Abstract
Introduction
It is reported that there are approximately 2100 000 new cases and 1770 000 new deaths of lung cancer each year globally. 1 And approximately 733 000 new cases and 610 000 deaths of lung cancer are observed in China annually. 2 As the most common category in lung cancer, nonsmall cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer. 3 Consequently, there are almost 623 000 new cases and 518 000 deaths of NSCLC in China annually. Own to the significant development of the therapeutic progress regarding the PD-1/PD-L1 blockades and targeted drugs for driver genes in recent years, NSCLC had become the most successful cancer in precision medicine. 4
Angiogenesis was reported to play an essential role in the development of numerous malignancies, which could be a vital therapeutic target for solid tumors. 5 Consequently, antiangiogenic targeted drugs were developed and exhibited promising efficacy for patients with advanced NSCLC. As an antiangiogenic humanized monoclonal antibody, bevacizumab demonstrated significantly improved PFS and OS in the first-line setting for advanced or metastatic NSCLC.6,7 With regard to the antiangiogenic multi-targeted tyrosine kinase inhibitors (TKI), anlotinib had become a new standard regimen as a third-line treatment for NSCLC in China since 2018 according to ALTER0303 clinical trial. 8
Unfortunately, to our knowledge, the objective response rate (ORR) of antiangiogenic targeted drugs was disappointing currently. The ORR of sorafenib, anlotinib, and apatinib monotherapy as the third-line treatment for patients with advanced or metastatic NSCLC was 4.9%, 9.2%, and 4.0%, respectively, 9 which suggested that great individual difference of antiangiogenic targeted-drugs existed at present. 10 As a result, potential biomarkers that might predict the clinical activity of anlotinib were needed urgently.
Interestingly, the vascular endothelial growth factor receptor 2 (VEGFR2) gene was located on chromosome 4q12 and contained 30 exons, which might be one of the most important therapeutic targets of anlotinib. 11 Additionally, previous exploratory research suggested that polymorphism of VEGFR2 might be a promising biomarker that was dramatically associated with the prognosis of patients who were treated with antiangiogenic TKI. 12 Additionally, a recent study indicated that polymorphism of −906T>C in the VEGFR2 gene might be used as a potential biomarker to predict the prognosis of patients with osteosarcoma who were treated with apatinib monotherapy. 13 Unfortunately, to our knowledge, there were still no valuable polymorphism biomarkers that were available to predict the efficacy of anlotinib monotherapy among patients with treatment-refractory advanced NSCLC currently.
As a result, this study aimed to explore the influence of VEGFR2 gene polymorphism on the clinical outcomes of anlotinib for patients with treatment-refractory advanced NSCLC. Additionally, an association between the polymorphism genotype status and VEGFR2 gene mRNA expression in peripheral blood mononuclear cell (PBMC) specimens was also investigated thoroughly.
Materials and Methods
Study Design and Eligibility Criteria
As a widely used antiangiogenic TKI clinically, anlotinib had been used among considerable patients with previously-treated advanced or metastatic NSCLC since June 2018. Consequently, it was appropriate to perform the real-world effectiveness analysis and the potential biomarkers study currently. Therefore, patients with previously treated advanced NSCLC from June 2018 to December 2020 in the Department of Medical Oncology of Peking Union Medical College Hospital were included in this study consecutively. Inclusion criteria were adopted according to the previous retrospective study. 14 The exclusion criteria included (1) newly diagnosed or unstable brain metastases; (2) concomitant with another tumor or serious diseases; (3) efficacy assessment data was not available. The study profile was illustrated in Figure 1. Ultimately, a total of 129 patients with treatment-refractory advanced or metastatic NSCLC was included in this study. The primary analysis of this study was the association between VEGFR2 polymorphism genotype status and ORR, progression-free survival (PFS), and overall survival (OS). The reporting of this study conformed to STROBE guidelines. 15

The flowchart of the retrospective study of anlotinib in the treatment for patients with treatment-refractory advanced nonsmall cell lung cancer (NSCLC).
