Abstract
Theranostic agents are promising due to their ability to diagnose, treat and monitor different types of cancer using a variety of imaging modalities. The advantage specifically of nanoparticles is that they can accumulate easily at the tumor site due to the large gaps in blood vessels near tumors. Such high concentration of theranostic agents at the target site can lead to enhancement in both imaging and therapy. This article provides an overview of nanoparticles that have been used for cancer theranostics, and the different imaging, treatment options and signaling pathways that are important when using nanoparticles for cancer theranostics. In particular, nanoparticles made of metal elements are emphasized due to their wide applications in cancer theranostics. One important aspect discussed is the ability to combine different types of metals in one nanoplatform for use as multimodal imaging and therapeutic agents for cancer.
Introduction
Nanotechnology is showing great promise in the field of medicine for diagnosis, imaging and treating cancer (ie, theranostics).1–4 Due to their size, chemical and physical stability at physiological conditions, nanoparticles can easily accumulate at tumor regions by passing through endothelial gaps in the tumor vasculature.5,6 Once attached or internalized in cancer cells, their multifunctional capabilities (ie, to load various imaging and therapeutic agents) can be used to effectively treat cancer. 7 Various imaging modalities (eg, ultrasound [US], magnetic resonance [MR], photoacoustic [PA], fluorescence, luminescence, computed tomography [CT], single-photon emission computerized tomography [SPECT] imaging) can be used to enhance contrast in images of tumors through the use of nanoparticles, while being able to be used for delivering therapeutic agents (ie, drugs, genetic material) and destroying cancer cells through generation of reactive oxygen species (ROS) and/or heat. 8 This theranostic capability of nanoparticles has proven to be successful in treating a wide variety of fast spreading cancers and is also considered a far better alternative than conventional chemotherapy treatment (eg, due to much lower systemic toxicity).9,10
Compared to nanoparticles made mainly of organic material, inorganic nanoparticles are advantageous for cancer theranostics because they are generally non-toxic, biocompatible, hydrophilic and highly stable. Inorganic metal-based nanoparticle systems provide greater variety of surface modification strategies, that can be used for loading of imaging and targeting/therapeutic molecules (eg, drugs, antibodies, peptides, genetic material) through molecular interactions (eg, electrostatic interactions, covalent bonding). The ability to design stimuli responsive metal nanoparticles allows for spatiotemporal selectivity and very low resistance from cancer cells during/after treatment. These types of particles can be synthesized to respond to specific stimuli (eg, light, magnetic field, US, x-ray, acidic pH, enzymes) for imaging and controlled drug release. In addition, the unique physicochemical properties (eg, optical, radioactive, magnetic, acoustic), of metal-based nanoparticles greatly facilitates cancer theranostics, providing in most cases the capability of multimodal imaging and therapy (ie, photodynamic therapy [PDT], thermal therapy and/or drug delivery).
Even though there have been significant developments in the field of cancer nanotheranostics over the past few decades, there is limited scientific literature covering the main aspects involved in imaging and therapy. This review focuses on the different cancer imaging and treatment techniques that are available when planning the construction of metal-based nanoparticles for cancer theranostics. It is mainly focused toward researchers interested or working in the field, providing an understanding of the differences in performance and targeting capabilities between different types of metal-based nanoparticles. The fundamental principles for the main techniques in cancer imaging and therapy are first provided, before the main signaling pathways used for cancer theranostics are discussed. This is followed by a summary of the different types of metal-based nanoparticles that have been used in non-clinical and clinical studies. Both in vitro and in vivo applications and results are provided for determining potential clinical implications. Finally, a summary with the main challenges that need to be overcome for metal-based nanoparticles in cancer theranostics is briefly provided. The main concepts involved in this review are an important resource for researchers developing more advanced metal-based nanoparticles for cancer theranostics.
Imaging Techniques for Detection and Monitoring
A wide variety of imaging modalities can be used for imaging tumors that have accumulated nanoparticles. These include techniques involving both non-ionizing (eg, optical imaging [OI], PA imaging, US imaging, MR imaging) and ionizing radiation (eg, CT imaging, SPECT imaging, positron emission tomography [PET] imaging). The choice of imaging modality/modalities depends on a variety of factors such as the dose for nanoparticles permissible, required resolution and sensitivity for providing high contrast images for analysis and the penetration depth required for specific cancer(s).
Optical Imaging
OI is one of the most common imaging modality used. After light absorption by nanoparticles (ie, either intrinsic or through the addition of dyes/fluorophores), photons are emitted in the form of luminescence or fluorescence. Advantages of using this imaging modality include the high spatiotemporal resolution and the use of low energy photons (ie, non-ionizing radiation), which are typically detected in the visible to NIR region. However, a main drawback of using this modality for imaging tumors is the poor tissue penetration (ie, up to few to several millimeters) 11 and high noise in images due to scattering of photons (ie, in the visible light region). 12 In addition, significant background signals can be a result of tissue autofluorescence and light absorption by proteins, heme groups (ie, mainly from red blood cells), and water. 13 To overcome these challenges, nanoparticles need to be constructed that are able to absorb and emit photons in the NIR range.
Photoacoustic Imaging
PA imaging is a non-invasive imaging modality, based on the PA effect and production of sound waves from the absorption of light. 14 Laser pulses lasting several nanoseconds are used at the site of interest (eg, tumor), leading to localized heating, thermo-elastic expansion and production of US waves. The PA effect requires light absorption from particles (eg, optical absorber/dye containing nanoparticles)15–17 or endogenous absorbers (eg, red blood cells) for investigating the anatomical (eg, size, structure) and/or biochemical features (eg, proteins upregulated in cancer) of tumors and microvasculature. For investigating the biochemical processes involved in cancer using nanoparticles, targeting moieties (eg, small molecules, peptides, aptamers, antibodies) are usually attached to the surface. The main advantages of PA imaging include the exceptional depth of penetration (ie, up to 5 cm), spatial resolution (ie, can be ∼10 μm or smaller) and temporal resolution (ie, can be ∼30 frames per second or above). Imaging can be performed over a wide range of depths and resolution, depending on the system setup (eg, optical resolution PA microscopy, acoustic resolution PA microscopy, acoustic resolution PA tomography/macroscopy). 18 A disadvantage of PA imaging is the requirement for a coupling medium, between tissue surface and transducer, to facilitate the transmission of US energy from the tissue to the transducer. Also due to endogenous absorption from skin and tissues (eg, from hemoglobin, melanin, lipids), 19 the contrast in PA images can be reduced, making it difficult to distinguish tumors from healthy tissues.
Ultrasound Imaging
US imaging utilizes the properties and behavior of high frequency sound waves as they travel and interact with biological tissues. US signals are influenced by the acoustic impedance of the medium (eg, density and speed of sound in tissue). 20 Clinical US imaging systems usually operate between 1 and 20 MHz, with pre-clinical systems reaching up to 55 MHz. 21 US transducers send and receive sound waves, with US waves being reflected from the interface between tissues or scattered from US contrast agents (eg, gas filled bubbles). The waves that are detected by the transducer are converted into electrical signals and processed for displaying US images. Since most metal-based nanoparticles have poor acoustic properties, they are usually combined with other contrast agents such as phase shift emulsions or bubbles that are able to provide strong US signals.22–24 The advantages of US imaging include its good temporal resolution (ie, seconds to minutes) and sensitivity (ie, picogram level), when bubbles (ie, microbubbles, nanobubbles) are used. Because PA and US imaging detect sound waves, they share similar advantages and limitations. The spatial resolution can range from 0.01 to 2 mm, with a penetration depth from a few millimeters to centimeters, depending on the US frequency. Air filled and dense anatomical structures (eg, lungs, bones) transmit sound waves poorly and are difficult to image. To improve the US contrast, bubbles are usually used, with a coupling medium (eg, gel) required to direct the US energy. However, at high US energy (ie, high frequencies), microbubbles can get damaged, reducing the contrast in images.
Magnetic Resonance Imaging
MRI can be used for visualizing the size, structure and morphology of tumors, using magnetic and radiofrequency (RF) energy for imaging atomic nuclei.25,26 Nuclei (eg, 1H, 13C, 31P, 19F, 17O2) used for MRI have angular momentum and due to mass, spin and charge of protons, produce a magnetic field. The ratio of the angular momentum to magnetic moment is known as the gyromagnetic ratio and is unique for each nucleus used in MRI. When placed in an external magnetic field, the magnetic moments of nuclei align either parallel or anti-parallel to the field. The MR signal generated for imaging is proportional to the net difference in the number of magnetic dipoles that align parallel versus those that align anti-parallel (ie, polarization), concentration of nuclei and the gyromagnetic ratio (ie, g-factor). RF pulses are responsible for altering the alignment of the magnetic dipoles, for providing different types of imaging (eg, longitudinal T1 relaxation, transverse T2 relaxation contrast imaging). Because the amount of magnetically active endogenous nuclei provide weak signals with lower sensitivity compared to other imaging modalities, exogenous MRI contrast agents (eg, nanoparticles) are added for enhancing image contrast. Advantages of MRI include unlimited depth of penetration and high spatial resolution (ie, 25-100 μm pre-clinical, ∼1 mm clinical). At the same time, physiological data can be collected using specialized techniques such as dynamic contrast-enhanced MRI (DCE-MRI) 27 or metabolic data collected using nuclear magnetic resonance (NMR) spectroscopy, 28 sharing fundamental principles with MRI. Limitations of MRI include the need to use high concentrations of contrast agents for imaging, which can have pharmacological effects and the high instrumental and maintenance costs.
Computed Tomography
CT imaging can provide contrast and distinguish tumors from healthy tissues based on differences in the degrees of x-ray attenuation. 29 Radiopaque material (eg, nanoparticles) can be used for enhancing contrast, with the attenuation coefficient dependent on the atomic number and electron density of elements. Elements such as iodine, 30 bismuth 31 or gold 32 are commonly used in nanoparticles for enhancing contrast in CT imaging, as they are electron dense elements with high atomic number. High contrast images of sections of tumors can be acquired, where regions that strongly absorb x-ray appearing white while regions that poorly absorb appear black. Advantages of using CT imaging include good spatial resolution (ie, 0.05-0.2 mm for pre-clinical imaging, 0.5-1.0 mm for clinical imaging), fast acquisition time and depth of penetration, due to the use of high energy, ionizing x-ray radiation. Having said this, high exposure and penetration of x-ray radiation through the body can be harmful, limiting the number of scans that can be taken. Furthermore, contrast agents, which usually need to be used at high concentrations can be toxic and cause adverse effects in organs.
Single-Photon Emission Computerized Tomography
SPECT imaging is a radionuclide-based modality that detects γ-ray emissions.33–35 For imaging, radiopharmaceuticals (ie, containing radionuclides such as 99mTc, 123I, 111In) with short half-life values are used, which decay via the emission of single gamma rays (ie, with differing energies). Significantly strong signals can be detected due to the large energies from gamma rays, while limiting background signals. A reconstruction is obtained by rotating the gamma camera at numerous positions. SPECT imaging is advantageous because multiple radionuclides can be imaged at the same time, providing high sensitivity and limitless depth of penetration. Unike other imaging modalities, SPECT imaging requires small amounts of imaging agents, which reduces toxicity and pharmacological effects. However, due to the use of ionizing radiation (ie, also in CT imaging) which is harmful at high doses, the number of scans that can be taken is limited. A similar imaging modality, PET imaging, can also be used for cancer imaging, with the main difference being that small particles called positrons (ie, particles with the same mass as electrons but opposite charge) are produced from the decay of the radiotracers.36,37 The benefit of using SPECT and PET imaging is that they can be combined with other imaging modalities such as MRI or CT imaging, to provide greater amount of scientific information.
Techniques and Pathways in Treatment
Different therapeutic agents (eg, drugs, genetic material) can be loaded in nanoparticles for cancer treatment. Particles can be constructed for specialized therapies such as PDT and photothermal therapy (PTT), using photosensitizers for the production of ROS and generation of heat from light absorption, respectively. Although the latter can be used for thermal tumor ablation, cancer theranostic nanoparticles can use intrinsic and/or extrinsic cell death pathways for apoptosis. These pathways involve the production of a number of intracellular signals by a variety of membrane and cytoplasmic proteins.
Therapeutic Delivery
Therapeutic agents such as drug molecules and/or genetic material (eg, paclitaxel, doxorubicin, siRNA) can be delivered using nanoparticles. Therapeutics can be delivered with nanoparticles using various non-covalent interactions, for example through entrapment,38–40 electrostatic interactions41,42 or affinity-based interactions (eg, avidin/biotin or biotin/streptavidin). 43 Therapeutic agents can also be attached to particles through covalent linking,44,45 which can improve their residence time and bio-distribution and reduce systemic toxicity. 46 Two strategies enabling nanoparticles to target tumors efficiently are passive and active targeting. Nanoparticles that use passive targeting rely on their physical and chemical characteristics such as size and charge to accumulate in tumors, by taking advantage of the tumor biology (eg, vascularity, leakiness, lack of lymphatic drainage). Due to the enhanced permeability and retention (EPR) effect and fenestrations in tumor vessels (ie, usually 200-800 nm size of gaps), nanoparticles are able to extravasate into tumors.47,48 Active targeting on the other hand involves the attachment of targeting moieties (eg, antibodies, peptides, aptamers)49,50 on nanoparticles for binding to specific tissue (eg, tumors). These decorated particles are specifically designed to target specific proteins/receptors overexpressed on tumor cells, for increasing the amount and penetration of therapeutic agents in tumors. A major barrier in designing effective theranostic nanoparticles is the reticuloendothelial system (RES), responsible for the fast degradation and clearance of many particles in organs (eg, liver, spleen). The size, charge and morphology of nanoparticles are important parameters/factors that can improve the pharmacokinetics and bio-distribution, for greater accumulation in tumors. 51
The main cancer cell internalization pathways for nanoparticles 52 are macropinocytosis, clathrin-dependent endocytosis, caveolin-dependent endocytosis, clathrin/caveolin-independent endocytosis and receptor-mediated endocytosis. In macropinocytosis, vesicles with membranes lacking specific proteins, called macropinosomes (ie, up to 5 μm in size in cells with membrane ruffling) are used to internalize large nanoparticles and micro-particles. Particles of approximately 100 nm size are internalized at a high rate through clathrin-mediated endocytosis as well as internalization of some important molecules such as essential nutrients and iron occur through this pathway. Vesicles are coated with cytosolic proteins (ie, mainly clathrin), with the protein dynamin used to pinch off vesicles from membranes. Caveolae are 50 to 80 nm invaginations for internalization of nanoparticles less than 100 nm, through caveolin-dependent endocytosis. Caveolae formation is due to binding of the caveolin protein to cholesterol. An advantage of this pathway is that the caveolae escape from fusion with lysosomes, which are organelles responsible for degradation of nanoparticles in cells. Clathrin/caveolin- independent pathways are divided into dynamin-dependent and dynamin-independent pathways, with vesicles being long and wide. Receptor-mediated endocytosis is used for internalizing nanoparticles loaded with therapeutic agents and moieties targeting overexpressed receptors (eg, folate, transferrin, epidermal growth factor, low density lipoprotein receptors) on cancer cells. 53 In most cases, multiple pathways are used when internalizing nanoparticles in cancer cells, as the size distribution of particles is often not monodisperse.
