Abstract
Introduction
Nasopharyngeal carcinoma (NPC) is a common head and neck malignancy with unique epidemiological and pathological features and accounts for 20 to 30 newly diagnosed cases per 100,000 individuals. 1 Although the prevailing standard of care, intensity-modulated radiotherapy (IMRT) has significantly improved the treatment outcomes, especially the localized control, approximately 10%-15% of NPC patients still suffer from local/regional recurrence following the initial treatment.2–4
Recurrent NPC (rNPC) continues to be a challenge for clinical oncologists with no conclusive evidence on the best treatment. 5 Aggressive treatments such as surgery, salvage radiotherapy (RT), palliative chemotherapy, immunotherapy, and so on may aid in improving the long-term survival of certain patients. 3 Though salvage surgical resection can achieve 5-year survival rates ranging from 50% to 77.1%, the confined anatomic space remains the main limitation of surgery.6,7 Chemotherapy-based comprehensive treatment seems to be a better choice for rNPC patients.2,8 Despite the lack of consensus, chemotherapy is often used in combination to reduce the tumor volume and act as a radiosensitizer before or during re-RT. 9 Nevertheless, the efficacy of the combined chemotherapy and RT in rNPC has not yet been fully evaluated.
It remains controversial whether chemotherapy can yield further survival benefits for rNPC patients with re-RT. 10 Several studies focusing on induction chemotherapy (IC) before re-RT have indicated good response rates in salvaging rNPC patients.11,12 The results of some retrospective trials of concurrent radiochemotherapy (RCT) have also been found promising in treating patients with rNPC.13,14 However, there have also been reports which demonstrated that the use of chemotherapy with re-RT may not confer further benefits in rNPC. These observations may be attributed to the treatment toxicity counteracting the benefits of chemotherapy.5,15 As a potential treatment approach, the combination of re-RT and chemotherapy for the treatment of rNPC needs a fine balance between its efficacy and treatment-related toxic reactions. 5 Therefore, it is crucial to establish a prognostic model for rNPC patients in terms of potential benefits from chemotherapy or salvage RT that may help select the optimal treatment mode for realizing personalized treatment.
In this study, we included 406 rNPC patients receiving salvage RT and evaluated the impact of combination chemotherapy. We aimed to identify the potential factors related to the curative effect of salvage RT and established a prognostic model for further predicting the survival and guiding treatment.
Methods
Patients
A total of 406 rNPC patients who were treated with salvage RT from October 2006 to September 2019 in our center were retrospectively considered, among which, 340 patients were eligible for the present study. The criteria for patients’ inclusion were as follows, (1) patients who had previously completed radical therapy but still had local/regional recurrence supported by pathological or radiological evidence; (2) patients who received salvage IMRT; and (3) patients with no distant metastases. Exclusion criteria included patients who received other local therapy like surgery and the cases who did not follow-up. The patients included were treated with salvage RT individually or RT combined with chemotherapy according to the physician's treatment protocol. To protect patient privacy, data are reported in an anonymized format. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16 Ethical approvals for this retrospective study were obtained from Fujian Cancer Hospital (K2022-030-01) and we obtained the subject's written informed consent.
Diagnosis and Treatment
The NPC recurrence in all cases was confirmed by histology or radiology. Each patient received a series of pretreatment evaluations before the recurrence treatment, including a complete history, physical examination, complete hematologic function examination, fiberoptic nasopharyngoscopy, computed tomography (CT) of the chest, ultrasound of the abdomen, magnetic resonance imaging (MRI) of the region of head and neck, and so on. The TNM staging of all NPC patients involved was carried out by at least 2 professional radiation oncologists based on the 8th staging system of UICC/AJCC.
