Abstract
Background
Adenoid cystic carcinoma of head and neck (ACCHN) is an uncommon head and neck cancers, whose predilection age is 40 to 60. Some studies have revealed that early-onset cancers, such as colorectal cancers and esophageal adenocarcinoma, might present some unique clinicopathological features and have different prognosis with late-onset ones. However, little is known about the early-onset ACCHN. This study aimed to develop a prognostic nomogram for overall survival (OS) of patients younger than 40 with ACCHN.
Methods
Cases with ACCHN from 1975 to 2016 were retrieved from SEER-18 program. Demographic, clinical, and survival outcomes data of patients were identified for further analysis. The caret package was used to randomly divide early-onset patients into a training cohort and a validation cohort. A prognostic nomogram was constructed based on the univariate and multivariate Cox analysis. The discriminative ability and calibration power of the nomogram was evaluated by the concordance index (C-index), calibration curve, and receiver operating characteristic (ROC) curve.
Results
A total of 5858 cases with ACCHN were selectively retrieved from SEER program in this study. The number of patients younger than 40, which was defined as early-onset ACCHN in this study, was 825. Based on the outcomes of multivariate analysis, tumor size, chemotherapy, surgery, and stage were selected for the construction of nomogram to predict 10-year OS. The C-index was 0.792 (95%CI 0.760-0.823) and 0.776 (95%CI 0.720-0.832) in the training and validation set, respectively. The area under the ROC curve values were 0.875 (95%CI 0.810-0.940) and 0.833(95%CI 0.754-0.912). The calibration plot indicated that this nomogram had proper calibration in both the training and validation cohorts.
Conclusion
A novel prognostic nomogram for early-onset ACCHN was constructed and validated in this study. This nomogram could be applied for assisting clinicians to more accurately assess the prognosis of young patients, which might facilitate clinical decision-making and subsequent follow-up.
Introduction
ACC of head and neck (ACCHN) is the second common salivary gland carcinoma which typically originates within the salivary glands, especially the minor salivary glands. 1 In addition, other sites of head and neck such as paranasal sinuses and pharynx are also potential origins of ACCHN. 2 This tumor could occur in any age but be more susceptible in middle-aged and older patients. 3
Recently, many researches demonstrated that a certain tumor in different age group might exhibit different clinical features and prognoses. In colorectal cancer, Mauri et al 4 found that patients younger than 30 had the worst prognosis, while the survival of patients between 40 and 50 showed a comparable or even better outcomes than that of those older than 50 years old. Compared with older adults,individuals with early-onset esophageal adenocarcinoma presented a more advanced stage and might encounter a worse prognosis. 5 Early-onset oral squamous carcinoma showed a high recurrence rate which was likely to be associated with its specific molecular and immune landscape. 6 The causes that lead to different prognoses in early-onset cancers still remained unclear. Evidence suggests an aetiological role of risk factor exposures in early life and young adulthood. 7 Some studies found that patients with early-onset tumors tended to harbor mutations causing hereditary cancer-predisposing syndromes or had familial history. 8 Early-onset cancers were often defined as cancers diagnosed in adults <50 years of age, which usually occupied a small part of the whole group. However, the most common age of ACC was 40 to 60. As supplementary Table 1 shown, the ACC patients aged 40 to 50 accounted for 15.74%. And the incidence dramatically declined when patients < 40 years old. Therefore, we defined cases < 40 as early-onset ACCHN. At present, there lacks a clear definition of early-onset ACCHN and the relevant studies about features and survival of early-onset ACCHN are scarce. Surveillance, Epidemiology and End Results program, which is called SEER for short, is an authoritative cancer statistic database monitored by American National Cancer Institute (NCI). This study used SEER-18 database (1975-2016) which encompass 18 cancer registers across American and 20 geographic regions representing 28% of the population in United States. This database provide adequate cases for our further analysis.
Characteristics of Adenoid Cystic Carcinoma of Head and Neck (ACCHN) Patients in 2 Age Groups.