Hyperthermia
Cancer cells can be treated using nanoparticle-based thermal therapy, using electromagnetic radiation (eg, using NIR, RF energy) to excite nanoparticles. For PTT, nanoparticles that are used usually have high absorption properties, or have such properties improved in nanoparticles by loading optical absorbers (eg, cyanine, IR dyes). 54 When using metallic nanoparticles, PTT depends on the surface plasmon resonance (SPR) frequency, which can be tuned to the NIR region for high penetration depth, by changing the physical characteristics of particles (eg, core–shell thickness, aspect ratio). 55 In general, PTT nanoparticles have good energy/photothermal conversion efficiencies for converting sufficient amounts of energy into heat (ie, from light absorption) for cancer cell death. Another way to generate heat is by exposing metallic nanoparticles (eg, superparamagnetic iron oxide nanoparticles) to an alternating magnetic field (AMF). 56 The magnetic polarity of particles is flipped and the loss from magnetic hysteresis/relaxation during this process leads to the production of heat. 57 This can be used for increasing the local temperature of tumors for treatment, after site specific accumulation of particles. During excitation of hyperthermia agents, when the temperature reaches 41 °C, cells change their expression of certain genes, with enhanced production of heat shock proteins for providing stability during stress.58,59 Antitumor effects are mainly a result of direct thermal ablation (ie, at temperatures over 42 °C) 60 with irreversible tissue damage occurring at 42 °C and apoptosis (ie, programmed cell death) 61 occurring at approximately 46 °C. As the temperature increases above 49 °C from tissue heating, protein denaturation, cell membrane disruption, inflammation, cytoskeleton damage and inhibition of DNA synthesis occurs. Hyperthermia can be combined with chemotherapy and radiation (ie, for radiotherapy), by introducing radionuclides in nanoparticles that emit ionizing radiation (eg, gamma rays, beta energy, charged particles) for damaging DNA in cancer cells. Synergistic effects from such treatments are due to the sensitivity of cells when exposed to hyperthermia. 62 Some main advantages of nanoparticle-based hyperthermia treatment include its non-invasiveness, strong tissue penetrating capability, and reduced systemic side effects due to localized treatment (ie, areas where magnetic field or laser is applied/focused).
Photodynamic Therapy
Nanoparticle-based PDT is a technique involving the generation of ROS (eg, hydrogen peroxide [H2O2], reactive superoxide anion radicals [O2−], hydroxyl radicals [•OH]). 63 The ROS can be generated from interaction with the surrounding molecular oxygen and molecules, after laser irradiation of photosensitizers contained in nanostructures. Increased ROS production leads to oxidative stress in cells and attack of biomolecules (eg, carbohydrates, nucleic acids, unsaturated fatty acids, proteins, amino acids, vitamins). 64 ROS generated from nanoparticles can lead to outer membrane lipid peroxidation, 65 and irreversible damage due to increased cell permeability and rupturing of membranes in organelles. 66 ROS can directly break bonds and interfere with protein function as well as react with nucleobases, causing gene mutations.67,68 The modes of cancer cell death from PDT include necrosis, apoptosis and mitotic cell death. 69 Necrosis involves cytoplasmic swelling, leading to release of intracellular contents and inflammation. Apoptosis is characterized by cell shrinkage, with fragmentation of cells into multiple vesicles. Cells undergoing mitotic cell death exhibit retardation at G(2)-M phase of growth, increased cell volume and multi-nucleation. The advantages of nanoparticle-based PDT are similar to those of nanoparticle-based hyperthermia treatment. Attaching photosensitizers to nanoparticles reduces or prevents undesirable side effects due to much greater accumulation of photosensitizers at tumor sites compared to accumulation of sensitizers in healthy tissues. 70
Intrinsic Cell Death Pathway
The intrinsic apoptotic pathway is activated in response to intrinsic stresses (eg, mutations, direct DNA damage) within the cell and by the sensor protein p53.71–73 Under normal conditions, p53 is directly phosphorylated and stabilized by DNA checkpoint proteins, ATM (ie, ataxia telangiectasia mutated protein) and Chk2 (ie, checkpoints factor-2). However, under stressful conditions, MDM2 (ie, mouse double minute-2 homolog) binds to and mediates ubiquitination of p53. The p53 protein can no longer function and initiates apoptosis by activating pro-apoptotic Bcl2 family members (eg, BAX, BID, NOXA, PUMA) and repressing anti-apoptotic Bcl2 proteins and CIAPs (ie, cellular inhibitor of apoptosis proteins). The p53 transcription factor also increases expression of other genes responsible for apoptosis, including PTEN (ie, phosphatase and tensin homolog deleted on chromosome-10), APAF1, Perp, p53AIP1 (ie, p53-regulated apoptosis-inducing protein-1), and genes that lead to increases in ROS. These ROS lead to oxidative damage in cells, including damage to mitochondrial DNA and disruption of mitochondrial oxidative phosphorylation.74,75 Proteins which are released from damaged mitochondria and are important to the intrinsic apoptosis pathway include SMAC (ie, second mitochondria-derived activator of caspase), Diablo, Arts, and Omi/HTRA2 (ie, high temperature requirement protein-A2), as they counteract the effects from IAPs (ie, inhibitor of apoptosis proteins). This enables a family of cysteine proteases, named caspases (eg, caspase-3) to be activated, that are involved in dismantling cells. Other proteins such as the nuclease, EndoG (ie, endonuclease-G) and protein AIF (ie, apoptosis-inducing factor) are important as they induce DNA fragmentation and initiate chromatin condensation.76,77
Extrinsic Cell Death Pathways
The main types of extrinsic cell death pathways are receptor-mediated and cellular stress-mediated apoptotic pathways.78,79 Receptor-mediated pathways are activated by death ligands such as Apo2L (ie, Apo2 ligand), Apo3L (ie, Apo3 ligand), FasL (ie, Fas ligand) and tumor necrosis factor (TNF)-related apoptosis-inducing ligand (ie, TRAIL), that can be loaded in nanoparticles.80–82 These ligands bind to cell surface death receptors (DRs) such as Fas, TNFR1 (ie, tumor necrosis factor receptor-1), Apo2 and Apo3, for transmitting apoptotic signals. This leads to structural change of the receptor (ie, oligomerization), clustering of death domains and activation of death caspases (DCs). Adapter molecules like FADD (ie, Fas-associated via death domain), TRADD (ie, tumor necrosis factor receptor-1-associated death domain), or Daxx contain death domains for transmitting apoptotic signals. The co-factor function of FADD gets blocked due to interaction with the regulator FLIP (ie, FLICE inhibitory protein). Upon recruitment by FADD, pro-caspase-8 oligomerization drives its activation through self-cleavage. Active caspase-8 then activates downstream caspases such as caspase-3 and caspase-7, for executing cell apoptosis. Cytochrome c is released after capsase-8 cleaves BID (ie, Bcl2 interacting protein) and the COOH-terminal part of caspase-8 translocates to mitochondria. Caspase-9 is activated when cytochrome c binds to APAF1 (ie, apoptotic protease activating factor-1), dATP and pro-caspase-9. Caspase-3 is activated after caspase-9 or caspase-8 cleaves pro-caspase-3. Caspase-3 is responsible for cleaving ICAD (ie, inhibitor of caspase-activated DNase), a DNA fragmentation factor. The active CAD oligomer causes the inter-nucleosomal DNA fragmentation and chromatin condensation, which are primary indicators of apoptosis. A nuclear pathway is also linked to apoptosis, where ZIP kinase (ZIPK) triggers apoptosis from nuclear PODs (ie, PML [promyelocytic leukemia] oncogenic domains), working alongside Daxx and Par-4 (ie, prostate apoptosis response protein-4). The TNFR1 is a common receptor for apoptosis, where after activation various death domain containing proteins can form distinct complexes (ie, possessing protein TRADD, TRAF2 [ie, TNF receptor associated factor-2], CIAP1 [ie, cellular inhibitor of apoptosis-1], and the kinase RIP1 [ie, receptor-interacting protein-1]), assembled at the plasma membrane. This is required to recruit IKK (ie, I-KappaB-kinase) and activate NF-KappaB (ie, nuclear factor-KappaB). After the TRADD-based complex dissociation from the receptor, complex II is formed for the recruitment of FADD and the initiator caspase-8. FLIP, an inhibitor of caspase-8 is responsible for the balance of effects by complex I versus complex II. Adequate FLIP is expressed to inhibit caspase-8 of complex II, when complex I NF-KappaB activation is sufficient. Apoptosis can be mediated by complex II only when Complex I-mediated NF-KappaB activation is insufficient.
Besides the use of DRs for apoptosis, growth factors can influence apoptosis via PI3K (ie, phosphatidylinositde-3 kinase) and the Akt pathway (ie, v-Akt murine thymoma viral oncogene homolog).83,84 PI3K is activated when growth factors bind to growth factor receptors, also leading to Akt activation. Akt is very important in regulating the pro-apoptotic member of Bcl2 family, BAD (ie, Bcl2-antagonist of cell death), involved in mitochondrial apoptosis, with other proteins such as PKC (ie, protein kinase-C) and ribosomal-S6 kinases also important. In addition to receptor-mediated apoptosis, cellular stresses (eg, from gamma, UV-radiation) can induce apoptosis, involving altering mitochondrial permeability, subsequent cytochrome c release (ie, regulated by Bcl2 family proteins) and formation of the apoptosome (ie, multiprotein platform for caspase-9 activation).85,86 Caspase-9 cleaves to caspase-3, leading to downstream events in cell death. BAX (ie, Bcl2-associated X-protein), BID (ie, BH3 interacting death domain) and BIM (ie, Bcl2-interacting protein) translocate to mitochondria to induce apoptosis, either by forming pores in mitochondria directly or by binding via BH3 domains to Bcl2, Bcl-XL, and Bfl1, and antagonizing these anti-apoptotic proteins.
Metal-Based Nanoparticles Used for Cancer Theranostics
Metal-based cancer theranostic nanoparticles contain one or more metal element(s), with each type having unique properties that can be used for specific imaging and treatment techniques. These include nanoparticles made of gold, silver, copper, platinum, iron, lanthanide and quantum dots (QDs). There have been extensive studies conducted using metal-based theranostic nanoparticles for passive or active targeting, for treating various cancer types. In addition, hybrid nanoparticles have shown to be promising in improving multifunctional and overall theranostic effectiveness/efficacy. This section provides examples of the various metal-based nanoparticles that can be used for cancer theranostics, with results also shown in tabular format (Table 1).
Theranostic Metal-Based Nanoparticles for Cancer Therapy and Imaging.

OI, optical imaging; DD, drug delivery; PDT, photodynamic therapy; MRI, magnetic resonance imaging; PTT, photothermal therapy; CT, computed tomography; RT, radiotherapy.
Gold Nanoparticles
Gold nanoparticles (AuNPs) have received attention for theranostics due to their chemical inertness, good biocompatibility, tunable morphology (ie, size, shape), surface chemistry, SPR and luminescence.87,88 Zhao et al developed AuNP-based theranostic system (ie, FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs-Verte) for PDT and imaging of cervical cancer. 89 Folic acid (FA)-conjugated block copolymers, attached to citrate stabilized AuNPs through a linker, were used for targeting folate receptors of cancer cells. The photosensitizer (PS), verteporfin (Verte), which has previously been used for treatment,90,91 was covalently attached to the surface of particles, for PDT (ie, through generation of ROS) and fluorescence imaging (FI). Modification with FA-PEG-P(Asp-Hyd)50-DHLA copolymers enhanced the colloidal stability of AuNPs in solution at various pH values, mainly due to the steric repulsion of the copolymers. The FA-PEG-P(Asp-Hyd)50-DHLA copolymer-modified AuNPs maintained the red color indicating minimal aggregation of particles, with no significant changes in the UV–Vis absorption spectra under various acidic and basic pH conditions. Dynamic light scattering (DLS) size measurements confirmed surface modification of AuNPs, with average particle sizes of 17.6, 56.7, and 110.3 nm, for citrate-stabilized AuNPs, FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs, and FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs-Verte, respectively. The FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs with Verte showed rapid generation of ROS, as a function of light exposure (ie, at 0.4 mW/cm2 with a He–Ne laser at 708 nm), while FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs without Verte did not generate any ROS. From flow cytometry studies, uptake in HeLa cervical cancer cells of FA-PEGP(Asp-Hyd)50-DHLA-AuNPs-Verte particles was approximately 99%. The intracellular uptake of AuNP conjugates was enhanced, compared to uptake of Verte only, which was approximately 19%. The attachment of FA ligands likely enhances receptor-mediated endocytosis. TEM images of HeLa cancer cells showed FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs-Verte particles found in cytosolic vesicles (eg, endosomes, lysosomes). Using concentration of 10 μg/mL for Verte in FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs-Verte nanoparticles and 0.4 mW/cm2 laser intensity, the phototoxicity was enhanced, compared to dark toxicity. Viability decreased with increase in laser exposure time (ie, <10% after 30 minutes laser exposure time) for cervical cancer cells with Verte only and FA-PEG-P(Asp-Hyd)50-DHLA-AuNPs-Verte.