Radiation was delivered by radical IMRT technique with simultaneous integrated boost using 6 MV photons. The details of the IMRT treatment plan are previously described.17,18 All patients were immobilized in the supine position with thermoplastic masks. The CT/MRI images were fused to assist in target volume delineation and determine the organs at risk (OARs). The gross tumor volume (GTV) included all gross diseases such as nasopharyngeal primary tumors (GTVnx) and positive lymph nodes (GTVnd) noted on imaging, clinical examination, and endoscopic findings. The clinical target volume (CTV) was defined as subclinical lesions including GTV and GTV plus a 6 to 10 mm margin. The planning target volume was defined as a safety boundary of GTV/CTV with an additional 3 mm margin to allow positional errors and intrinsic organ movements. The prescribed doses for NPC were delineated according to our center's guidelines. The prescribed radiation doses were 54-70 Gy and 50-64 Gy, given in 30 to 35 fractions of 1.67-2.2 Gy/dose, once per day, 5 times per week for the planning target volumes derived from GTV, and CTV, respectively. The doses of OARs in the initial treatment were set according to the Radiation Therapy Oncology Group (RTOG) 0225 protocol. 19 The re-irradiation doses in OARs were set at less than 70% of the tolerated dose criteria for the OARs in the RTOG 0225 protocol.
Given that the standard regimens of RCT were not available for rNPC patients, chemotherapy was administered at the oncologists’ discretion with the consideration of patients’ clinical characteristics. The platinum-based IC was given for 1-4 cycles and commonly combined with gemcitabine (1.0 g/m2, d1 and d8), paclitaxel (135-175 mg/m2, d1), docetaxel (75 mg/m2, d1) + 5-fluorouracil (600 mg/m2, d1-5), or other regimens every 3 weeks. Ninety-four patients received 1-3 cycles of platinum-based concurrent chemotherapy. The various schedules of chemotherapy are detailed in Supplemental Table S1. The application of adjuvant chemotherapy was based on the physician's judgment and tumor status. Targeted agents (Nimotuzumab, Cetuximab, or Endostar) were used as adjuvants given concurrently with RT during treatment. Targeted anti-angiogenesis therapy has a broad prospect of applying in NPC. 20 Endostar, recombinant human endostatin injection, has been independently developed based on the anti-angiogenesis activity of endostatin by Chinese researchers. 21 A multicenter phase II clinical study showed that Endostar combined with chemotherapy and sequential IMRT had significant survival benefit and tolerance in patients with locally advanced NPC. 22 Tumor response to recurrent treatment was classified according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 assessed by 2 independent radiation oncologists.
Follow-up and Outcomes
All patients were examined every week during their course of treatments. Following the treatment, the patients were followed up with a visit every 3 months for the first 2 years, every 6 months for the next 3 years, and an annual visit after 5 years. In each follow-up visit, complete examinations, including medical history, physical examination, nasopharyngeal endoscopy, chest CT, abdominal ultrasonography, MRI of the head-and-neck region (once every 6 months) or a whole-body bone emission computed tomography (ECT) (once every year), were routinely required. By using clinical records or telephone communication, the survival, tumor status, and treatment adverse effects were documented. The latter was secondary to the course evaluated based on the radiation morbidity scoring scheme of the RTOG at each follow-up. Other toxic side-effects of the treatment were graded per Common Toxicity Criteria for Adverse Events (CTCAE) v5.0 which is not mentioned by RTOG. The final follow-up was conducted on December 19, 2021. In addition, the clinical information of patients, including age at recurrence, gender, recurrent TNM category, clinical stage, disease-free interval (DFI), the application of chemotherapy at the first course, details of radiotherapy or chemotherapy regimens at the recurrent course, and tumor response to recurrent therapy, was collected. DFI was defined as the interval following the completion of the first course of treatment to the date of diagnosis of relapse. 23
The primary outcome was overall survival (OS, defined as the interval time from the completion of re-RT to death or the time of the last follow-up). The secondary endpoints included progression-free survival (PFS, defined as the time following completion of re-RT to the onset of local or regional recurrence, distant metastasis, or death from any cause), loco-regional progression-free survival (LRPFS, defined as the interval from the completion of re-RT to the onset of local or regional progression again), and distant metastasis-free survival (DMFS, defined as the interval from the completion of re-RT to the onset of distant metastasis).