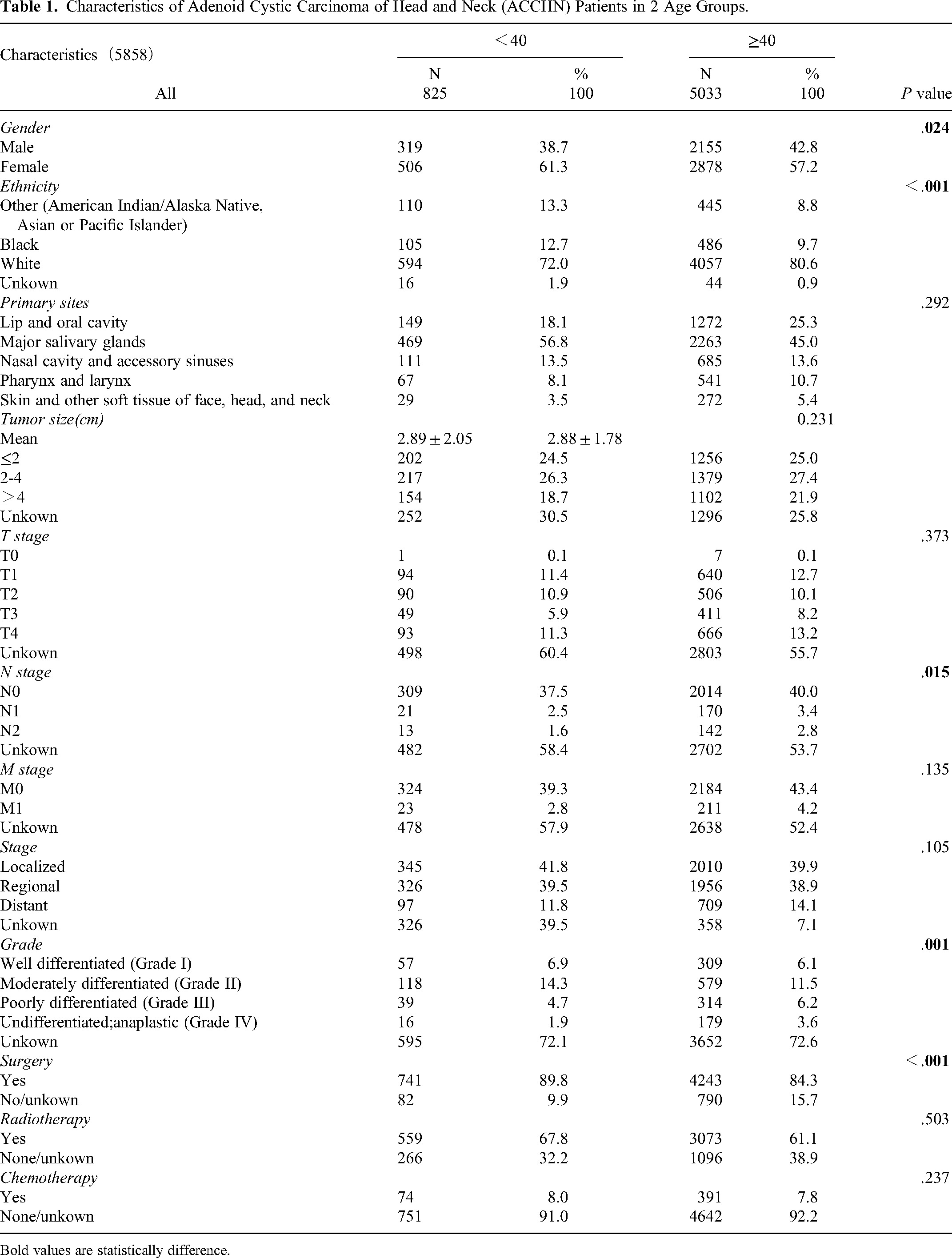
Bold values are statistically difference.
Nomogram is constructed based on regression analysis, which has been accepted as efficient prognostic tool in the last decades. It has been established to predict the prognosis of various types of cancers.9–12 This study aimed to probe into the potential independent risk factors and build an effective nomogram for early-onset ACCHN, which might help clinicians in the process of diagnosis and treatment.