This study was approved by the ethics committee of Peking Union Medical College Hospital (approval number: JS-2616). Each enrolled patient was provided with written informed consent. We had de-identified all patient details and the medical data of all patients were confidential at the highest level. And the main information of the consent informed the patient that their medical data during anlotinib administration would be analyzed and approximately 4 mL of peripheral blood specimens would be collected for pharmacogenomics study of anlotinib.
Administration of Anlotinib and Efficacy Evaluation Protocol
The initial dosage of anlotinib was mainly 12 mg (some patients were treated with 10 mg initially) per day orally, half an hour after meals for 2 weeks, and discontinued for 1 week, every 21 days was 1 therapeutic cycle. All the patients continued the treatment until disease progression or intolerable adverse reactions. The dosage of anlotinib was adjusted according to the hematological or nonhematological toxicity during anlotinib administration, 10 mg or 8 mg was the available dosage for the patients. RECIST 1.1 criteria were adopted to perform the efficacy assessment. 16 The changes of target lesions were evaluated radiologically every 2 cycles or depended on the actual situation of the patients (clinical symptoms of the patients were getting worse) with CT or MRI regularly.
Detection of Genotype Status of
VEGFR2
Polymorphisms
Almost 4 mL of peripheral blood specimens prior to anlotinib administration was collected in this study individually and genomic DNA was extracted subsequently. Regarding the analysis of VEGFR2 gene polymorphism, the single nucleotide polymorphism included in this study were collected from the NCBI database with the minor allele frequency >10% among the Chinese population or the previous study that had reported the preliminary clinical significance of the polymorphisms. Accordingly, polymorphisms included were 4397T>C, 889C>T, and 18553C>T. As shown in Table 1, only 4397T>C was dramatically associated with PFS.
Association Analysis Between Polymorphisms of

Genotype status of 4397T>C polymorphism was detected according to the previous exploratory study. 17 Additionally, genotyping results for 4397T>C were confirmed in some randomly selected samples using the ionization-time-of-flight mass spectrometry (Sequenom, SanDiego, CA) method. And the 2 genotyping approaches reached a 100% agreement.
Determination of
VEGFR2
Gene mRNA Expression
Peripheral blood mononuclear cell (PBMC) specimens were initially provided from 77 randomly selected subjects from the 129 patients with advanced or metastatic NSCLC. Unfortunately, 6 patients were unqualified for the PBMC specimens and 5 patients failed for the RNA extraction. A total of 66 mRNA specimens were available for the subsequent analysis and preserved in liquid nitrogen ultimately. The procedure of RNA extraction and mRNA expression experiment of VEGFR2 gene was adopted according to the previous exploratory study. 18
Statistical Analysis
All the data in this study were analyzed using SPSS version 25.0 (IBM, USA). Hardy-Weinberg equilibrium test was adopted for the 4397T>C genotypes status using χ2 test method. Statistical significance of proportion and continuous variables according to 4397T>C genotype status was carried out with χ2 test and Mann-Whitney U-nonparametric test, respectively. The primary analysis was the association between PFS and 4397T>C genotype status. Kaplan-Meier survival curves were drawn using Stata 14.0 to produce the PFS and OS survival curves according to 4397T>C genotype status. And log-rank test was used to estimate the significant difference. PFS and OS were defined based on the description of the previous study. 14 Cox regression model was constructed for PFS and OS in the multivariable analysis. P < .05 was considered suggestive.
Results
Baseline characteristics of the 129 patients with advanced NSCLC and the prevalence of
Eventually, a total of 129 patients with advanced or metastatic NSCLC were included in this study. Demographic and clinical characteristics were exhibited in Table 2, the median age of the patients was 57 years (range: 19-82 years). A total of 84 patients were male and 45 patients were female. The pathological stage of IV was observed in 121 patients (93.8%). ECOG 0 score was noted in 31 patients. Nonsmoker/former smokers were found in 103 patients (79.8%). Histology of adenocarcinoma and squamous cell carcinoma was found in 99 cases (76.7%) and 23 cases (17.8%), respectively. The number of metastases sites with ≤ 3 and > 3 was observed in 76 and 53 cases, respectively. EGFR positive mutation was found in 45 patients (34.9%) and ALK abnormal was detected in 4 patients (3.1%). Treatment lines of anlotinib with third-line and further-line were noted in 17 patients and 112 patients, respectively. A total of 76 patients (58.9%) had received targeted drug therapy previously. It should be noted that 11 patients (8.5%) had been administered anti-vascular TKI treatment previously. Furthermore, a total of 33 patients were treated with immunotherapy previously. Additionally, anlotinib was administered with an initial dosage of 12 and 10 mg in 111 and 18 patients, respectively.
