Abstract
Keywords
Introduction
Head and neck adenoid cystic carcinoma (HNACC) is an uncommon malignant tumor mainly derived from the salivary glands, comprising approximately 1% of all head and neck carcinomas and 10% of salivary gland neoplasms.1,2 Although HNACC can occur in various sites, such as the nasal cavity, throat, and oral cavity, it is characterized by its distinctive cystic features and glandular differentiation. HNACC typically exhibits a slow and infiltrative growth pattern, displaying high invasiveness and local infiltration capability. 3
Treatment of HNACC is often tailored based on the tumor's size, location, and stage. Surgical resection is the primary treatment modality aimed at completely removing the tumor tissue. Radiation therapy can be used to control tumor growth and prevent recurrence.4,5 However, the outcomes of HNACC treatment remain unsatisfactory. The heterogeneity of HNACC poses a challenge for radiotherapy, and the complex anatomical location of head and neck tumors limits the effectiveness of surgery or postoperative radiation therapy. 6 The survival rate for systemically treated HNACC patients is only 50% at 10 years after diagnosis, mainly due to treatment failure from local recurrence and metastatic spread.7,8 Consequently, HNACC presents significant obstacles in terms of treatment.
Particle therapy, a promising and evolving radiotherapy technique, shows potential in treating tumors that are resistant to conventional photon beam radiotherapy. When contrasted to traditional radiation, it has unique biological and physical benefits. The “Bragg peak,” a sharp and localized peak of dose, is used in particle treatment to precisely administer radiation to the tumor while effectively preserving normal organs.9,10
Currently, there are 2 main types of particle radiation therapy: proton radiation therapy and heavy ion radiation therapy. These modalities vary in their biological effects and are often compared using the relative biological effect (RBE), which measures the biological effect dose relative to 250 kV X-rays. The RBE increases with higher linear energy transfer (LET) under similar conditions. 11 Proton beams exhibit a LET considered intermediate between photons and heavy ions, categorizing them as low LET radiation. In clinical practice, the RBE of protons is typically set at 1.1. 12 The primary advantage of proton beams lies in their dose precision rather than their biological effect. Following proton radiotherapy, the majority of DNA damage consists of repairable single-strand breaks. 13 In contrast, carbon ions possess higher LET and RBE. In comparison to proton beams, carbon ion beams not only generate dose tails at extended distances 14 but also exhibit smaller lateral scattering and scattering ranges. 15 Radiobiological damage induced by carbon ion beams predominantly involves double-strand breaks, which are relatively challenging to repair. Moreover, carbon ion beams demonstrate an oxygen enhancement ratio of 1, resulting in a heightened killing effect on hypoxic, G0-phase, and S-phase cells. Consequently, they are viewed as having greater potential for treating radioresistant tumors. 13
Particle therapy holds unique advantages for treating HNACC. Its beam characteristics enable precise radiation dose delivery to the tumor, minimizing the impact on critical structures and reducing treatment-related side effects, ultimately improving patients’ quality of life.16,17
Due to the increasing interest in the efficacy and safety of particle therapy for HNACC, we conducted a systematic review and meta-analysis to assess the current impact of particle beam therapy on HNACC patients.
Materials and Methods
Protocol Registration
The study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines, and the review protocol was registered in PROSPERO (CRD42023399464). 18
Search Strategy
Our search strategy was determined based on PRISMA guidelines and recommendations. We conducted extensive searches on 5 databases, including Pubmed, Cochrane Library, Web of Science, Embase, and Medline, from the establishment date of each database until December 31, 2022. The interventions included the use of particle beam therapy (including proton beam therapy (PBT), carbon-ion radiation (CIRT), or their combination with photon therapy) for HNACC. Detailed search strategies for each database are provided in the appendix.
Inclusion and Exclusion Criteria
We included studies conducted after 1990 that met the following criteria: (1) Patients with adenoid cystic carcinoma (ACC) of the head and neck; (2) patients receiving either charged particle therapy or a combination of charged particle and photon therapy; (3) Reported relevant outcomes such as tumor control, survival, and complications; (4) Data from original studies, including randomized controlled trials, nonrandomized clinical trials, case series or observational studies; (5) The complete text has been released in English.
We excluded studies that met any of the following criteria: (1) Reviews, commentaries, or other nonoriginal studies; (2) sample sizes of less than 5 patients; (3) duplicate patient data; (4) inability to extract survival data of interest for HNACC; (5) median follow-up less than 6 months.
Data Extraction
Two reviewers (C.Y.L and D.T.Q) independently selected the literature, extracted the data, and evaluated the risk of bias for eligible studies. The results were cross-checked by a third reviewer (Z.Q.N). Any disagreements were resolved through discussion among the 3 investigators until a consensus was reached. The data extraction included the following information: (1) First author, journal information, publication year, research institution, study design, and study period; (2) follow-up time, number of patients, sex, age, tumor site, tumor size, tumor stage, doses, and fractionation regimens; (3) primary endpoints: Overall survival (OS), progression-free survival (PFS), and local control (LC). Secondary outcomes: Treatment-related toxicity; (4) evaluation indicators for quality and bias assessments;
Quality and Risk of Bias Assessment
Each study's quality was independently evaluated by 2 reviewers (W.Q and W.X). In case of disagreement, the third author (Z.Q.N) made the final decision. The risk of bias for all included studies was assessed using Cochrane's Risk of Bias in nonrandomized studies of interventions tool. 19 The risk of bias was categorized as low, moderate, severe, or serious.
Statistical Analysis
Descriptive statistics were used to compile baseline parameters and toxicity incidence. Frequencies and percentages were used to represent dichotomous data, whereas means with standard deviations or medians with interquartile ranges were used to describe continuous data. Since the case series studies were conducted in various settings, a random-effects model was employed to generate a broad summary estimate. Proportions with 95% confidence intervals (CIs) were calculated to determine the effect sizes for continuous outcomes. All analyses were performed using STATA version 17.0 (StataCorp).
