Abstract
Introduction
44% of the new gastric cancer cases are diagnosed in China. Gastric cancer has become China's malignant tumor with an incidence rate of second and a mortality rate of third.1, 2 The early diagnosis of gastric cancer is relatively difficult, and thus most of patients are diagnosed in advanced stage. At present, the standard first-line chemotherapy regimen for advanced or metastatic gastric cancer is fluorouracil and platinum, and the second-line treatment usually chooses the drugs which were not used in the first-line treatment, such as taxanes and irinotecan.3–5 However, patients have limited benefits from the use of such traditional chemotherapy drugs, and the efficacy of chemotherapy has reached a bottleneck.
As a highly heterogeneous tumor, understanding of the molecular mechanism of gastric cancer tumorigenesis and progression is deepening in recent years. The new treatment model continues to mature,,including targeted therapy and immunotherapy,6–8 based on human epidermal growth factor receptor-2 (HER2) and vascular endothelial growth factor (VEGF) pathway.9, 10 Apatinib has been approved in the third-line treatment of advanced or metastatic gastric cancer. In recent years, a number of clinical studies have confirmed the efficacy of apatinib in advanced or metastatic gastric cancer.11–13
Exploring the efficacy of anti-angiogenic tyrosine kinase inhibitor (TKI) in combination with other targeted drugs, chemotherapy, or immune checkpoint inhibitors (ICIs), and at the same time moving the treatment time forward to the second or even the first line, is a new research direction. This study aimed to evaluate the real-world clinical data of anti-angiogenic TKI plus chemotherapy versus chemotherapy in advanced or metastatic gastric cancer.
Materials and Methods
Patient Population
This was a retrospective and propensity score-matched cohort study. From November 2017 to April 2020, patients with advanced or metastatic gastric cancer who have failed prior treatment were collected in this study, the patients received apatinib combined with irinotecan or irinotecan monotherapy as a second-line or above therapy regimen. We have de-identified all patient details. In this study, the patients who received apatinib combined with irinotecan were consecutively selected, propensity score matching was used to select patients who received irinotecan therapy.
Treatment Regimen
The patients received apatinib combined with irinotecan regimen or irinotecan monotherapy. In the combination therapy group, the dose of apatinib was 250 mg daily, and irinotecan was given at the dose of 125 mg/m2 on d1 and d8 every three weeks until disease progression, unacceptable toxicity, or death. If grade III or above adverse events associated with apatinib occurred, the dose of apatinib could be reduced. After six cycles of treatment, if the clinical evaluation is effective (CR + PR + SD), apatinib monotherapy is used for maintenance treatment, without irinotecan therapy. In the irinotecan monotherapy group, irinotecan was administered intravenously at the same dose.
Efficacy and Safety
The response evaluation criteria in solid tumors (RECIST version 1.1) was used to evaluate the clinical efficacy based on imaging examination, which was performed every two cycles of therapy, including complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). The proportion of CR + PR was objective response rate (ORR), and the proportion of CR + PR and SD was disease control rate (DCR). Common Terminology Criteria for Adverse Events (version 4.0) was used to assess the drug toxicity.
Statistical Analysis
The reporting of this study conforms to STROBE guidelines. 14 Propensity score matching was used to select patients to achieve a balance of baseline clinicopathological characteristics between the two treatment groups. Included variables were age, gender, ECOG PS, primary tumor site, metastatic site, number of metastatic sites, and treatment line. Progression-free survival (PFS) was defined as starting apatinib with irinotecan or irinotecan treatment to disease progression or death. Overall survival (OS) was defined as starting apatinib with irinotecan or irinotecan to patient death or last follow-up. PFS and OS were plotted using Kaplan–Meier method, and the follow-up period is up to June 1, 2021. Pearson's chi squared test or Fisher's exact test was used to compare differences between groups. Receiver operating characteristics (ROC) analysis was applied to determine the cut-off value of neutrophil-to-lymphocyte ratio (NLR) and mean platelet volume (MPV). Univariate analysis was performed to seek predictive factors for PFS and OS. SPSS 22.0 software (SPSS Inc., IL, US) software was used and P < .05 was considered to be significant.
