Abstract
Nano-constructs of biocompatible polymers have drawn wide attention owing to their potential as theranostics for simultaneous therapy and detection of cancer. The present mini review summarizes various nano-architectures of polymers that have been developed as theranostic agents for the simultaneous treatment and diagnosis of cancer in a single platform. Additionally, research prospects of polymeric cancer theranostics for the future have been highlighted.
Introduction
Theranostic platforms that assimilate both therapeutic and investigative competences in a single agent, bestows real-time identification of diseased region location, monitor of drug distribution/accumulation and visualization of therapeutic outcomes. 1 Among the deadly diseases, cancer (also termed as malignancy) represents an aggressive killer that kills millions of people every year and thus, has become a critical global healthcare problem.2,3 Intense research work, therefore, has been focused on the development of techniques to combat this life threatening disease, and theranostic management of cancer has gained wide attention in this context. Till date, numerous diagnostic modalities have been employed for the detection of cancer such as near-infrared (NIR) fluorescence, positron emission tomography (PET), single-photon emission computed tomography (SPECT), magnetic resonance imaging (MRI), computed tomography (CT), and photoacoustic imaging 4 On the contrary, therapeutic modalities include chemotherapy, photothermal therapy (PTT), immunotherapy, gene therapy, photodynamic therapy (PDT), radiation, hyperthermia, etc. (Figure 1). Theranostics for cancer, therefore, integrates the combination of 2 or more of these modalities within a single platform.