Statistics
The normality of the distribution of continuous variables was assessed using Kolmogorov–Smirnov tests. Two-independent sample t-tests and Mann–Whitney U tests were used to analyze normally and non-normally distributed measures, respectively. Chi-squared tests were performed to evaluate the difference in categorical variables between the RT and the RCT groups. Survival curves were drawn using the Kaplan-Meier method with a log-rank test to compare the differences. Univariate and multivariate Cox regression analyses were performed to explore the independent significance of potential prognostic indicators for survival outcomes using the “survival” and “survminer” R packages. Candidate predictor factors for survival outcomes included gender, age at recurrence, recurrent T (rT) category, recurrent N (rN) category, recurrent stage (rStage), DFI, initial treatment regimen (RT alone or RCT), and the present treatment regimen (salvage RT or RCT).
To develop a model to predict the benefit of rNPC patients from salvage RT or RCT, not only the factors that had prognostic significance were considered, but also those that may have a role in heterogeneous treatment effects. Multivariate Cox analyses were conducted on candidate variables to establish a prognostic risk signature using the Akaike information criterion (AIC) for model fitting, and the variable with the smallest AIC was selected as the final prognostic one. 24 The model's predictive probabilities in predicting OS were evaluated with the calibration curve. R software v3.6.2 (https://www.r-project.org/) and SPSS Statistics v25.0 were utilized for all statistical analysis. The threshold of significance was set at a P-value < .05.
Results
Patient Characteristics
A total of 406 patients with rNPC were treated in our center between October 2006 and September 2019. Among them, 13 cases utilized other RT technology (5 with 2-dimensional conformal RT and 8 with 3-dimensional conformal RT), 7 cases presented recurrence with distant metastases, 6 cases abandoned treatment, 15 cases received surgery, and 25 cases did not follow up after treatment. They were all excluded from this study. The flowchart for the inclusion of patients is shown in Figure 1. The final study sample consisted of 340 patients who received salvage RT (n = 111) or RCT (n = 229) depending on the physician's discretion. The detailed characteristics of patients were well balanced between the 2 groups (P > .05), and are presented in Table 1. The median follow-up time was 56 months (range 2-174 months).

The flowchart of the patient inclusion.
Baseline Characteristics of Patients in the RT Group and RCT Group.

Abbreviations: CCRT, concurrent chemoradiotherapy; CR, complete response; DFI, disease-free interval; IC, induction chemotherapy; PR, partial disease; RCT, radiochemotherapy; RT, radiation therapy; SD, progressive disease.
Initial treatment, received RT alone or RCT therapeutic regimen in first course of treatment.
There was a female predominance of 76.6% (85 patients) in the RT group and 76.0% (174 patients) in the RCT group. A total of 82% of patients received RT combined with chemotherapy at the first course of treatment (88 patients in the RT arm and 191 patients in the RCT arm). Among the 340 patients, 332 patients presented local recurrence according to histopathological investigation or radiological confirmation. Regional recurrence was confirmed in the other 8 patients (1 patient in the RT group and 7 patients in the RCT group) through lymph node biopsy. Further, 85.6% and 72.9% of patients had stage rN0 disease in the RT group and the RCT group, respectively. Most patients were in rStage IV in both the RT group (42.3%) and the RCT group (42.8%).
Treatment Outcomes and Efficacy
The patients completed the planned irradiation with a median dose of 60 Gy in the RT group and 60.8 Gy in the RCT group. Six patients prematurely discontinued RT because of acute side effects and accumulated doses between 25.2 and 49.5 Gy (5 patients in the RT group and 1 patient in the RCT group). The 5-year OS, PFS, LRPFS, and DMFS were 55.8% versus 45.7%, 38.6% versus 41.2%, 63.9% versus 66.7%, and 88.9% versus 87.4%, respectively, with no significant differences between RT group and RCT group (P > .05). The survival curves are displayed in Figure 2. While performing this analysis, a total of 43 (38.7%) of the 111 patients in the RT group and 113 (49.3%) of the 229 patients in the RCT group were deceased at their last follow-up (P = .066). Among the deceased patients, 22 (19.8%) passed away due to local and/or regional failures, 5 (4.5%) due to distant metastasis, and 4 (3.6%) due to locoregional and distant recurrence in the RT group. While in the RCT group, 44 (19.2%) patients were deceased due to local and/or regional failures, 10 (4.4%) due to distant metastasis, and 9 (3.9%) due to locoregional and distant recurrence (P > .05, Table 2).