Demographic and Clinical Characteristics.

Abbreviations: NSCLC, nonsmall cell lung cancer, NA, not available, ECOG, Eastern Cooperative Oncology Group, EGFR, Epidermal Growth Factor Receptor, ALK, Anaplastic Lymphoma Kinase, TKI, Tyrosine Kinase Inhibitor.
The prevalence of 4397T>C polymorphism among the 129 patients with advanced NSCLC were: TT genotype 78 cases (60.5%), TC genotype 43 cases (33.3%), CC genotype 8 cases (6.2%), the minor allele frequency of 4397T>C is 0.23. The distribution of the 3 genotypes was in accordance with Hardy-Weinberg equilibrium (P = .531). Given that the frequency of CC genotype was relatively rare, CC and TC genotypes were merged subsequently. As exhibited in Table 2, patients with TT and TC/CC genotypes were comparable to similar baseline characteristics.
Association Between Genotype Status of 4397T>C Polymorphism and Clinical Outcomes of the 129 Patients With Advanced NSCLC
Best overall response during anlotinib administration indicated that complete response was observed in 0 case, partial response was noted in 12 patients, stable disease was found in 89 patients, progressive disease was reported in 28 patients, which yielded an ORR of 9.3% (95% confidence interval [CI]: 4.9%-15.7%) and disease control rate (DCR) of 78.3% (95% CI: 70.2%-85.1%). As illustrated in Figure 2, the waterfall plot for the best percentage change in target lesion according to 4397T>C genotype status was presented. ORR of patients with TT genotype and TC/CC genotype was 10.3% (95%CI: 4.5%-19.2%) and 7.8% (95%CI: 2.2%-18.9%), respectively (P = .645). DCR of patients with TT genotype and TC/CC genotype was 80.8% (95%CI: 70.3%-88.8%) and 74.5% (60.4%-85.7%), respectively (P = .399).

The waterfall plot of best change (reductions in sum of target lesion diameters) from baseline of the 129 patients with treatment-refractory nonsmall cell lung cancer (NSCLC) who received anlotinib monotherapy.
The data cut-off date of this study was March 2021. The median follow-up duration of all patients was 9.6 months (follow-up range: 0.3-30 months). Prognostic data indicated that progression or death events were observed in 104 patients when the data was cut-off. As a result, the maturity of PFS data of this retrospective study was 80.6%. As illustrated in Figure 3, the median PFS of the 129 patients with NSCLC was 4.1 months (95% CI: 2.84-5.36). Furthermore, OS was evaluated simultaneously. A total of 91 patients were noted of death events at the date of data cut-off. Therefore, the maturity of OS data of this retrospective study was 70.5%. As exhibited in Figure 3, the median OS of the 129 patients with NSCLC was 10.1 months (95% CI: 8.58-11.62).