According to the definition of cancers in adolescence and young adults by the National Cancer Institute, in this study, the age of patients with early-onset ACCHN is stipulated as younger than 40. 13 By using the data from the SEER (surveillance, epidemiology, and end results) database, our study compared the clinical features of ACCHN younger than 40 and older than 40 and built a nomogram based on the information of ACCHN patients <40 to predict their survival outcomes.
Materials and Methods
The reporting of this study conforms to RECORD guidelines. 14
Patient Population
All information was extracted by SEER*stat software (version 8.3.9) of the National Cancer Institute. Patients were identified according to the special codes of International Classification of Diseases for Oncology, third edition (ICD-O-3) as following: (1) histological code: 8200/3 adenoid cystic carcinoma; (2) primary sites labeled: lip (C000-C006, C008-C009), base of tongue (C019), tongue excl base of tongue (C020-C024, C028-C029), gum, floor of mouth, and other mouth (C030-C031, C039-C041, C048-C052, C058-C062, C068-C069), salivary gland (C079-C081, C088-C089), oropharynx (C090-C091,098-104, 108-109), nasopharynx excl posterior wall (C110, C112-C113,C118-C119), posterior wall of nasopharynx (C111), hypopharynx (C129-C132, C138-C139), pharynx (C140, C142, C148), nasal cavity including nasal cartilage (C300), accessory sinuses NOS (C319), larynx (C320-C323, C328-C329), skin of head, face and neck (C440), connective and soft tissue of face, head and neck (C490). All pathologically diagnosed ACCHN were analyzed based on race, age at diagnosis, sex, primary site, grade, ICD-O-3 histologic type, SEER historic stage A (1973-2015), tumor size, T stage, N stage, M stage, surgery, radiation, chemotherapy, survival time and vital study at study cutoff. The diagram of case selection was shown in Figure 1. As to SEER historic stage, “localized” means the cancer is restrained to the primary site without spreading; “regional” refers to that cancer has invaded its nearby lymph nodes or organ and tissues; “distant” suggests that metastasis of distant organ and distant lymph is examined.

Diagram for case selection.
Statistical Analysis
All cases from SEER program were divided into 2 groups: younger than 40 and older or equal to 40. The features of ACCHN were analyzed with descriptive statistics. Across the comparison group, continuous variables and categorical variables were processed by using Student's t test and Chi square respectively. Kaplan–Meier plots and log-rank tests were used to compare the difference in survival among groups. The caret package was used to randomly divide early-onset patients into a training cohort and a validation cohort. To figure out the risk factors of prognoses, a Cox proportional hazard model was constructed for univariate and multivariate analyses, with hazard ratio (HR) and 95% CI. The discriminative ability and calibration power of the nomogram was evaluated by the concordance index (C-index), calibration curve, and receiver operating characteristic (ROC) curve. IBM SPSS statistics 25.0 and R (version 4.1.1) was used for data analysis and plotting. P < .05 was considered as statistically significant.
Ethics
This is a population study based on SEER database which is publicly available, all information of patients throughout the study is de-identified. Because the information in the SEER database does not require the patient's explicit consent, the study is waived from ethical approval. The informed patient consent is not required due to the retrospective nature of the study.
Results
Comparisons of Characteristics Between Younger and Older Patients with ACCHN
5858 cases of ACCHN were recorded in the SEER database from 1975-2016 and we divided them into 2 groups according to their ages: <40 and ≥40. These 2 age groups showed a statistically significant difference in gender, ethnicity, N stage, grade, and surgery. Compared to early-onset ACCHN, the proportion of males was slightly higher in the older group. White people occupied the majority in 2 groups, but the ratio of non-White was higher in early-onset ACCHN. The percentage of patients in N0, N1 and N2 stage were all smaller than its older counterpart. This might because surgeons were more inclined to perform nodal examination in older patients. The tumor size of 2 age groups were similar (<40: 2.89 ± 2.05 cm; ≥ 40: 2.88 ± 1.78 cm). As to the pathological grade, the higher grade (grade III and grade IV) accounted for 5.6% in early-onset ACCHN while when patients older than 40, this climbed to 9.8%. Most of the patients with ACCHN would undergo surgical resection, but the percentage was moderately higher in patients younger than 40 (89.8%).