Few modalities of diagnosis and therapy in cancer theranostics.
Till date, theranostic platforms for cancer that have been developed include nano-formulations of polymers, 5 dendrimers, 6 liposomes, 7 proteins, 8 carbon nanotubes, 9 metals, 10 quantum dots, 11 etc Among these nano-formulations, polymers have gained wide significance owing to their excellent biocompatibility, good biodegradability, ease of synthesis, high drug loading capacity and long blood circulation time. 2 Additionally, polymeric theranostics exhibit improved specificity, triggered or sustained release, water solubility, bioavailability and targeting properties. 12 Nano-constructs of both natural and synthetic polymers have been widely developed and explored as theranostic platforms for cancer. These nano-constructs include various architectures such as vesicles, micelles, hyperbranched polymers, nanoparticles, and hydrogels. (Figure 2). These architectures improve the conjugation of diagnostic and therapeutic agents onto the polymeric nano-constructs and aids in accomplishing their theranostic aspects. The current mini-review, therefore, focuses on the numerous nano-constructs of polymers (both synthetic and natural polymer) that have been developed and studied as theranostic platforms for the simultaneous detection and treatment of cancer. This mini-review will provide state-of-the-art literature to the researchers working in the area of polymeric theranostics for cancer.
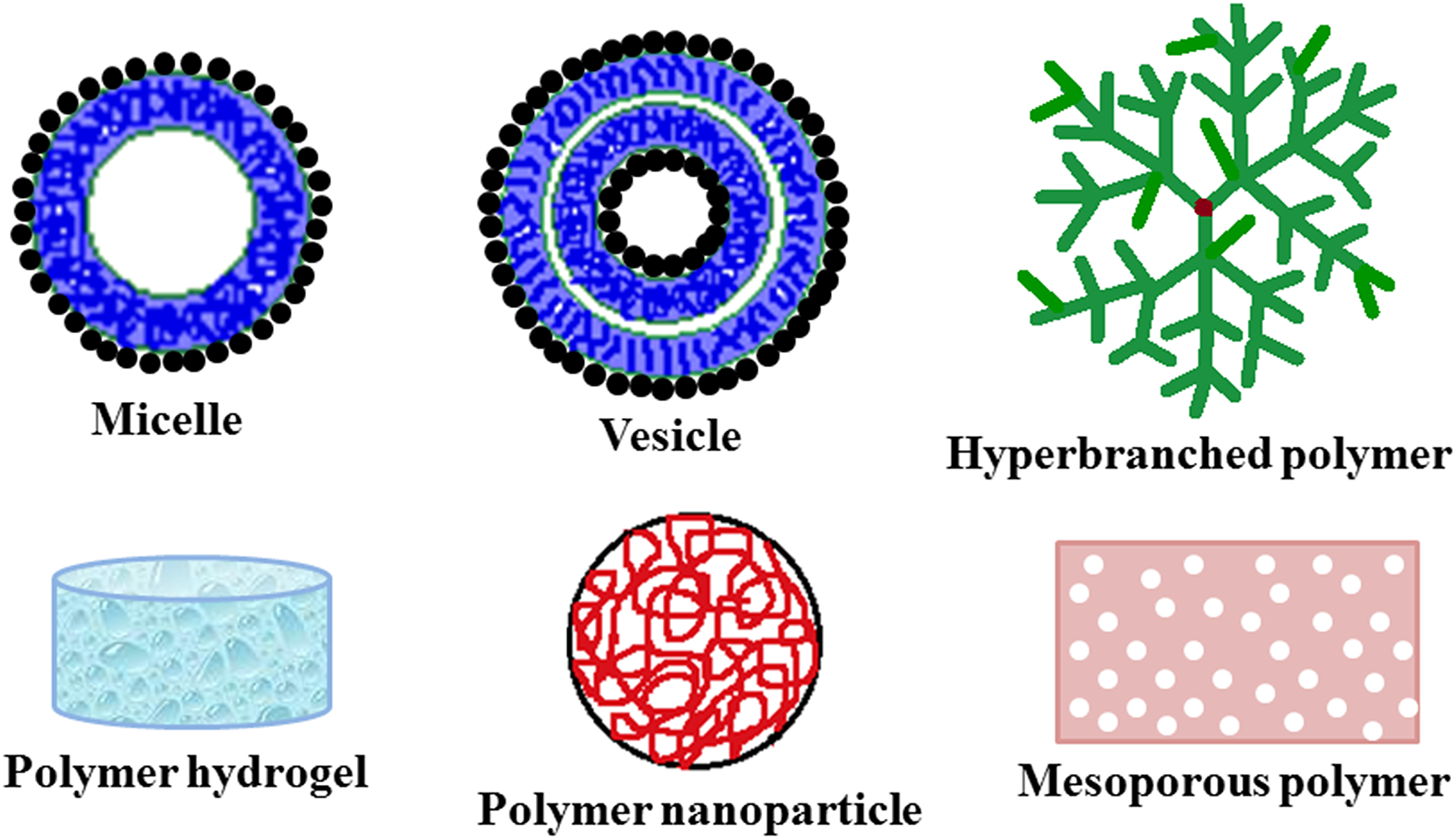
Various polymer nano-constructs in cancer theranostics.