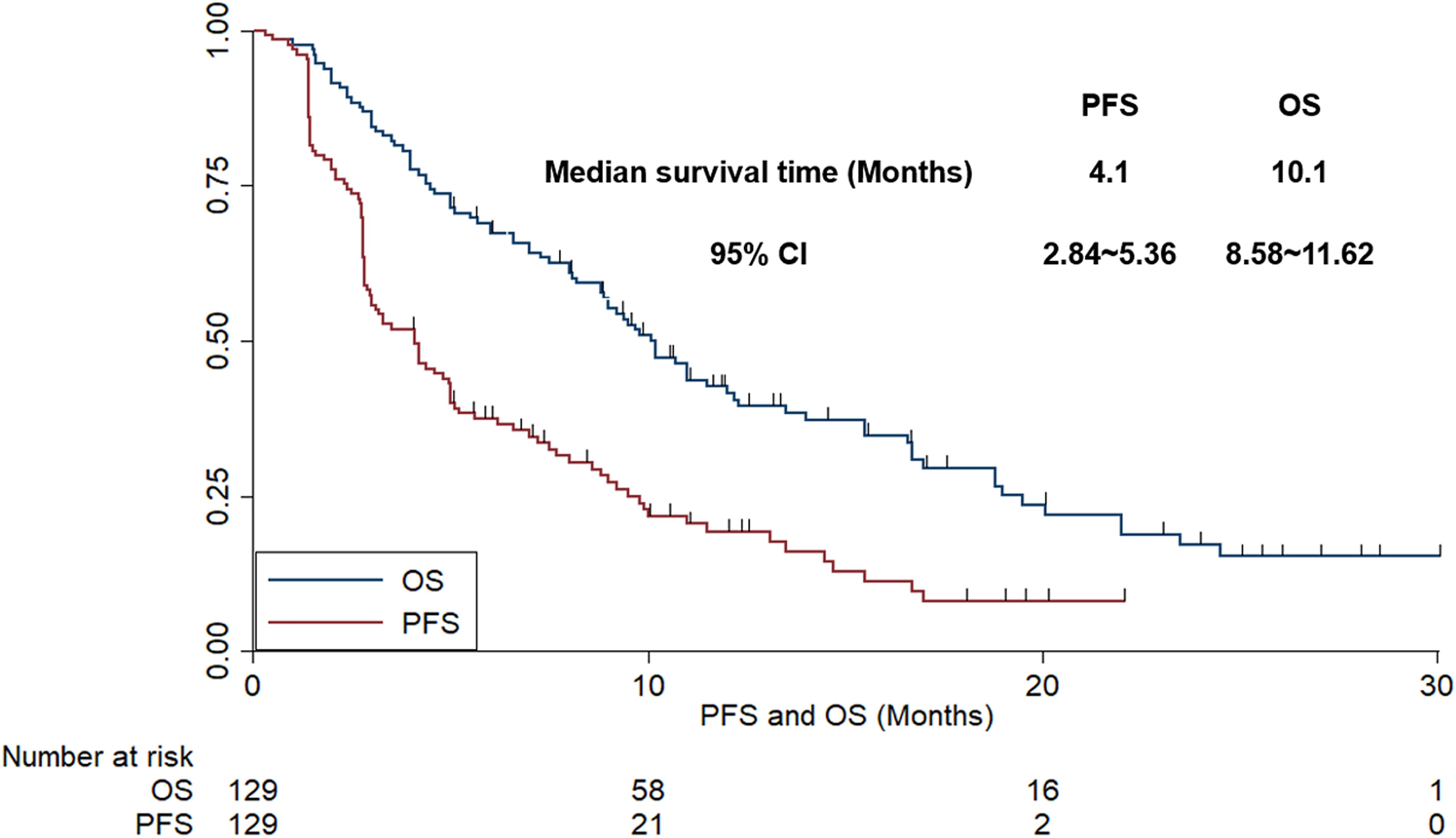
The progression-free survival and overall survival of the 129 patients with treatment-refractory nonsmall cell lung cancer (NSCLC) who received anlotinib monotherapy.
Additionally, we analyzed the association between PFS and baseline characteristic subgroups subsequently. And the univariate analysis for PFS according to baseline characteristics was exhibited in Table 3. ECOG score and the number of metastases sites were associated with PFS significantly, which suggested that the median PFS of the patients with ECOG 0 score was longer compared with those of the 1-2 score (5.1 vs 3.5 months, P = .015). Additionally, patients with the number of metastases sites ≤ 3 conferred superior PFS compared with patients with the number of metastases sites > 3 (median PFS: 4.8 vs 3.3 months, P = .021).
Univariate Analysis Between PFS and Baseline Characteristic Subgroups.

Abbreviations: ECOG, Eastern Cooperative Oncology Group, EGFR, Epidermal Growth Factor Receptor, ALK, Anaplastic Lymphoma Kinase, TKI, Tyrosine Kinase Inhibitor.
