Abstract
Background:
Although the landmark INT-0116 trial and National Comprehensive Cancer Network (NCCN) guidelines recommended pT3-4Nx gastric cancer (GC) patients to receive chemoradiotherapy, the role of radiotherapy has not been distinguished from chemoradiotherapy.
Methods:
GC with behavior of metastasis-indolent in lymph node (MILN) being confirmed with more than 15 examined LNs after gastrectomy were identified using the Surveillance, Epidemiology and End Result (SEER) database. The cancer-specific survival (CSS) of subgroups for radiotherapy, chemotherapy, chemoradiotherapy and non-adjuvant-treatment were compared. Propensity score matching (PSM) was performed between radiotherapy and non-radiotherapy subgroups to further distinguish the role of radiotherapy from chemoradiotherapy. Cox regression was performed to identify whether radiotherapy or chemotherapy could independently improve prognosis.
Results:
We identified 690 MILN GC patients in SEER database. 5-year CSS was 71.9% in radiotherapy subgroup and 75.1% in non-radiotherapy subgroup(HR = 1.013, 95% CI = 0.714-1.438, p = 0.940), 75.6% in chemotherapy subgroup and 68.5% in non-chemotherapy subgroup(HR = 0.616, 95% CI = 0.430-0.884, p = 0.008), 52.5% in radiotherapy-alone subgroup and 71.9% in non-adjuvant treatment group (HR = 1.604, 95% CI = 0.575-4.471, p = 0.360), 72.9% in chemoradiotherapy subgroup and 79.5% in chemotherapy-alone subgroup (HR = 1.365, 95% CI = 0.859-2.172, p = 0.185), respectively. Further, PSM markedly improved balance of variables between radiotherapy subgroup and non-radiotherapy subgroup. After PSM, the role of the variables of radiotherapy and chemotherapy in contributing to improving CSS are consistent with that before PSM. Cox regression showed chemotherapy, tumor size, tumor invasiveness and Lauren classification were independent prognostic factors, but not including radiotherapy.
Conclusions:
Chemoradiotherapy confers superior prognosis to MILN GC patients compared with surgery alone might only be attributed to chemotherapy rather than radiotherapy.
Introduction
Gastric cancer (GC) is a global health problem, with more than 1 million people newly diagnosed with GC worldwide each year. 1,2 Currently, surgery remains the cornerstone of treatment for local advanced GC (LAGC) 3 –5 , and systematic chemotherapy has been demonstrated that it conferred superior prognosis after gastrectomy. 6 -9 However, the role of radiotherapy was only investigated in combining with chemotherapy, as a part of chemoradiotherapy in Western practices.
Chemoradiotherapy has become the standard treatment and been successfully translated to the community in Western. 10,11 Similarly, the National Comprehensive Cancer Network (NCCN) guidelines recommended that both pT3-4Nx and pTxN+ GC patients who undergo R0 resection should receive chemoradiotherapy. 12 As early as 2001, the landmark INT-0116 trial in the United States established the role of adjuvant chemoradiotherapy in the multidisciplinary approach to the management of LAGC. 10,13 Then, the INT0116 trial laid the foundation for the popularity of chemoradiotherapy for curatively resected GC with primaries T3 or greater and/or positive nodes in North America. Additionally, Kozak et al. found that the release of the INT 0116 trial likely reflected the increased use of chemoradiotherapy, which has been associated with improved survival in GC patients, suggesting that the improved outcome seen in this trial has been successfully translated to the community. 11 Consistently, some retrospective studies with large sample sizes have also shown the survival advantage of chemoradiotherapy. 14 -16 However, these studies evaluated chemoradiotherapy versus surgery alone and thus could not distinguish the effect of chemotherapy and radiotherapy from chemoradiotherapy in prolonging survival in Western practices.