Survival Analysis of Early-Onset ACCHN
As shown in Figure 2b and c, White people had a better prognosis than black people and other races (P = .003), and female patients showed a longer survival time compared to the male ones (P = .013). Figure 2e indicated that ACCHN originated from the skin and other soft tissue of face, head and neck had the best survival outcomes, followed by major salivary glands and lip and oral cavity subsequently (P < .001). This might be because these sites will be diagnosed earlier and are also much more resectable due to access and lack of other vital structures in the area. As shown in Figure 2d, f, and g, a higher grade, a more advanced stage and a larger tumor size were all associated with worse survival (P < .001). In Figure 2h, i, and j, the prognoses of patients who took surgery (P < .001) was much better than people who did not, while the outcomes of patients applied with chemotherapy (P < .001)was completely opposite, for chemotherapy was commonly used for advanced disease or disease that was unresectable due to size or co-morbidities preventing surgery. Meanwhile, there was little difference between group with and without radiotherapy (P = .803).

Kaplan–Meier curve analysis for selected variables of patients with early-onset ACCHN. (a) survival outcomes of adenoid cystic carcinoma of head and neck (ACCHN) patients younger or older 40-years old. (b) Survival analysis for early-onset ACCHN patients of different races. (c) Survival outcomes of female and male patients with early-onset ACCHN. (d) Survival outcomes of early-onset ACCHN patients with different tumor sizes. (e) Survival outcomes of patients with early-onset ACCHN originated from different locations. (f) Survival outcomes of patients with early-onset ACCHN in different grades. (g) Survival outcomes of patients with early-onset ACCHN at different stages. (h) Survival curves of early-onset ACCHN patients treated with or without surgery. (i) Survival curves of early-onset ACCHN patients treated with or without radiotherapy. (j) Survival curves of early-onset ACCHN patients treated with or without chemotherapy.
Risk Factors of Overall Survival of Early-Onset ACCHN
As listed in Table 2. in univariate Cox Regression analysis, races other than white and black, male sex, pharynx and larynx, nasal cavity and accessory sinuses, more advanced stage, larger tumor size, no surgery, and chemotherapy performed were all associated with worse prognosis. All factors that were statistically insignificant were excluded. In multivariate analysis, some independent risk factors for overall survival (OS) rate were observed. ACCHN at the localized stage(HR = 0.383, 95%CI 0.227-0.646,P < .001) showed a significantly better prognosis than that of the regional stage, while distant stage (HR = 2.083 , 95%CI 1.296-3.346,P = .002) had the worst survival outcome among 3 stages. Patients who underwent surgery (HR = 0.403, 95%CI 0.237-0.688,P < .001) had longer survival time than those who did not. However, chemotherapy-performed patients (HR = 2.138, 95%CI 1.297-3.525,P = .003) showed worse prognosis. Moreover, larger tumor size also significantly related to worse survival(HR = 1.426, 95%CI 1.075-1.893, P = .014).
Univariate and Multivariate Analysis for Patients With Early-Onset Adenoid Cystic Carcinoma of Head and Neck (ACCHN).

Bold values are statistically difference.