Furthermore, an association between baseline characteristic subgroups and OS was also carried out in this study meanwhile. And the univariate analysis for OS according to baseline characteristic subgroups was shown in Table 5. Obviously, ECOG score, number of metastases sites, and history of immunotherapy were significantly correlated with OS, which demonstrated that the median OS of the patients with ECOG 0 score was longer than that of patients with the 1-2 score (12.0 vs 7.3 months, P = .011). Additionally, the median OS of patients with the number of metastases sites ≤ 3 was better than that of patients with the number of metastases sites > 3 (11.5 vs 7.0 months, P = .008). Interestingly, the median OS of patients who had received immunotherapy previously was longer than that of patients who failed to receive immunotherapy (11.0 vs 8.2 months, P = .033).
In terms of polymorphism analysis, as illustrated in Figure 4, the median PFS of patients with TT and TC/CC genotype of 4397T>C polymorphism was 5.0 (95% CI: 3.69-6.31) and 2.8 (95% CI: 2.68-2.94) months respectively (χ2 = 6.821, P = .009). Furthermore, a multivariate Cox regression analysis was adopted for PFS including the variables that were significant in the univariate analysis. As shown in Table 4, a statistically significant difference was still observed of VEGFR2 4397T>C polymorphism for PFS, which indicated that 4397T>C was an independent factor for PFS (odds ratio [OR] = 0.61, P = .012). Additionally, as illustrated in Table 4, ECOG score (OR = 0.69, P = .027) and the number of metastases sites (OR = 0.72, P = .029) were also independent factors for PFS, respectively.
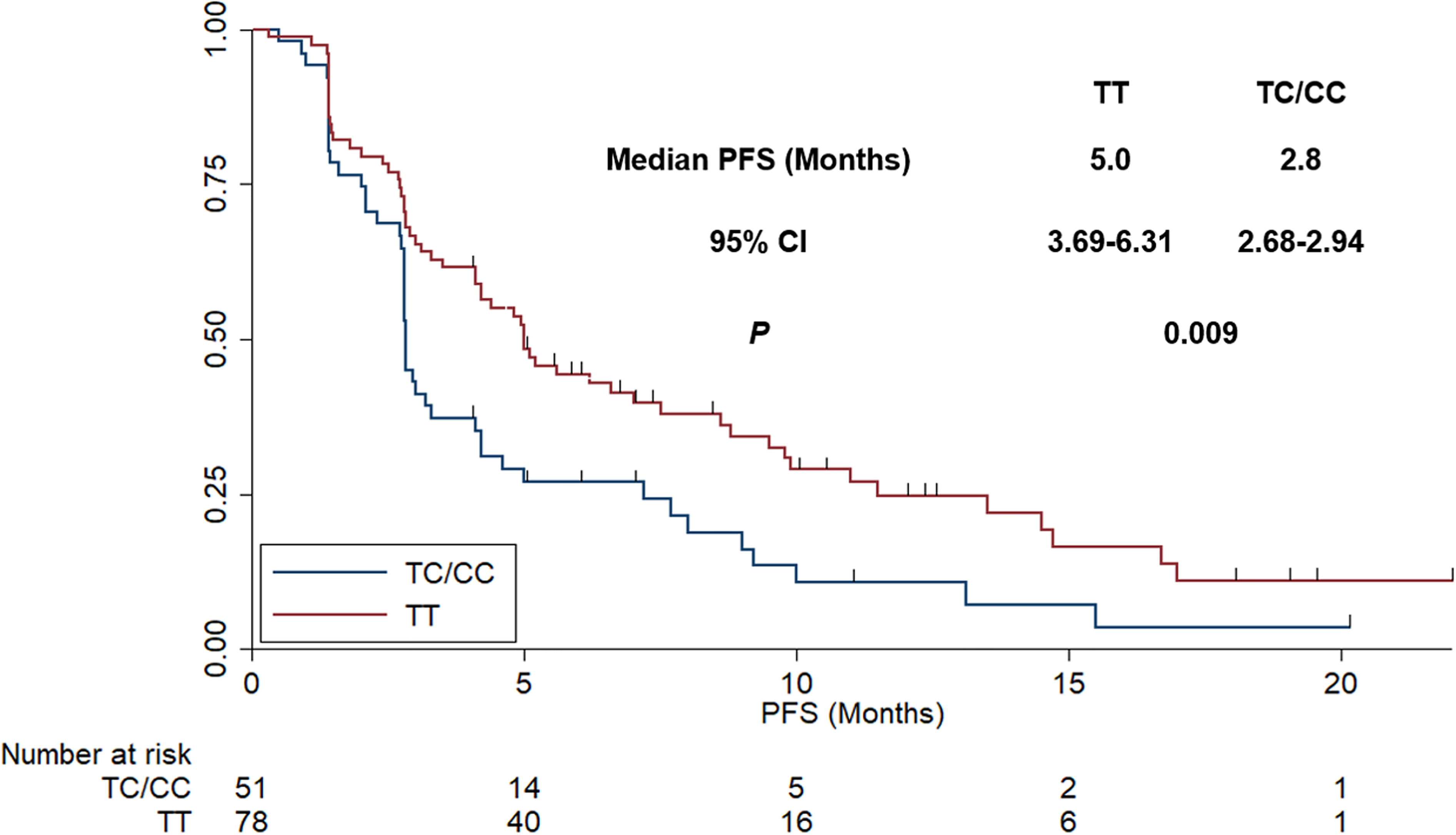
The progression-free survival of the 129 patients with treatment-refractory nonsmall cell lung cancer (NSCLC) who received anlotinib monotherapy according to VEGFR2 4397T>C genotype status.
Multivariate Cox Analysis for PFS According to Baseline Characteristic Subgroups and 4397T>C Polymorphism Status.