Results
Search Results and Selection
A total of 307 potential articles were found in 5 databases and were thoroughly searched. After removing 125 duplicate papers, 182 items remained. Through title and abstract screening, 101 articles were eliminated. Among the remaining 81 studies, 67 were disregarded for either being old studies overlapping with the new study sample (n = 24) or not reporting any predetermined outcomes (n = 43). Finally, 14 studies were included in this meta-analysis. Our study consisted of 1 prospective study, 1 multicenter retrospective study, and the rest were single-center retrospective studies. The procedure for selecting studies for review is described using the PRISMA flowchart(Figure 1).

Flow chart of study selection.
Features of the Included Research
The included research spanned the years 2006 to 2022, with publications from different regions. Among the 14 included studies, 2 were from the USA,20,21 3 from China,22–24 4 from Japan,25–28 4 from Germany,29–32 and 1 from Switzerland. 33
Participants
A total of 1297 patients with ACC of the head and neck were included in all 14 studies, with the majority of cases located in the sinus region. The median age of the patients ranged from 45 to 60 years, and the median follow-up period was between 27.2 and 56 months. Seven studies involved patients with primary lesions, 6 involved patients with primary lesions and recurrence, and 1 involved patients with primary lesions and metastatic lesions. The characteristics of the 14 included studies are summarized in Table 1.
Baseline Characteristics of Included Studies.
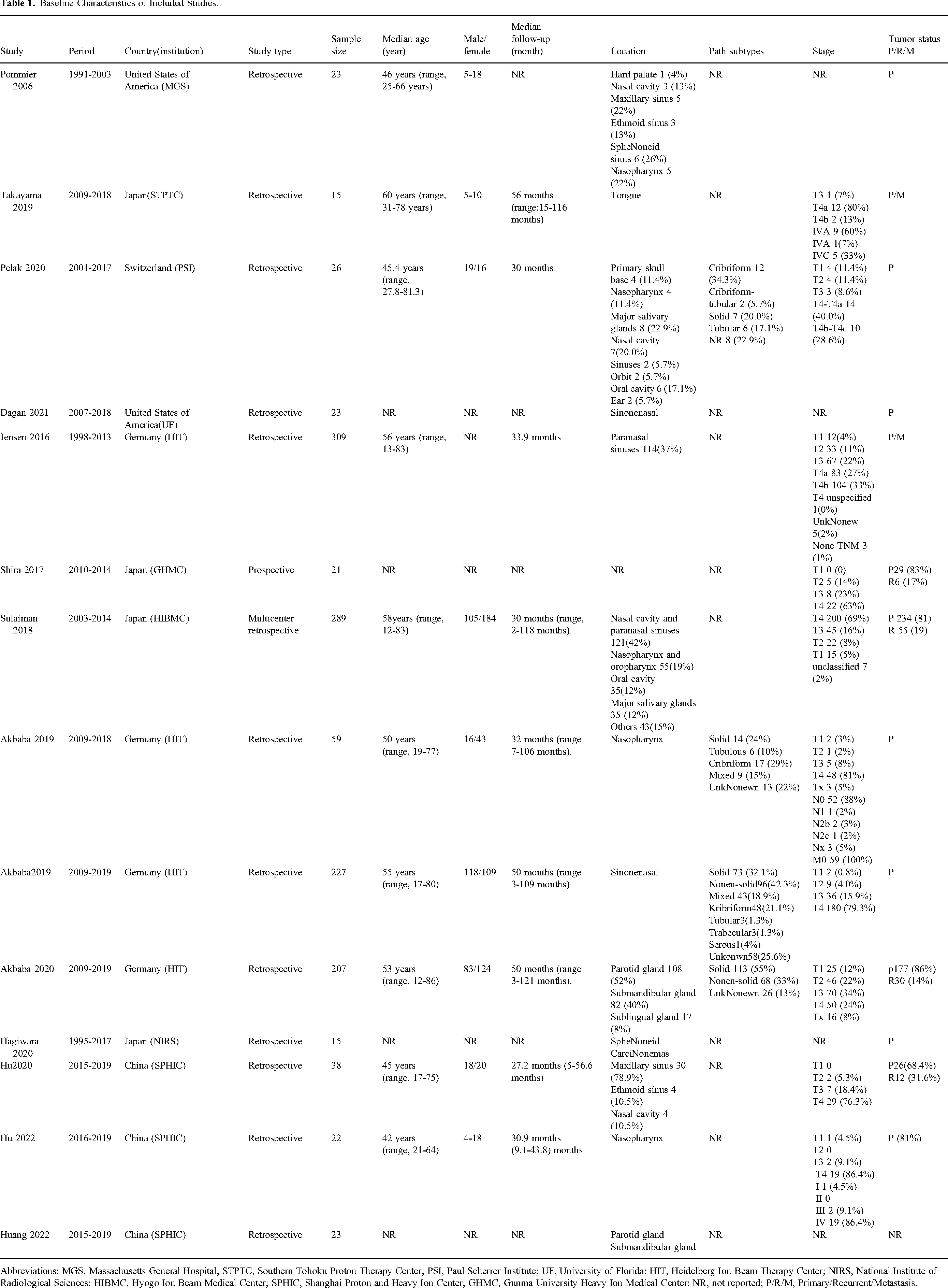
Abbreviations: MGS, Massachusetts General Hospital; STPTC, Southern Tohoku Proton Therapy Center; PSI, Paul Scherrer Institute; UF, University of Florida; HIT, Heidelberg Ion Beam Therapy Center; NIRS, National Institute of Radiological Sciences; HIBMC, Hyogo Ion Beam Medical Center; SPHIC, Shanghai Proton and Heavy Ion Center; GHMC, Gunma University Heavy Ion Medical Center; NR, not reported; P/R/M, Primary/Recurrent/Metastasis.
