Abstract
Objective
Small cell carcinoma of the cervix (SCCC) is rare but extremely aggressive and resistant to current therapies. We herein evaluate the efficacy of bevacizumab, apatinib, and anlotinib in recurrent/metastatic SCCC patients in a real-world setting.
Methods
Recurrent/metastatic SCCC patients were recruited between January 2013 and July 2020. Baseline characteristics were extracted from medical records, and patients were divided into an anti-angiogenic group and non-anti-angiogenic group. The efficacy of treatments was determined using Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria. Kaplan–Meier analysis was performed for survival analysis.
Results
Sixteen patients received anti-angiogenic drugs after tumor recurrence/metastasis; of them, 10 cases received them as first-line treatment, 5 cases as second-line treatment, and 1 case as fourth-line treatment. Another 23 patients received traditional therapies, including surgery, chemotherapy, and radiotherapy. The use of anti-angiogenic drugs in first-line treatment significantly prolonged progression-free survival (PFS) compared to the controls, with a median PFS of 8 months (2-20 months) and 3 months (1-10 months), respectively (P = .025). This trend was also notable in patients who started anti-angiogenic treatment after the second-line recurrence/metastasis. However, there was no benefits for overall survival (OS) in either the 10 first-line cases or all 16 cases (P = .499 and .31, respectively). Both bevacizumab and small molecule drugs (apatinib and anlotinib) presented similar efficacy in SCCC patients.
Conclusions
At present, this is the largest cohort study that provides real-world data, showing that anti-angiogenic regimens could significantly prolong PFS in recurrent/metastatic SCCC. Aside from bevacizumab, the novel oral small molecule drugs provide more choices with similar efficacy. These findings warrant further validation in well-designed future studies.
Introduction
Small cell carcinoma of the cervix (SCCC) is a rare and extremely aggressive subtype of cervical cancer. 1 SCCC is characterized by its significant resistance to current therapies and a high incidence of early metastasis, even in cases where the primary tumor is relatively small. 1–3 For early SCCC, the 5-year overall survival (OS) is 81.8% (stage IA) and 55.4% (stage IB), while for advanced tumors, the 5-year OS sharply drops to less than 25% in stage IIB and IIIB patients and 10% in stage IV patients. 4 This is much poorer than the survival rates of patients with squamous cell or adenocarcinoma of the cervix. 5
Due to its rarity, multicenter, randomized studies exploring optimal therapies for SCCC are challenging to perform. Current evidence is primarily obtained from small observational studies, and is inconsistent and sometimes controversial. 6,7 Similar to treating other cancers, a combination of surgery, chemotherapy, and radiation is most frequently used for early SCCC, while chemotherapy plus radiation is the preferred choice for late SCCC. 8,9 Regarding chemotherapeutic regimens, etoposide + cisplatin (EP) is the first recommendation, and taxol + cisplatin/carboplatin (TP/C) is a possible substitution when EP is not applicable. 10 For recurrent/metastatic cases, no consensus has been reached regarding the best chemotherapeutic regimens. A large group of drug candidates such as topotecan, irinotecan, pemetrexed, gemcitabine, vincristine, and doxorubicin have been tested in different studies, but have limited efficacy. 6,11,12
Angiogenesis is a classic characteristic of most human cancers and has been demonstrated to be a potent therapeutic target. 13 Bevacizumab was the first anti-angiogenic drug approved by the Food and Drug Administration (FDA) in 2004. 14 This drug specifically binds with vascular endothelial growth factor (VEGF) and blocks the VEGF/VEGFR signaling pathway. 14 In metastatic colorectal cancer, the first phase 3 clinical trial proved that the addition of bevacizumab to chemotherapy achieved significant survival benefits, which has been supported by several other studies. 15–17 For non-small-cell lung cancer (NSCLC), the efficacy of bevacizumab is also promising. A key study (E4599) reported an elongated median OS (10.3-12.3 months) in NSCLC patients who received a combination treatment of bevacizumab and paclitaxel + carboplatin. 18 Further studies demonstrated that bevacizumab was beneficial for NSCLC either as the first-line or maintenance treatment. 19–21 Recently, the encouraging efficacy of bevacizumab was reported in a large panel of human cancers, making it the first approved and most widely used anti-angiogenic drug. 22–24 In the common types of cervical cancer, anti-angiogenic drugs have presented notable anti-tumor effects either in single use or combined with other regimens. Based on the encouraging results from the GOG-240 study, the FDA approved the use of bevacizumab in recurrent, metastatic, and persistent cervical cancers. 25,26