On the basis of INT0116 study, the phase 3 Adjuvant chemoradiation Therapy in Stomach cancer (ARTIST) trial further showed that radiotherapy in addition to capecitabine/cisplatin chemotherapy after radical resection did not improve the 5-year survival rate (73% vs. 75%). Nevertheless, in the subgroup of patients with pathological lymph node(LN) metastasis, who received chemoradiotherapy experienced superior disease-free survival(DFS) to those who received chemotherapy alone .Then, the subsequent trial, ARTIST2, was conducted to investigate whether radiotherapy is beneficial in LN-positive GC. However, the interim analysis of the recent ongoing ARTIST 2 study showed that radiotherapy did not provide further benefit in patients with stage II-III lymph node-positive GC after D2 radical resection. 17 Thus, currently, radiotherapy is not considered a postoperative adjuvant therapy for GC performed with D2 lymph node dissection in eastern Asia, especially in LN-negative patients. However, the effect of radiotherapy differed between Eastern and Western trials might be attributed to the discrepancy of surgical quality assurance since 54% of patients in the INT0116 trial had D1 lymphadenectomy or less 10 while most patients in ARTIST underwent D2 lymphadenectomy. 18,19 Notable, chemotherapy has been demonstrated to be effective in improving prognosis both in Eastern and Western practices. 6 -9 Taking into account the difference of the setting of chemoradiotherapy and chemotherapy, we hypothesized that chemoradiotherapy conferred a better prognosis compared with surgery alone in Western practices in previous studies 10,11 might mainly be attributed to chemotherapy alone rather than radiotherapy.
To explore this hypothesis, we assessed it in selected subgroups GC with the behavior of metastasis-indolent in lymph node (MILN: T3-4N0M0). Therefore, we investigated whether radiotherapy contributes to improving the prognosis of MILN GC patients who underwent gastrectomy in Western practices.
Methods
Data Source and Data Selection
This retrospective cohort study assessed the role of radiotherapy in MILN GC (T3-4N0M0) patients whose data were abstracted from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) population-based data registry. 20 The SEER database is the most comprehensive cancer registry in the United States and provides information from cancer registries that comprise approximately 28% of the country’s population. SEERStat software version 8.3.6 was published by SEER and was used to identify eligible patients in this study. The software was obtained from the official network (https://seer.cancer.gov/).
Patient Selection
Within the SEER database, we identified 44528 patients with GC confirmed by pathological examination and with active follow-up from 2010 to 2016. The cut-off date in this study was 12/31/2016.
We excluded patients who met the following criteria: TNM stage without being confirmed by gastrectomy; local LN metastasis (N+) or distant metastasis (M+); and age <18 years old or >85 years old at the time of diagnosis.
Depending on the system, 4417 GC patients were identified. Then, to ensure that the patients with N0 status were MILN, the patients with <16 LN examined or primary tumor invasion less than T3 were also excluded. Then, 690 patients were diagnosed with T3/T4, including T3, T4a, T4b, and T4 Nos, N0, and M0 tumors were abstracted and analyzed. The patients who underwent gastrectomy were grouped into the radiotherapy group (n = 288) and non-radiotherapy group (n = 402). To control confounding factors by different indications for radiotherapy between arms, we performed a matched analysis, a total of 218 patients in the radiotherapy group and 218 patients in the non-radiotherapy group were matched at a 1:1 ratio (data extraction flowchart is shown in Figure 1).

Flow chart showing study cohort. GC gastric cancer, No.LN number of lymph nodes.
The following patients’ information was used in our study: Baseline demographics including sex, age, race, marital status, insurance situation; Tumor features including primary tumor invasion, node status, metastasis status, grade, tumor location, tumor size and Lauren classification; Treatment information including gastrectomy, radiotherapy and chemotherapy. Cancer stage was determined or recoded according to the AJCC/UICC TNM staging system (seventh version). 21 Age was categorized into groups of < 60 and ≥ 60 years based on statistical and clinical consideration. Tumor location was categorized as the esophagogastric junction and stomach. Tumor size was recoded as <5 cm, 5-10 cm, >10 cm. More details can be obtained from SEERStat software version 8.3.6 and SEER manual 2016.
The endpoint of this study was CSS(Cancer-specific Survival), which was defined as the period from the date of diagnosis to the date of gastric cancer caused death, and patients who survived to the latest follow-up were censored.
Statistical Analysis
Descriptive statistics were used to calculate the absolute number and frequency among patients. The χ2, t, or Fisher’s exact test was used for intergroup comparisons where appropriate. The CSS was computed by the Kaplan-Meier method and tested by log-rank test. Survival-associated factors were evaluated using univariate analysis and multivariable Cox proportional hazards (PH) regression, adjusting for sex, age, race, marital status, insurance situation, primary tumor invasion, Lauren classification, grade, tumor location, tumor size, chemotherapy and gastrectomy.