Furthermore, the association between 4397T>C genotype status and OS was illustrated in Figure 5, the median OS of the patients with TT and TC/CC genotype of 4397T>C polymorphism was 11.5 (95% CI: 9.64-13.36) and 7.3 (95% CI: 3.82-10.78) months, respectively (χ2 = 5.799, P = .016). Similarly, a multivariate Cox regression analysis was constructed for OS including the variables that were significant in the univariate analysis. And the univariate analysis and multivariate analysis results were shown in Table 5. Obviously, VEGFR2 4397T>C was still an independent factor for OS (OR = 0.65, P = .021) after multivariate Cox adjustment analysis. Additionally, ECOG score (OR = 0.62, P = .015), number of metastases sites (OR = 0.58, P = .011) and history of immunotherapy (OR = 0.72, P = .041) were also independent factors for OS.

The overall survival of the 129 patients with previously-treated nonsmall cell lung cancer (NSCLC) who received anlotinib monotherapy according to VEGFR2 4397T>C genotype status.
Association Analysis Between Overall Survival and Baseline Characteristic Subgroups in Univariate Analysis and Multivariate Analysis.

Abbreviations: ECOG, Eastern Cooperative Oncology Group, EGFR, Epidermal Growth Factor Receptor, ALK, Anaplastic Lymphoma Kinase, TKI, Tyrosine Kinase Inhibitor.
Correlation Between Genotype Status of 4397T>C Polymorphism and
VEGFR2
mRNA Expression
The mRNA expression of VEGFR2 was an analysis based on the 66 PBMC specimens. Firstly, the prevalence of VEGFR2 4397T>C polymorphism in the 66 specimens was: TT genotype 41 cases (62.1%), TC genotype 21 cases (31.8%), CC genotype 4 cases (6.1%), which was comparable with the genotype frequency among the 129 patients. Similarly, CC and TC genotypes were combined as well. As shown in Figure 6, patients with TC/CC genotype were associated with higher expression of VEGFR2 mRNA in PBMC specimens than that of patients with TT genotype (3.973 ± 0.488 vs 2.760 ± 0.682, P < .001).

The relative expression level of VEGFR2 mRNA in peripheral blood mononuclear cell specimens according to VEGFR2 4397T>C genotype status.