In a panel of SCCC tissues, it was demonstrated that VEGF is overexpressed in 94.4% SCCC, indicating that SCCC patients might benefit from anti-VEGF therapies. 27 Indeed, there have been a few publications reporting on the positive effects of bevacizumab in treating SCCC. In 2018, Takeshi Hirasawa et al. reported the successful treatment of a recurrent refractory SCCC patient using TP + bevacizumab. 28 This patient first experienced tumor recurrence in the pelvic cavity, which rapidly progressed to multiple liver metastases. The combination of TP and bevacizumab surprisingly achieved complete remission (CR) of the tumor in the pelvic cavity and partial remission (PR) for the liver metastases. After 11 cycles of chemotherapy, the patient discontinued the therapy, and tumor progression was observed in the liver 2 months later. Similarly, another case study reported by Yuri Nakao et al. involved an SCCC patient who presented with para-aortic lymph node recurrence 3 months post-surgery and adjuvant chemotherapy treatment (EP, 6 cycles). The patient was treated with 8 cycles of TC + bevacizumab and achieved CR with a progression-free survival (PFS) of more than 24 months. 29 Although the evidence from case reports is weak, their results indicate the potential benefits of anti-angiogenic drugs in SCCC treatment.
To better understand the role of anti-angiogenic regimens in recurrent/metastatic SCCC, we performed this retrospective study to evaluate the efficacy of anti-angiogenic drugs (bevacizumab, anlotinib, and apatinib) in patients treated in our center.
Materials and Methods
Patients
We retrospectively collected data from all recurrent/metastatic SCCC patients (n = 39) in the First Affiliated Hospital and Cancer Hospital of Zhengzhou University between January 2013 and December 2021. Pathologic diagnoses were reconfirmed by an experienced pathologist. The baseline characteristics of each patient are provided in Table 1. All patients had undergone radical hysterectomy ± chemotherapy ± radiotherapy as a preliminary treatment and later experienced tumor recurrence. Sixteen patients were treated with anti-angiogenic therapies where the other 23 were given routine treatments. This protocol was approved by the ethics committee of the Cancer Hospital of Zhengzhou University (approval number: 2022-197-001, Zhengzhou, China) and the First Affiliated Hospital of Zhengzhou University (approval number: 2022-KY-1433-002, Zhengzhou, China), and informed consent was obtained from all the patients. Our study abides by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement, 30 and we have de-identified all patient details.
Patients’ Baseline Characteristics.

Statistical analysis was not performed due to the unknown HPV status in too many cases.
Treatment Response Evaluation and Follow-up
The responses to treatments were evaluated every 2 months or suspected disease progression. Pelvic examination and plasma tumor markers were routinely performed each time. Imaging tests including computed tomography (CT), positron emission tomography-computed tomography (PET-CT), single-photon emission computed tomography (SPECT), and magnetic resonance imaging (MRI) were used as appropriate to determine the size of tumors. The Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 was used to determine CR, PR, stable disease (SD), or progressive disease (PD). During the nontreatment period, follow-up was performed every 3 months until death or the patient was censored. Disease-free survival (DFS) was defined as the period from the date of surgery to the diagnosis of first recurrence/metastasis, and OS was defined as the period from the beginning of treatment after first recurrence/metastasis to the death, with patients alive at last follow-up censored on that date. PFS was defined as the period from the treatment beginning after first recurrence/metastasis to the second recurrence/metastasis or death, with patients censored if alive and with no evidence of tumor recurrence/metastasis.