Results
Patient
A total of 55 patients were included in this study. 26 patients received apatinib combined with irinotecan and 29 patients received irinotecan. Table 1 summarized the patient and treatment characteristics. In the combination therapy group, the median age was 53 years (range from 26–70), with 21 male and 5 female patients. 19 and 7 patients were diagnosed as advanced gastric cancer and gastroesophageal junction (GEJ) adenocarcinoma, respectively. Lymph node (73.1%), liver (50.0%), lung (19.2%), and peritoneum (19.2%) were common metastatic sites. In this study, 16 patients (61.5%) and 10 patients (38.5%) received apatinib combined with irinotecan regimen as second line or third- to forth-line treatment, respectively. Of the cases, 92.3% were identified as HER-2 negative, only two cases were HER-2 positive.
Patient and Treatment Characteristics.

Abbreviations: ECOG, Eastern Cooperative Oncology Group performance status; GEJ, Gastroesophageal Junction Tumors.
In the monotherapy group, the median age was 48 years (range from 25–68), with 19 male and 10 female patients. 17 and the other 12 patients had advanced gastric cancer and GEJ adenocarcinoma, respectively. Of the cases, 82.8% were identified as HER-2 negative, and the other five cases were HER-2 positive. Lymph node (58.6%), liver (51.7%), peritoneum (27.6%), and lung (20.7%) were common metastatic sites. 21 patients (72.4%) and 8 patients (27.6%) received apatinib combined with irinotecan regimen as second line or third to forth-line treatment, respectively. There were no statistically significant differences in baseline index between the two groups.
In this study, all the patients received the guideline-recommended first-line treatment regimen, including chemotherapy for HER2-negative patients, which was based on fluorouracil in combination with platinum and/or taxane, and chemotherapy plus trastuzumab for HER2-positive patients.
Efficacy
No patients gained CR in this study. In the combination therapy group, 7, 14, and 5 patients were evaluated as PR, SD, and PD. The overall ORR and DCR were 26.9% (7/26) and 80.8% (21/26), respectively (Table 2). In the irinotecan monotherapy group, 5, 11, and 13 patients were evaluated as PR, SD, and PD. The overall ORR and DCR were 17.2% (5/29) and 55.2% (16/29), respectively. There was no significantly differences in ORR between the two groups (26.9% vs 17.2%, P = .385). However, the DCR in the apatinib combined with irinotecan group was higher than in irinotecan population (80.8% vs 55.2%, P = .043). In the combination therapy group, median PFS and OS were 4.2 months (95% CI = 2.3–6.1) and 8.0 (95% CI = 1.8–14.2) months, respectively (Figure 1). In two patients, the OS reached more than 2 years, of which one patient was 27 months and the other 34 months. The 1-year survival rate was 46.2%. In the monotherapy group, median PFS and OS were 3.3 months (95% CI = 2.1–4.5) and 5.9 (95% CI = 4.7–7.1) months, respectively. The one patient with longest treatment period with irinotecan monotherapy was 6.8 months. Patients who received apatinib combined with irinotecan obtained better PFS and OS than irinotecan monotherapy. Subgroups analysis showed that PFS and OS were generally consistent across sex, age, number of metastatic sites, and primary tumor site (Figure 2). However, for patients with ECOG PS 2, apatinib combined with irinotecan did not show a survival advantage in PFS compared with irinotecan monotherapy (HR = 1.24, 95% CI = 0.43–3.60, P = .696). Simultaneously, patients with ECOG PS 2 who received irinotecan monotherapy obtained better OS (HR = 4.19, 95% CI = 1.19–14.27, P = .026).