Interventions and Controls
In 3 studies, PBT alone was used as an intervention, and in 4 studies, CIBT alone was used. The remaining 7 studies used mixed beam methods, with 4 combining photons with either PBT or CIBT and 3 combining PBT with CIBT.
Outcomes
All investigations recorded survival outcomes such as OS, LC, and PFS. Additionally, toxic adverse effects were mentioned. The treatment regimens’ main results of all included studies are summarized in Table 2.
Treatment Regimens Main Results of all Included Studies.

Risk of Bias Assessment
Out of the 14 studies, 12 were classified as having a moderate risk of bias, while 2 were considered low risk. The majority of studies were classified as moderately risky due to the “Bias due to deviations from intended interventions” option. This classification was influenced by the uneven distribution of postoperative and definitive radiation patients, as well as the varying doses of particle treatment in the investigations.
Results of Meta-Analyses
Efficacy
Primary ACC
Only 3 studies (2 using PBT and 1 using CIRT) reported the 5-year OS for a total of 105 patients. The pooled 5-year OS rate was 78% (95%CI = 66-91%), with high interstudy heterogeneity (I2 = 60.7%, p = .078). The pooled 5-year PFS rate was 62% (95%CI = 47-77%), also showing heterogeneity (I2 = 57.6%, p = .094) (Figure 2).

Random-effects meta-analysis of 5-year OS and PFS among primary HNACC patients. Abbreviations: OS, overall survival; LC, local control; PFS, progression-free survival; HNACC, head and neck adenoid cystic carcinoma.
OS Rate for Particle Therapy
For particle therapy, the OS rate at 2.5 years was 86.1% (95%CI = 95%-100%, 5 studies, n = 419), and at 5 years, it was 77% (95%CI = 73-82%, 7 studies, n = 925). Specifically, for PBT, the 5-year OS rate was 83% (95%CI = 73-92%, 3 studies, n = 61), while for CIRT, the OS rate at 3.5 years was 83% (95%CI = 67-99%, 4 studies, n = 595), and at 5 years, it was 76% (95%CI = 71-81%, 4 studies, n = 864) (Figure 3).

Random-effects meta-analysis of 5-year OS of PBT and CIBT. Abbreviations: OS, overall survival; PBT, proton beam therapy; CIRT, carbon-ion radiation.
LC Rate for Particle Therapy
The LC rate for particle therapy at 2.5 years was 92% (95%CI = 84-100%, 5 studies, n = 419), and at 5 years, it was 73% (95%CI = 61-85%, 6 studies, n = 902). Specifically, for PBT, the 5-year LC rate was 92% (95%CI = 83-100%, 2 studies, n = 38), while for CIRT, the LC rate at 5 years was 66% (95%CI = 52-79%, 4 studies, n = 864) (Figure 4).

Random-effects meta-analysis of 5-year LC of PBT and CIBT. Abbreviations: LC, local control; PBT, proton beam therapy; CIRT, carbon-ion radiation.
PFS Rate for Particle Therapy
The PFS rate for particle therapy at 2.5 years was 76% (95%CI = 68-84%, 5 studies, n = 419), and at 5 years, it was 55% (95%CI = 48-62%, 6 studies, n = 915). Specifically, for PBT, the 5-year PFS rate was 67% (95%CI = 47-88%, 2 studies, n = 46), while for CIRT, the PFS rate at 3 years was 69% (95%CI = 64-75%, 3 studies, n = 574), and at 5 years, it was 52% (95%CI = 46-59%, 4 studies, n = 864) (Figure 5).
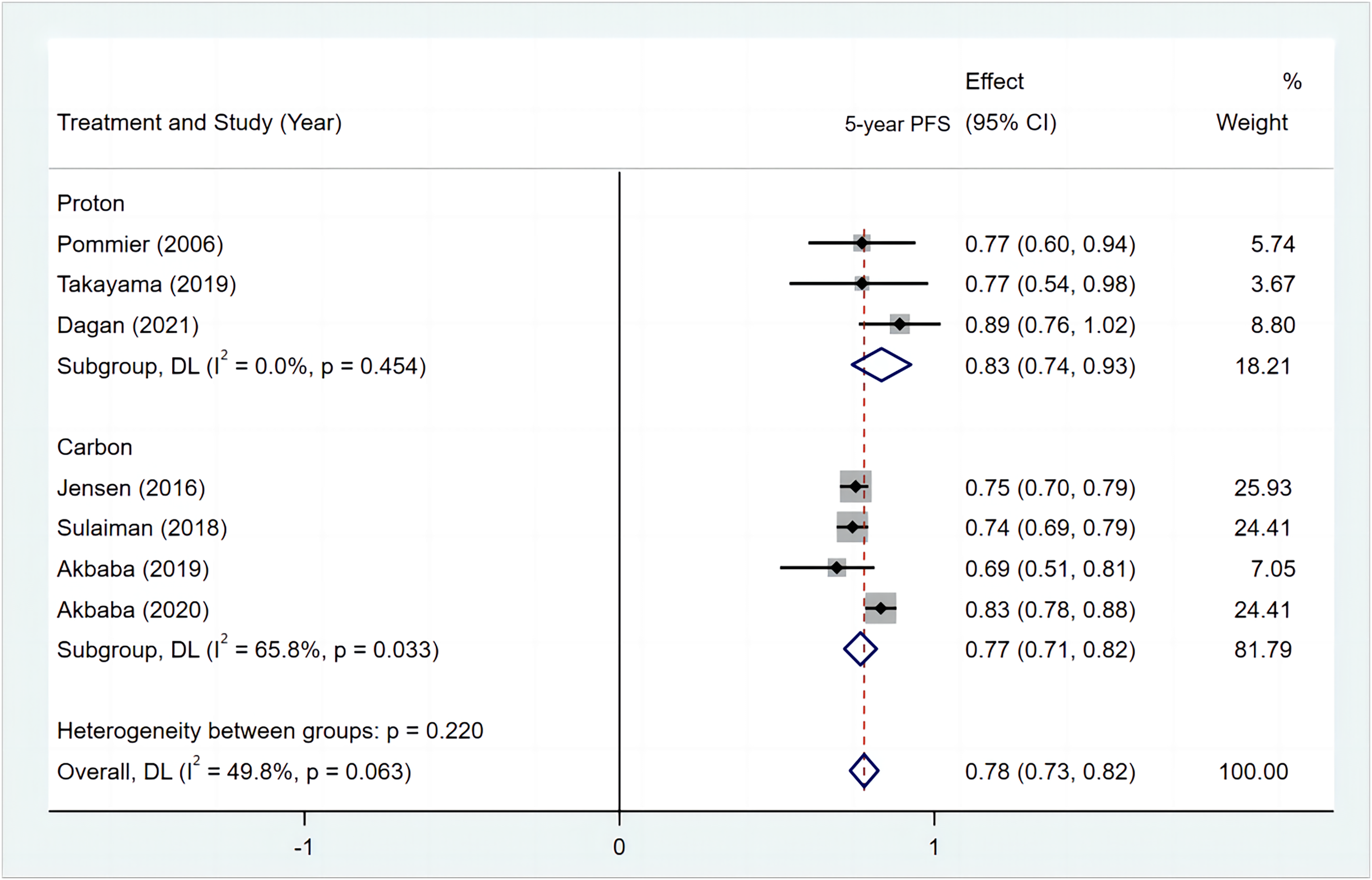
Random-effects meta-analysis of 5-year PFS of PBT and CIBT. Abbreviations: PFS, progression-free survival; PBT, proton beam therapy; CIRT, carbon-ion radiation.