Statistical Analysis
A chi-square test, Fisher's exact test, and ANOVA were used to compare the baseline characteristics of patients in the different groups as appropriate. Kaplan–Meier analysis was used to estimate the effects of anti-angiogenic drugs on OS and PFS. All statistical analyses were performed using SPSS 21.0 (IBM). P < .05 was considered statistically significant.
Results
Patient Characteristics
All 39 patients experienced tumor recurrence/metastasis after the primary treatment of surgery ± chemotherapy ± radiotherapy. After the first tumor recurrence/metastasis, 16 patients were treated with anti-angiogenic drugs in combination with routine therapies (the anti-angiogenic group), while the other 23 patients only received routine therapies (the non-anti-angiogenic group). There were no differences between the 2 groups in patient age (P = .053), body mass index (P = .729), stage at diagnosis (P = .917), primary treatments (P = .504), depth of invasion (P = .803), lymphovascular invasion (P = .812), or lymphatic metastasis (P = .725) (Table 1). No specific alterations of plasma tumor markers (including CA-125, CA-199, CEA, AFP, SCC, and HE4) were identified in either primary diagnosed or recurrent SCCC patients. The median DFS in the anti-angiogenic group was 14.5 months (ranging from 5 to 45 months) and 12 months (ranging from 4 to 39 months) in the non-anti-angiogenic group (P = .377, Figure 1).
There was no significant difference in disease-free survival (DFS) between the 2 groups (P = .377).
Efficacy of Anti-Angiogenic Regimens in Recurrent/Metastatic SCCC
In the anti-angiogenic group, 10 patients received the anti-angiogenic regimen as first-line treatment after the first recurrence/metastasis, 5 cases started as the second-line treatment, and 1 case started as the fourth line. All 16 patients received routine chemotherapy regimens, most commonly EP (n = 7) and TC (n = 5). Two patients also received local radiotherapy. One patient had surgery for a single metastasis in the brain, followed by chemotherapy. These details are supplied in Table 2. In the control group (no anti-angiogenic drugs used), 21 patients received chemotherapy (13 EP, 3 TP, 2 TC, and 3 other combinations) as the primary treatment after first recurrence/metastasis (Table 3). Among these, 6 patients also had local radiotherapy as a supplement. Moreover, 2 patients with a single metastasis to the brain underwent surgery plus radiotherapy as the primary treatment after first recurrence/metastasis. In addition to lesion sites, maximum diameters of main metastatic lesions are supplemented in Tables 2 and 3. There was no significant difference in the metastasis sites and diameters between the 2 groups.
Treatment Details of Patients Received the Anti-Angiogenic Drugs.

Abbreviations: An, anlotinib; Ap, apatinib; Bev: bevcizumab; C, carboplatin; Cap, Capecitabine; D, docetaxel; Do, doxorubicin; E, etoposide; G, gemcetabine; If, ifosfamide; Ir, irinotecan; P:pemetrexted; P, cisplatin; T, axol; To, Topotecan; Vinc, Vincristine; Vino, vinorelbin.
Treatment Details of Patients in the non-Anti-Angiogenic Group.