Additionally, the prognostic significance of VEGFR2 mRNA expression was also analyzed subsequently. VEGFR2 gene mRNA expression was classified into VEGFR2 high expression (VEGFR2-H) and VEGFR2 low expression (VEGFR2-L) based on the median VEGFR2 gene mRNA expression. Patients with VEGFR2-H and VEGFR2-L were observed in 34 cases and 32 cases, respectively. As illustrated in Figure 7, patients with VEGFR2-H had a trend for worse PFS compared with patients with VEGFR2-L (median PFS: 3.0 [95% CI: 2.77-3.23] versus 5.0 [95% CI: 3.97-6.04]), even the difference failed to be statistically significant (χ2 = 3.093, P = .079).

The progression-free survival of the 66 patients with treatment-refractory nonsmall cell lung cancer (NSCLC) who received anlotinib monotherapy according to VEGFR2 mRNA expression status.
Discussion
Firstly, our work highlighted the real-world evidence for the efficacy of anlotinib monotherapy among patients with treatment-refractory advanced NSCLC. Secondly, polymorphism analysis exhibited that patients with TC/CC genotype of 4397T>C were associated with inferior prognosis. Thirdly, mRNA expression analysis indicated the mRNA expression level of VEGFR2 was significantly correlated with 4397T>C genotype status. Collectively, VEGFR2 polymorphism 4397T>C might be used as a valuable biomarker to predict the prognosis of patients with NSCLC who received anlotinib administration.
The efficacy results of this study indicated that the ORR and DCR of the 129 patients with advanced NSCLC were 9.3% and 78.3%, respectively. No CR patients were observed. The efficacy data in our study were consistent with that of anlotinib monotherapy in the solid tumors reported previously. 19 Prognostic data indicated the median PFS was 4.1 months, which was inferior to that in the phase III clinical trial (Alter0303) of anlotinib in advanced NSCLC initiated by Han et al 8 (ORR = 9.2%, DCR = 81.0%, and median PFS = 5.4 months). The discrepancy might be attributed to the retrospective design of our study. To our knowledge, the management and adherence of patients were poor in the retrospective study, which had been proved in a previous retrospective study. 20 It seemed that a great gap existed objectively between prospective clinical trials and real-world studies. 21 Furthermore, patients with ECOG PS 2 score were also included in our study and the multivariate Cox analysis of our study indicated that patients with ECOG 1-2 scores conferred worse prognosis compared with those with 0 score. And it was noteworthy that the clinical outcomes of previous retrospective analysis of anlotinib in the treatment of NSCLC were basically consistent with the results of our study. 14 Interestingly, a median OS of 10.1 months was observed in our study, which was slightly longer than that of the study of anlotinib in NSCLC reported recently. 22 We thought the reason might be the fact that numerous PD-1/PD-L1 blockades were approved in China since 2018, patients with NSCLC had the opportunity to select PD-1/PD-L1 blockades in the subsequent treatment, which was efficacious and brought the patients with survival benefits consecutively. 23
To the best of our knowledge, our study was the first study that identified the implication of VEGFR2 polymorphism on the clinical outcomes of patients with previously-treated NSCLC who received anlotinib treatment. As we know, the relevance analysis between VEGFR2 polymorphism and the clinical outcomes of antiangiogenic targeted drugs was a hotspot recently. A previous study by Song et al 24 included 135 patients with NSCLC and explored the clinical implication of KDR genetic variation among patients with NSCLC who received apatinib monotherapy. The conclusion highlighted that the prognosis of apatinib might be impacted by KDR 4397T>C polymorphism, which preliminarily confirmed the clinical significance regarding pharmacogenomics in the clinical outcomes of patients with NSCLC who received antiangiogenic targeted drugs therapy. The results were basically consistent with that of the present study. Subsequently, another retrospective study initiated by Liu et al 13 recruited a total of 105 patients with metastatic osteosarcoma and explored the clinical significance of VEGFR2 gene polymorphism among patients with metastatic osteosarcoma who received apatinib administration. Results indicated that the TT genotype of 4397T>C was dramatically associated with PFS and OS of the patients. Furthermore, similar research initiated by Yan et al 17 included 118 patients with metastatic ovarian cancer and investigated the clinical significance of VEGFR2 4397T>C polymorphism. Conclusions highlighted patients with TC/CC genotype of 4397T>C was correlated with worse prognosis compared with those with TT genotype. Additionally, another research initiated by Geng et al 18 included 128 patients with chemotherapy-refractory SCLC and investigated