Construction and Validation of Nomogram Related to Overall Survival
The prognostic nomogram was constructed based on the independent risk factors selected by the multivariate Cox regression analysis. As shown in the Figure 3, this nomogram for 10-year OS comprised 4 variables: tumor size, chemotherapy, surgery and stage. The instruction of this nomogram would be elaborated as follows:
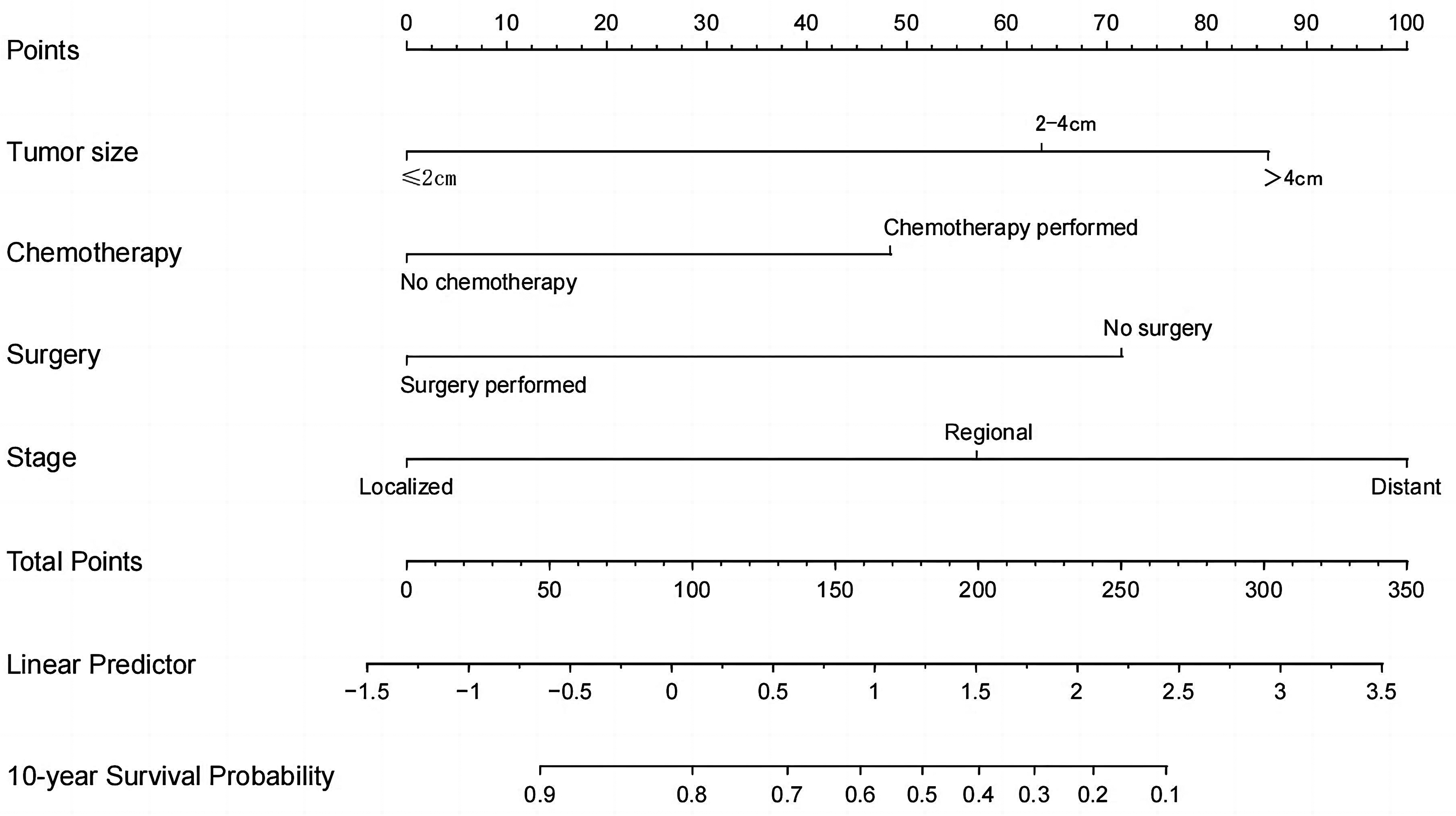
Nomogram for 10-year survival probability.
According to the clinical characteristics of each patient, we could draw a vertical line from a certain variable to the horizontal axis named “Points” on the top of the nomogram. The number this vertical line passed through in the “Points” axis was the score of this variable. A total score could be obtained by summing up all the scores of these 4 variables. Then, we found the corresponding points on the axis marked with “Total Points” and draw a vertical line from this point to “10-year Survival Probability”. The number we got was the predicted 10-year OS value.