Kaplan-Meier curves of survival outcome between radiotheraoy (RT) and chemoradiotherapy (RCT) group. (A) Overall survival (OS), (B) progression-free survival (PFS), (C) loco-regional progression-free survival (LRPFS), and (D) distant metastases-free survival (DMFS).
Failure Patterns of RT Group and RCT Group.

Abbreviations: RT, radiotherapy; RCT, radiochemotherapy.
In the RCT group, 196 (85.59%) patients received IC, and 97 (42.36%) patients received concurrent chemotherapy. Given that most patients received platinum combined with gemcitabine (GP, 55.10%) or paclitaxel (TP, 36.73%) for IC regimen, they were included in further analysis. We found there were no significant differences in 5-year OS, PFS, LRPFS, and DMFS between TP group and GP group (Supplemental Figure S1, P > .05). In the RT group, 8 (7.2%) patients achieved complete response, 77 (69.4%) patients had a partial response, 22 (19.8%) patients had progressive disease, and 4 (3.6%) patients could not be evaluated. In the RCT group, 18 (7.9%) patients achieved complete response, 176 (76.9%) had a partial response, 30 (13.1%) had progressive disease, and 5 (2.2%) were not evaluable. The disease control rate was similar for both groups (P = .336).
Toxicities
Most of the patients involved in the study tolerated their established re-RT well, except for 6 patients. During the treatment, the incidence of mild to moderate hematological toxicities including leukopenia, thrombocytopenia, anemia, serum alanine transaminase (ALT) elevation, and serum aspartate transaminase (AST) elevation were significantly higher in the RCT arm than those reported in the RT arm (P < .05, Table 3). Moreover, the observed proportion of serum creatinine was rare in the 2 groups (P = .778). The common acute toxic reactions during RT, including xerostomia and dermatitis were considered tolerable (P > .05), whereas patients would suffer from more oral mucositis (P = .014) and gastrointestinal reactions in the RCT group (P = .007).
Common Treatment-Related Adverse Events.
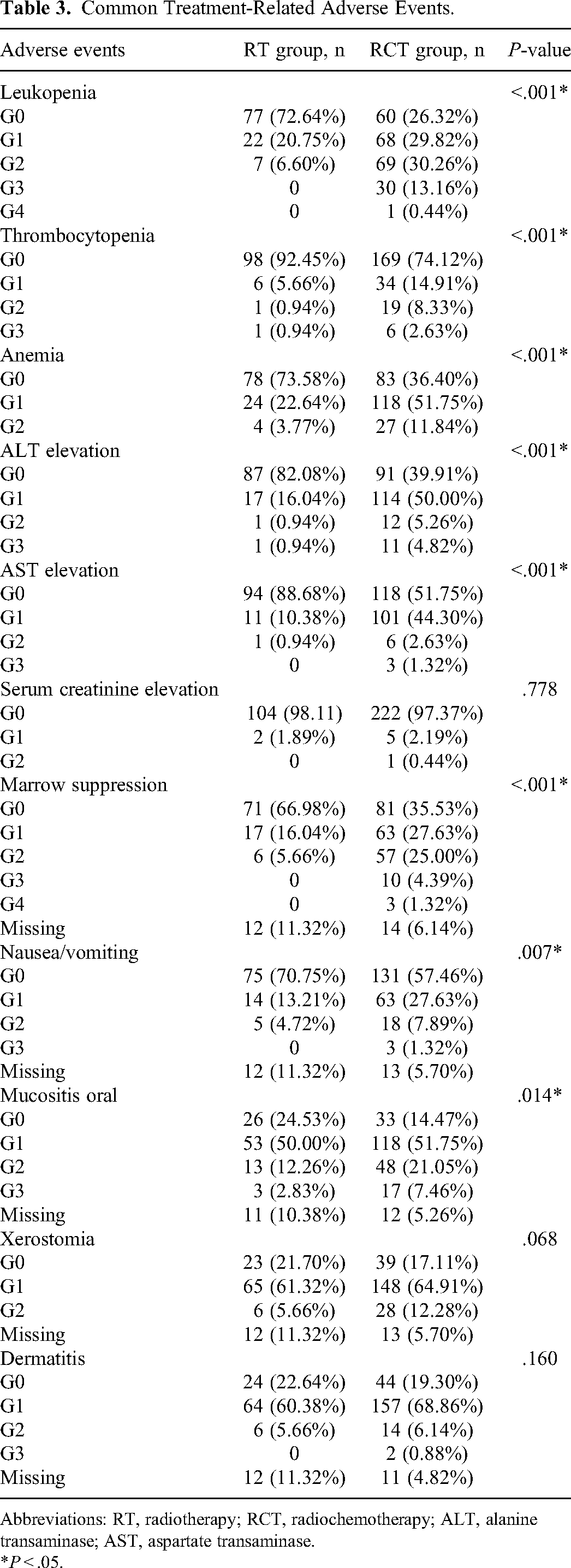
Abbreviations: RT, radiotherapy; RCT, radiochemotherapy; ALT, alanine transaminase; AST, aspartate transaminase.