To control confounding factors by different indications for radiotherapy between arms, we performed a matched analysis. In the case-matched analysis, which aimed to balance high-dimensional observed covariates, propensity score matching (PSM) was applied. The matching factors were the independent prognostic factors found to be unbalanced between the 2 groups confirmed by univariate Cox PH regression. Race, Lauren classification, tumor size and chemotherapy were included.
The PH and linearity assumptions for 25 continuous variables were examined using restricted cubic splines. Continuous variables were transformed into adequate forms for fitting the assumptions as appropriate. For categorical variables, log-log survival plots were used to identify the PH assumption, and all variables were fitted to the assumption. Results were considered statistically significant at a 2-sided P < 0.05. Data analysis were performed using IBM SPSS 25.0.0.0(IBM, Armonk, NY).
Nomogram Construction and Validation
Nomogram construction and validation analysis were performed using R 3.4.1. 22 Foreign, Hmisc, survival and rms packages were used. Data were read with the foreign package. 23 Variables were selected using the backward stepwise selection method in the Cox regression model with the survival package. 24 Based on the predictive models with the identified prognostic factors, nomograms were constructed to predict 3- and 5-year CSS. Nomogram validation consisted of discrimination and calibration. Discrimination was evaluated using Harrell’s concordance index (C-index) with the rms package. 25 Nomogram validation consisted of discrimination and calibration. Discrimination was evaluated using Harrell’s concordance index (C-index) with the rms package and Hmisc package. 26 Generally, a higher C-index value indicates better discrimination, with a value of 0.7 indicating moderate discrimination. A time-dependent receiver operating characteristic (ROC) curve was drawn to evaluate the accuracy of the nomogram. Validation was performed by comparing the means of predicted survival with those of actual observed survival estimated by the Kaplan-Meier method. To evaluate the efficacy of the nomogram better, stratification strategy was adopted, the X-tile program (Yale University School of Medicine, New Haven, CT, USA) was used to define the optimal cut-off points for the log-rank test and the highest specificity and sensitivity.
Results
Baseline Characteristics and Long-Term Survival
Together, 690 MILN GC patients who underwent gastrectomy between January 2010 and December 2016 were prospectively enrolled. As shown in Table 1, 288(41.7%) patients received radiotherapy and 402(58.3%) patients not; 487(70.6%) patients received chemotherapy and 203(29.4%) patients not; 193(28.0%) patients received no adjuvant treatments, 209(30.3%) patients receive chemotherapy alone, 10 patients (1.4%) received radiotherapy alone and 278(40.3%) patients received chemoradiotherapy. Regardless of their assignment in the radiotherapy subgroup or non-radiotherapy subgroup, individuals younger than 60 years of age were more likely to receive radiotherapy than those older than 60 years [48.5% (97/200) vs. 38.9% (191/490), p = 0.043]. The tumor size and Lauren classification were unbalanced between the 2 subgroups. The patients who did not receive chemotherapy were generally less likely to receive radiotherapy than those who received treatment with chemotherapy [4.9% (10/203) vs. 57.08% (278/487), p < 0.001]. No significant differences were found in terms of other variables.
Patient and Tumor Characteristics.