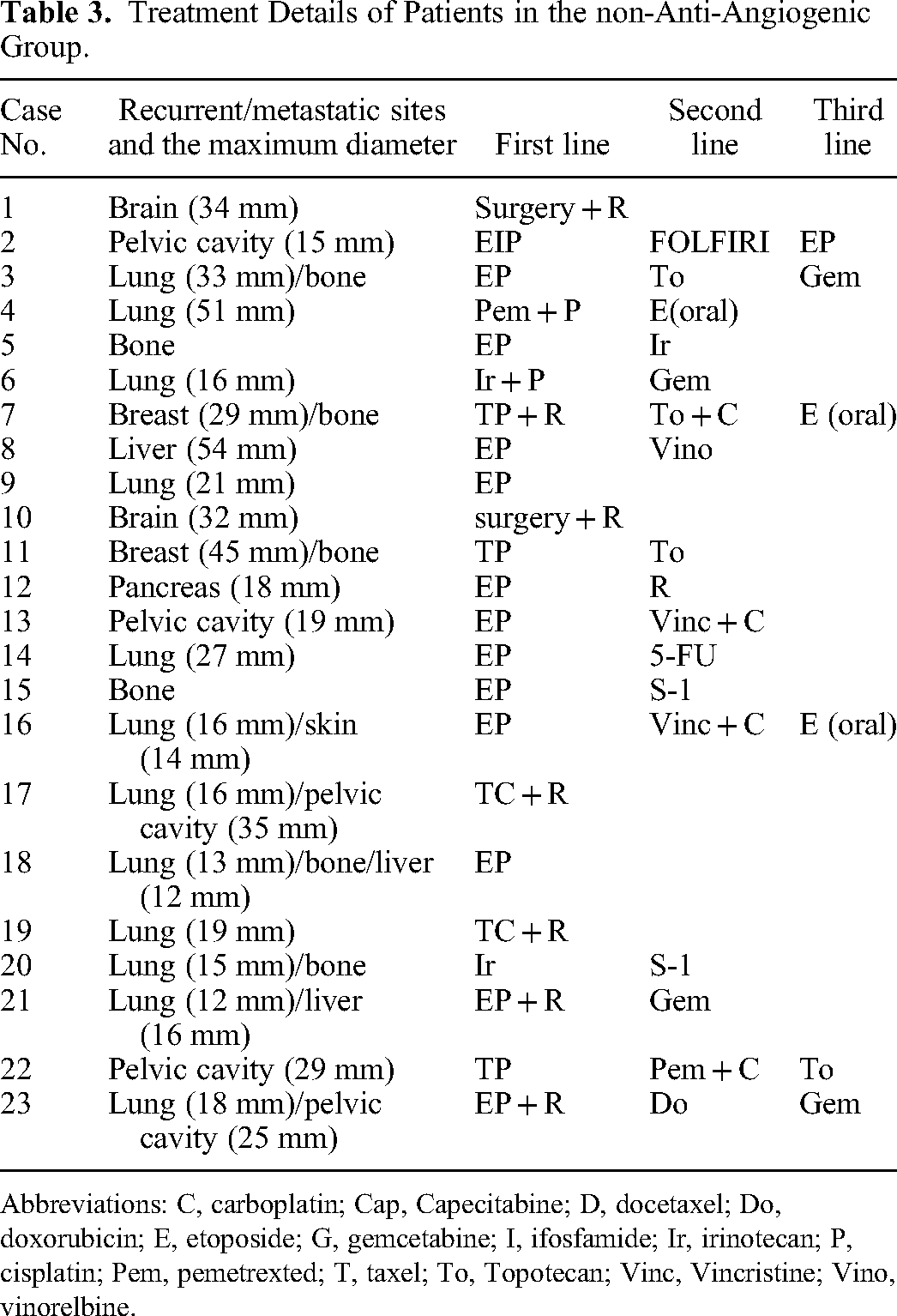
Abbreviations: C, carboplatin; Cap, Capecitabine; D, docetaxel; Do, doxorubicin; E, etoposide; G, gemcetabine; I, ifosfamide; Ir, irinotecan; P, cisplatin; Pem, pemetrexted; T, taxel; To, Topotecan; Vinc, Vincristine; Vino, vinorelbine.