Polymer Vesicles as Cancer Theranostics
Nanoscale architectures, viz. vesicles and micelles exhibit at least 1 dimension in 1 to 100 nm range. Polymer vesicles (or polymersomes) are nano-sized hollow spherical structures with a hydrophilic cavity and a hydrophobic membrane. Unlike polymer micelles that exhibit simple core–shell nanostructures, polymersomes possess more complicated structure. 13 Prepared by self-assembly technique, polymer vesicles are well-developed nanoplatforms that exhibits several excellent features such as versatile functionalities, smart and rapid response to different stimuli and high drug loading capacity. 14 Polymer vesicles have been considered to be promising theranostic platforms for cancer and research work on them have been highlighted in several journals. For instance, bone-targeting polymer vesicle was prepared by the self-assembly of poly(ε-caprolactone)67-b-poly[(L-glutamic acid)6-stat-(L-glutamic acid-alendronic acid)16] in water (size ∼219 nm; zeta potential −49.9 ± 0.6 mV to −45.2 ± 1.3 mV) and subsequently studied as theranostic platform for treatment and diagnosis of malignant bone tumors. 15 The alendronic acid (ADA) unit in the vesicle exhibits high affinity toward bone tissues by chelating Ca2+ ions in hydroxyapatite that are highly presented in bone tumors. Additionally, ADA-99mTc chelation renders it a superior bone scintigraphic agent for bone-tumor-targeted SPECT imaging. The poly(ε-caprolactone) membrane circumvents premature removal of encapsulated chemotherapeutic drug (doxorubicin hydrochloride) in the lumens and, thus, effectively elongates blood circulation time of the drug resulting in increased drug concentration at the tumor site. In another approach, self-assembled lipid-polymer theranostic vesicle of 1,2-dipalmitoyl-sn-glycero-3-phosphatidylcholine and triblock copolymer Pluronic F127 have been developed with covalently attached 5(6)-carboxyfluorescein (for fluorescence imaging) and studied for photodynamic applications against glioblastoma multiforme cell lines (T98G). 16 The presence of F127 results in the formation of 100 nm sized small unilamellar vesicles with excellent solution stability, low polydispersity and zeta potential of 2.88 ± 1.01 mV. The photoactivatable PDT property of the vesicles was achieved using verteporfin as photosensitizer. The theranostic properties of the vesicle are completely photoactivatable and could be trigged at the wavelength of 470 nm. The presence of only 1.0 μmol L−1 of verteporfin in the vesicle showed 98% reduction in the viability of T98G cells, thereby inferring the theranostic potency of the developed lipid-polymer vesicle for cancer treatment. In a different work, self-assembled polymeric theranostic vesicle of amphiphilic block copolymers were developed containing quinone trimethyl lock-capped self-immolative side linkages and covalently bonded photosensitizers, viz. Nile Blue and coumarin and their theranostic potency was studied against A549 and HeLa cell lines. 17 The developed theranostic vesicles (size 89-150 nm; zeta potential −8 mV to −3 mV) were NQO1 [NAD(P)H:quinone oxidoreductase isozyme 1] responsive that triggers both PDT and NIR fluorescence emission potency upon cellular internalization of the vesicles in tumor cells that contain overexpressed NQO1. The intracellular NQO1 enzymes trigger self-immolative quinone linkage cleavage and release of photosensitizers that eventually results in the simultaneous turn-on of NIR fluorescence emission and activated PDT. This process eventually transforms vesicles into cross-linked micelles which possess small size and hydrophilic cores that could be monitored directly through enhanced MRI signals. Polymer vesicles have been similarly prepared and studied as theranostic platforms for the diagnosis and treatment of breast cancer, 18 liver tumor, 19 cervical cancer, 20 etc.
Polymer Micelles as Cancer Theranostics