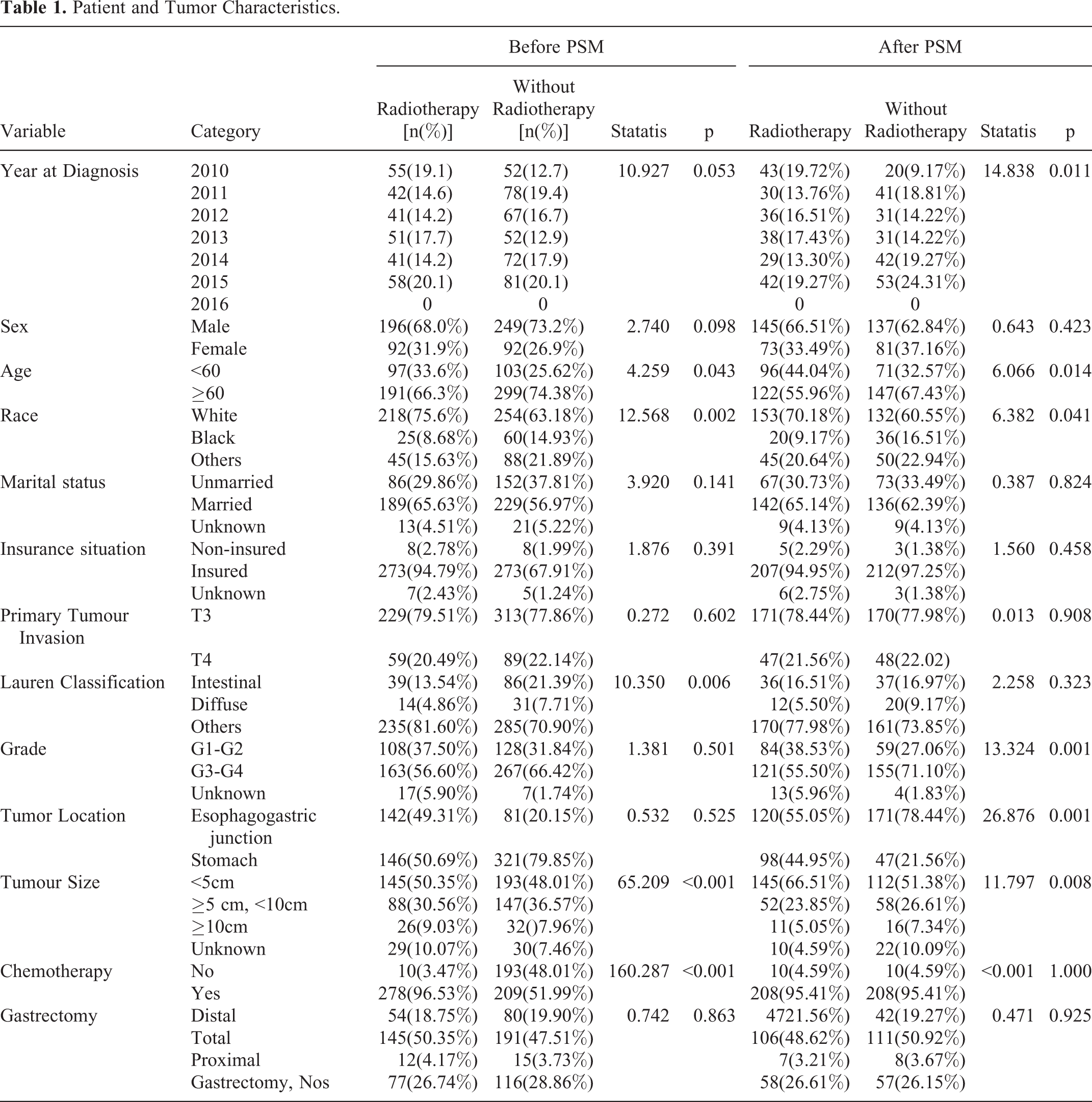
The balance of variables between the subgroups was markedly improved after PSM. Especially, for the variable of chemotherapy, which has been explicit demonstrated to be associated with prognosis in previous trials 6 -9 and our study and be related to the conducting of radiotherapy in our study, the significance of difference (p value) between radiotherapy subgroup and non-radiotherapy subgroup was changed from 0.001 into >0.500.The baseline characteristics of patients included in this study are listed in Table 1.