The predictive accuracy of this nomogram was assessed by discrimination and calibration. In our study, the concordance index (C-index) and ROC curve were applied to estimate the discriminative ability. And calibration curve was plotted to evaluate the calibration of this prognostic nomogram. Moreover, validation was performed internally and externally in the training cohort and validation cohort. The C-index was 0.792 (95%CI 0.760-0.823) in the training set and 0.776 (95%CI 0.720-0.832) in the validation set. In the ROC analysis, the area under the ROC curve (AUC) values were 0.875 (95%CI 0.810-0.940) and 0.833(95%CI 0.754-0.912), respectively. Both C-index and AUC exceeded 0.75, which indicated that this nomogram had a good discrimination ability for 10-year OS. The Y-axis of calibration plot referred to the real OS and X-axis meant the predicted OS of nomogram. As shown in Figure 4c and d, the calibration curves of 10-year OS rate were close to the 45-degree ideal diagonal reference line both in the training set and validation set. This reflected that this nomogram had a good concordance between the predicted and observed probabilities.
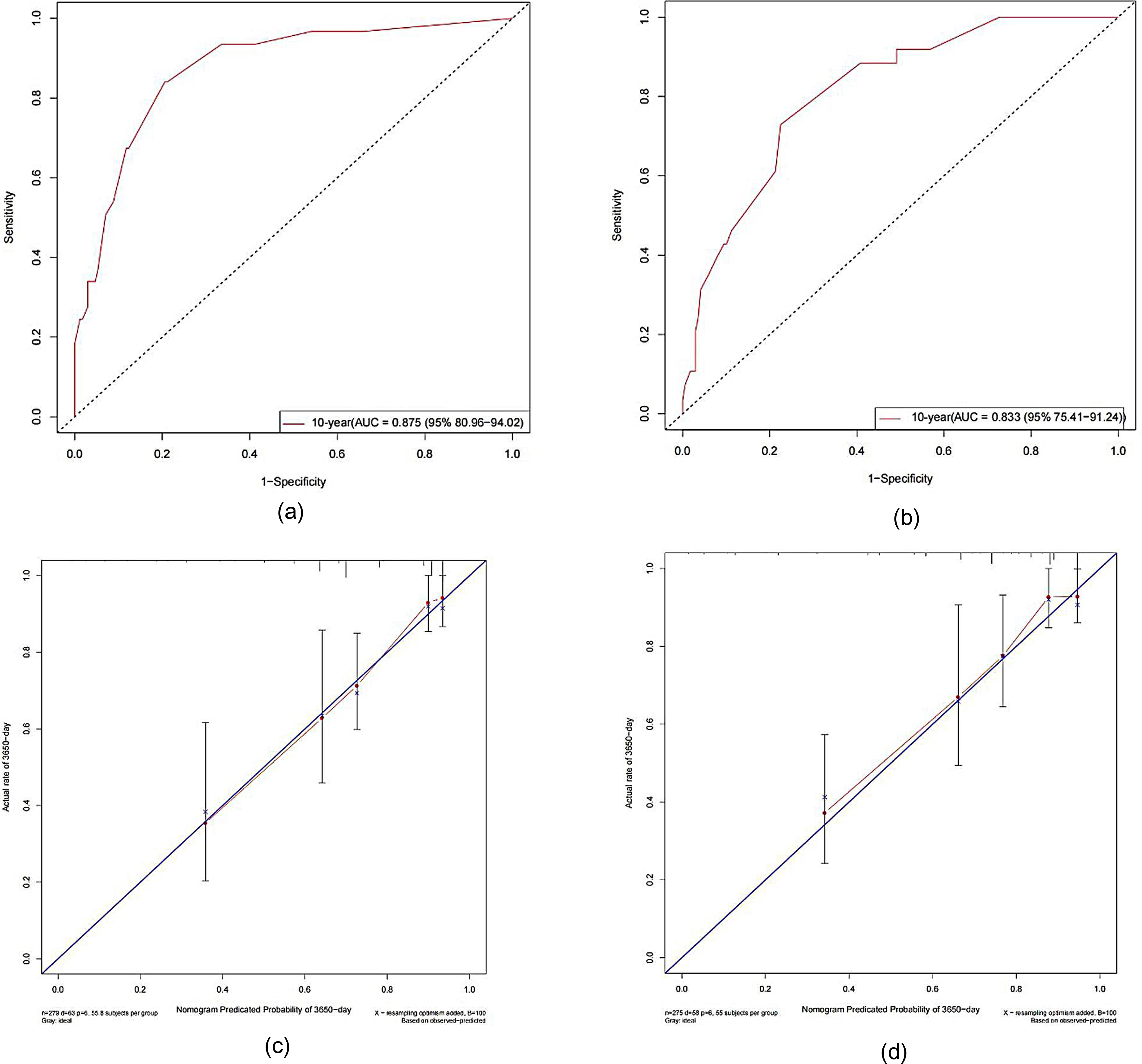
Receiver-operating characteristic curves (ROC) and calibration curves of the nomogram in training and validation cohort. (a) ROC curves in the training cohort. (b) ROC curves in the validation cohort. (c) Calibration plot of training cohort. (d) Calibration plot of the validation cohort.
Discussion
ACCHN is a rare head and neck cancer which is most commonly originates within minor and major salivary glands. 2 In the previous study, the predilection age of ACC was 40-60. 3 However, as shown in Supplemental Figure S1 and Table S1, the percentage of patients varying from 60 to 69 was significantly larger than that of 40 to 49 in both males and female groups. And the proportion of 70 to 79 group was also higher than that of the 40 to 49 group, especially in female. Thus, it might be reasonable to redefine the susceptible age group of ACCHN as 40 to 79. Recently, some researchers have found that the age of patients with some types of cancers was associated with the clinicopathological characteristics and ultimately influenced the prognoses. Colorectal cancer in individuals younger than 50, which is defined as early-onset colorectal cancer (EO-CRC), has been well-studied. Compared to CRC