Safety
Eight studies reported grade 3 and above acute toxicity. The incidence of grade 3 and above acute toxicity was 22% (95%CI = 13%-32%) (Figure 6). Acute toxicity is mainly manifested as mucositis, dermatitis, hearing loss, and other effects caused by different radiotherapy sites. Among the 3 studies that reported late toxicity, the incidence was 8% (95%CI = 3-13%) (Figure 7). Late toxicities mostly included dysphagia, radiation-induced osteonecrosis, and hemorrhage.

Grade 3 and above acute toxicity.
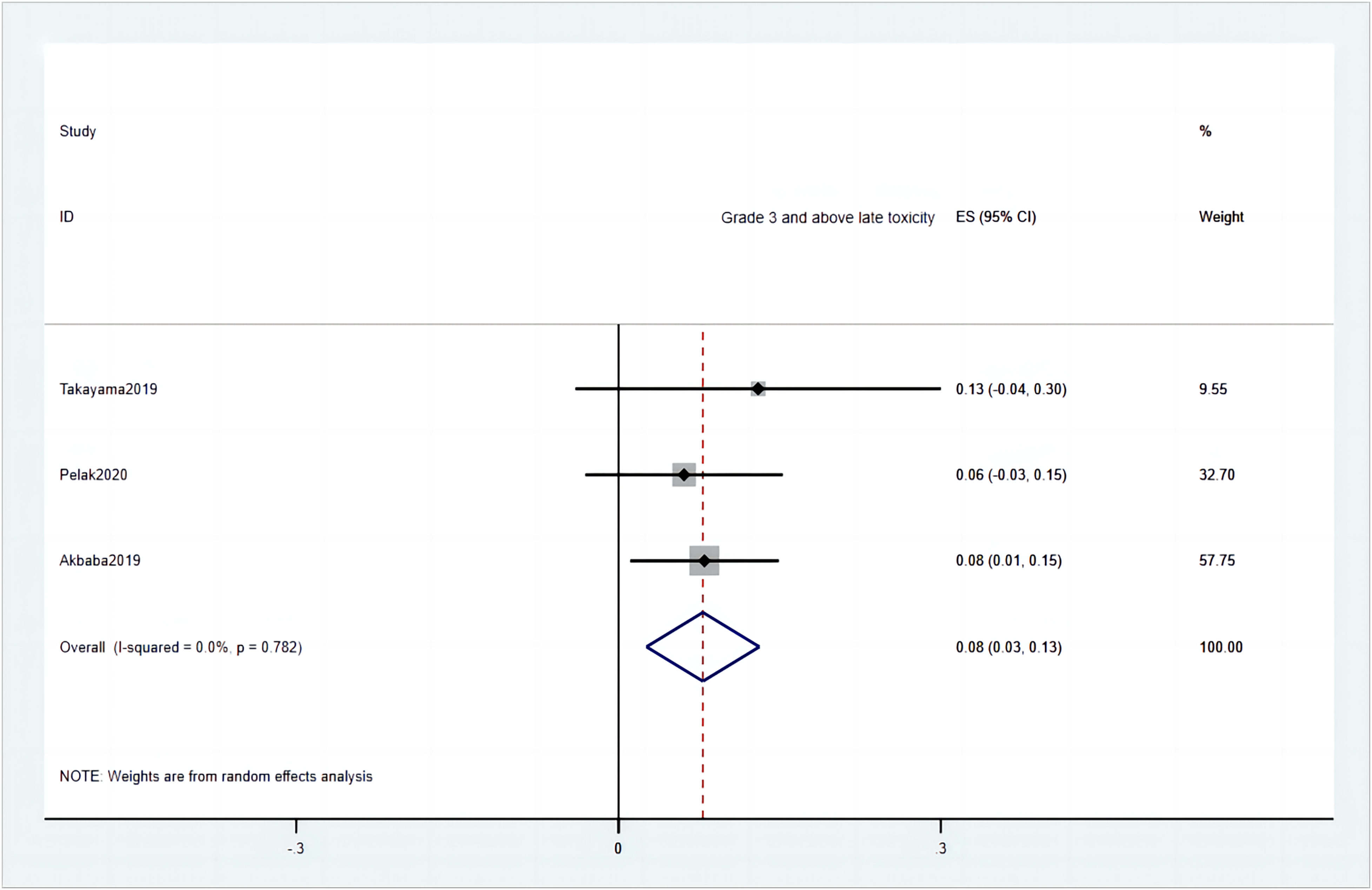
Grade 3 and above late toxicity.