Non-spherical AuNPs such as nanocages (AuNCs) can be constructed with hyaluronic acid (HA), anti-Glypican-1 (anti-GPC1) antibody, oridonin (ORI), gadolinium (Gd), and cyanine-7 (Cy7) dye (ie, GPC1-Gd-ORI@HAuNCs-Cy7 NPs or ORI-GPC1-NPs). 92 Compared to other nanoparticles, AuNCs have unique properties including inner hollow space, ease of surface functionalization, and the capacity to transport and deliver various cargo.93–95 The anti-GPC1 antibody on nanoparticles improves the blood circulation time and half-life and reduces the clearance rate, while promoting accumulation of particles at the tumor site. 96 ORI is a natural compound, having detoxification, antibacterial, anti-inflammatory, and anti-tumor effects,97,98 while Gd and Cy7 are MRI and FI agents, respectively. The HA coating of nanoparticles provides binding sites for the anti-GPC1 antibody and enables encapsulation of high amounts of ORI for triggered drug release. Qiu et al showed ORI from ORI-GPC1-NPs (ie, in 10% fetal bovine serum [FBS] containing media) was released at greater amounts at pH 5.5 and in the presence of enzymes (ie, hyaluronidase), compared to release of ORI from the same nanoparticles at pH 5.5 (ie, without hyaluronidase). This shows the triggered release capability of nanoparticles under acidic conditions and in the presence of enzymes present at the tumor site. Using 100 μg/mL of ORI-GPC1-NPs, the cell viability values in PANC-1 and BXPC-3 pancreatic cells (ie, expressing high levels of GPC1), were <50% after 24 h incubation. The IC50 values (ie, half maximal inhibitory concentrations) after 24 hours incubation with ORI-GPC1-NPs were more than 3-fold lower for PANC-1 and BXPC-3 cancer cells, compared to 293T cells (ie, cells with lower expression of the GPC1 proteins). This shows the higher potency and specific targeting ability of ORI-GPC1-NPs for GPC1 positive cells for theranostics. Furthermore, the apoptosis rate in cells from the ORI-GPC1-NPs group was much greater than control, ORI and ORI-NPs groups, with downregulation in the expression of bcl-2 protein and upregulation in the expression of cleaved-caspase-3 protein (ie, protein markers for apoptosis). Treatment with ORI-GPC1-NPs led to greater inhibition of migration of pancreatic cells, compared to when treated with ORI and ORI-NPs. Compared to ORI-NPs, fluorescence intensity from ORI-GPC1-NPs was more than 4 times greater at 48 hours post-injection in tumors, with signals peaking at 24 hours post-injection. The MRI intensity from ORI-GPC1-NPs in tumors after 24 hours post-injection was more than 2 times greater, compared to tumors with ORI-NPs. Treatment with ORI-GPC1-NPs led to the strongest inhibitory effect on tumor growth, with the tumor size being the smallest out of all the treatment groups (ie, negative control, gemcitabine, ORI, ORI-NPs, ORI-GPC1-NPs).
Gold nanorods (AuNRs) when combined with optical absorbers (eg, IR780 NIR dye) can maximize light absorption (ie, through optical interaction effect) and can be used as PA contrast agents. Ge et al synthesized ultrasmall AuNRs with a thick coating of NIR dye (IR780) and anticancer drug doxorubicin (DOX) (ie, AuNR@IR780/DOX-RGD-PEG) for synergistic chemo-photothermal cancer therapy. 99 The ligand arginine-glycine-aspartic acid (RGD) was attached to the surface of polyethylene glycol (PEG) coated AuNRs, for specific targeting to positive αvβ3-expressing cancer cells and for enhancing cancer cell imaging.100–102 Absorption spectra of the approximately 35 nm AuNR@IR780/DOX-RGD-PEG showed that both DOX and IR780 were loaded on the particle, with characteristic peaks of IR780 at 780 nm and DOX at 490 nm. Compared to temperature increment from laser irradiation for IR780 (∼19.5 °C) and AuNR (∼14 °C), the temperature increment for theranostic AuNR@IR780/DOX-RGD-PEG was more than 2.5 times higher (ie, ∼60 °C), with PA intensity about 3 times higher at the same optical density value (ie, OD808 at 808 nm). Theoretical studies showed that the AuNR@IR780/DOX NPs were found to have 3.5 times higher electromagnetic (EM) field (ie, due to increased light absorption efficiency) than that of the bare AuNRs, resulting in amplified PTT and PA imaging performance of AuNR@IR780/DOX-RGD-PEG. In vitro drug release profiles showed pH-responsive drug release ability of theranostic nanoparticles with rapid DOX release observed at pH 5.5 (ie, over 70% of the DOX released after 15 hours), with minimal DOX released from nanoparticles at pH 7.4 (Figure 1A). The IC50 value in U87MG brain cancer cells treated with AuNR@IR780/DOX-RGD-PEG NPs at pH 6.5 and laser irradiation (808 nm, 0.25 W/cm2 for 5 minutes) was 0.52 mg/mL, lower than that from cells treated with DOX only (4.2 mg/mL) (Figure 1B). The PA signal from AuNR@IR780/DOX-RGD-PEG in tumors reached a maximum at 20 hours post-injection (Figure 1C), and was greater than PA signals from AuNR@IR780/DOX-RGD and AuNR@IR780/DOX-PEG (Figure 1D), showing the importance of PEG protection and RGD in increasing the circulation time and cancer cell targeting, respectively. Furthermore, the tumor accumulation efficiency of AuNR@IR780/DOX-RGD-PEG NPs (ie, with 11.5 injected dose per gram tumor tissue) was 2.4 and 3.5 times higher than that of the AuNR@IR780/DOX-RGD NPs (4.8 ID per g) and AuNR@IR780/DOX-PEG NPs (3.3 ID per g), respectively. With laser irradiation of AuNR@IR780/DOX-RGD-PEG NPs, tumor growth was significantly inhibited without regrowth in tumor bearing mice, showing excellent synergistic chemo-photothermal effect.

Drug-gold nanorod nanoparticles (AuNR@IR780/DOX-RGD-PEG) for photoacoustic imaging and enhancing anticancer efficacy. In vitro DOX cumulative release profiles (A) of the theranostic NPs in different pH conditions and cytotoxicity in U87MG brain cancer cells (B) were determined without/with laser irradiation (ie, 808 nm, 0.25 W/cm2 for 5 minutes). In vivo PA imaging (C) and PA amplitude (D) of the tumor treated with AuNR@IR780/DOX-RGD-PEG NPs at different post-injection time points revealed maximum signal 20 hours after injection of particles (scale bar: 1 mm). Republished with permission of the Royal Society of Chemistry (RSC Advances) from 99 (distributed under Creative Commons Attribution-NonCommercial 3.0 Unported (CC BY-NC 3.0) License at https://creativecommons.org/licenses/by-nc/3.0/ with no changes). The gold nanorods were synthesized using a seedless method, using cetyltrimethylammonium bromide (CTAB), gold(III) chloride trihydrate (HAuCl4·3H2O), AgNO3, HCl, ascorbic acid and NaBH4. The theranostic AuNR@IR780/DOX-RGD-PEG NPs were prepared by self-assembly using AuNR, polyphenol modified IR780, and DOX in DMSO, in the presence of water and ferric ions (Fe3+). The polyphenol modified RGD peptide and methoxy-poly(ethyleneglycol) Meo-PEG-polyphenol with an acid-labile β-thiopropionate linker were then added in the mixed solution, forming the theranostic NPs.
AuNPs can also be used for targeted imaging and gene therapy of lung cancer by loading epidermal growth factor receptor (EGFR) small interfering RNA (siRNA) onto the collagen modified surface (C-Au-EGFR si). 103 RNA interference (RNAi)-based gene silencing provides ability to knockdown target genes involved in pathogenesis, with several studies reporting on many cancer associated genes targeted by RNAi.104–107 Yu et al showed by using endosomal inhibitors and fluorescence microscopy experiments that C-Au nanoparticles entered A549 and BEAS-2B lung cancer cells through clathrin-mediated endocytosis and macropinocytosis, with lysosomes degrading the particles. 108 Cell viability from treatment with C-Au-EGFR si was more than 1.5-fold lower, compared to cells only and cells with C-Au (ie, after 48 hours incubation). Confocal fluorescence microscopy experiments showed maximum internalization of FITC labelled C-Au nanoparticles after 24 h incubation with lung cancer cells. There was also a decrease in transmembrane protein (ie, EGFR) expression level, after treatment with C-Au-EGFR si. Results from in vivo experiments revealed no significant change in tumor volume over treatment time period (ie, 30 days) with C-Au-EGFR si (ie, ∼40 mm3), with volumes for tumor bearing mice with phosphate buffered saline (PBS), C-Au, and EGFR si being approximately 150 mm3, approximately 140 mm3 and approximately 100 mm3, respectively after the same time. The tumor weight was more than 2 times reduced with C-Au-EGFR si, compared to PBS and C-Au groups. The overall survival of mice was prolonged due to treatment with C-Au-EGFR si.
Silver Nanoparticles
Like AuNPs, silver nanoparticles can be shaped into specific morphologies, making them promising agents for cancer therapy and imaging. For example, Zeng et al developed a “all-in-one” silver nanoprism platform for targeted tumor theranostics, 109 with inherent features such as strong SPR bands, high photothermal conversion efficiency, and great x-ray attenuation ability.110,111 The polydopamine (PDA) nanoshell serves as a coating on silver particles for providing biocompatibility, drug delivery, enhanced photothermal stability and therapy (ie, PTT). The Ag@PDA nanoparticles can be decorated with RGD peptides (ie, Ag@PDA-R) for targeting cancer cells and loaded with chemotherapeutics (ie, DOX) for combined photochemotherapy. The RGD peptide targets integrins (ie, αvβ3 integrin), that are highly expressed in many kinds of cancer cells.112,113 The absorption from Ag@PDA nanoprisms shifts to the NIR region after addition of RGD peptides, with the Ag@PDA-R particles having a hydrodynamic size of approximately 160 nm and a zeta potential of -5 mV (ie, negatively charged). The Ag@PDA particles (ie, at 10 μg/mL concentration) showed photothermal stability under five irradiation/cooling cycles (ie, at 808 nm, 1 W/cm2), with peak temperature of approximately 55 °C (ie, with initial temperature of ∼20 °C). Viability of 4T1 breast cancer cells in vitro after treatment with 150 μg/mL Ag@PDA-R particles and laser irradiation at 1 W/cm2 for 5 minutes (ie, at 808 nm) was approximately 10%, with viability being approximately 75% at the same concentration of Ag@PDA-R particles but without laser irradiation (Figure 2A). Confocal fluorescence images from DOX loaded particles (ie, Ag@PDA-R&D) in 4T1 cells showed signals being enhanced with increased incubation time, with more DOX molecules accumulating in the nucleus with time (Figure 2B). To show the ability of particles to be used for PA imaging, PA amplitudes were determined. The PA signals from Ag@PDA-R particles were stable for 8 hours after administering particles to 4T1 tumor-bearing mice, with PA images showing the intra-tumor accumulation of particles. The maximum PA amplitude from Ag@PDA-R in 4T1 tumors was at 800 nm, close to the absorption maximum of these nanoparticles. 3D in vivo images of tumors showed strong CT signals from Ag@PDA-R, with no signals seen before injection of particles (Figure 2C). Tumor ablation using Ag@PDA-R nanoparticles resulted from cell apoptosis caused by boosted temperature, which was further verified using histological (ie, hematoxylin and eosin, H&E) and fluorescence terminal deoxynucleotidyl transferase mediated deoxyuridine triphosphate nick end labeling (TUNEL) staining of tumor tissue. There was no significant increase in the tumor volume with time after treatment with Ag@PDA-R nanoparticles and laser irradiation (ie, using 808 nm, 1 W/cm2, 5 minutes). Immunological responses after chemo-PTT with Ag@PDA-R&D showed elevated production of interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor (TNF), interleukin-12p70 (IL-12p70) and interferon-γ (IFN-γ) proteins in the sera of mice (ie, determined 15 days after treatment) (Figure 2D), with activation-associated cytokines detected, confirming augmented antitumor immune response. 114 Also detected were significant increases in maturation of dendritic cells (DCs), functioning as messengers between innate and adaptive immunities. 115 Results from evaluation of the immune response after photochemoimmunotherapy (Ag@PDA-R&D + laser + anti-PD-1 blockade antibody) showed greater suppression of primary tumor growth and improved survival rate of mice, compared with that of the PBS and Ag@PDA-R&D groups.
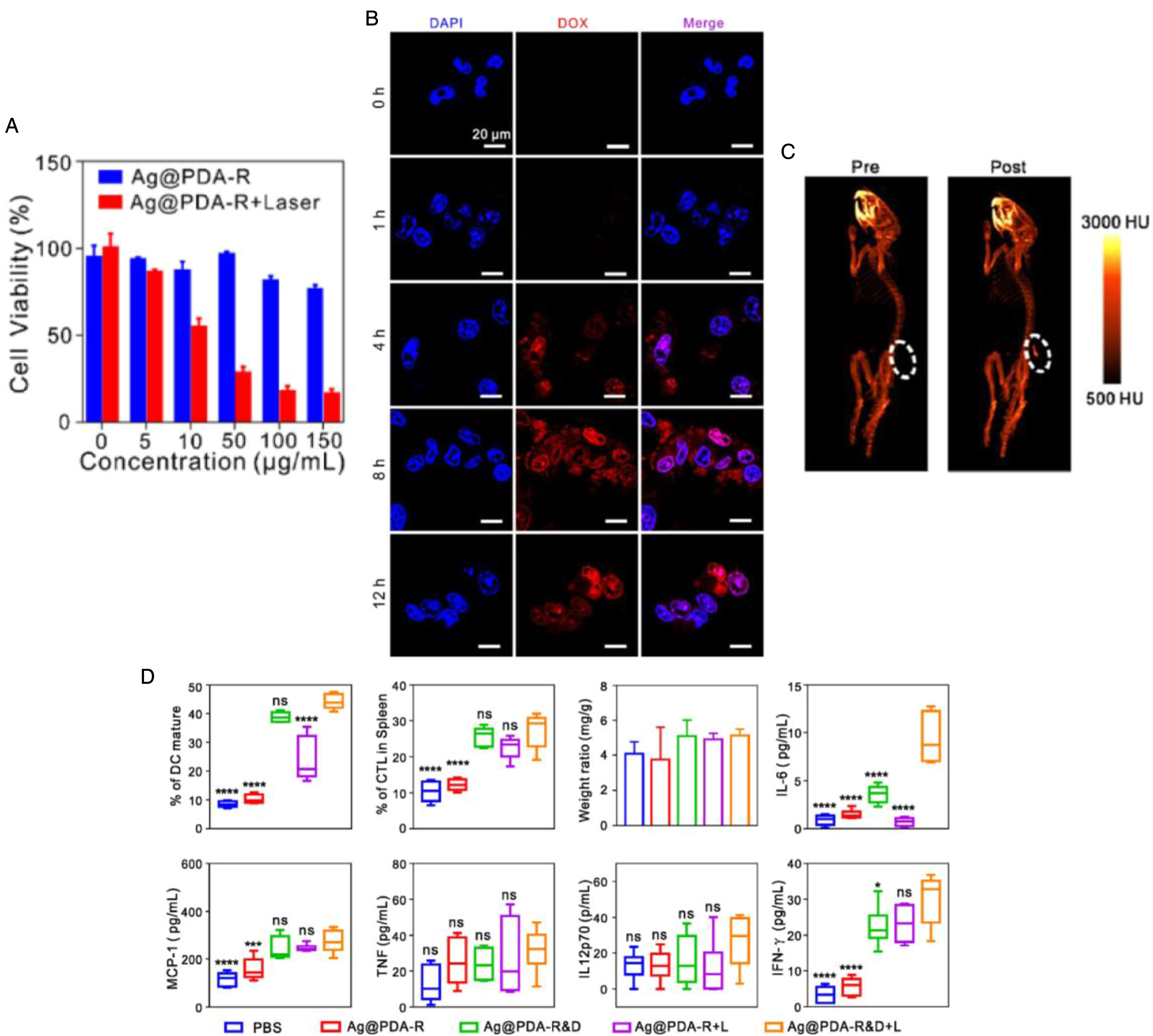
Silver nanoprisms (Ag@PDA-R, Ag@PDA-R&D) for cancer therapy and imaging. In vitro viability of 4T1 breast cancer cells was determined for different concentrations of Ag@PDA-R (after 24 hours incubation) in darkness or under laser irradiation at 1 W/cm2 for 5 minutes and further 24 hours of incubation (A). Fluorescence confocal images of 4T1 cells treated with Ag@PDA-R&D at different time points were acquired (B) (blue and red represent DAPI and DOX fluorescence, respectively) showing internalization of drug loaded particles. (C) 3D in vivo CT images of 4T1 tumor bearing mice were obtained before (left) and after injection of Ag@PDA-R (right) to show localization of nanoparticles in tumor site. The white dot circle demarcates the tumor site. Pre: before particle administration. (D) Frequency data of the flow cytometric examination of mature DCs (CD11c + CD80 + CD86+) in the lymph glands and cytotoxic T lymphocytes CTLs (CD4–CD8+) in the spleen is presented. The weight ratio was defined as the spleen weight (mg) compared to the counterpart body weight while the production of IL-6, MCP-1, TNF, IL12p70, and IFN-γ proteins were determined from the sera of mice 15 days after various treatments. *p < 0.05, ***p < 0.001, ****p < 0.0001, and ns: not significant (p > 0.05), analyzed by one-way ANOVA, followed by Dunnett's multiple comparison test. Data are represented as mean ± standard deviation. Reprinted with permission from Ref. 109 Copyright (2020) American Chemical Society. The silver nanoprisms were synthesized using sodium citrate, H2O2, NaBH4 and AgNO3. For 11-mercaptoundecanoic acid (MUA) passivation, silver nanoprisms were mixed with sodium citrate and MUA with a PDA coat produced using dopamine. For loading RGD peptide to form Ag@PDA-R particles, the Ag@PDA particles were first mixed with streptavidin before adding biotin-RGD peptide. Further loading of drug in nanoparticles was carried out by mixing DOX with Ag@PDA-R to form Ag@PDA-R&D.