*P < .05.
Prognostic Factors
The results of univariate Cox regression analysis revealed that higher age at recurrence (HR = 1.028, 95% CI: 1.013-1.042, P < .001), higher rT stage (HR = 1.139, 95% CI: 1.002-1.295, P = .047), and initial treatment of RT alone (HR = 0.641, 95% CI: 0.450-0.915, P = .014) were adverse prognostic factors for OS in rNPC patients. It was validated that age at recurrence was also significantly correlated with PFS (HR = 1.014, 95% CI: 1.001-1.028, P = .033), as shown in Table 4. However, none of the candidate variables were identified to be independent prognostic factors for PFS, LRPFS, and DMFS.
Univariate Analysis of Prognostic Factors in rNPC.

Abbreviations: DFI, disease-free interval; DMFS, distant metastases-free survival; HR, hazard ratio; LRPFS, loco-regional progression-free survival; OS, overall survival; PFS, progression-free survival; RT, radiotherapy; RCT, radiochemotherapy.
Initial treatment, received RT alone or RCT therapeutic regimen in first course of treatment.
Present treatment, received salvage RT or RCT treatment in recurrent course.
*P < .05.
The multivariate model enrolled 2 prognosis factors, including age at recurrence (HR = 1.028, 95% CI: 1.013-1.043, P < .001) and rT category (HR = 1.143, 95% CI: 1.005-1.299, P = .042) with the smallest AIC to further establish a model predictive of whether rNPC patients would benefit from salvage RT combined with chemotherapy. The patients were divided into 2 subgroups based on median risk scores calculated by the model. The Kaplan-Meier curves presented remarkably worse OS in the high-risk group than in low-risk one (HR = 1.87, 95% CI: 1.36-2.57, P < .001, Figure 3A). The median survival time was 2.5 years in the high-risk group, while it was not reached for low-risk one. It was revealed that salvage RT alone may be a superior treatment option than RCT in the low-risk rNPC (patients with lower age at recurrence and early rT category) (RCT group vs RT group, HR = 1.89, 95% CI: 1.11-3.20, P = .038, Figure 3B). A calibration plot was determined to validate the model performance, which indicated good concordance as shown in Figure 3C. What's more, the results of multivariate Cox regression analysis also showed the role of age at recurrence and rT category as independent predictors of OS.

The stratification and prediction of overall survival (OS) based on the constructed prognostic model. (A) Kaplan-Meier curves of OS between low- and high-risk group. (B) Kaplan-Meier curves between radiotheraoy (RT) and radiochemotherapy (RCT) group with low risk score. (C) The calibration plot for validation of the model.
Discussion
Herein, the long-term treatment outcomes and toxicities of re-RT combined with chemotherapy in rNPC patients were estimated. A total of 340 patients were included for analysis. It was observed that chemotherapy combined with RT offered similar local control rates and survival outcomes compared with those with RT alone. Moreover, it presented more incidence of treatment toxicity. It was observed that the age at recurrence and rT category are independent predictors for OS. A predictive model was established for rNPC patients’ stratification that showed that patients with high-risk scores presented worse OS. When compared with treatment outcomes for RT combined with chemotherapy during the recurrent stage, salvage RT alone may benefit for low-risk patients with lower recurrence age, or those who are at an early rT category.