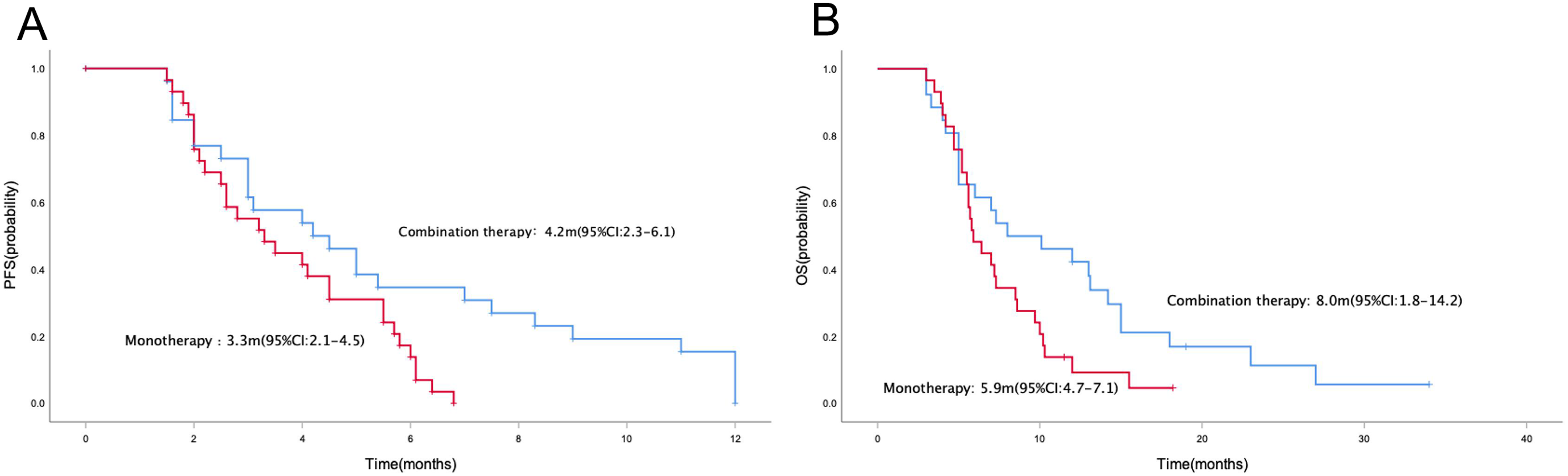
Kaplan–Meier curve of PFS (A) and OS (B) in the whole population.

Subgroup analysis of PFS (A) and OS (B) according to clinicopathologic factors, including sex, age, ECOG PS, number of metastatic sites, and primary tumor site.
Efficacy of Apatinib Combined with Irinotecan in Patients with Advanced or Metastatic Gastric Cancer.

Bold represents P < .05. Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, overall response rate; DCR, disease control rate; PFS, progression free survival; OS, overall survival.
Safety
The toxicity was well tolerated (Table 3). The incidence of Grade 3-4 AEs was 23.1% and 20.7% in the apatinib combined with irinotecan group and irinotecan group, respectively. Neither treatment-related death or no unexpected side effects occurred. The most common treatment-related AEs were hematological toxicity, including decreased neutrophil count, anemia, decreased white blood count, decreased platelet. The incidence of hematological toxicity between the two groups was similar. The adverse events that were attributed to apatinib were secondary hypertension (in seven patients, 26.9%), hand-foot syndrome (5,19.2%) and proteinuria (5, 19.2%). Other non-hematological toxicity was also similar between the two groups, including fatigue, nausea or vomiting and diarrhea.
Treatment-Related AEs (TRAEs).

Abbreviation: AE, adverse event.
Exploratory Analysis of Predictive Factors
In the combination therapy group, an univariate analysis of predictive factor was carried out, including gender, ECOG, age, number of metastatic sites, lymph node metastasis, peritoneal metastasis, visceral metastases, primary tumor site, treatment line, prior surgery, MPV, NLR, Grade 3 or 4 AEs, secondary hypertension, and hand-foot syndrome. However, none of these clinicopathologic factors were associated with PFS (Table 4). Univariate analysis for OS demonstrated that only secondary hypertension was considered to be a favorable factor (P = .040) for OS (Figure 3).

Kaplan–Meier curve of PFS (A) and OS (B) in apatinib combined with irinotecan therapy population with or without secondary hypertension.
Exploratory Univariate Analysis of Factors to Predict PFS and OS of Apatinib Combined with Irinotecan Treatment.