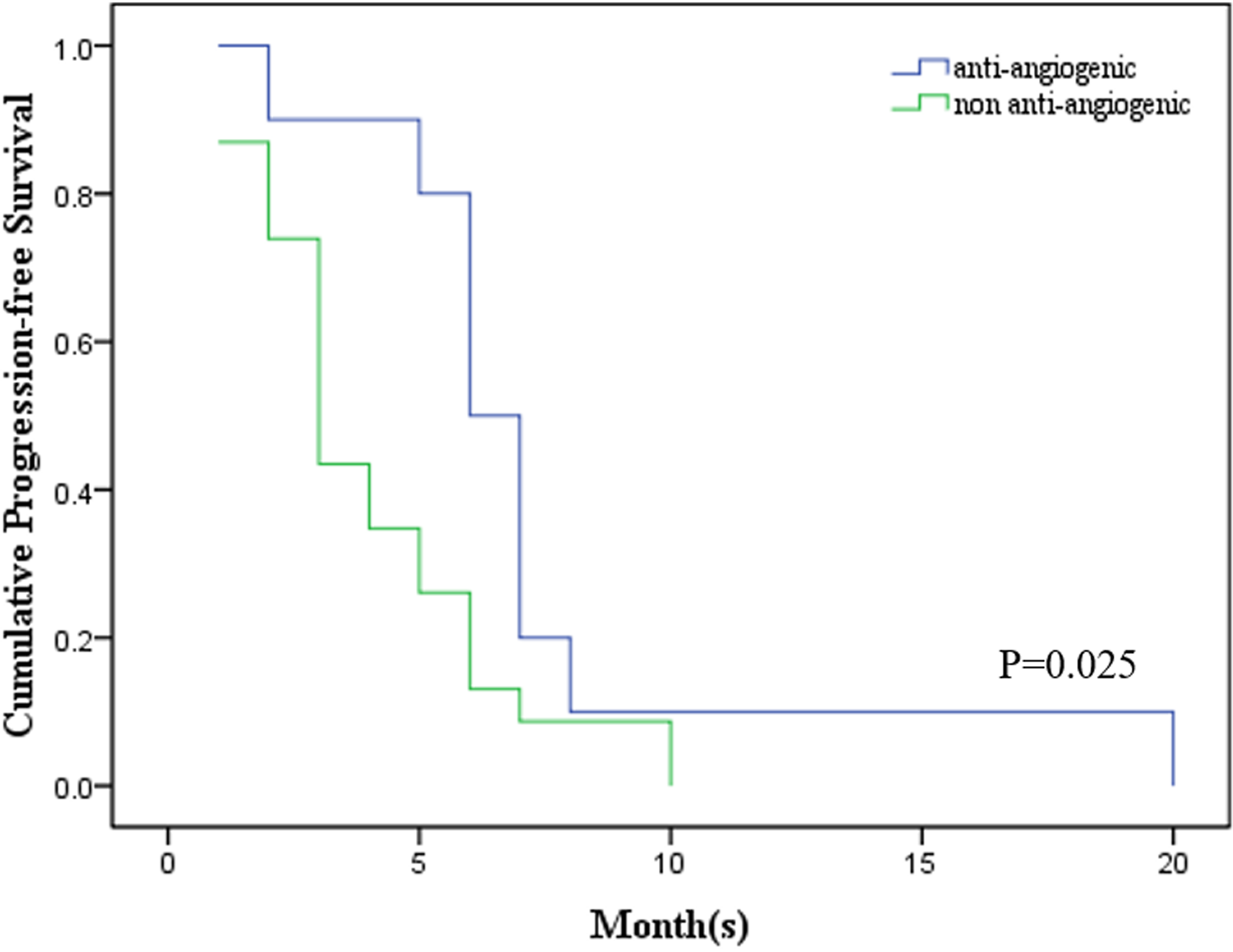
For the 10 patients who started anti-angiogenic treatments after the first recurrence/metastasis, the median PFS was 8 months (2-20 months), which was significantly longer than those in the control group which had a median PFS of 3 months (1-10 months) (Figure 2, P = .025). Five patients started the anti-angiogenic treatments until the second tumor relapse, which was already resistant to multiple therapies. The median PFS for these patients was 4 months (1-6 months, Supplementary Table S1), while most patients in the control group presented no definitive PFS after the second recurrence. Although statistical analysis could not be performed due to the limited number of cases, there was a trend suggesting that even later addition of the anti-angiogenic therapy might provide benefits for recurrent SCCC, which deserves further investigation.