IMRT has been the most important salvaging treatment for rNPC, which provides long-term tumor control as reported in our previous study. 17 NPC is sensitive to chemotherapy and a combined regimen of cisplatin and gemcitabine is the standard first-line regimen of rNPC/metastatic NPC.8,25 However, whether the addition of chemotherapy can bring survival benefits for rNPC patients with re-RT remains controversial. Until now, only a few studies have evaluated the long-term survival outcome of combined chemotherapy with re-RT in rNPC.14,15 Several trials focusing on IC before re-RT have indicated good response rates in treating rNPC patients, especially the general approach of using cisplatin and gemcitabine.12,26 A prospective study has shown 70% partial response when 3-cycle IC with cisplatin and gemcitabine were administered, with curative effects superior to those without IC. 11 The results of a phase II trial showed significant improvements in OS rates when weekly cisplatin was added to IMRT as compared to that of IMRT alone. Notably, concomitant chemoradiotherapy can improve survival outcomes, especially in the rNPC patients with advanced stage category (III-IV) and rT category (rT3-4), with DFI > 30 months, and larger tumor volume (> 26 cm3). 27 A previous study on 184 rNPC patients treated with re-RT by IMRT showed that a combination of chemotherapy significantly improved only DMFS, but not OS. 28 Contrary to the above views, a meta-analysis had shown no significant impact of chemotherapy on tumor control and prognosis, which may be attributed to the treatment toxicity counteracting the benefits of chemotherapy. 15 The authors observed a high incidence of grade 5 treatment toxic reactions, reaching up to 33%. 15 In addition, it has been reported that the tolerance of patients to re-RT would reduce when combined with chemotherapy. 29 Consistent with the results from previous studies, our results suggest that chemotherapy may be unwarranted for some rNPC patients. However, the curative effect of chemotherapy on rNPC with salvage RT cannot be simply excluded. A prospective study should be conducted in the future to verify the findings.
It is vital to explore the optimal curative strategies focusing on improving therapeutic effects and minimizing treatment toxicities in rNPC. Reirradiation with conventional RT techniques is associated with a considerable risk of severe complications. However, IMRT is an advanced technique that enables the delivery of high-dose radiation to the target volumes, thereby potentially enhancing the therapeutic ratio with tolerable toxicity. 4 Unfortunately, radiation toxicity is still a serious problem even in patients in the rT1-2 stage. 30 Previous studies reported that nearly 55% of rNPC patients exhibited late radiation injuries more severe than grade 3 injuries, whereas 30% exhibited grade 5 injuries including nasopharyngeal necrosis, necrosis of the internal carotid artery, or other vessels, temporal lobe necrosis, and so on.15,31 Some of these severe toxicities may be related to the large fraction size used in previously heavily irradiated normal tissues. In the current study, all patients were initially treated with RT including conventional RT and IMRT. To minimize the risk of excessive long-term adverse effects, reirradiation volume was limited including the recurrent foci with an expanded margin, and lymphatic drainage sites were not covered in the CTV. Further, a limited number of studies have explored the possibility of chemotherapy to increase toxicity. On the one hand, the use of chemotherapeutics such as cisplatin and docetaxel in RCT was regarded as a radiosensitizer for head and neck cancers, while on the other, it may increase the occurrence of toxicity in re-RT cases.9,29,32 Zong et al reported that the rT category and chemotherapy are associated with the incidence of severe late radiation injury. 31 It was demonstrated that rNPC patients receiving RCT suffered more late toxicity than those treated with RT alone. 33 Previous studies have indicated that chemotherapy was the major contributor to severe grade 3 acute hematological toxicities. 31 In addition, some studies demonstrated that the use of chemotherapy at any phase can lead to significant toxicities. 31 Our results show that radiation damage and acute hematological toxicities were more likely to be found in the RCT group. Although the late radiation injuries noted in the RCT group were not significantly different as compared with the RT group, 34 thus, the addition of chemotherapy to re-RT in rNPC patients needs careful consideration given the apparent increase in toxicities.