Tan et al constructed spherical indocyanine green-loaded silver nanoparticle@polyaniline core/shell theranostic nanocomposites (ICG-Ag@PANI) for PA/NIR FI-guided and single-light-triggered therapy (ie, PTT and PDT). 116 Polyaniline is a conductive polymer that has received significant attention as an agent for photothermal cancer ablation, 117 while making particles biocompatible. ICG is a NIR dye approved by the Federal Drug Administration (FDA), 118 and used to produce local heating and ROS for PTT and PDT, respectively. 119 Upon photo-irradiation (eg, 808 nm), the ICG is released and de-quenched from the surface of Ag@PANI particles for hyperthermia and NIR FI. Simultaneously, cytotoxic ROS generation from ICG is used for further cell death. Averaged 2′,7′-dichlorodihydro-fluorescein (DCF) fluorescence intensity of individual samples measured from intracellular ROS detection imaging showed more than 6-fold increase in ROS production from ICG-Ag@PANI particles with laser irradiation (ie, at 808 nm, 1.0 W/cm2, 5 minutes), compared to Ag@PANI particles only. NIR laser irradiation (ie, at 808 nm, 1.0 W/cm2, 5 minutes) of ICG-Ag@PANI in HeLa cervical cancer cells led to viability of approximately 10%, compared to cell viability of approximately 30% to 40% when treated with other nanoparticles/molecules (ie, ICG, PANI, Ag@PANI) and laser irradiation. The PA intensity from ICG-Ag@PANI in tumors of HeLa tumor-bearing mice was more than 2-fold greater than signals from PBS, free ICG, PANI and Ag@PANI groups.
Coating silver (Ag) nanoparticles with a nanometric shell layer of a semiconductor (eg, zinc oxide, ZnO) can enhance the plasmonic features of Ag, with plasmon-exciton coupling 120 important for photoluminescence (PL) imaging. In addition, upon ultraviolet (UV) irradiation, the nanoparticles can generate radicals and ROS, 121 triggering activation of pro-apoptotic enzymes for cancer cell death. 122 Ghaemi et al demonstrated intracellular ROS induction by Ag@ZnO core–shell nanoparticles (Ag@ZnO NPs) through permanent optically active holes on nanoparticles123,124 that can be used for breast cancer theranostics. 125 The cell viability in MDA-MB231 breast cancer cells after UV irradiation of 30 μg/mL Ag@ZnO nanoparticles was approximately 20%, lower than viability in cells with nanoparticles only (ie, ∼60% cell viability). The amount of lactic dehydrogenase (LDH) released (% control) using nanoparticles and UV radiation was more than 4-fold higher, compared to cells incubated with Ag@ZnO only. This indicates that the antioxidant capacity of cancer cells is not sufficient for protection against a high amount of ROS, leading to cell membrane disruption and apoptosis.126,127 There was greater generation of intracellular H2O2 and O2• (ie, indicated by dichlorofluorescein (DCF) and hydroethidine (HE) fluorescence), using Ag@ZnO nanoparticles and UV radiation in breast cancer cells, compared to AgNPs plus radiation and ZnO NPs plus radiation groups. The oxidative stress in cancer cells and their resulting oxidative damage 128 increased after treatment with Ag@ZnO NPs and UV radiation, compared to Ag@ZnO NPs only, as measured by glutathione reductase (GR) and superoxide dismutase (SOD) levels (ie, measure of enzyme activity). The generation of ROS after UV irradiation of Ag@ZnO NPs triggers the upregulation of p53 protein and activation of pro-apoptotic proteins, increasing the amount of Bax and decreasing the amount of Bcl2 proteins. This in turn leads to mitochondrial membrane leakage, release of cytochrome c, activation of caspase-3 and DNA fragmentation for apoptosis. Flow cytometry results were consistent and showed higher amount, level and activity of cytochrome c, p53 and caspase-3 respectively, after UV irradiation of Ag@ZnO NPs. To show the bio-imaging ability, images were acquired of particles in phantoms showing greater enhancement in CT and optical contrast for Ag NPs and Ag@ZnO NPs, compared to water (ie, negative control).
Copper Nanoparticles
The use of copper can increase the light absorption in nanoparticles and enhance bio-imaging (ie, with ultralow-background) and PTT (ie, with high photothermal conversion efficiency).129,130 Chen et al designed a NIR persistent luminescence nanoparticle (PLNP)/copper sulfide (CuS)-based nanoprobe for luminescence imaging-guided PTT. 131 The PLNP (Zn1.1Ga1.8Ge0.1O4:Cr3+) and CuS are connected through a matrix metalloproteinase (MMP)-specific peptide substrate (H2N−GPLGVRGC−SH), making the nanoparticles MMP activatable optical probes. MMPs have important functions in tumor metastasis, invasiveness and angiogenesis 132 and are overexpressed in the area of cancer. Once the PLNP/CuS nanoprobes accumulate at the tumor region, they can be activated for cancer theranostics. The tumor targeting can be improved by further bio-conjugation of succinimidyl carbonate-poly-(ethylene glycol)thiol (SC−PEG−SH) and peptide c[Arg-Gly-Asp-(D-Tyr)-Lys] (c(RGDyK)). To keep minimal background signal of the activatable probe, the PLNP−CuS−RGD nanoprobe is able to provide nearly 94% luminescence quenching. The PLNP−CuS−RGD showed good stability in physiological environment (ie, in PBS, serum) after 24 hours, with no significant change in the size distribution. To demonstrate the activatable capability (ie, by enzymes) of particles, increasing the concentration of MMP-2 enzyme led to increase in luminescence of PLNP−CuS−RGD nanoprobe. With increasing concentration of MMP-2, the size distribution of the PLNP−CuS−RGD probe changed, suggesting it was successfully cleaved by MMP-2. This was further confirmed by in vitro studies, where in MMP-2 enzyme positive SCC-7 squamous cancer cells luminescence signals from LNP−CuS−RGD were observed, with no signals found in MMP-2 negative 293T cells. In vivo images showed significant luminescence in the tumor area of SCC-7 tumor-bearing mice after intravenous injection of PLNP–CuS–RGD for at least 2 hours, indicating the enhanced tumor targeting and effective MMP activation of the probe (ie, using PLNP−CuS−RGD). In contrast, the control tumor-bearing mice group with MMP-2 inhibitor gave much reduced signal intensity in the tumor. The uptake of PLNP−CuS−RGD by SCC-7 tumor also increased with time, for improving therapeutic outcome. To show the ability of PLNP−CuS−RGD to destroy cancer cells, in vitro cell viability studies were carried out in MMP-2 enzyme positive SCC-7 and MMP-2 negative 293T cells. After 24 h incubation with PLNP−CuS−RGD (ie, 200 μg/mL concentration) and 10 min irradiation with 1 W/cm2 (ie, at 808 nm), the cell viability percentages in SCC-7 and 293T cells were approximately 10% and approximately 40%, respectively, with viability approximately 80% with particles only. In SCC-7 tumor-bearing mice, the tumor volume showed 25-fold, 25-fold, and 37-fold increase (ie, compared with the original volume), for control groups with treatment of nanoprobe (PLNP−CuS−RGD) alone, with laser irradiation alone, and with no treatment, respectively. However in the PLNP−CuS−RGD plus laser group (ie, using 808 nm laser at 1.5 W/cm2, after 2 hours post-intravenous injection), the tumor volume did not change much compared with primary volume, showing photothermal therapeutic effectiveness of these nanoparticles. Furthermore, H&E staining showed no side effects (eg, in organs).
Hexagonal copper nanoparticles can be constructed for improving cancer theranostic capability. For example, Wang et al constructed hollow hexagonal Cu9S8 nanoparticles for PA imaging and enhancing chemodynamic performance. 133 Compared to solid Cu9S8 nanoparticles, the hollow Cu9S8 nanostructures improve the specific surface area, increasing the number of active sites (ie, for catalysis of the Fenton-like reaction) for improving therapeutic performance.134–137 Copper sulphide can destroy cancer cells through hydroxyl radicals generated from endogenous H2O2, under the catalytic conditions of copper ions. 138 Absorption spectra for hollow Cu9S8 nanostructures showed strong absorbance in the NIR region. Using a concentration of 200 μg/mL and 808 nm laser (ie, at a power density of 1.0 W/cm2 for 10 minutes), the change in temperature was approximately 20 °C for hollow Cu9S8 nanoparticles. The PA signal intensity increased linearly with concentration of hollow particles, reaching a value of approximately 12,000 (a.u.) at 200 μg/mL. In 4T1 breast cancer cells, hollow Cu9S8 nanoparticles had a stronger fluorescence intensity than solid Cu9S8 nanoparticles. When Cu9S8 (100 μg/mL) or 100 μM H2O2 plus Cu9S8 (100 μg/mL) was added to the culture medium after 15 minutes incubation, green fluorescence was observed suggesting that Cu9S8 could catalyze the production of hydroxyl radicals in intracellular or extracellular H2O2 through a Fenton-like reaction (ie, with a greater extent in hollow particles). Furthermore, 4T1 breast cancer cells treated with hollow Cu9S8 nanoparticles plus H2O2 and laser irradiation led to approximately 5% viability, compared to approximately 60% viability with particles with H2O2 but without laser irradiation. In vivo PA images and signals from 4T1 tumors after injection of hollow Cu9S8 nanoparticles revealed PA signal intensity peaking at 4 h post-injection, with signals from particles found throughout the tumor. Tumors treated with hollow Cu9S8 particles and 808 nm laser irradiation had the lowest relative tumor volumes, compared to other treatment groups, with hematological assay and H&E staining showing strong biocompatibility of particles.
Copper sulfide nanoparticles can be loaded (ie, forming core) inside a mesoporous silica shell along with perfluorocarbon (ie, perfluoropentane, PFP) for PA and US dual modality-guided photothermal combined immunotherapy. Zhang et al developed the CuS@mSiO2-PFP-PEG nanoparticles named as CPPs, with size of approximately 100 nm. 139 Under NIR light irradiation, through absorption and heat generation, the CPPs produce localized hyperthermia for destroying cancer cells, while being able to be used for PA imaging (ie, through nanoparticle absorption and acoustic wave generation). The simultaneous production of microbubbles from vaporization of PFP can be used for US imaging, making the nanoparticles multimodal imaging agents. The destruction of cancer cells leads to the release of tumor-associated antigens, with anti-PD-1 checkpoint blockade therapy used for inhibiting tumor growth.140–142 Cell viability of breast cancer cells was approximately 20% after 4 hours incubation with CPPs and laser irradiation for 4 min (Figure 3A), with >75% cell viability with particles only. This was consistent with flow cytometry results (Figure 3B) and co-staining of living/dead cells (Figure 3C), with significant number of MDA-MB-231 cells showing strong red fluorescence signals (ie, from propidium iodide, PI) after treatment (ie, after 4 hours incubation with CPPs and 808 nm laser irradiation). In contrast, weak red fluorescence signals were observed from cells with CPPs only and PBS plus laser. In vivo PA images (Figure 3D) showed that after intravenous injection of CPPs, the signals peaked at 24 hours post-injection at 808 nm laser irradiation (Figure 3E). The PFP bubbles generated from laser irradiation were sufficient for providing contrast-enhanced US images of tumors. Mean fluorescence intensity (MFI) values (ie, from 1,1′-dioctadecy1-3,3,3′,3′-tetramethylindotricarbocyanine iodide, DIR-labelled particles) were more than 2 times greater in tumors, compared to other organs, after 24 hours post-administration of CPPs and laser irradiation. The relative tumor volume for CPPs plus laser irradiation treatment group decreased with time due to temperature increase (eg, from ∼35 to ∼50 °C after 5 minutes laser irradiation) due to therapy (ie, PTT), while the tumor volumes for other groups (ie, control, control plus laser, CPPs) increased. Flow cytometry results showed that following treatment using CPPs, laser irradiation and anti-PD-1, the number of T cells increased for enhancing activation of innate immune response, through the production of interferon and interleukin proteins.143–145 Immunohistochemistry (IHC) analysis showed an increased expression of these proteins as well as in tumor necrosis factor 146 after using CPPs, laser irradiation and anti-PD-1.