Anti-angiogenic treatments prolonged progression-free survival (PFS) in patients who started the therapy after the first recurrence/metastasis (P = .025).
For OS, neither the 10 patients that received first-line anti-angiogenic treatment (median OS = 9.5 months, 5-31 months) nor the whole anti-angiogenic group (median OS = 9.5 months, 5-31 months) showed superiority to the controls (median OS = 8 months, 3-64 months) (Figures 3 and 4, P = .499 and .31).

No benefits for overall survival (OS) were detected between the group that received anti-angiogenic drugs as the first-line treatment and the controls (P = .499).
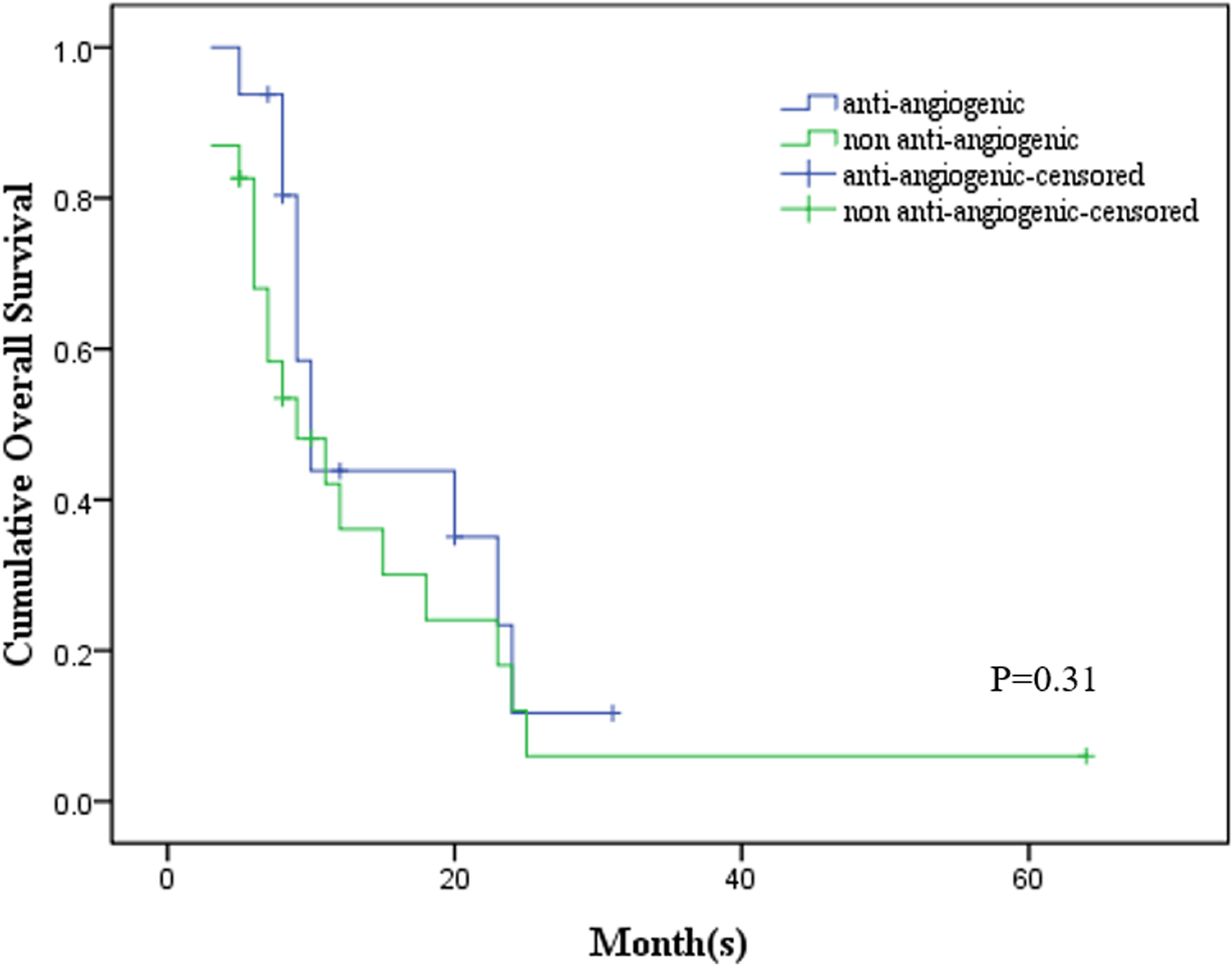
No benefits for overall survival (OS) were detected between the anti-angiogenic group and the non-anti-angiogenic group (P = .31).