It is necessary to recognize the rNPC candidates who may benefit from re-treatment. Several models have been constructed for the treatment stratification of patients.22,35–37 A validated salvage radiotherapy outcome score was proposed that identified a low-score subgroup of the female sex, who had previous RT-induced grade 3 and higher-grade late toxicities, and suboptimal performance status. They had a significantly better survival benefit after receiving palliative CT alone than receiving salvage RT. 35 Li et al presented a prognostic index for OS including rT category, age, and late toxicities from prior RT, re-IMRT dose, and recurrence tumor volume. 23 A total of 531 patients were included to conduct and validate a prognostic model based on a recursive partitioning analysis that divided patients into 3 groups. 36 Salvage RT could benefit rNPC patients in the low- and intermediate-risk groups, while not in the high-risk group. In this study, a predictive model for screening patients who could benefit from salvage RT was developed. Accordingly, salvage RT alone induced a significant survival benefit in patients with lower age at recurrence and early rT category. Both age at recurrence and rT category also had significant prognostic values. The model established by Li et al also confirmed that the important factor, age at recurrence, may affect the prognosis of rNPC. 23 This observation is consistent with the current clinical practice too. Older patients may have lower tolerance owing to multiple comorbidities, and poor organ function or performance status. 38 Further, the late disease stage indicates a higher incidence of toxicity.
The present study has its limitations. First, the selection bias was unavoidable because of the retrospective analysis. Since the cohort in this study included patients from 2006 to 2019, limitations of detection methods are inevitable leading to missing plasma Epstein-Barr virus (EBV) DNA data, which is an important indicator associated with clinical outcomes during the treatment.33,39 In addition, due to the retrospective nature, this study lacked data on late radiation-induced adverse events in different treatment methods. Moreover, the relevant analyses of re-RT dose were not involved in the current study, given the complexity of radiobiology. Furthermore, although we collected clinical features of each patient involved and established a predictive model, an external cohort needs to be used to validate the clinical utility of the findings.
Conclusion
In conclusion, chemotherapy did not appear to improve the effectiveness of salvage RT in rNPC patients. A predictive model to identify patients who can benefit from salvage RT to personalize treatment recommendations is proposed. Therefore, further treatment strategies are needed for a reasonable balance between tumor control and toxicity in rNPC patients and to improve survival outcomes.
Supplemental Material
sj-doc-1-tct-10.1177_15330338231155721 - Supplemental material for Impact of Radiotherapy Combined With Chemotherapy on Long-Term Outcomes of Patients With Recurrent Nasopharyngeal Carcinoma
Supplemental material, sj-doc-1-tct-10.1177_15330338231155721 for Impact of Radiotherapy Combined With Chemotherapy on Long-Term Outcomes of Patients With Recurrent Nasopharyngeal Carcinoma by Ying Li, Wei Liu, Yun Xu, Lan-Yan Guo and You-Liang Weng, Zong-Wei Huang, Xiao-Chuan Chen, Ting Lin, Jun Lu, Su-Fang Qiu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
We thank all investigators and patients who participated in the study. We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Author Contributions
LY and LW: manuscript preparation; XY and WY: quality control of data; GL and HZ: data collection; CX and LT: statistical analysis; LJ and QS: study design and manuscript review. All authors read and approved the final manuscript.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fujian Clinical Research Center for Radiation and Therapy of Digestive, Respiratory and Genitourinary Malignancies, Major Research Projects for Young and Middle-aged Health Researchers of Fujian Province, China (grant number 2021ZQNZD010), National Natural Science Foundation of China (grant number 82072986), National Clinical Key Specialty Construction Program (grant number 2021), Science and Technology Program of Fujian Province, China (grant number 2018Y2003), Wu Jieping Medical Foundation (grant number 320.6750.2021-01-27), Joint Funds for the innovation of science and Technology, Fujian Province (grant number 2018Y9105), Fujian Provincial Clinical Research Center for Cancer Radiotherapy and Immunotherapy (grant number 2020Y2012).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