Bold represents P < .05. Abbreviations: ECOG, Eastern Cooperative Oncology Group performance status; GEJ, Gastroesophageal Junction Tumors; MPV, Mean Platelet Volume; NLR, Neutrophil-to-Lymphocyte Ratio; AE, adverse event; PFS, progression free survival.
Discussion
Although a variety of chemotherapy drugs are used, there is still a lack of effective treatment options after first-line treatment failure.15, 16 WJOG 4007 trial was a randomized, phase III clinical study, which compared irinotecan and paclitaxel in advanced gastric cancer, no statistically significant difference was observed in PFS and OS between paclitaxel and irinotecan. 4 At present, taxanes or irinotecan monotherapy was the standard second-line treatment regimen for metastatic gastric cancer. However, the median PFS was only 3.6 and 2.3 months for paclitaxel and irinotecan in WJOG 4007 trial. And therefore, it is necessary to search for new treatment strategies for metastatic gastric cancer.
There are sufficient evidence-based medical evidences on targeted therapy for gastric cancer, but currently, only the first-line of anti-HER-2 therapy and the second-line or third-line of anti-angiogenic therapy have entered clinical practice. Recently, apatinib has been proved to be effective in a variety of solid tumors.17–20 Preclinical studies have shown that apatinib exerts antitumor effects in gastric cancer through a variety of mechanisms.21–23 A previous study that enrolled 273 patients with gastric cancer who progressed after chemotherapy, demonstrated that apatinib treatment significantly improved median PFS and DCR compared with the placebo group. 24 Apatinib has been approved as a third line treatment option for advanced or metastatic gastric cancer or GEJ in China.
The best administration mode of apatinib is still being explored. In addition to the single-drug treatment scheme, apatinib combined chemotherapy is also the preferred treatment strategy. Apatinib has a synergistic effect with chemotherapy and can reverse resistance to chemotherapy.23, 25, 26 Previous researches revealed that apatinib combined with chemotherapy is more effective compared with apatinib or chemotherapy alone.13, 27, 28 The combined chemotherapy regimen includes oxaliplatin, FOLFOX, FBC, S-1, taxanes, FOLFIRI, etc. There is no evidence for using apatinib alone in the second-line treatment. In this study, we also used the programme of apatinib combined with chemotherapy. Nevertheless, the clinical efficacy of apatinib combined with irinotecan, which was the standard second-line regimen, has not been reported yet.
Our study evaluated the efficacy and drug toxicity of apatinib combined with irinotecan versus irinotecan. The main scheme of the second-line treatment for advanced or metastatic gastric cancer is single drug chemotherapy with paclitaxel and irinotecan or a combination of two drug chemotherapy schemes according to clinical guidelines. Therefore, in this study, we choose chemotherapy alone as standard control group. In the combination therapy group, the overall ORR and DCR were 26.9% (7/26) and 80.8% (21/26). No statistical difference in ORR was found, but the DCR in the combination group was significantly higher than in monotherapy population. The median PFS and OS reached 4.2 and 8.0 months, respectively, compared with irinotecan monotherapy, apatinib combined with irinotecan obtained better PFS and OS. Although the PFS and OS in our study is similar with previous second-line trials, the difference is that 38.5% patients received apatinib plus irinotecan as third-fourth-line therapy in this study. So apatinib plus irinotecan therapy still demonstrated well efficacy as late-line treatment regimen.
However, except for ECOG PS 2, PFS and OS were generally consistent across subgroups by sex, age, number of metastatic sites, and primary tumor site. Our present study demonstrated that for patients with ECOG PS 2, apatinib combined with irinotecan did not show a survival advantage in PFS compared with irinotecan monotherapy, and simultaneously, patients with ECOG PS 2 who received irinotecan monotherapy obtained better OS. This may be related to the increased incidence of drug-related toxicity caused by combination therapy. And thus, for patients with advanced or metastatic gastric cancer, the choice of treatment should fully take into account the patient's performance status and accompanying diseases. For patients with better physical condition, the combination regimen is better than monotherapy. However, for patients with poor physical status, monotherapy maybe preferred option.