in other age groups, EO-CRC presents unique features in epidemiology, clinical presentation, molecular etiology, and hereditary.4,15,16 Bergquist et al 17 discovered that although the incidence of gastric cancer in American declined over decades, the incidence of early-onset gastric cancer (EOGC) was rising. Of note, EOGC showed genetic and clinical differences from traditional gastric cancer, with a more worrisome trend and a more aggressive phenotype. Individuals at any age are possible to contract ACCHN, but elderly person is more susceptible. 3 At present, there lacks a consensus on the definition of early-onset ACCHN, and relative research is scarce. However, some studies have revealed that ACCHN patients in different age groups exhibited distinct survival outcomes. Shen et al 18 declared that age was a strong risk factor in patients with ACCHN, and older patients were more likely to die of ACCHN. Mu et al 19 discovered that in patients older than 60, the possibility of ACC-specific death was 1.719 times as its younger counterparts. Ganly et al 20 found that younger age was associated with not only a higher OS rate, but also a lower 10-year recurrence probability. Thus, it is necessary to construct a more reliable model to accurately predict the prognosis of young patients. In this study, we defined early-onset ACCHN as cases younger than 40 years old. Early-onset ACCHN occupied approximately 14% of all patients and showed a dramatically better OS rate (Figure 2a). The independent risk factors were selected by Cox regression model: tumor size, stage, surgery, and chemotherapy. Then, a prognostic nomogram was constructed based on these selected variables. The predictive accuracy of this nomogram was internally and externally invalidated by C-index, ROC, and calibration plot.
The biological behavior of ACC is relatively indolent and the growth of tumor mass is commonly slow. 2 At present, clinicians generally hold a positive attitude to the short-term survival rate of patients with ACC. DeAngelis et al 21 reported a retrospective analysis of 24 patients with ACC derived from the minor salivary glands, and the 5-year survival rate was as high as 92%. However, the long-term survival rate dropped obviously for its persistent and recurrent growth pattern and late onset of metastases. 2 Thus, we chose to put attention to the 10-year OS rate in this nomogram.
