the occurrence or development of EMT and promoted the invasion and migration of CRC.
Previous studies on MARCH2 have focused on its function in the immune system.7,11 However, its role in malignant tumors is less studied. The results of the present study were slightly different from those of the previous studies; a previous study reported that MARCH2 was associated with the depth of interstitial invasion and lymph node metastasis, 9 while the current study suggested that MARCH2 was not associated with the presence or absence of full-thickness intestinal wall invasion and distant metastasis. This might be due to the limited sample size and insufficient progression at the time of tumor resection. Furthermore, these results should be validated in larger clinical trials. However, the current study showed that MARCH2 was associated with lymph node metastasis, suggesting that MARCH2 might be related to the invasion and migration ability of CRC. Therefore, this speculation and correlations between MARCH2 and CRC invasion and migration were further confirmed using cell-based experiments.
Despite the fewer studies on the role of MARCH2 in malignancies, numerous researchers have shown that the MARCH family is closely related to tumors. For example, MARCH8 was abnormally expressed in esophageal tumors and non-small cell lung cancer and promoted the invasion and migration of tumors.12,13 The high expression levels of MARCH1, MARCH5, and MARCH7 could promote the invasion and migration of ovarian cancer cells.14–16 MARCH7 could also promote the proliferation and invasion of cervical cancer. 17 These findings suggested a potential role of MARCH2 in the invasion and migration of malignant tumors.
CRC can invade adjacent tissues as well as distant tissues, causing distant metastasis, which mainly depends on the invasion and migration ability of tumor cells and involves a variety of complex mechanisms. EMT is one of the key factors for the metastasis of epithelial-derived tumors as well as a developmental program, in which cancer cells acquire invasion and metastasis ability.18,19 Epithelial cells have the characteristics of mesenchymal cells, such as decreased cell adhesion, increased motility, and changes in cytoskeleton structure. It is usually accompanied by the downregulation of the epithelial immune marker E-cadherin and upregulation of the mesenchymal immune marker Vimentin. 20 The current study found that MARCH2 was negatively correlated with E-cadherin in the CRC tissues. Silencing the MARCH2 gene in SW480 cells resulted in the upregulation of E-cadherin and the downregulation of Vimentin. These results suggested that MARCH2 could promote the process of EMT in CRC.
This study confirmed a role of silencing MARCH2 in preventing the transition of EMT. However, this study only performed the invasion and migration experiments in one type of cell. Therefore, a more rigorous approach should be adopted using multiple mutant cell systems and in-vivo experiments. The mechanism of MARCH2, regulating EMT, was still unclear. In future studies, we will further explore the role of MARCH2 in the invasion and metastasis of CRC using in-vivo experiments as well as the specific pathways regulating EMT. Numerous studies have shown that EMT enables tumor cells to adapt to the changing microenvironment and enhances drug resistance in the tumor cells.21,22 EMT is an important mechanism, mediating drug resistance in CRC. The in-depth studies of its regulatory mechanism might explore the potential of MARCH2 as a clinical indicator and therapeutic target as well as its correlation with drug resistance in CRC tissues.
In conclusion, the high expression level of MARCH2 in CRC was an independent prognostic marker, which was unfavorable for the patient's survival. MARCH2 might affect the invasion and metastasis of CRC through EMT, thus a promising target for its treatment.
Footnotes
Acknowledgments
The authors thank Zixi Yang, Zijiang Lu, Qiang Chen, and Bo Yang for their help in obtaining and analyzing article data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The ethical approval for this study was obtained from the Clinical Trials Ethics Committee of the Affiliated Hospital of Southwest Medical University (Luzhou, China) (IRB: KY2021300) on December 24, 2021. All the patients signed an informed consent form before participating in the clinical study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the major cultivation project of Sichuan Provincial Department of Education (grant no. 18CZ0044) and the joint research project of Luzhou Municipal People's government and Southwest Medical University (grant no. 2016JLZXNYD-J17).