Akin to polymersomes, nano-sized polymer micelles are also prepared by self-assembly technique (Figure 3). The most important parameter that determines their thermodynamic stability is critical micelle concentration, which usually occurs in the μm range. 21 However, factors such as pH, ionic strength, temperature and high shear forces during circulation in physiological systems, sometimes, might affect the stability of polymeric micelles that might lead to their subsequent dissociation after intravenous administration. Various research groups have studied the applicability of polymer micelles as cancer theranostics. Pan and co-workers have developed polymeric micellar nanoparticles using an amphiphilic triblock copolymer, viz. Pluronic F127 [poly(ethylene oxide)106-block-poly(propylene oxide)70-block-poly(ethylene oxide)106] that encapsulates hydrophobic NIR heptamethine cyanine dye me-IR825 inside the core of the micelles. 22 The developed theranostic micellar nanoparticles [size 19.5 ± 10.2 nm; zeta potential +37.4 ± 2.2 mV (in pure water) and +0.8 ± 0.1 mV (in 10 mM phosphate-buffered saline)] exhibit dual fluorescence emissions at ∼610 nm (for in vitro mitochondrial fluorescence imaging, cancer/normal cell differentiation, and early stage cancer detection) and 845 nm (for in vivo NIR fluorescence imaging), in addition to showing in vitro and in vivo mitochondria-targeted PTT and in vivo photoacoustic imaging upon excitation at NIR region (808 nm). In vivo experiments in tumor-bearing nude mice revealed that the micelles could target tumor tissues after intravenous injection through enhanced permeability and retention (EPR) effect and be maintained at the tumor site for prolonged period. Furthermore, the degraded products of the micellar nanoparticles after PTT were observed to be biocompatible, inferring the posttreatment biosafety of the prepared micelles. In another work, polygalacturonic acid-polyacrylic acid conjugated hybrid nano-micelles were fabricated as nano-elastic carrier for oral and injected dosage in the treatment of liver cancer with resistance to acidic milieu of upper gastrointestinal tract and alkaline pH of colon, along with cancer cell targeting (using folic acid as targeting agent), anti-immune recognition, high diffusion and drug loading capacity (loading efficiency 23%). 23 The nano-micelle (size 100-200 nm) successfully encapsulates LY2157299 (an anti-tumor molecule) and inhibits TGF-β to treat hepatocellular carcinoma cancer cells. Polymeric theranostic micelle of N-(2-Hydroxypropyl)methacrylamide polymer conjugated pyropheophorbide-a have also been synthesized (size of ∼200 nm) and studied for PDT (at ∼420 nm) and photodynamic diagnostics (at ∼680 nm) for cancer treatment. 24

Schematic diagram for the preparation of (a) micelle and vesicle, (b) hyperbranched polymer, and (c) polymer nanoparticles.
Hyperbranched Polymers as Cancer Theranostics
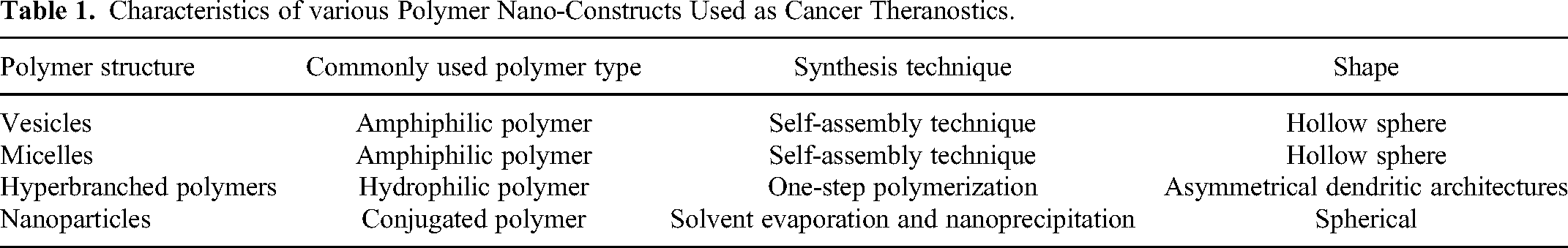
Hyperbranched polymers are 3D dendritic architectures with irregular asymmetrical branching, low polydispersity and a branching degree of 0.5 to 0.66. They were originally reported by Kim and Webster in the late 1980s. 25 They have been widely recognized as theranostic platform for their excellent multifunctionalization ability, biocompatibility, aqueous solubility, compact structure, low viscosity, and high stability. Hyperbranched polymers could be easily synthesized through one-step polymerization reactions with good yields and desired physicochemical features, while their large number of terminal functional groups facilitate in achieving the desired chemical modification. The large specific surface area of hyperbranched polymers significantly enhances their therapeutic loading capacities, 26 whereas improved chain flexibility and deformability enhances blood circulation duration drastically. 