Light-responsive core–shell CuS@mSiO2-PFP-PEG (CPPs) nanoparticles for bimodal imaging-guided photothermal therapy-primed cancer immunotherapy. Viability of breast cancer cells (A) was determined after incubation with CPPs and PBS and after laser irradiation for 1, 2, 3, and 4 minutes. Apoptosis analysis was carried out after different treatments using flow cytometry (B). Confocal laser scanning microscopy (CLSM) images showed live/dead cells (C) using Calcein AM/propidium iodide labelling for different treatment groups with in vivo US/PA imaging (D) showing signals peaking at 24 hours post-injection (E) (0 hours is pre-injection). Scale bar: 50 μm. Annexin V FITC and PI were used for detecting cancer cell apoptosis. Data are represented as mean ± standard deviation. Reprinted with permission from Ref. 139 Copyright (2020) American Chemical Society. The CuS@mSiO2 nanosystem was prepared by mixing CuCl2, sodium citrate, and Na2S. CuS-cetyltrimethylammonium chloride solution (CTAC) nanoparticles were mixed with CTAC and triethanolamine (TEA), with methoxy PEG silane added to form PEG coated CuS@mSiO2 nanoparticles. To make CuS@mSiO2-PFP-PEG (CPPs) nanoparticles, perfluoropentane (PFP) was added.
Copper-palladium alloy tetrapod nanoparticles (TNP-1) can also be used to induce pro-survival autophagy for optimizing therapy (ie, PTT) of drug-resistant cancer. Zhang et al demonstrated that the particles have unique sharp-tip structure for providing superior NIR photothermal conversion efficiency. 147 Autophagy can be inhibited when combining particles with 3-methyl adenine (3-MA) or chloroquine (CQ). If induced autophagy is pro-survival, inhibitors of autophagy can be used to enhance cancer cell death.148,149 Synergistic effect on TNP-1-mediated PTT in triple-negative (4T1), drug-resistant (MCF7/MDR) and patient-derived breast cancer models was seen when autophagy was inhibited with 3-MA or CQ. It was found that the shape and composition of TNP-1 were important for inducing autophagy, as neither particles with the same composition but different shape (ie, SNP) nor particles with the same shape but different composition (ie, TNP-2) induced autophagy under the same circumstances. The underlying mechanism for the ability of TNP-1 to induce autophagy is the enhanced production of intracellular ROS, particularly ROS generated in mitochondria. In MCF-7/MDR model, tumor weight of mice on day 15 in TNP-1 plus CQ and NIR laser irradiation treatment group was more than 10 times reduced compared to PBS plus NIR treatment group, with tumor volume unchanged at approximately 100mm3 during the 15-day therapeutic period. The tumor size and growth could be determined using PA imaging (ie, due to the optical absorption and acoustic properties of TNP-1), with signals peaking at 24 h post-injection in 4T1 breast tumors.
Platinum Nanoparticles
Platinum nanoparticles can be used to overcome limitations such as uncontrolled drug release behavior, targeting deficiencies, and hypoxia-associated poor PDT efficacy in solid tumors. Their inherent advantages include efficient catalytic activity and high stability for PDT,150,151 enabling particles to be used for synergistic treatment of cancer. 152 Hao et al constructed polymeric nanoparticles with ROS-responsive prodrug and platinum nanozyme for enhanced chemophotodynamic therapy of colon cancer. 153 The production of ROS is possible due to the introduction of a ROS-responsive prodrug (ie, CPT-TK-2-(1-hexyloxyethyl)-2-devinyl pyropheophorbide-a (HPPH)), consisting of thioketal bond linked camptothecin (CPT) and photosensitizer HPPH, which can effectively decompose ROS-responsive covalent bonds (eg, thioketal, arylboron ester, thioether, diselenide ether, peroxyacetate).154–156 The HPPH photosensitizer has good photodynamic activity, a high penetration rate for tumor tissue, and low phototoxicity, making it ideal for cancer therapy. 157 Under 660 nm laser irradiation, the CPT-TK-HPPH produced ROS, effective in enhancing PDT and controlled release of the CPT for treatment of colon cancer. In addition, due to the light absorption and fluorescence properties of HPPH, the CPT-TK-HPPH/Pt NPs can be used for fluorescence and PA imaging. The platinum nanozyme (PtNP) is important in nanoparticles (ie, CPT-TK-HPPH/Pt NPs), for efficiently catalyzing the decomposition of H2O2 into oxygen for relieving hypoxia in tumor tissue. The cumulative drug release (ie, using 660 nm, 200 mW/cm2 irradiation) after 7 days (ie, in PBS, 37 °C) from approximately 180 nm negatively charged (ie, -41 mV zeta potential) CPT-TK-HPPH/Pt NPs with laser irradiation was approximately 60%, and further increased to approximately 100% with the addition of 5 mM H2O2 at the same time point. By quantifying the fluorescence intensity from CPT-TK-HPPH/Pt NPs in CT26 colon cancer cells, it was found that nanoparticles were internalized in cells with a time-dependent manner. Compared to the no-laser groups, the intracellular ROS levels (ie, using 2′,7′-dichlorofluorescin diacetate (DCFH-DA) as the sensor) from treatment groups using CPT-TK-HPPH NPs and CPT-TK-HPPH/Pt NPs with 660 nm laser irradiation were 1.5- and 2.3-fold higher. Cell viability of CT26 cancer cells after treatment with CPT-TK-HPPH/Pt NPs (ie, after 24 hours incubation) was approximately 60%, with viability decreasing to approximately 10% upon addition of 660 nm laser irradiation (ie, at 200 mW/cm2 for 5 minutes). At the same concentration for nanoparticles (ie, 4.50 μg/mL HPPH and 2.45 μg/mL CPT), and after 48 h incubation, the viability from using CPT-TK-HPPH/Pt NPs was approximately 50% with the addition of laser irradiation decreasing the viability to approximately 5%. Flow cytometry analysis showed more than 7-fold increase in apoptosis rate in CT26 cells due to the addition of laser irradiation with CPT-TK-HPPH/Pt NPs, compared to CPT-TK-HPPH/Pt NPs only, with a greater proportion of cells in late apoptosis. FI of CPT-TK-HPPH/Pt NPs in CT26 tumor-bearing mice showed fluorescence intensity more than 3-fold higher than signals from HPPH in tumors, at 48 h post-injection. In vivo PA imaging at 680 nm showed tumor signals from CPT-TK-HPPH/Pt NPs increasing during first 6 hours after injection of particles, with signals being stable from 6 to 48 h post-injection. Both the tumor volume (100 mm3) and weight (100 mg) from CPT-TK-HPPH/Pt NPs plus laser irradiation treatment group were significantly reduced compared to other treatment groups and control group without treatment (ie, with ∼1250 mm3 tumor volume, 1000 mg tumor weight).
He et al tailored platinum(IV) amphiphiles for self-targeting all-in-one assemblies as precise multimodal theranostic nanomedicine. 158 The Pt(IV) amphiphiles can spontaneously self-assemble into stable supramolecular nanostructures (ie, micelles M(Pt) or vesicles V(Pt)). The Pt(IV) prodrugs are amphiphilic due to existence of the hydrophobic Pt(IV) core and hydrophilic cell targeting ligand, lactose. Lactose can efficiently target hepatoma cells overexpressing asialoglycoprotein receptors via receptor-mediated endocytosis,159–162 while Pt can be used for CT imaging, due to its high x-ray absorption coefficient. 163 The nanostructures produced can also be used for NIR FI due to the introduction of cyanine-7.5 (Cy7.5) optical absorber. The Pt(IV) complexes were designed by introducing two additional ligands to the axial positions, which can decompose to the corresponding toxic Pt(II) drugs under photo-irradiation or chemical reduction, for treating cancer cells.164–171 The IC50 values in A549, HeLa and HepG2 cancer cells from treatment with Pt(IV), M(Pt) and V(Pt) and laser irradiation (365 nm, 10 mW/cm2 for 15 minutes) were < 100 μM after 72 h incubation, with IC50 values > 200 μM with agents only (ie, without irradiation) (Figure 4A). Tumor volumes were lowest with V(Pt) and laser irradiation treatment group (∼100 mm3) at day 24, compared to tumors with V(Pt) only, without irradiation (∼500 mm3) (Figure 4B). The tumors were treated with 365 nm wavelength, 10 mW/cm2, for 30 min on days 1, 4, and 7. Strong fluorescence signals in the liver and orthotopic liver tumor were seen even at 24 hours post-i.v. injection of V(Pt/Cy7.5) particles. There was more than 2 times Pt concentration in the liver (ie, where the tumor exists), compared to Pt concentrations in organs (ie, heart, lung, kidney, spleen) at all time points measured (ie, from 1 to 24 hours post-injection) (Figure 4C). Furthermore, ex vivo CT images before and after V(Pt) injection for 12 hours showed highest signals in the liver where the tumor was present, with particles found well distributed (Figure 4D). Results from immunofluorescence of phosphorylated histone H2AX (γH2AX) protein expression in tumor tissues at day 24 showed up-regulation of γH2AX protein (ie, positive fluorescence signals from the γH2AX protein) from treatment with V(Pt) and laser irradiation, indicating increased number of tumor cells undergoing apoptosis.

Platinum(IV) amphiphile nanoparticles for cancer theranostics. IC50 values of M(Pt) and V(Pt) were determined against HepG2, HeLa, and A549 cancer cells (A) after 72 hours incubation without and with irradiation. Tumor growth inhibition curves in terms of tumor volume were calculated for different treatments (B) with biodistribution of platinum (Pt) (C) detected using inductively coupled plasma optical emission spectrometer (ICP-OES) and mass spectrometer (ICP-MS) after V(Pt) injection (***p < 0.001). To show capability of particles to be used for imaging, ex vivo CT images (D) were captured before and after V(Pt) injection for 12 hours. Data are represented as mean ± standard deviation. Reprinted with permission from Ref. 158 Copyright (2018) American Chemical Society. The platinum amphiphiles with different hydrophilic–hydrophobic balance self-assembled into different nanostructures. The Pt(IV)-1 amphiphile with one lactose molecule formed micelles (M(Pt)), while Pt(IV)-2 amphiphile with two lactose molecules formed vesicles (V(Pt)) above the critical aggregation concentrations (CAC). The Pt(IV)-3 precursor refers to c,c,t-[Pt(NH3)2(N3)2(OH)2].
Iron Nanoparticles
Nanoparticles containing iron are attractive due to their magnetic properties and ability to be used for MRI. Khaniabadi et al developed 4 to 10 nm trastuzumab conjugated porphyrin-superparamagnetic iron oxide nanoparticles (IONs-PP-TZ) for therapy (ie, PTT) and MRI. 172 The iron oxide nanoparticles (IONPs) were first conjugated with protoporphyrin using carbodiimide chemistry and then trastuzumab attached to the surface through glutaraldehyde crosslinking. Trastuzumab (TZ) is a monoclonal antibody which binds to HER2 receptor and can improve cell targeting when using nanoparticles. 173 Porphyrin (eg, protoporphyrin, PP) on the other hand has strong photosensitizing properties and ability to generate singlet oxygen for PDT and heat upon light absorption in the therapeutic window (650-1350 nm) for PTT. 174 The cell viability in MCF-7 breast cancer cells at 1 mg Fe/mL and 10 minutes laser irradiation (ie, using 808 nm, 200 mW) from using IONs-PP-TZ was approximately 65%, compared to viability of approximately 250% at the same concentration for IONs-PP-TZ but before irradiation. The cells were destroyed after 5 minutes irradiation with IONs-PP-TZ using 0.25 to 1 mg Fe/mL. The signal intensity from T2-weighted MR images decreased with IONs-PP-TZ concentration, with the transversal relaxation rates increasing.
Iron-based nanoparticles can also carry chemotherapeutic agents. Zhang et al designed iron-gallic acid (GA) network-based nanoparticles (Fe-GA@PEG-PLGA) with copolymer poly(lactic-co-glycolic acid) (PLGA) for MRI-guided chemo-photothermal synergistic therapy. 175 GA is a naturally occurring compound which stimulates apoptosis and leads to suppression of migration of tumor cells.176,177 The temperature of solutions containing Fe-GA network-based nanoparticles (ie, Fe-GA@PEG-PLGA) rose to approximately 70 °C from approximately 25 °C using concentration of 25 μg/mL and 808 nm laser irradiation (ie, using 1.6 W/cm2 and after 6 minutes). The nanoparticles showed excellent photothermal stability, as the temperature differences between adjacent peaks were within 4 °C after 4 cycles of laser irradiation. Under acidic pH of 5, nearly 70% of GA was released, greater than the cumulative percent release at pH 6.8 and 7.4. As the tumor microenvironment pH is acidic, the nanoparticles can release significant amounts of therapeutic agents once accumulated in tumor regions. In vitro experiments in 4T1 breast cancer cells using coumarin-6 labeled Fe-GA@PEG-PLGA nanoparticles showed that the cellular uptake was time dependent. The 4T1 cell viability was approximately 5% after incubation with 25 μg/mL Fe3+ of nanoparticles and laser irradiation (ie, at 1.6 W/cm2), compared to approximately 40% viability under no laser irradiation (ie, at the same concentration for nanoparticles) (Figure 5A). For HepG2 liver cancer cells, the viability was approximately 10% with laser and Fe-GA@PEG-PLGA nanoparticles and approximately 40% without laser but with particles (Figure 5B). After annexin V-FITC/PI staining, the percentage of late apoptotic cells was approximately 85% with Fe-GA network-based nanoparticles (ie, with 12.5 μg/mL Fe3+ concentration) and laser irradiation and approximately 20% with nanoparticles (Fe-GA@PEG-PLGA) only. After injection of Cy7-loaded Fe-GA network-based nanoparticles in tumor bearing mice, the signal intensity was at a maximum at 24 h post-injection (Figure 5C), with relative T1 MR signal value highest at 24 h post-injection (ie, for Fe-GA@PEG-PLGA), for the time points used for measurements (Figure 5D). The average volume of tumors after treatment with Fe-GA@PEG-PLGA and laser (5 minutes, 808 nm, 1.6 W/cm2) was significantly reduced at day 12, compared to control with PBS only (Figure 5E). Furthermore, H&E staining of organs and hematological studies (eg, of white blood cells, red blood cells, platelets count) revealed the nanoparticles as biocompatible and not toxic to main organs.