Failure Patterns
Eight trials (PBT = 2, CIBT = 6) indicated a trend of HNACC therapy failure. In 1 PBT research, disease progression occurred in 13 (37.1%) patients (local failures: 4, distant metastases: 6, both: 3). 33 Another PBT trial 20 reported 2 local failures (6%) within the high-dose region. Among the CIBT studies, a research study 32 revealed that 66 patients (32%) experienced disease progression, with 25 patients (12%) encountering local relapses. In a separate CIBT investigation, 131 patients (43%) showed disease progression, with 52% experiencing local failures, 74% developing distant metastases, and 26% experiencing both. 29 Further analysis of the 68 patients with local recurrences revealed that 46% had new tumor activity within the high-dose region, 22% along the field border, 10% outside the treatment field, and 1 case in local lymph nodes. 29 Another CIBT study reported 45 patients (16%) with local recurrences, with 41 instances (15%) occurring within the high-dose region, 4 cases (1%) along the field border, and 5 cases (2%) in the neck. 27 Three additional investigations reported in-field recurrence rates of 2 (5%), 8 (42%), and 27 (44.3%) individuals, respectively.23,30,31 Across all the reports, the most common site of distant metastasis was the lungs.
Treatment-Related Death
Two studies recorded treatment-related deaths. In 1 study, 2 individuals had nasopharyngeal ACC with T4 tumors that almost encircled the carotid artery. All patients showed rapid tumor shrinking in response to C-ion radiation but ultimately passed away due to ulceration and bleeding at the tumor site. 26 In another study, 1 case of sinonasal ACC passed away from hemorrhage 10 months after CIRT reirradiation as salvage treatment. 23
Discussion
The majority of studies included in this systematic review were conducted between 2018 and 2022. The combination of CIBT and photon beams is widely used for treating HNACC, with Germany, Japan, and China having the highest concentrations of carbon ion therapy. Our study revealed the following findings: (1) The 2-year and 5-year OS rates for particle beam therapy in HNACC are 97% and 77% respectively; (2) the 2-year and 5-year LC rates are 92% and 73%; (3) the 2-year and 5-year OS and PFS rates for particle beam therapy in primary HNACC are 78% and 62%; (4) the 5-year OS, LC, and PFS rates for CIBT in HNACC are 77%, 65%, and 77%, respectively. For PBT, the corresponding rates are 83% for OS, 92% for LC, and 83% for PFS;(5) particle therapy for HNACC is generally safe, with an acute toxicity rate of G3 at 22% and a late toxicity rate of 8%.
This study compared the 5-year survival rates of CIBT and PBT and found no significant differences, except for LC. The variation in LC can be attributed to factors such as the higher proportion of patients with T4 stage and differences in tumor irradiation site. For instance, in 1 study, a majority of patients were classified as T4a/b stage (60%) and underwent radiotherapy due to a high rate of subtotal resection (72%), resulting in a large radiation fields. 31 Another study revealed that patients with T4 stage accounted for 81% of the total cases, and subgroup analysis indicated a 5-year LC rate of only 35% for T4 cases, leading to low LC. 30 Moreover, it is worth noting that CIBT constituted a significant proportion (93%) of all patients included in this study, which likely reflects the practicality of particle therapy.
In the study, it was concluded that there is a frequent occurrence of local tumor recurrence within the high-dose region. For instance, Sulaiman's study 26 reported that out of the 45 cases of local recurrence, 41 (91%) occurred in the high-dose region. Notably, this study utilized CIBT alone, with a median radiotherapy dose of 64 Gy (RBE), and a significant proportion of patients (25%) received doses below 60 Gy (RBE). This raises the question of whether the limitation of normal tissue dose in the high-dose region contributes to an increased likelihood of internal recurrences. Another study conducted by Gao 34 focused on intensity-modulated radiotherapy (IMRT) for HNACC, where they found that 8 of 10 had recurrent lesions located within the radiotherapy regions. This finding suggests that recurrence within the high-dose region is also common in conventional IMRT approaches. In contrast, HIT29–32 utilized an IMRT + C12 treatment modality, where patients received a median total dose greater than 74 Gy after 25 + 8 fractions. However, despite the higher treatment weighting of C12, approximately 40% of the recurrences still occurred within the radiotherapy region. This raises the question of whether increasing the treatment weighting of C12 is effective in reducing recurrences within the radiotherapy region, which warrants further investigation.
Three studies from Japan25–27focused on CIRT alone and reported 2-year OS, LC, and PFS rates of 94%, 88%, and 68%, respectively. The 4-year OS and LC rates were 81.5% and 46.4% respectively. The estimated 5-year OS, LC, and PFS rates were 74%, 68%, and 44% respectively. One study reported 43 cases (15%) of late grade 3 or higher toxicity, with osteonecrosis of the jaw being the most common. Two patients treated with ACC in the nasopharynx died from bleeding ulcers at the tumor site.
It is important to note that among all the research, only the 3 studies from China22–24 used PBT + CIRT. However, only 1 article reported the toxicity time for the PBT + CIRT treatment group. 23 Out of 19 acute grade 2 toxic events, including mucositis, dermatitis, and dry mouth, 10 patients received PBT and CIRT combined therapy. Unfortunately, they did not report survival rates for PBT + CIRT separately. Nine of the 15 patients who experienced grades 1 to 2 advanced toxicity events underwent combined PBT and CIRT treatment. The risk of toxic effects with the combination of PBT + CIRT is tolerable due to the lack of grade 3 and above acute and late tertiary toxic responses.