27 Furthermore, the enormous number of branches allows the successful attachment of a range of diagnostic and therapeutic agents for efficient cancer treatment. One of the most commonly used polymer to develop hyperbranched polymer theranostics is polyethylene glycol (PEG) which readily forms hyperbranched architectures with excellent biocompatibility. Pearce and co-workers developed PEG hyperbranched polymers (size 6.2 nm) through RAFT polymerization, which featured glutamate urea targeting ligand for overexpressed Prostate-Specific Membrane Antigen (PSMA) to target prostate cancer cells. 28 The hyperbranched polymers were incorporated with fluorescent cyanine-5 for fluorescent optical imaging and further loaded with chemotherapeutic drug, doxorubicin (loading efficiency ∼90%), via hydrazone formation that maintains drug release from the polymer in in vitro endosomal conditions with an eventual controlled drug release of 90% over 36 h. The drug-loaded polymer was localized into the cancer cells through receptor mediated internalization, which is followed by its release into the nucleus after hydrazone degradation. The same group in another work developed hyperbranched polymer of PEG (size 6.7 ± 1.6 nm) and covalently attached 8-mer peptide aptamer to it for targeting heat shock protein 70 (Hsp70) biomarker that is overexpressed in breast cancer cells. 29 The presence of 8-mer peptide aptamer provides both targeting and potential chemosensitization functionality to the theranostic agent. Additionally, the hyperbranched polymers were loaded with doxorubicin as a chemotherapeutic drug (via hydrazone bond that enables pH sensitive drug release at the mild acidic conditions in lysosomes/endosomes of tumor cells) and cyanine-5 as an optical imaging agent. The targeted hyperbranched polymer was observed to get accumulated in vesicles of live MDA-MB-468 cells, while in vivo studies in mice showed longer retention at the tumor site, possibly due to ligand-receptor interaction. Recently, carbon dot-drug conjugate (viz. carbon dot-hydrazine-doxorubicin) were synthesized and subsequently co-self-assembled with PEG-based hyperbranched polymer (acts as both gatekeeper and surfactant) to obtain pH/reduction dual-triggered theranostic agent (size 220 nm) for cancer treatment. 30 Another important class of hyperbranched polymer cancer theranostics is hyperbranched polyglycerols which exhibits good chemical stability, water solubility and inertness under biological conditions.31,32 Other hyperbranched polymer theranostics for cancer includes hyperbranched polyesters 33 and hyperbranched polyamidoamine. 34 Hyperbranched semiconducting polymer 35 has also been developed as cancer theranostics (Table 1).
Characteristics of various Polymer Nano-Constructs Used as Cancer Theranostics.
Polymer Nanoparticles as Cancer Theranostics
Nanoparticles of polymers have been widely studied as cancer theranostics, till date, particularly due to their tunable size and multifunctionalization ability that aids in achieving aqueous solubility and attachment of both therapeutic as well as diagnostic modalities onto them. Among these polymeric nanoparticles, fluorescent nanoparticles especially conjugated polymer nanoparticles has gained enormous attention as they usually eliminates the requirement of external diagnostic (fluorescent) agents, vital in safe and inexpensive fluorescence imaging techniques. Additionally, conjugated polymer nanoparticles exhibit some excellent features such as desirable optical characteristics, small size, low toxicity, good photostability, ease of modification, high quantum yield, and excellent biocompatibility. 36 Conjugated polymer based NIR-II nanoparticles, viz. co-benzobisthiadiazole-dithienosilole (size 40-60 nm; zeta potential ∼−39 mV) with narrow bandgap was synthesized as cancer theranostics with superior photostability and photothermal conversion efficiency of 65%. 37 The nanoparticles exhibit significant photoacoustic imaging contrast augmenting capacity (1064 nm laser irradiation), excellent active targeting ability and showed good PTT efficiency with a no recurrence tumor removal efficiency of 100% (in both in vitro and in vivo).