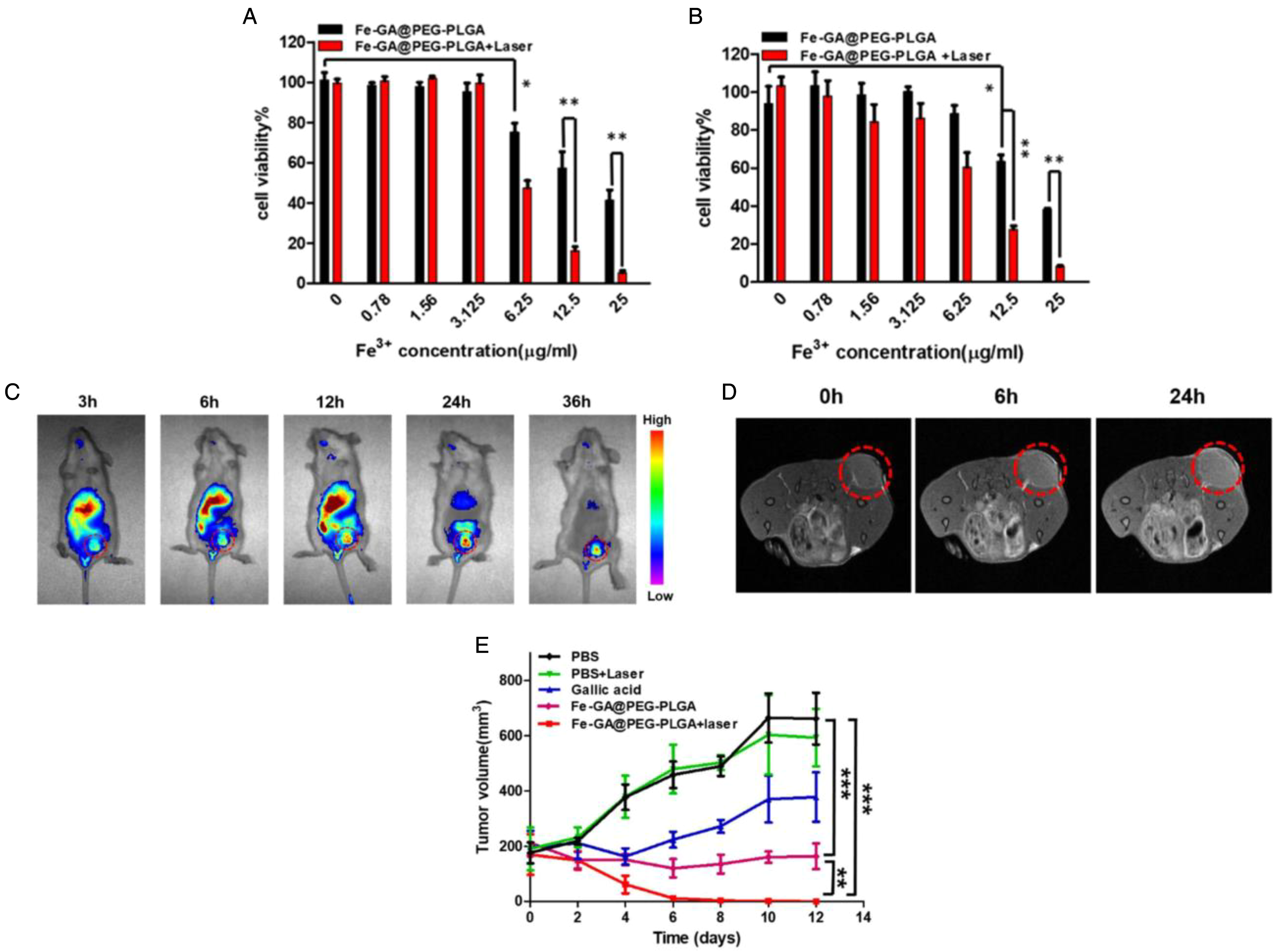
Theranostic capabilities from iron-gallic acid network-based nanoparticles (Fe-Ga@PEG-PLGA). The cancer cell viability of 4T1 breast cancer cells (A) and HepG2 liver cancer cells (B) after incubation for 24 hours with Fe-GA@PEG-PLGA nanoparticles, without or with NIR laser irradiation at power density of 1.6 W/cm2 (*p < 0.05, **p < 0.01). The in vivo distribution of Cy7-loaded Fe-GA@PEG-PLGA nanoparticles in tumor bearing mice was determined at various time points using fluorescence imaging (C) after intravenous injection of particles (1 mg/kg Cy7). The in vivo T1 MRI contrast from the Fe-GA network-based nanoparticles (D) was also determined at different time points post-intravenous injection (0 hours means pre-injection). After 24 hours post-injection of Fe-GA@PEG-PLGA nanoparticles, the tumors of mice were exposed to 808 nm laser (1.6 W/cm2) for 5 minutes and the resulting tumor volumes (E) were determined after for several days. Tumor volumes were also determined for different treatment groups for comparing results (**p < 0.01, ***p < 0.001, two-tailed Student's t-test). Data are represented as mean ± standard deviation. Reprinted from Nanomedicine: Nanotechnology, Biology and Medicine from Ref. 175 Copyright (2019), with permission from Elsevier. Fe-GA network-based nanoparticles were prepared by adding PEG-PLGA to Fe-GA network with Cy7 or coumarin-6 added to solutions during synthesis for fluorescence capability.
Saesoo et al fabricated approximately 160 nm liposomes consisting of superparamagnetic iron oxide nanoparticles (SPIONs) functionalized with anti-CD20 (rituximab, RTX) for the treatment and imaging of lymphoma (ie, Lip/PEG/Tween80/SPIONs-PVA/RTX). 178 Liposomes (Lip) have been shown to be effective for blood brain barrier (BBB) penetration and for improving drug delivery.179–181 Rituximab is commonly used in therapies for a broad variety of B-cell malignancies and induces apoptosis through CD20 binding.182,183 The Lip/PEG/Tween80/SPIONs-PVA/RTX exhibited superparamagnetic behavior with a saturation magnetization value of 0.4 emu/g. For Z138c lymphoma cells, the ratio values for quantifying cellular uptake in terms of SPION:Hoechst and Lip:Hoechst using Lip/SPIONs/RTX were greater than 2 and 2.5, respectively, while for Granta lymphoma cells the ratio values were greater than 4. The internalization of Lip-RTX in Z138c and Granta cells gradually increased with incubation time for time points between 30 minutes and 4 hours, and was more than 2-fold higher, compared to Lip. This confirms that the RTX antibody selectively binds to CD20 antigen receptor in lymphoma cells for improving theranostics. After annexin and propidium iodide staining of cells, flow cytometry results showed that Lip/PEG/Tween80/SPIONs-PVA/RTX induced apoptosis by a higher degree (ie, ∼65% and ∼43% in Granta and Z138c cells, respectively), compared to free RTX (ie, ∼18% and ∼17% in Granta and Z138c cells, respectively). Permeability of the BBB was determined by measuring the trans-endothelial electrical resistance (TEER), using bEnd3 endothelial cells and seeding cells onto 0.4 μm pore size membrane inserts. Granta lymphoma cells were seeded at the bottom of the culture dish for cell viability studies from the use of Lip/PEG/Tween80/SPIONs-PVA/RTX. Results revealed that free RTX is not readily transported through the BBB membrane, but the use of liposomes for loading promotes cross-barrier transport. RTX-conjugated liposomes resulted in toxic effects after 5 hours exposure with lymphoma cells, with no therapeutic effects from free RTX during the time periods used for experiments. After 48 hours following intravenous injection of anti-CD20-liposome-SPIONs, hypo-intense signals were observed for both T1 and T2-weighted sequences in the lymphoma xenografts.
Jiang et al developed SPIO nanoparticles that can be loaded in nanoparticles, such as micelles.
184
The size range of SPIO-loaded poly(ethylene glycol)-co-poly(D,

Stability, drug loading content, efficiency and bioimaging capabilities of SPIO-loaded micelles. Dissociation of SPIO-free and SPIO-loaded micelles (A) were determined from fluorescence intensity (ie, from pyrene, lem = 390 nm, lex = 333 nm) at the concentration of lower critical micelle concentration (CMC) (8 μg/mL) at 37 °C (**p < 0.05). The effect of SPIO-clustering on drug loading of PEG-PLA polymeric micelles was determined by calculating the drug loading content (DLC, %) (B) and drug loading efficiency (DLE, %) (C). DLE (%) was calculated using the following equation: [estimated Doxo loading] / [theoretical Doxo loading] × 100 (***p < 0.01, **p < 0.05). T2 relaxation rates (1/T2, s− 1) of the collected H2009 cells (D) were determined after treatment with Doxo-SPIO-micelles, with the inset showing T2 maps of cell samples. Data are represented as mean ± standard deviation. Reprinted from Biomaterials Research from Ref. 184 Copyright (2022), with permission from Springer Nature under Creative Commons Attribution 4.0 International (CC BY 4.0) License (https://creativecommons.org/licenses/by/4.0/) with no changes. Crystalline and monodisperse SPIO nanoparticles were synthesized from Fe(acetyl acetonate)3 in an aryl ether with PEG-PLA surfactant molecules used to encapsulate SPIO and Doxo for the micelles. The SPIO nanoparticles and DOX molecules were loaded in polymeric micelles by a solvent-evaporation method.
Chitosan coated magnetic iron oxide nanoparticles (ie, IR820-CS-Fe3O4) can be synthesized as a theranostic platform for melanoma. Hou et al showed that the approximately 25 nm IR820-CS-Fe3O4 nanoparticles have absorbance in the 700 to 900 nm region, with the peak absorption of IR820 red-shifted after grafting on CS-Fe3O4. 186 The IR820-CS-Fe3O4 nanoparticles have good stability in aqueous solution, with the peak absorbance value not changing significantly after 8 days. On the other hand, the absorption of IR820 alone decreased from peak absorbance value of 1.035 at day 1 to 0.688 at day 8. The nanoparticles were taken up by A375 melanoma cells by endocytosis and located in the lysosomes, with the uptake (ie, in terms of mean fluorescence signals) of FITC-CS-Fe3O4 nanoparticles in cells concentration dependent. The T1 and T2 relaxation rates of IR820-CS-Fe3O4 showed a linear relationship with increasing Fe concentration, with changes in MRI signals from images of A375 cells seen with different concentrations of IR820-CS-Fe3O4. Upon laser irradiation (808 nm, 8 W/cm2) of IR820-CS-Fe3O4, both the temperature and singlet oxygen production increased, with change of temperature of approximately 10 °C and oxygen production of approximately 104 after 5 min irradiation. As the IR820-CS-Fe3O4 concentration increased, the number of dead cells increased after laser irradiation (ie, all cells were destroyed at concentration of 16 μg/mL for IR820-CS-Fe3O4 with laser irradiation). These results show the potential of IR820-CS-Fe3O4 nanoparticles for PTT and PDT due to the addition of IR820 NIR dye,187,188 with the ability also to be used as imaging agents for cancer.
Some studies report the use of magnetic nanoparticles where the use of RF radiation and MR can be used for both imaging and therapy. Ashokan et al developed spherical approximately 125 nm iron doped single-phase calcium phosphate biomineral nanoparticles (nCP:Fe) for image-guided RF ablation of cancer. 189 Increasing the Fe3+ dopant concentration from 1.5% to 6% resulted in T2 contrast intensity enhancement, with maximum T1 contrast at 4.1%. Significant reduction in viability for nCP:Fe treated N1S1 hepatoma cells was seen after non-invasive RF treatment (ie, 100 W, 5 minutes exposure). Cell viability was approximately 5% using 300 μg/mL of nCP:Fe and RF treatment, compared to approximately 30% viability with same concentration, but with un-doped nCP. This is due to significant change (ie, rise) in temperature of approximately 15 °C, compared to approximately 7.5 °C for un-doped nCP. From optical images, cells treated with nCP:Fe and RF radiation were found to have lost membrane integrity and disintegrated. Results from experiments in rats showed clear T2 weighted MRI contrast enhancement in the liver, 1 hour after nCP:Fe injection.
Lanthanide Nanoparticles
Lanthanide particles can serve as important theranostic agents for biomedical applications,190–192 with attractive physical and chemical characteristics, including long-lived luminescence, large Stokes or anti-Stokes shifts, resistance to photo-bleaching, narrow emission bands and low toxicity. Addisu et al developed mixed lanthanide oxide nanoparticles coated with alginate (Alg)-polydopamine (PDA) as multifunctional nanovehicles for dual modality (ie, targeted imaging and chemotherapy). 193 Two systems of nanoparticles were investigated, with Alg-PDA complexed with either terbium/europium or dysprosium/erbium oxide NPs (ie, Tb/Eu@AlgPDA or Dy/Er@AlgPDA NPs). For specific targeting of cancer cells, Tb/Eu@AlgPDA nanoparticles can be functionalized with FA, with the ability to load anticancer drug doxorubicin (FA-Tb/Eu@AlgPDA-DOX NPs). Alg is biocompatible, biodegradable and has functional groups for chemical modification,194,195 while PDA can be used for loading chemical dyes or drugs on its surface via π−π stacking and/or hydrophobic−hydrophobic interactions. 196 Under low pH (ie, in tumors), the interaction between drug (eg, DOX) and AlgPDA nanocomposite can be disrupted, making nanoparticles containing AlgPDA pH sensitive drug releasing vehicles. 197 After 24 hours at pH of 5 or 6 (37 °C, PBS), the cumulative drug release was approximately 42% or approximately 23%, respectively, from FA-Tb/Eu@AlgPDA-DOX particles. After 4 days at pH 5, the DOX release from particles increased to approximately 60%, with only approximately 13% DOX release in the first 24 hours at pH 7.4. The DOX release did not surpass approximately 14% after 5 days at pH 7.4, showing the ability of particles to carry and delivery the majority of loaded therapeutic agents to tumors. In HeLa cervical cancer cells, the viability was approximately 10% with 18 μg/mL DOX in FA-Tb/Eu@AlgPDA-DOX nanoparticles, with viability approximately 20% when treated with DOX only (ie, at same concentration of 18 μg/mL). In zebrafish with FA-Tb/Eu@AlgPDA-DOX NPs, fluorescence signals were found in different body parts, showing the potential of particles for clinical imaging. Growth curves of HeLa cell spheroids after treatment with FA-Tb/Eu@AlgPDA-DOX NPs for 48 h showed the growth ratio at approximately 0.7 at 18 μg/mL DOX concentration, compared to approximately 1.05 growth ratio with no treatment (ie, after 48 hours). The growth ratio from FA-Tb/Eu@AlgPDA-DOX NPs was smaller than when spheroids were treated with DOX only (∼0.9), showing the anticancer efficacy of FA-Tb/Eu@AlgPDA-DOX NPs. Fluorescence microscopy images showed that the signals from FA-Tb/Eu@AlgPDA and DOX from FA-Tb/Eu@AlgPDA-DOX NPs were found throughout HeLa cell spheroids.