Currently, IMRT stands as the predominant radiotherapy treatment technique. IMRT is a three-dimensional and conformal treatment method administered through linear accelerators equipped with either static multileaf collimators (MLC, in step-and-shoot IMRT) or dynamic leaf MLCs, Tomotherapy(Tomo) machines, or volumetric arc modulated therapy (VMAT). This approach effectively optimizes volumetric irradiation of irregular shapes. 35
Recent trials of postoperative HNACC patients treated with IMRT have also shown positive outcomes for OS and LC. Xu 36 retrospectively examined clinical data from 55 HNACC patients who underwent postoperative IMRT from 2007 to 2016. The patients were divided into R0, R1, and R2 surgical resection groups, with 22 (40.0%), 20 (36.4%), and 13 (23.6%) patients, respectively. Among them, 54.5% of the patients received adjuvant chemotherapy, and the median total radiation dose was 62 Gy (range 46-72 Gy). The 5-year OS, distant metastasis-free survival (DMFS), and local-regional recurrence-free survival (LRRFS) rates were 93.9%, 75.3%, and 82.5%, respectively. Myelosuppression (5.5%), mucositis (9.1%), and dermatitis (1.8%) were the most prevalent acute toxicities of grade 3 or higher. Another study by Chen 22 reviewed 227 patients with nonmetastatic HNACC treated with postoperative radiotherapy from 2000 to 2017.IMRT was administered to 213 patients, of whom 92% received a total dose of 60 to 72 Gy to the GTV and/or involved lymph nodes. The 5- and 10-year LRRFS, DMFS, and OS rates were 89%, 68%, 79% and 63%, 88%,71% respectively. Choi 37 examined 126 patients with nonmetastatic HNACC who underwent postoperative radiation between 2005 and 2019. A median dose of 63.0 Gy (range 36.0-70.4) of IMRT was given to 100 patients at the primary location. The 5-year LRRFS, PFS, and OS rates were 69.6%, 46.6%, and 81.1%, respectively. However, it should be noted that patients with HNACC treated with postoperative radiotherapy generally have earlier tumor stages and higher KPS scores, which lead to better survival outcomes.
Now, let's focus on definitive radiation for HNACC. In a retrospective analysis of 44 HNACC patients treated with radiation therapy from 1993 to 2017, 20 patients received definitive radiotherapy, while 22 patients received postoperative radiotherapy. The 5-year OS rates for these 2 groups were 85.3% and 79.7%, respectively, and the 5-year LC rates were 82.5% and 83.1%, respectively. There were no significant differences in OS or LC rates between the 2 groups (p = .4392, p = .0904, respectively). Grade 3 mucositis affected 5 patients (11.4%) as an acute adverse event. 38 Another study by Swain 39 retrospectively analyzed 23 patients with locally advanced HNACC who underwent definitive chemoradiotherapy from 2011 to 2018. 74% of these patients received IMRT. The 3- and 5-year OS, LRRFS, and DMFS rates were 78%, 79.7%, 67.4%, and 63%, 50.9%, 48.6%, respectively. Three patients experienced acute grade 3 mucositis. However, the results were somewhat disappointing. Unfortunately, we could not find studies on definitive IMRT in inoperable HNACC patients, so relevant data were lacking. More comprehensive and precise information on CIBT in HNACC may come from the findings of Phase II clinical study NCT04214366, which is currently investigating postoperative or definitive CIBT alone in HNACC.
However, despite the emergence of newer techniques such as Tomotherapy, VMAT, MR-linac, and FLASH, the percentage of VMAT or Tomotherapy and its associated variances have not been addressed in published IMRT studies of HNACC. Nevertheless, insights gleaned from radiation therapy for other head and neck tumors shed light on the distinctions among several emerging treatment modalities. For instance, a study comparing Tomotherapy with conventional IMRT techniques in head and neck cancer treatment demonstrated that Tomotherapy enhances the homogeneity of dose distribution within the tumor region and shows promise in safeguarding critical organs. 40 Another study, involving 10 patients with head and neck tumors, compared separate Tomotherapy and VMAT treatment plans and concluded that Tomotherapy exhibited superior overall treatment plan quality in cases where satisfactory target coverage was achieved with both techniques. 41 Furthermore, while magnetic resonance imaging (MRI) simulation is gaining traction in radiation therapy, CT remains the primary imaging tool for simulation and treatment planning due to the quantitative electron density information necessary for dose calculation. By integrating methods like volume density distribution and synthetic MRI to estimate electron density on MRI, MRI-only workflows can be realized, reducing patient exposure to ionizing radiation and eliminating potential alignment errors. 42 Although MRI-based head and neck radiation therapy has demonstrated feasibility, 43 its future trajectory warrants further exploration. In recent years, the novel FLASH technique has garnered significant interest from radiation oncologists. Named for its use of electron pulses at higher dose rates (>40 Gy/s), FLASH supersedes the traditionally employed dose rate (approximately 0.05 Gy/s). 44 Additional experiments have indicated that FLASH yields equivalent tumor-killing efficacy as conventional radiotherapy while offering superior normal tissue protection. In clinical practice for head and neck tumors, the application of the FLASH technique has proven most effective in dosimetric studies for reducing doses to the brainstem, spinal cord, and parotid gland. 45 These innovative techniques are poised to propel the field of radiation therapy forward as it continues to evolve.
It is worth mentioning that ACC is less likely than other head and neck tumors to spread to the lymph nodes. 46 However, perineural invasion (PNI) is quite common even in the early stages of HNACC.47,48 PNI is an independent risk factor for developing HNACC and is significantly associated with worse rates of OS, LC, and disease-free survival, as reported by Ju. 49 A recent study investigated the effects of carbon ion and photon irradiation on PNI-associated neurotrophic factor-3 (NT-3) in primary mucosal melanoma (PMM) and pancreatic ductal adenocarcinoma (PDAC) cells in vitro. The results showed that carbon ion preference usually reduces cell viability, proliferation, and migration and that the increase in NT-3 after irradiation has the opposite effect. 50 Particle therapy may have more advantages than conventional photon radiotherapy in improving PNI.