To our knowledge, this is the first real-word study evaluating the efficacy of apatinib combined with chemotherapy in advanced or metastatic gastric cancer. A previous real-world study evaluated the clinical efficacy of apatinib combined with chemotherapy in advanced or metastatic gastric cancer. 13 Apatinib showed a favorable efficacy, the DCR was 82.72% and 81.82% in second-line and third-line or above treatment, respectively. For survival outcomes, the mPFS and OS was 5.0 and 7.5 months in second-line and above treatment population. The effectiveness of apatinib combined with irinotecan in our present study is consistent with this study. But the difference is that a significant proportion of patients in this study received combined chemotherapy regimen, including XELOX, EOX, and DCF. Apatinib combined with irinotecan single-agent chemotherapy has achieved similar clinical effects in our present study.
Adverse events (AEs) of irinotecan and anti-angiogenesis therapy observed in this study were consistent with the known safety data in previous study.29, 30 Overall, the toxicity was well tolerated, and most of the AEs were in grades 1–2. Compared with irinotecan monotherapy, apatinib plus irinotecan increased the incidence of adverse events that were attributed to apatinib, including secondary hypertension, proteinuria, and hand-foot syndrome. The inhibition of VEGFR in vascular endothelial cells by apatinib will reduce the production of nitric oxide and prostacyclin, leading to an increase in blood pressure. The occurrence of hand foot syndrome is related to a variety of mechanisms and shows a dose dependence. The proteinuria is related to the inhibition of VEGF in glomerular mesangial cells by apatinib. Most of apatinib related AEs were in grades 1–2, and only one patient developed ≥ Grade3 of hand-foot syndrome. The incidence of hematological toxicity and other non-hematological toxicity was similar between the two groups. Because the number of cases in the dose reduction group is small, we did not carry out statistical analysis between the standard treatment group and dose reduction group. We will continue to explore the impact of dose reduction on therapeutic efficacy and patient prognosis. In previous clinical trials, the adverse events included hyperbilirubinemia and bleeding. However, patients in these studies received 850 mg of apatinib, or used a combination chemotherapy regimen. In our present study, the patients received apatinib plus single drug chemotherapy, and we did not observe significant hyperbilirubinemia and bleeding.
Anti-angiogenesis therapy including apatinib have great individual differences in clinical efficacy due to tumor heterogeneity. An exploratory univariate analysis of was carried out in our study to seek possible predictors in the combination therapy group. Unfortunately, we did not find an effective predictor for PFS. Multiple previous studies had shown that the presence of apatinib-related AEs including hand-foot syndrome, hypertension or proteinuria were biomarkers of antitumor efficacy.13, 31 Consistently, the results of our study showed that secondary hypertension was considered to be a potential predictive factor for longer OS. Generally speaking, adverse events caused by anti-angiogenic TKI represent a reaction of the body to drug therapy, which may indicate a better clinical therapeutic efficacy. Therefore, these adverse events may be used as a simple and intuitive biomarker for anti-angiogenic TKI therapy. However, the value of these biomarkers still needs further prospective clinical researches to verify.
Our study had several limitations, including an observational study obtained from a single center, and not very large patient cases. Thus, we will continue to accumulate clinical data and design prospective clinical studies to confirm the value of apatinib combined with irinotecan for advanced or metastatic gastric cancer as second-line or above therapy.
Conclusion
Anti-angiogenic TKI plus chemotherapy showed better PFS, OS and DCR compared with chemotherapy alone for advanced or metastatic gastric cancer as second-line or above therapy regimen, with a tolerable and manageable safety.
Footnotes
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
This study was carried out in accordance with the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the ethics committee of the Affiliated Cancer Hospital of Zhengzhou University (KY-0192). Written informed consent was obtained from all patients for the use of the medical records for research purposes.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 1000 Talents Program of Central plains, Young and Middle-aged Health and Technology Innovation Leading Talent Project of Henan Province, Sate Key Laboratory of Esophageal Cancer Prevention & Treatment, Medical Science and Technique Foundation of Henan Province (Medical Science and Technique Foundation of Henan Province, (grant number 204200510023, YXKC2020008, Z2020000X, 212102310623, SB201901101).