the clinical significance of VEGFR2 rs2305948 polymorphism. The conclusion suggested that CT/TT genotype of rs2305948 was correlated with an inferior prognosis than that of TT genotype among patients with extensive-stage SCLC. Additionally, a recent study initiated by Bai et al 12 included 108 patients with metastatic colorectal cancer and performed the exploration of the clinical impact of VEGFR2 4397T>C polymorphism. Results exhibited that patients with TT genotype of 4397T>C polymorphism predicted superior PFS and OS in patients with colorectal cancer who received apatinib administration. All the above researches suggested that 4397T>C genetic variation of VEGFR2 might relate to the disposition and treatment of antiangiogenic targeted drugs, which was in line with the conclusion in our study basically. Interestingly, it should be noted that all the studies found that 4397T>C polymorphism was associated with the prognosis instead of the ORR and DCR. The further prospective clinical trial was needed to confirm this conclusion subsequently. Besides, results from the studies regarding the association between VEGFR2 polymorphism and clinical outcomes of other drugs supported our conclusion to some extent. A previous study by Sullivan et al 25 recruited a total of 170 patients with metastatic NSCLC who were treated with first-line platinum-based chemotherapy and explored the prognostic significance of VEGFA/VEGFR2 polymorphism. Results indicated that VEGFA rs2010963 and VEGFR2 4397T>C were dramatically correlated with OS. It seemed that patients with 4397T>C major genotype were correlated with superior prognosis, which was in concert with our results similarly. Furthermore, a previous phase III clinical trial initiated by George et al 26 included 286 patients with renal cell cancer who received sunitinib as adjuvant therapy. The conclusion suggested that longer DFS was observed with sunitinib versus placebo for VEGFR2 4397T>C TT patients. The stratification analysis according to 4397T>C genotype status indicated that the median DFS was longer of the patients with TT genotype than the total patients when patients received sunitinib treatment, which was consistent with our study as well. Collectively, VEGFR2 4397T>C might be used as a potential biomarker to predict the clinical outcomes of patients with previously-treated NSCLC who received anlotinib monotherapy.
Additionally, the potential mechanism of the clinical significance of 4397T>C polymorphism was also carried out in our study meanwhile. PBMC specimens were used for the mRNA expression analysis, which suggested that mRNA expression of the VEGFR2 gene was significantly higher among patients with TC/CC genotype. Interestingly, it was noteworthy that patients with VEGFR2 high expression might be associated with worse PFS compared with patients with VEGFR2 low expression. In our opinion, VEGFR2 was the most important receptor to connect with VEGFA. 27 And the previous study indicated that higher expression of VEGFR2 predicted a higher chance to regenerate blood vessels and relapse or metastasize of tumor cells.28,29 Therefore, patients with the higher expression levels of VEGFR2 were associated with a shorter prognosis among patients with NSCLC 30 and gastric cancer, 31 respectively. Therefore, these studies were basically consistent with our study. Collectively, VEGFR2 polymorphism 4397T>C might be used as a valuable biomarker to predict the prognosis of patients with NSCLC who received anlotinib administration.
Obviously, limitations were observed in our study. Firstly, our study was designed as a real-world study and potential bias existed inevitably and objectively. Secondly, the sample size was relatively limited as a retrospective study. However, the prognostic significance of the polymorphism was fully evaluated and the VEGFR2 gene mRNA expression was performed in our study to interpret the potential mechanism of the polymorphism. Therefore, we thought our study was of potential clinical significance for patients with treatment-refractory advanced NSCLC who received anlotinib monotherapy.
Footnotes
Abbreviations
Acknowledgments
The authors would like to express sincere gratitude to the patients and their families for participating in this study. We would thank all the staff who took part in this study.
Authors’ Note
XL and MH designed the research and prepared this manuscript. BZ and MG collected the data and conducted the statistics. YC and PY participated in the experiments and follow-up. All authors participated in the reviewed of the manuscript and approved the final manuscript.
Ethical Approval
This study was approved by the ethics committees of the ethics committee of Peking Union Medical College Hospital (approval number: JS-2616).
Informed Consent
All patients in this study provided the written informed consent.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