Conjugated polymers have also been combined with other nanoparticulate species to further improve their theranostic properties. For instance, poly(p-phenylene-co-cyclodextrin)-g-poly(ethylene glycol) conjugated polymer theranostics (size 114.4 ± 48.48 nm; zeta potential −31.8 ± 11.1 mV) was developed for use as fluorescent imaging and radiotherapy agent in cancer treatment (using U87 human primary glioblastoma cell lines). 38 The graft copolymer was further conjugated with Au nanoparticles which were observed to induce considerable enhancement in the radiotherapy efficiency, where otherwise unconjugated sample showed very less therapeutic efficiency. In another instance, conjugated polymer nanoparticles based theranostic nanomedicine (size ∼105 nm; zeta potential +0.8 mV) with photoacoustic, NIR fluorescence and MRI-guided cancer PTT was developed by chelating gadolinium to the conjugated polymer of poly (fluorene-thiadiazoloquinoxaline)-poly(ethylene glycol), where chelated Gd3+ ions introduced MRI characteristics to the conjugated polymer nano-theranostic. 39
Other Polymer Nano-Constructs as Cancer Theranostics
Apart from these important nano-architectures, other polymeric nano-constructs such as hydrogels, 40 nanogels, 41 and mesoporous, 42 have also been studied as cancer theranostics. For instance, thermoresponsive injectable hydrogels of supramolecular poly(N-acryloyl glycinamide-co-acrylamide) (size 40 ± 5.3 nm; zeta potential ∼-11 mV) was fabricated for reconstructive mammoplasty and local therapy of breast cancer. 40 The hydrogel, additionally, bears polydopamine-coated gold nanoparticles and doxorubicin that delivers excellent photothermal and chemotherapeutic effects, respectively. The presence of Au nanoparticles assists in CT imaging. Gelation of the injected supramolecular polymer nanocomposite sol could be achieved in the resected cancerous breast cavity, thus acting as breast filler, avoiding the recurrence of breast cancer, and facilitating breast reconstruction. In a different approach, a special “in situ framework growth” technique was adopted to develop hollow mesoporous phototheranostic nanoparticles (size ∼40 nm) for cancer treatment that involves co-hydrolysis of PEG2000-perylene diimide-silane and silica/organosilica precursors during the preparation of hollow mesoporous organosilica. 42 In situ polymerization in the hollow structure produces thermosensitive polymer in the cavities that significantly enhances the drug loading capacity (hydrophobic drug SN38) and their release under NIR laser irradiation. The hollow mesoporous phototheranostics showed synergistic photothermal/chemotherapy effects and exhibit excellent fluorescence and photoacoustic imaging capabilities. Additionally, chemical chelation of organosilica shell with 64Cu isotope bestows PET imaging abilities. In similar approach, Hanafy and co-workers developed multi branched/flower like shaped 43 and elongated spherical shaped 44 theranostic systems comprising of curcumin with mucoadhesive polymer (carboxymethyl cellulose) and chitosan-polyelectrolyte pairs (protamine and dextran) for treatment of liver and breast cancer, respectively.
The tremendous scientific research in the area of polymer nano-construct theranostics has inspired for their clinical investigation to progress in the direction of cancer treatment. Clinical trials involve several steps (viz. Phases I, II, III, and IV) as discussed in the review article by Essa et al. 45 Table 2 lists few polymer nano-constructs that have completed or are undergoing clinical trial studies for cancer treatment.
Few Clinical Trial Studies of Cancer Treatment Using Polymeric Nano-Contructs.

Conclusions
In conclusion, biocompatible polymers with nanoscale architectures have been a rising star of cancer theranostics. The future is also bright as tremendous research efforts have been focused on their design, synthesis and assessment of their applicability as theranostic platforms that could eventually be translated into the clinic. We envisage that this mini review presents an impetus to the scientific community and provides novel perspectives into polymeric theranostics for cancer treatment.
Footnotes
Abbreviations
Author’s Contribution
HK wrote the manuscript. MP reviewed and edited the manuscript. All authors revised the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our study did not require an ethical board approval because it did not contain human or animal trials.