Apart from the slow progress and the long course, ACC is also characterized by its aggressive infiltrative ability. 22 It was not rare to detect perineural invasion and lymphovasular invasion in ACC and the incidence of distant metastasis was as high as 40% to 50%.3,23 Based on these features, ASCO recommended that postoperative radiotherapy should be offered to all patients with resected ACC. 24 The effects of adjuvant radiotherapy on locoregional control have been validated in some studies.22,25,26 However, local and distant recurrence would eventually occur despite combined treatment with surgery and radiotherapy in some patients. 27 In this study, there was no statistical difference between patients with or without radiation (Figure 2i). This was might because there was too much-censored data and only 14 cases were confirmed that they refused radiotherapy and 254 patients were unknown if they were performed. Moreover, some researchers postulated that radiotherapy might only delay rather than prevent a recurrence.28,29 Some side effects might have a negative impact on patients’ survival such as necrosis of the soft tissues, craniofacial bones, and the temporal brain lobe, as well as cervical myelopathy. 30 Therefore, further investigation is needed to explore how to employ radiotherapy and minimize its side effects. Currently, data is insufficient to support that the use of chemotherapy benefits the survival of patients. In the latest ASCO guideline, concurrent chemotherapy is not recommended as a routine treatment. 24 In our study, young patients who took chemotherapy had a shorter survival time (Figure 2j). In the nomogram, chemotherapy also brought negative effects on 10-year survival probability. In accordance with these results, doctors might be supposed to hold a more prudent attitude toward the application of chemotherapy. Dos Santos found that ACC from major salivary glands showed a longer OS in comparison to other sites. 31 As shown in Table 2, we had similar results in univariate analysis: ACC from major salivary showed better survival outcomes while ACC from pharynx and larynx, nasal cavity, and accessory sinuses showed worse prognoses. But primary sites showed no statistically significant in multivariate analysis. This might be because primary site was a confounding factor that could be influenced by other factors.
However, there existed some limitations in our study. We chose patients’ information from 1975 to 2016 during which the regimens of surgery, radiotherapy, and chemotherapy changed significantly. For example, as to surgery, radical resection and local resection might lead to different outcomes. So, it is necessary to collect more data and stratify patients more clearly according to their therapeutic plans. Moreover, pathological type has been verified to be closely related to the prognoses of ACCHN patients. Because of the lack of pathological information in SEER database, we did not contain this part in our nomogram model.
Conclusion
We developed a prognostic nomogram model for early-onset ACCHN and validated this model using an independent patient cohort. This model showed an application prospect for predicting the OS of ACCHN patients which were helpful for the clinician in decision-making and personalized medicine.
Supplemental Material
sj-docx-1-tct-10.1177_15330338231163026 - Supplemental material for Prognostic Model and Nomogram for Early-Onset Adenoid Cystic Carcinoma of Head and Neck: A Retrospective SEER-Based Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338231163026 for Prognostic Model and Nomogram for Early-Onset Adenoid Cystic Carcinoma of Head and Neck: A Retrospective SEER-Based Analysis by Li Dai, Shi-zhou Zhang and Yi-hua Wu in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
The authors gratefully acknowledge the cooperation of Dr Luo xiaojie and Dr Yang xudong in preparing this article.
Contribution
Yi-hua Wu and Shi-zhou Zhang made a substantial contribution to the conception of the article and revised it critically. Li Dai was responsible for the acquisition and analysis of the data and drafted the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
This is a population study based on the SEER database (SEER*stat software, version 8.3.9) which is publicly available, and all information of patients throughout the study is de-identified. Because the information in the SEER database does not require the patient's explicit consent, the study is waived from ethical approval. The informed patient consent is not required due to the retrospective nature of the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the science project of the Shandong province, (grant number 2011GSF11842).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