Furthermore, we compared the differences in drugs among the 10 patients who received anti-angiogenic treatment after first recurrence/metastasis. In this subgroup, 4 patients received bevacizumab and the other 6 patients received the small molecules (4 had apatinib and 2 had anlotinib). There was no apparent significant difference in PFS and OS between the 2 groups, although statistical analyses were not carried out due to the small sample size (Supplementary Table S2).
Discussion
Recently, anti-angiogenic regimens have shown satisfying efficacy in many types of cancers and are widely used in the clinic, either alone or combined with other therapies. 14,31,32 Aside from blocking neovascularization in the tumor mass, these regimens can also remodel the tumor environment and metabolism, which then enhances sensitivity to chemo-/radiotherapy, immunotherapy, and targeted therapy. 33–35 Since the release of the GOG-240 results, bevacizumab has gradually been accepted as a preferred choice for cervical cancer patients with tumor recurrence or metastasis. Indeed, as subsequent studies revealed, the efficacy of bevacizumab was universal in common cervical cancers including squamous carcinoma, adenocarcinoma, and adenosquamous carcinoma patients, independent of race/ethnicity, country/area, or economic levels. 36–39 Thus, it is rational to propose that anti-angiogenic regimens might shed light on the dilemma of treating SCCC.
In addition to the 2 case reports mentioned above, a retrospective study evaluated the efficacy of TPB (topotecan + taxol + bevacizumab) in a panel of 13 recurrent SCCC patients. 40 The median PFS was 7.8 months in the TPB groups, which was significantly longer than patients who received chemotherapy alone (median PFS = 4 months). 40 These results support our findings that adding anti-angiogenic regimens to routine chemotherapy may significantly prolong PFS after recurrence/metastasis. Through stratification analysis, we found that the anti-angiogenic treatment prolonged PFS both as a first-line and second-line therapy, which further enhances the proposal that anti-angiogenic treatment is crucial for treating recurrent SCCC.
Unfortunately, we found that the anti-angiogenic drugs exerted limited influence on OS, despite the time of starting treatment. Similar results were also reported in the above study focused on TPB therapies. 40 There are 2 possible explanations for this phenomenon: first, several patients discontinued the anti-angiogenic drugs after CR/PR/SD due to notable adverse effects or financial difficulty, which could accelerate further recurrence/metastasis; second, the innate characteristics of SCCC might endow it with the ability to adapt to the stress of anti-angiogenic treatments and rapidly gain acquired resistance.