Nanoparticles can exhibit radioactive characteristics by including the lanthanide element 177Lu and can be used for SPECT imaging and radiotherapy (RT). Viana et al constructed 177Lu-labeled Eu-doped mesoporous silica (SiO2) nanoparticles as a theranostic radiopharmaceutical for colorectal cancer. 198 177Lu is an ideal dual-modality radionuclide, as it emits medium energy beta radiation for RT and low energy gamma rays for SPECT imaging. 199 On the other hand, the inclusion of silica (SiO2) in nanoparticles, improves biocompatibility, enabling surface modification and loading of therapeutic agents (eg, drugs, targeting molecules/ligands).200,201 Exponential decay curves for the EuDPA/SiO2–NH2 nanoparticles revealed a low radiative decay rate (Arad = 228 s−1), and high non-radiative decay (Anrad = 2297.8 s−1), with a reduction in lifetime (τ = 0.31 ms). This indicated a recovery of quantum efficiency of emission (η) equal to 7%. The EuDPA/SiO2–NH2 nanoparticles showed good in vitro uptake in HT-29 colorectal cancer cells, with strong cytoplasmic fluorescence due to accumulation of particles. Cell viability results with particles only revealed biocompatibility of nanoparticles, with no significant changes with increase in concentration after 24 or 48 hours incubation. Evaluation of bio-distribution from experiments indicated that percentage injected dose per gram of tissue (%ID/g) in tumors was significantly greater from using 177Lu−EuDPA/SiO2−NH2 (177Lu-NP) nanoparticles (ie, ∼70% ID/g), compared to 177LuCl3 (ie, ∼10% ID/g), with lower accumulation in major organs. After treatment initiation (ie, using RT), the tumor volume was approximately 500 mm3 for the 177Lu-NP group and lower than the tumor volume (∼1750 mm3) for control EuDPA/SiO2–NH2 nanoparticles group (ie, at the same time point). These results show that the lutetium (177Lu) element in nanoparticles can be used as effective cancer theranostic agents.
Chen et al designed and synthesized luminescent core−shell Nd3+-sensitized NaY(Mn)F4:Yb/Er@NaYbF4:Nd UCNPs (CSUCNPs) for deep tissue imaging and multidrug resistance (MDR) tumor diagnosis, with minimized heating effect on healthy tissues. 202 The developed upconversion nanoparticles (UCNPs) have emission bands in the red to NIR spectral range (600-1100 nm) for deep tissue penetration,203–205 after excitation at 808 nm. As well, incorporation of D-α-tocopherol polyethylene glycol 1000 succinate (TPGS) in nanoparticles has the ability to overcome the MDR effect, through inhibition of the P-glycoprotein (P-gp)-mediated drug efflux.206–208 TPGS has an amphiphilic structure, and it can lead to formation of a hydrophobic layer for drug loading, after its lipophilic alkyl tail binds to oleic acid containing CSUCNPs. Greater drug release from CSUCNPs-TPGS-doxorubicin nanoparticles (D-CSUCT) at pH 5 was seen, compared to drug release at pH 7.4 (ie, 26%), with 94% DOX released during the period of 168 hours. At lower pH (ie, acidic) conditions, the number of hydrogen ions (H+) increases and positive charges weaken the hydrophobic interactions, increasing water solubility of DOX and its release from particles. The improved drug release is important for cancer treatment due to the acidic microenvironment of tumor tissue. Using two-photon confocal laser scanning microscopy (TCLSM) at 808 nm excitation, the upconversion luminescence (UCL) of CSUCNPs was observed for both DOX-sensitive MCF-7 (Figure 7A) and DOX-resistant MCF-7/ADR (Figure 7B) breast cancer cells, suggesting the particles can be used for different cancer bio-imaging applications. In vivo tumor imaging showed red luminescence (ie, from D-CSUCT) at the tumor site (Figure 7C), with TCLSM images of tumor tissue revealing weaker luminescence intensity from D-CSUCT at the denser tissue areas, and stronger intensity at the looser tissue areas with vacuoles. The mean intracellular DOX fluorescence intensity of D-CSUCT treated MCF-7/ADR cells was about 15 times larger than that of DOX treated cells and approximately 2 times larger than that of TPGS-DOX treated cells (Figure 7D), due to cellular uptake facilitation by D-CSUCT. Cell viability studies revealed that MCF-7/ADR cells were highly resistant to free DOX with approximately 60% viability at DOX concentration of 100 μg/mL (ie, after 24 hours incubation), which decreased to approximately 20% viability using D-CSUCT, at same concentration of DOX (Figure 7E). This is largely due to the inhibition effect of TPGS on P-gp which increases the enrichment of DOX, when cells are treated with D-CSUCT. The relative tumor weight from treatment with D-CSUCT was approximately 10 times reduced, compared to the tumor weight of control (ie, mice injected with PBS) and approximately 3 times less than the tumor weight of tumor bearing mice treated with DOX only (ie, relative tumor weight after 14 days) (Figure 7F). Immunohistochemical analyses of tumor tissues using caspase-3, TUNEL, Ki67 and CD31 staining revealed higher level of apoptosis and decreased proliferation of cancer cells after treatment with D-CSUCT, compared to other groups (ie, control with PBS, DOX, CSUCNPs-DOX). The increased efficacy of therapeutic activity from D-CSUCT was reflected by the lowest microvessel density (MVD).

Upconversion nanoparticles (UCNPs) for synchronous multidrug resistance tumor imaging and therapy. TCLSM cell imaging showed internalization of D-CSUCT after 1 hour incubation in MCF-7 (A) and MCF-7/ADR (B) breast cancer cells. Upconversion luminescence imaging (C) of tumor bearing mice after injection of D-CSUCT showed accumulation of particles at the location of tumors. There was significantly greater intracellular DOX fluorescence intensity seen with D-CSUCT (D) with reduction in cell viability of MCF-7/ADR cells with D-CSUCT (E) compared to other treatment groups (n = 3, *p < 0.05). Results were consistent showing reduction in relative tumor weight (F) after treatment with particles, after 14 days (n = 5, *p < 0.05). Reprinted with permission from Ref. 202 Copyright (2018) American Chemical Society. The CSUCNPs were synthesized through a liquid–solid–solution (LSS) strategy. For NaY(Mn)F4:Yb/Er core synthesis, MnCl2, YCl3·6H2O, YbCl3·6H2O and ErCl3 were added to a mixture containing NaOH, deionized water, ethanol and oleic acid (OA). Then NaF was added resulting in the colloidal solution. The fabricated Mn2+-doped UCNPs were then mixed with Yb3+, Nd3+, NaOH, deionized water, ethanol and OA, followed by addition of NaF. Surface modification of CSUCNPs with TPGS was carried out via hydrophobic interaction between OA ligands on CSUCNPs and hydrophobic end of TPGS. Drug molecules were loaded in nanoparticles by mixing CSUCT nanoparticles with DOX molecules. CSUCT refers to CSUCNPs with TPGS, CSUCNPs-DOX refers to CSUCNPs with DOX, TPGS-DOX refers to TPGS with DOX and D-CSUCT refers to CSUCNPs with both TPGS and DOX.
Lanthanide nanoparticles can also have unique structures and treatment properties for effective tumor targeting and imaging. For example, He et al synthesized lanthanide-peptide-derived bacterium-like nanotheranostic particles (LProd nanorods) for colorectal cancer. 209 The loaded dodecameric peptide (PMI) reactivates tumor suppressor protein p53 through antagonizing the interaction between p53-MDM2/MDMX.210–212 In the presence of glutathione (GSH) (ie, a tripeptide thiol found in the cytoplasm of cancer cells),213,214 the approximately 160 nm particles are able to disassemble into approximately 10 nm nanocrystals and release peptide cargo for therapy. With 10 mM GSH, 50% peptides (ie, PMI-Cys) were released at 0.64 hours, and >90% peptides could be released at 2 hours. On the other hand, >90% residual PMI-Cys was still present in LProd after a 12-hour GSH-free incubation in PBS buffer (pH 7.4). Under the same conditions, LProd nanorods showed an almost complete cellular uptake by cancer cells, relative to its cyclopeptide iNGR-free counterpart. Both spherical LPparticle and LProd nanostructures were found internalized and well distributed in HCT116 cancer cells, with higher cellular uptake and intracellular fluorescence intensity from LProd. In HCT116 tumor-bearing mice, the radiance flux at tumor sites from LProd was approximately 2.5 times greater, compared to spherical LPparticle nanostructures, at 12-hour post-injection. By loading CD13-specific binding cyclopeptide iNGR (CRNGRGPDC), the accumulation of LProd nanostructures further increased, due to specific targeting. 215 Immunohistochemical (IHC) assays in xenograft tumors showed levels of p53 and p21 significantly elevated in the LProd- or LPparticle-treated groups, compared to PBS and PMI-treated groups. Results from Ki67 staining of treated cells from the use of LP nanoparticles revealed p53 reactivation inhibited tumor growth. Compared to other groups (ie, with ∼20 relative tumor volume), tumor bearing mice treated with LP nanoparticles had much lower relative tumor volumes (ie, ∼2.5 for LProd, LPparticle).
Quantum Dots
QDs can be used as attractive theranostic nanoparticles due to their intrinsic properties, high versatility, cost-effectiveness, high quantum yield emission and flexibility in surface functionalization for a variety of biological applications. 216 Soheyli et al synthesized biocompatible AgInS-based/ZnS core/shell QDs for high-contrast FI and treatment of melanoma cancer cells. 217 The Ag-In-S QDs have long PL lifetime and large Stokes shifts, which improves bio-imaging due to reduction in self-absorption effects. By making moderate modifications in their chemical composition by the incorporation of Zn and Cu ions, the bandgap energy and emission spectra of QDs can be tuned for maximizing bio-imaging contrast. Up to a concentration of 50 ppm, the QDs are biocompatible, with the cell viability (ie, after 5 hours incubation) in melanoma cells at 50 ppm QDs decreasing by <10%, 25% and 20% for ZAIS/ZnS, AIS/ZnS, and CAIS/ZnS QDs, respectively. However, upon laser irradiation (ie, at 405 nm for 5 minutes) at 50 ppm QDs, there was >80% cell death indicating the potential application of particles for cancer treatment. Confocal fluorescence microscopy studies showed intracellular ROS generation (ie, by oxidation of DCFH into DCF by free H2O2) after laser irradiation of the QDs. Flow cytometry results showed 79%, 64%, and 60% ROS generation after 405 nm laser irradiation in cells for ZAIS/ZnS, AIS/ZnS, and CAIS/ZnS QDs, respectively. There was a high degree of uptake of QDs in melanoma cells, with an amount of 91%, 82%, and 88% intracellular fluorescence for ZAIS/ZnS, AIS/ZnS, and CAIS/ZnS QDs, respectively.
Yang et al developed ultrasmall bismuth sulfide QDs for boosting phototherapeutic efficiency (ie, using both PTT and PDT).
218
Bismuth is used because of its biocompatibility,
219
with Bi2S3 possessing high absorption coefficient and a direct band gap in the near infrared (NIR).220,221 Furthermore, the Bi2S3 QDs offer abundant photoelectrons for the rapid production of ROS, due to high reducing ability. Positively charged polymers (ie, poly-
Photoabsorption, photodynamic and cancer cell viability studies using PLL-Bi2S3 QDs. The UV–visible–NIR absorption spectra of PLL-Bi2S3 QDs (A) were determined at different concentrations with the ROS levels (B) and mitochondrial membrane potential measured by JC-1 staining (C) of HepG2 liver cancer cells without or with laser irradiation. The different groups include positive control group, PLL-Bi2S3 QDs (50 μg/mL, without laser irradiation), and PLL-Bi2S3 QDs (50 μg/mL) with NIR laser irradiation (808 nm, 0.8 W/cm2, 10 minutes). Cell viability of different cancer cells (MCF-7 breast, HepG2 liver, HeLa cervical) was determined after incubation with only PLL-Bi2S3 QDs (D) and with both PLL-Bi2S3 QDs and NIR irradiation (808 nm, 0.8 W/cm2) for 7 minutes (E) (**p < 0.01 and ***p < 0.001). Data are represented as mean ± standard deviation. Reprinted from Chemical Engineering Journal from Ref.
218
Copyright (2019), with permission from Elsevier. Bismuth sulfide QDs were synthesized by mixing and adding Bi2O3, glacial acetic acid to oleic acid (OA) to form bismuth oleate. A 1-octadecene solution was added to synthesize bismuth precursor solution. The precursor solution was mixed with 1-octadecene and OA before thioacetamide (TAA) and ethyl acetate were added. Poly-
Al-Ani et al constructed lead sulfide (PbS) QDs with small 6.5 kDa protein-based antibody replacements (ie, affibodies, specifically ZHER2:432 Afb) for targeting,225,226 specifically toward the cancer biomarker, human epidermal growth factor receptor 2 (HER2). The nanoparticles can also be loaded with zinc(II) protoporphyrin IX (ZnPP-Afb2C-PbS QDs) for combining imaging, targeting, and therapy capabilities. 227 Zinc(II) protoporphyrin IX (ZnPP) has been shown to have therapeutic anticancer activity, with inhibition of hemeoxygenase-1 enzyme228,229 in cancer cells and ability to induce apoptosis,230,231 through generation of singlet oxygen species from laser irradiation (ie, λex = 425 nm). After 72 hours incubation with Afb2C-PbS QDs, a mean growth inhibition (GI50) value of 26 μg/mL was achievable in HER2 positive SKBR3 breast cancer cells, while no growth inhibitory effects were observed in MDA-MB-231 cancer cells (ie, HER2 normal) < 100 μg/mL. In the presence of ZnPP in nanoparticles (ZnPP-Afb2C-PbS QDs), the normalized absorbance from using MTT assay was approximately 0.1 at concentration of 100 μg/mL in SKBR3 cells, which was lower than the absorbance value (∼0.25) in MDA-MB-231 cells after treatment using same particles at same concentration. This shows the enhanced cellular uptake of ZnPP-Afb2C-PbS QDs in HER2 positive cancer cells and ability to induce higher cytotoxicity, compared to HER2 normal cancer cells. Cell cycle studies after treatment with ZnPP-Afb2C-PbS QDs showed presence of significant pre-G1 events, indicative of DNA degradation and consequent cell apoptosis. 232 Confocal microscopy imaging (ie, at 425 nm excitation, 593 nm emission) showed intracellular fluorescence from ZnPP-Afb2C-PbS QDs, detected in SKBR3 cells, with presence of blebbing of cell membranes and indication of apoptosis, due to presence of ZnPP.