As of January 2024, there are currently 14 operational carbon ion processing centers globally, with an additional 5 under construction, marking a net increase of 2 operational centers compared to 2021. The number of proton centers surpasses this, with 117 currently operational and 28 under construction.51,52 Despite the inherent advantages of particle therapy, such as Bragg peaks and enhanced RBE, its full benefits are realized only when challenges related to beam delivery and target motion are mitigated. To address the drawbacks of particle therapy, new technologies that have progressively been applied to photon therapy, such as VMAT, FLASH, and MRI-guided radiotherapy, are now being increasingly integrated into particle therapy practices. Looking ahead, it is conceivable that faster, more precise, and more effective particle therapy techniques will emerge, potentially positioning it as the radiation therapy modality of choice in the future.
However, there are limitations to particle therapy. Firstly, particle therapy equipment is scarce, and many areas lack access to this advanced technology. Secondly, particle therapy is associated with higher costs, which may pose challenges for patients and healthcare institutions. Jensen 53 evaluated the cost-effectiveness of IMRT plus carbon ion (C12) boost for HNACC compared to IMRT alone. The results showed an additional total treatment cost of €18,076 (€13 416- €22 922) and a mean survival benefit of 0.86 years. Although LC was better, the experimental treatment also incurred higher follow-up expenditures. Li 54 assessed the cost-effectiveness of PBT for nasopharyngeal cancer patients and found that the cost of PBT is 3.2 to 4.8 times that of IMRT. PBT should provide at least ≥24% probability of normal tissue complications to be considered cost-effective. Therefore, when developing treatment plans, it is important to consider all relevant economic, practical, and medical aspects.
This meta-analysis has several limitations that should be acknowledged: (1) The included studies did not include randomized controlled trials; (2) the patient population was relatively small, especially in the PBT treatment group; (3) the median follow-up time was short, with no studies having a median follow-up time of more than 5 years; (4) some of the included studies involved multiple pathological types, and there was a lack of information extraction from various key sources; (5) the treatment regimens varied among the studies, with some using a combination of particle-based treatments with concurrent chemotherapy or targeted therapy while others did not have uniform systemic treatment regimens; (6) publication bias may be present as unpublished data were not included in the analysis.
Despite these limitations, the findings of this investigation suggest potential benefits of particle therapy for patients with HNACC. Future research efforts should focus on larger-scale multicenter studies with longer follow-up periods to further optimize and incorporate particle therapy into HNACC treatment. A multinational prospective randomized trial, NCT02838602, comparing carbon ion therapy with photon or photon and proton combination therapy as treatment options for unresectable or incompletely resected radiation-resistant tumors (including HNACC) is currently underway. These trials will provide more robust evidence regarding the effectiveness, long-term outcomes, and cost-effectiveness of particle therapy. Furthermore, in particle therapy, it is crucial to optimize the target volume and enhance the dose distribution within the target area and organs at risk. This optimization aims to maximize the dosimetry advantages offered by particle therapy.
Conclusion
Particle therapy shows promise as an emerging radiation treatment modality in the management of HNACC. It offers advantages such as higher LC rates, improved OS rates, and reduced treatment-related toxicities. However, the limited availability and cost considerations associated with particle therapy require further research and optimization. With further development and efforts to customize treatment approaches, particle therapy has the potential to enhance treatment outcomes and improve the quality of life for patients with HNACC.
Footnotes
Acknowledgments
Mingyu Tan expresses heartfelt gratitude to his mentor, Professor Wang Xiaohu, whose rigorous guidance, kindness, and humility have left an indelible mark. He also extends sincere thanks to his parents and his wife's family for their unwavering support and care throughout his life and career.
Author Contributions
Methodology, conceptualization, and writing—original draft were done by Mingyu Tan. Data curation was done by TianQi Du and Yanliang Chen. Investigation was conducted by Qian Wang and Xun Wu. Project administration was done by Qiuning Zhang and Xiaohu Wang. Writing—review and editing was done by Qiuning Zhang. Validation was done by Hongtao Luo. Software was handled by Zhiqiang Liu and Shilong Sun. Resources were handled by Jinhui Tian and Kehu Yang. Supervision was done by Xiaohu Wang.
Data Availability
Research data are stored in an institutional repository and will be shared upon reasonable request to the author Mingyu Tan(tanmingyudoc@126.com).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Key Research and Development Program of China (No.2022YFC2401500), Gansu Province Project of Science and Technologies (Grant No. 20JR10RA680, 22CX8JA149), Science and Technology Project of Lanzhou City (Grant No. 2023-1-9), Science and Technology Plan Project of Chengguan District of Lanzhou (No. 2020-2-2-5), and the Lanzhou heavy Ion Accelerator High-end user Project (HIR20GY007).
Ethics Statement
As this is a meta-analysis utilizing data from published articles, ethical approval is not necessary.