In the Chinese market, there are 2 small molecule drugs available which mainly function as anti-angiogenic drugs, apatinib and anlotinib. 41,42 Both drugs competitively bind with the VEGFR2 protein and block its bioactivities in promoting neovascularization. Moreover, they have other multiple targets like c-kit, Ret, and c-Src (apatinib) and FGFR, PDGFR, and c-kit (anlotinib).43,44 As reported in previous clinical trials, the 2 drugs exhibited anti-tumor activities in common cervical cancers, either in single use or combined with other treatments (details regarding the above studies are summarized in Supplementary Table S3). 45–49 In this retrospective study, there was no significant difference in PFS or OS in patients who received bevacizumab or the small molecule drugs. Although this is an interesting finding, the limited number of cases impairs the credibility of the current evidence and a larger study is underway to validate these results in our center. The key issue of combination therapy was whether it would induce more severe adverse effects. In our patients treated with anti-angiogenic drugs, the most common adverse effects were hypertension and proteinuria. These adverse effects were grades 1 and 2, which mainly occurred during the first 2 weeks of the treatment. This was consistent with a previous study reporting that bevacizumab treatment for advanced cervical cancer was not accompanied by notable influences on patients’ life quality. 50,51
In addition to anti-angiogenic regimens, another type of available therapy is immunotherapy targeting programmed cell death protein 1 (PD-1)/programmed cell death 1 ligand 1 (PD-L1). 52 However, 2 studies have demonstrated that the expression of PD-L1 was extremely low in SCCC. 53,54 Moreover, Carroll et al. also found that all 25 SCCC patients in their study all had microsatellite stability, which is also a negative predictor for immunotherapy. 53 Consistently, in a phase II trial, 6 recurrent SCCC patients were treated with pembrolizumab alone, regardless of their PD-L1 expression. Disappointingly, only 1 case had a short SD period, while the others progressed rapidly, causing researchers to conclude that this strategy showed minimal efficacy in women with recurrent SCCC. 55 Therefore, it is suggested that a combination regimen targeting angiogenesis (or other pathways) and PD-1/PD-L1 inhibitors might be a better option than the use of immunotherapy alone.
There were 2 limitations to this study. First, the small sample size could only provide moderate evidence to support the conclusions. Second, the heterogeneity of chemotherapy and radiotherapy treatments used in our cohort might exert an influence on patient response rates and introduce bias to the final outcomes. Thus, although it is challenging to recruit SCCC patients, a multicenter, randomized trial (possibly international) is warranted.
Conclusions
In summary, we provide real-world data suggesting that anti-angiogenic therapies were beneficial for recurrent/metastasis SCCC patients, even when applied after multiple lines of treatment. In addition, novel, oral small molecules (such as anlotinib and apatinib) provided additional choices beyond bevacizumab and exhibited similar efficacy and fewer adverse effects. These findings warrant further validation in future studies.
Supplemental Material
sj-xlsx-1-tct-10.1177_15330338231160393 - Supplemental material for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study
Supplemental material, sj-xlsx-1-tct-10.1177_15330338231160393 for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study by Haifeng Qiu, Ning Su, Shuping Yan and Jing Li in Technology in Cancer Research & Treatment
Supplemental Material
sj-xlsx-2-tct-10.1177_15330338231160393 - Supplemental material for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study
Supplemental material, sj-xlsx-2-tct-10.1177_15330338231160393 for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study by Haifeng Qiu, Ning Su, Shuping Yan and Jing Li in Technology in Cancer Research & Treatment
Supplemental Material
sj-xlsx-3-tct-10.1177_15330338231160393 - Supplemental material for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study
Supplemental material, sj-xlsx-3-tct-10.1177_15330338231160393 for Real-world Efficacy Data on Anti-Angiogenic Drugs in Recurrent Small Cell Cervical Carcinoma: A Retrospective Study by Haifeng Qiu, Ning Su, Shuping Yan and Jing Li in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This protocol was approved by the ethics committee of the Cancer Hospital of Zhengzhou University (approval number: 2022-197-001, Zhengzhou, China) and the First Affiliated Hospital of Zhengzhou University (approval number: 2022-KY-1433-002, Zhengzhou, China).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