Hybrid Nanoparticles
Combining metal elements to form hybrid nanoparticles can enhance reliability of cancer detection due to the ability of using more than one imaging modality. As well, hybrid particles can improve the therapeutic functionality and encapsulation efficiency of drugs for treating cancer. 233 For example, David et al developed and evaluated a multifunctional organic-inorganic nanotheranostic hybrid for pancreatic cancer. 234 The iron oxide-gold nanoparticles provide dual contrast enhancing ability for both MRI and micro-CT, with co-encapsulation of nucleotide analogue gemcitabine in a chitosan matrix (CGFe@Au). 235 Particles consist of a central iron (Fe) core, coated with iron oxide (Fe2O3), with gold (Au) nanoparticles on the surface. As the environment gets more acidic (ie, in tumors), there is faster release of drug, due to the higher swelling exhibited by the chitosan polymer from CGFe@Au particles.236,237 Encapsulation efficiency (∼96%) of gemcitabine was found to be highest at 3:2:1 carrier:drug:Fe@Au contrast agent ratio (ie, ∼40% entrapment of Fe@Au). A 10-°C increase in temperature was seen (ie, initial temperature ∼32 °C) using 3 mg/mL Fe@Au in 17 seconds in an AMF, while it took 165 and 724 seconds to achieve the same temperature increase in the CFe@Au and CGFe@Au NPs, respectively. In the 42 to 46 °C temperature range, hyperthermia can lead to cytotoxicity for treating cancer cells, with further temperature increase used for thermal ablation for greater cancer cell death. Phantoms with Fe@Au were used for determining MRI signals, as iron and its oxides can be used as negative contrast agents. Results revealed dose-dependent reduction in the MRI signal intensity from Fe@Au nanoparticles. Increasing the concentration of CGFe@Au NPs increased the signal intensity value, seen in micro-CT images. In vitro cytotoxicity studies showed that using a concentration of 100 μM CGFe@Au NPs resulted in approximately 25% viability in PanC-1 pancreatic cancer cells (ie, after 48 hours incubation).
Gold coating on lanthanide phosphate nanoparticles (ie, [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800@Ab) can provide ability for radiotherapy (RT) and SPECT imaging. 238 The nanoparticles combine the radiation resistance of lanthanide phosphate for stability, magnetic properties of Gd for separation during synthesis, and a gold coating for surface functionalization and attachment of targeting moieties (eg, antibodies, peptides). Sobol et al developed these theranostic agents for pulmonary metastatic diseases, using the monoclonal antibody mAb-201b for targeting thrombomodulin receptors in the lungs.239–241 The approximately 70 nm Lu0.5Gd0.5(PO4)@Au@PEG800@Ab nanoparticles accumulated in the lungs with 85% injected dose (ID) found after 1 hour post-injection of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800, with signification amount of particles still present after 24 hours from administration of nanoparticles in mice (ie, ∼55% ID). Nanoparticles were injected after delivery of clodronate liposomes in order to suppress the RES and increase the blood circulation time of the nanoconjugates.242,243 As the PEG polymer linker length increased, maximum lung uptake decreased and spleen uptake increased (Figure 9A). Identical staining patterns were seen for primary antibody as with the anti-thrombomodulin labeled nanoparticles from immunohistochemistry, demonstrating persistent antigen recognition and binding of the conjugated antibody. Compared to untreated mice (ie, without clodronate liposomes), uptake of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800@mAb201b nanoparticles increased from approximately 75% to approximately 100% ID (ie, at 1 hour post-administration) for mice treated with clodronate liposomes (ie, administered prior to injection of nanoparticles with antibody) (Figure 9B). With treated mice, liver uptake of mAb201b targeted nanoparticles was less than 10% ID with even less uptake seen in the spleen. Furthermore, SPECT/CT images, at 20 or 24 hours post-injection of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800@Ab nanoparticles revealed there was rapid uptake of the nanoconjugates in both the treated and untreated mice (Figure 9C). However, in the treated mice, the accumulation/uptake of the gold coated lanthanide phosphate nanoparticles was higher and more localized to the lungs, compared to accumulation/uptake in the controls.

Gold-coated 177Lu containing lanthanide phosphate nanoparticles for therapy and imaging of pulmonary metastatic disease. Biodistribution of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEGx (x = 800, 3400, or 5000) in mice treated with clodronate liposomes (A) in the lung, liver, spleen and kidney. Biodistribution of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800@mAb201b at 1 hour post-injection in both clodronate and untreated mice (B) (ns = not significant, ***p < 0.001, by 2-way ANOVA). SPECT/CT images (C) show accumulation of [177Lu]Lu0.5Gd0.5(PO4)@Au@PEG800@mAb201b over time in mice treated with clodronate (left) and untreated mice (right) (time points = 10, 20, 30, 40, 50, 60, 70, 1200, 1440 minutes from left to right). Reprinted from APL Bioengineering from Ref. 238 Copyright (2017), with permission from AIP Publishing under Creative Commons Attribution 4.0 International (CC BY 4.0) License (https://creativecommons.org/licenses/by/4.0/) with no changes. Nanoparticle cores were created by mixing LuCl3, Gd(NO3)3 and sodium tripolyphosphate (Na-TPP). Radioactive nanoparticles were prepared by adding 177Lu to a solution of HCl, with the volume of lutetium and gadolinium solutions in the reaction mixture altered. The nanoparticles were then added to a solution containing sodium citrate and NaAuCl4 for creating [177Lu]Lu0.5Gd0.5(PO4)@Au particles. Polyethylene glycol (PEG) linker was added to gold coated nanoparticles for stability and to facilitate attachment of antibody. Antibody conjugation of nanoparticles was achieved by EDC/NHS activation [1-ethyl-3-(3-dimethylaminopropyl)carbodiimide/N-hydroxysuccinimide].
Zhang et al synthesized platelet derived growth factor subunit B (PDGFB) targeting biodegradable FePt alloy assemblies for MRI guided starvation-enhancing chemodynamic therapy (CDT) (ie, using pH-sensitive liposome-coated FePt alloys and glucose oxidase, pLFePt-GOx). 244 GOx triggers simultaneous glucose and oxygen deprivation, which dramatically increases cellular acidity and H2O2 level. Heavy metals can be used to catalyze the conversion of endogenous H2O2 to highly toxic hydroxyl radicals (·OH), whose accumulation then triggers ROS-mediated oxidative stress and cell apoptosis.245–248 On the other hand, platelet derived growth factor subunit B/platelet derived growth factor receptor beta (PDGFB/PDGFR-β) axis is a vital oncogenic signal and potential therapeutic target for different types of carcinomas.249–251 The pLFePt-GOx nanoliposomes possess excellent superparamagnetism, providing significantly enhanced signals for T2-weighted MRI imaging. The intracellular H2O2 production in MDA-MB-231 breast cancer cells treated with pLFePt-GOx particles was more than 6 times greater than H2O2 production from cells with saline only and pLFePt. The cell viability percentages in MDA-MB-231 and 4T1 breast cancer cells were approximately 10% and approximately 5%, respectively after incubation with nanoparticles (ie, Pt concentration of 10 μg/mL). This was more than 2 times lower in viability than from cells treated with pLFePt, due to the ability of glucose oxidase to enhance CDT from oxygen starvation (ie, through glucose consumption and production of gluconic acid and H2O2) and ROS induced apoptosis of cancer cells. In tumor bearing mice at day 20, the tumor volume was more than 6 times lower, compared to tumor volume from mice with PBS only, and more than 4 times lower compared to mice treated with GOx only (ie, using dose of 5 mg/kg, every 2 days for a total of 11 times). Furthermore, the T2-weighted MRI signal intensity from pLFePt in tumors decreased with time after injection of particles, with results indicating enhanced accumulation and contrast for MRI imaging.
Khuyen et al studied the luminescence properties of a nanotheranostics based on a multifunctional Fe3O4/Au/Eu[1-(2-naphthoyl)-3,3,3-trifluoroacetone]3 nanocomposite, combining multiple metal elements. 252 The nanoparticles can be used for hyperthermia and photothermia therapy due to increase in temperature upon laser excitation. Europium is a rare earth element which can be used for providing strong emission of red light, with biocompatibility and luminescent properties for biomedical imaging.253–255 The effect of temperature on the luminescent properties of particles was observed through changes in shape and emission intensity. The luminescence intensity decreased with increase in temperature (ie, with measurements in 20-70 °C range) due to non-radiative transition and luminescent quenching. The results indicate that at physiological temperature (ie, at 37 °C), the nanoparticles can provide both strong luminescence for imaging as well as generate sufficient heat for cancer cell death. After conjugation of biocompatible nanocomposites with IgG antibody (FAEu(NTA)3–IgG), the particles can be used for imaging PC3 prostate cancer cells, showing the ability to observe the cancer cells through luminescence emitted from particles (ie, using UV light excitation).
Clinical use of Metallic Nanoparticles
A variety of gold-based nanoparticles have been used in clinical trials, such as AuroShell and NU-0129. Gold AuroShell particles, based on extensively studied nanoshells,263,264 are PEG-coated silica-gold core–shell particles that can be used for NIR light facilitated thermal ablation. The nanoparticles can absorb deep tissue penetrating NIR light (eg, delivered by optical fiber via bronchoscopy), converting light into heat for destroying primary or metastatic lung tumors and surrounding blood vessels (ClinicalTrials.gov identifier NCT01679470). AuroShell particles can also be used to treat prostate tumors, with results showing median prostate volume (ie, using MRI) decreasing from 49 cm3 at baseline to 42 cm3 at 3 months in patients. 265 This specific therapy (ie, AuroLase therapy, NanoSpectra Biosciences) targets only tumor regions where AuroShell particles have accumulated (ie, after intravenous injection of particles) and treated with focused laser, significantly reducing damage to adjacent healthy tissue. 266 NU-0129 is a formulation containing siRNAs conjugated gold nanoparticles targeting the Bcl-2-like protein 12 (Bcl2L12) sequence for antineoplastic activity (ClinicalTrials.gov identifier NCT03020017). Clinical trials using NU-0129 are ongoing for the treatment of glioblastoma multiforme (GBM) or gliosarcoma by targeting the Bcl2L12 gene. 267 The Bcl2L12 gene prevents apoptosis of tumor cells and by using NU-0129 to target this gene the tumor growth can be stopped. Preliminary results from patients after administration of NU-0129 revealed no significant toxicities from treatment, with high signals from gold (Au) observed in the tumor-associated endothelium, macrophages and tumor cells. 268 The percentages of initial Au level still present in 2 GBM tumors were 41% and 81%, from tumors recurred 159 and 174 days post-NU-109 trial enrollment. Uptake of NU-0129 in glioma cells led to significant reduction in tumor-associated Bcl2L12 protein expression, with elevated caspase-3 activation and increased level of wild-type p53. In addition, various nanomaterial-based sensor arrays have been developed for identifying gastric diseases, including gold-based sensors (eg, ClinicalTrials.gov identifier NCT01420588). Experiments have been carried out to use such nanosensor arrays for non-invasive screening to distinguish gastric cancer and related precancerous lesions. 269 The sensors contain layers of gold nanoparticles with different organic ligands for identifying and characterizing volatile organic compounds (VOCs) present in exhaled breath samples.
IONPs are iron-based nanoparticles that have been used clinically for cancer therapy and imaging. A specific magnetic hyperthermia therapy based on magnetic nanoparticles (ie, NanoTherm, MagForce Nanotechnologies AG) has been approved, which can be used in combination with MRI or CT imaging for monitoring tumor growth/regression. The aminosilane-coated IONPs are activated by an externally applied, rapidly AMF, for the generation of heat from particles (ClinicalTrials.gov Identifier NCT05010759). The temperature increase from the heat generated is significant enough to destroy cancer cells or sensitize them to additional treatment approaches such as radiotherapy and/or chemotherapy. 270 Results from using NanoTherm therapy and radiotherapy revealed sustained necrosis with no evidence of tumor activity. Follow-up imaging after 2 months revealed no signs of tumor progression. In another study, patients with GBM received thermotherapy treatments with the magnetic fluid, followed by radiotherapy with minor or no side effects. 271 The intratumoral temperature from treatment was between approximately 42 and approximately 50 °C, with signs of local tumor control. Similar to nanoparticles used in NanoTherm therapy, Magnablate is a formulation containing iron nanoparticles that has recently been investigated in clinical trials (ClinicalTrials.gov Identifier NCT02033447) with 12 prostate cancer patients. Other metal-based nanoparticles such as hafnium oxide nanoparticles (NBTXR3, Nanobiotix) can be stimulated with external radiation to enhance tumor cell death via electron production (ClinicalTrials.gov Identifier NCT01946867). The nanoparticles are activated by radiotherapy, and when combined improve antitumor efficacy, improving percentage survival more than 2-fold compared to other treatment groups (eg, 5% glucose vehicle, NBTXR3 only). 272 Preliminary clinical results are encouraging as the nanoparticles alone show low toxicity and good antitumor effects for treating sarcoma. 273
Conclusions
The current review highlights some of the recently studied theranostic nanoparticles containing metal elements. Years of development and research has gone into developing these facile and reliable agents for cancer, possessing unique properties (eg, absorption, scattering, attenuation, radioactive, magnetic) for effective treatment and imaging. Having said this, to have even more breakthroughs in the development of these types of nanoparticles and have greater clinical translation, various limitations/drawbacks must be overcome and addressed in future studies. Many metal-based nanoparticles have low biodegradability and further studies are required to examine their toxicity, pharmacokinetic and pharmacodynamic properties. High systemic toxicity due to accumulation of nanoparticles in healthy or non-malignant tissues can lead to undesired side effects (ie, oxidative stress, metabolic and macromolecular changes) that can outweigh the benefits of using metal nanoparticles for cancer theranostics. Even though a large number of nanoparticles are used for in vivo experiments, less than 1% of injected nanoparticles are internalized in tumor cells with most of the rest captured or taken up by macrophages, extracellular matrix and tumor-associated fibroblasts. To enhance the efficacy of metal nanoparticles for cancer theranostics, important parameters of nanoparticles such as type, physicochemical (eg, size, morphology, charge) and functional (ie, coating(s), loading of drugs and/or targeting moieties) characteristics have to be carefully evaluated. For example, to improve tumor accumulation, nanoparticles can be coated with polymers (eg, through PEGylation) to reduce clearance by the immune system while enabling imaging, therapeutic and targeting agents (eg, small molecules, proteins, nucleotides) to be conjugated on the surface of particles. In addition, designing stimuli-responsive nanoparticles (eg, sensitive to pH, temperature and/or radiation) provides delivery specifically to tumors for treatment and imaging. As more of these engineered nanoparticles are being designed and developed there will be greater control in completely treating cancer.
Footnotes
Authors’ Contribution
Donald Fernandes contributed and was involved in all aspects of the work.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