Abbreviations
Appendix
Pubmed, Web of scince
Patient
#1 “Salivary gland”OR“nasopharynx” OR “nasopharyngeal” OR “paranasal Sinus” OR “paranasal” OR “oropharyngeal” OR “oropharynx” OR “oral cavity” OR “pharynx” OR“hypopharynx”
#2“cylindroma”OR“adenoid cystic”OR“ACC”
#3 #1 AND #2
#4 (“Cancer” OR (neoplasm*)OR(malignan*) OR (oncol*) OR (carcinoma*[mesh]) OR “tumor” OR “tumour”)
#5 ((Head and neck neoplasms [mesh]) OR (salivary gland neoplasms [mesh]) OR (oropharyngeal neoplasms[mesh]) OR (nasopharyngeal neoplasms [mesh]) OR(pharyngeal neoplasms [mesh]) OR (paranasal sinus neoplasms [mesh]))
#6= (#3) AND (#4 OR #5)
Intervention and comparison
#7(((((((Charged Particle) OR (Charged Particle Therapy)) OR (Proton)) OR (Proton Therapy)) OR (Proton Radiation Therapy)) OR (Carbon ion)) OR (Carbon ion Therapy)) OR (Carbon ion Radiation Therapy)
Outcome
#8 “Mortality”OR (adverse effect*) OR (adverse event*) OR(side effect*) OR (toxicit*) OR “diseasefree survival” OR “overall survival” OR “local control”OR “tumor control” OR “tumour control”
#9 (Mortality [mesh]) OR (head and neck neoplasms mortality [mesh]) OR (treatment outcome [mesh]) OR(adverse effects [subheading]) OR (survival rate [mesh])
#10= #8 OR #9
PICO = #6 AND #7 AND #10
EMBASE Medline
#1(Salivary gland)/br OR (nasopharynx) OR (nasopharyngeal) OR (paranasal Sinus) OR (paranasal) OR (oropharyngea) OR (oropharynx) OR (pharynx) OR (oral cavity) OR (hypopharynx)
#2(cylindroma)/br OR (adenoid cystic) OR (ACC)
#3(Cancer)/br OR (neoplasm*) OR (tumor) OR (tumour) OR (malignan) OR (oncol*) OR (carcinoma*) OR ((Cancer)/br OR (neoplasm*) OR (tumor) OR (tumour) OR (malignan) OR (oncol*) OR (carcinoma*))
#4'charged particle’ OR (charged AND particle) OR (charged AND particle AND therapy) OR proton OR (proton AND therapy) OR (proton AND radiation AND therapy) OR (carbon AND ion) OR (carbon AND ion AND therapy) OR (carbon AND ion AND radiation AND therapy)
#5(Mortality)/br OR (adverse effect*) OR (adverse event*) OR (side effect*) OR (toxicit) OR (diseasefree survival) OR (overall survival) OR (local control) OR (tumor control) OR (tumour control) OR ((mortality)/br OR (head and neck neoplasms mortality) OR (treatment outcome) OR (adverse effects) OR (sickness impact profile) OR (survival rate))
('salivary gland'/exp OR 'salivary gland’ OR (salivary AND ('gland'/exp OR gland)) OR nasopharynx OR nasopharyngeal OR (paranasal AND sinus) OR paranasal OR oropharyngea OR oropharynx OR pharynx OR (oral AND cavity) OR hypopharynx) AND ('cylindroma'/exp OR cylindroma OR (adenoid AND cystic) OR acc) AND ('cancer'/exp OR cancer OR neoplasm* OR tumor OR tumour OR malignan OR oncol* OR carcinoma*) AND ('charged particle’ OR (charged AND particle) OR (charged AND particle AND therapy) OR proton OR (proton AND therapy) OR (proton AND radiation AND therapy) OR (carbon AND ion) OR (carbon AND ion AND therapy) OR (carbon AND ion AND radiation AND therapy)) AND (adverse AND effect* OR (adverse AND event*) OR (side AND effect*) OR toxicit OR (diseasefree AND survival) OR (overall AND survival) OR (local AND control) OR (tumor AND control) OR (tumour AND control) OR 'mortality'/exp OR mortality OR (head AND neck AND neoplasms AND mortality) OR (treatment AND outcome) OR (adverse AND effects) OR (sickness AND impact AND profile) OR (survival AND rate))
Cochrane
Patient
#1 Salivary gland OR nasopharynx OR nasopharyngeal OR paranasal Sinus OR paranasal OR oropharyngeal OR oropharynx OR oral cavity OR pharynx OR hypopharynx
#2cylindroma OR adenoid cystic OR ACC
#3 #1 AND #2
#4 (Cancer OR (neoplasm)OR(malignan) OR (oncol) OR (carcinoma[mesh]) OR tumor OR tumour) OR (Head and neck neoplasms) OR (salivary gland neoplasms) OR (oropharyngeal neoplasms) OR (nasopharyngeal neoplasms) OR(pharyngeal neoplasms) OR (paranasal sinus neoplasms))
Intervention and comparison
#5′charged particle’ OR (charged AND particle) OR (charged AND particle AND therapy) OR proton OR (proton AND therapy) OR (proton AND radiation AND therapy) OR (carbon AND ion) OR (carbon AND ion AND therapy) OR (carbon AND ion AND radiation AND therapy)
Outcome
#6 MortalityOR (adverse effect) OR (adverse event) OR(side effect) OR (toxicit) OR diseasefree survival OR overall survival OR local controlOR tumor control OR tumour control
('salivary gland'/exp OR 'salivary gland’ OR (salivary AND ('gland'/exp OR gland)) OR nasopharynx OR nasopharyngeal OR (paranasal AND sinus) OR paranasal OR oropharyngea OR oropharynx OR pharynx OR (oral AND cavity) OR hypopharynx) AND ('cylindroma'/exp OR cylindroma OR (adenoid AND cystic) OR acc) AND ('cancer'/exp OR cancer OR neoplasm* OR tumor OR tumour OR malignan OR oncol* OR carcinoma*) AND ('charged particle’ OR (charged AND particle) OR (charged AND particle AND therapy) OR proton OR (proton AND therapy) OR (proton AND radiation AND therapy) OR (carbon AND ion) OR (carbon AND ion AND therapy) OR (carbon AND ion AND radiation AND therapy)) AND (adverse AND effect* OR (adverse AND event*) OR (side AND effect*) OR toxicit OR (diseasefree AND survival) OR (overall AND survival) OR (local AND control) OR (tumor AND control) OR (tumour AND control) OR 'mortality'/exp OR mortality OR (head AND neck AND neoplasms AND mortality) OR (treatment AND outcome) OR (adverse AND effects) OR (sickness AND impact AND profile) OR (survival AND rate))
Risk-of-Bias Summary for the Studies Included in the Meta-analysis, Using Cochrane Risk-of-Bias Tool ROBINS-I.