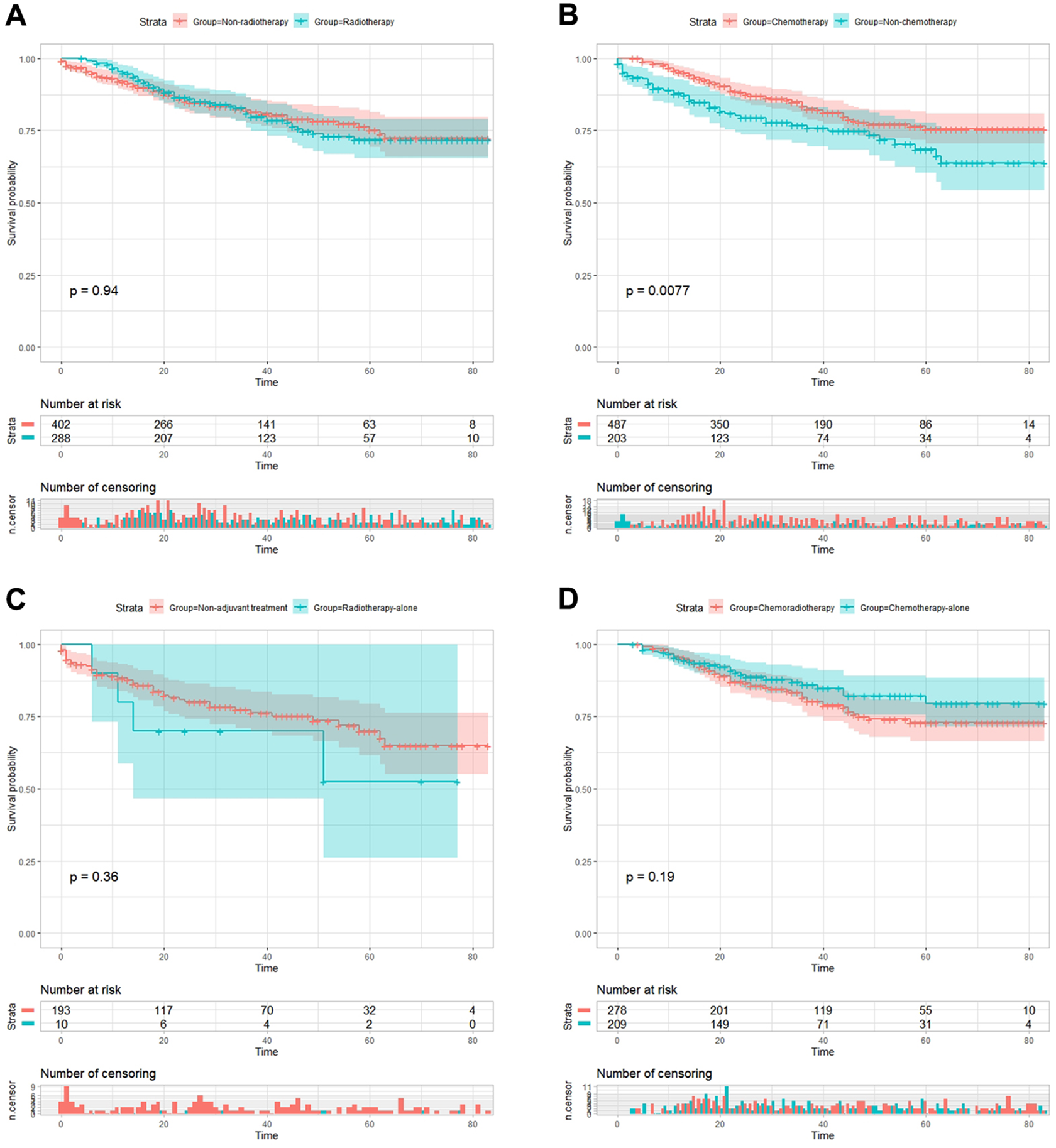
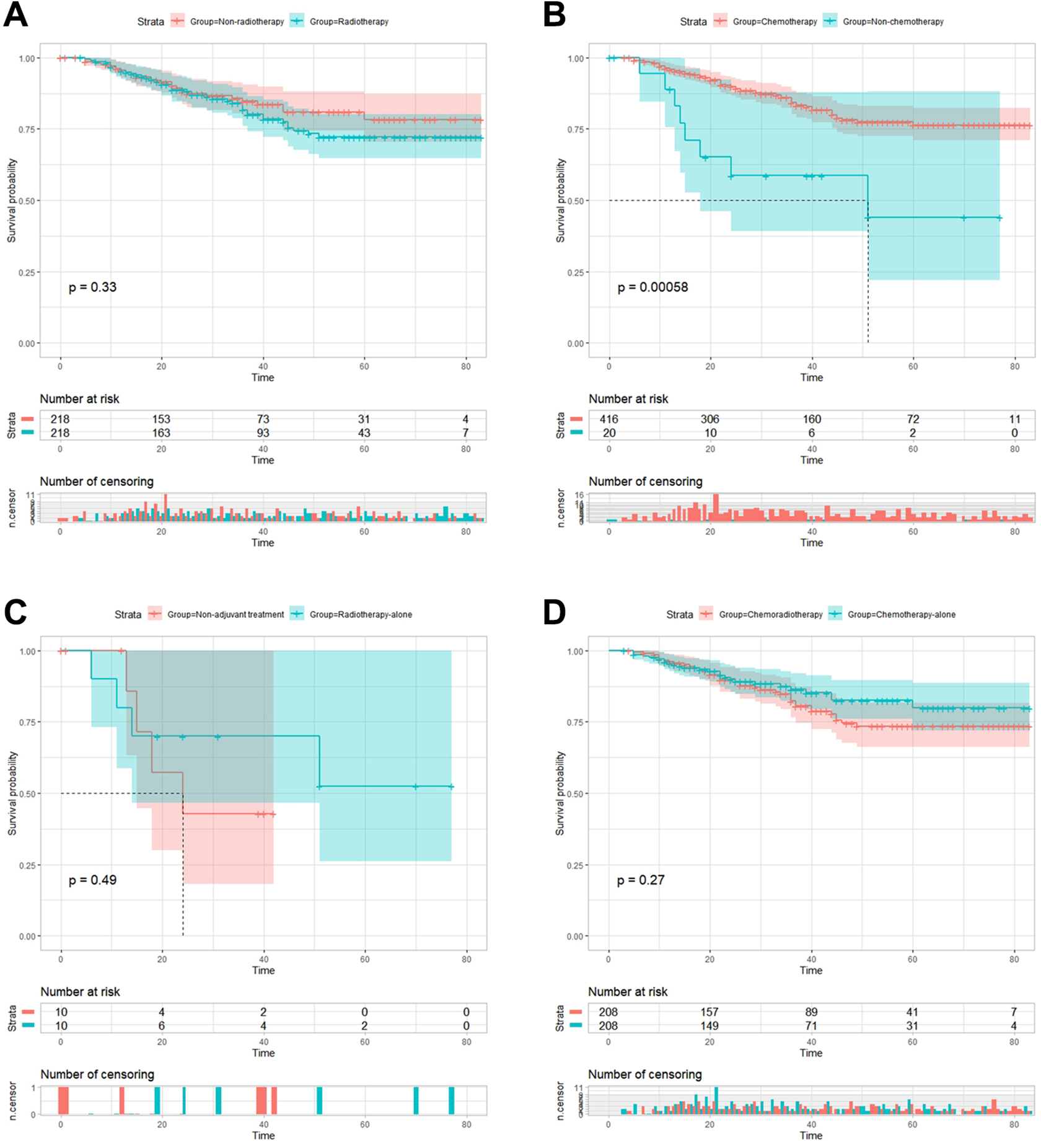
After PSM, Kaplan-Meier analysis showed that 5-year CSS was 72.1% in radiotherapy subgroup and 78.3%in non-radiotherapy subgroup [HR = 1.259, 95% CI = 0.788-2.012, p = 0.333](Figure 2A), 76.4% in chemotherapy subgroup and 44.0% in non-chemotherapy subgroup (HR = 0.297, 95% CI = 0.143-0.621, p = 0.001)(Figure 2B), 52.5% in radiotherapy alone subgroup and 42.9% in non-adjuvant treatment subgroup(HR = 0.591, 95% CI = 0.132-2.647, p = 0.487)(Figure 2C), 73.4% in chemoradiotherapy subgroup and 79.8% in chemotherapy alone subgroup(HR = 1.325, 95% CI = 0.804-2.182, p = 0.266)(Figure 2D). Result of the cohort before PSM were shown in Figure 3, overall, survival outcome between these subgroups was essentially the same before and after PSM.
Comparison of cancer-specific survival (CSS) after propensity score matching. A. 5-year CSS was 72.1% in radiotherapy subgroup and 78.3%in non-radiotherapy subgroup [HR = 1.259, 95% CI = 0.788-2.012, p = 0.333]; B. 76.4% in chemotherapy subgroup and 44.0% in non-chemotherapy subgroup (HR = 0.297, 95% CI = 0.143-0.621, p = 0.001); C. 52.5% in radiotherapy alone subgroup and 42.9% in non-adjuvant treatment subgroup(HR = 0.591, 95% CI = 0.132-2.647, p = 0.487); D. 73.4% in chemoradiotherapy subgroup and 79.8% in chemotherapy alone subgroup(HR = 1.325, 95% CI = 0.804-2.182, p = 0.266).
Comparison of cancer-specific survival (CSS) before propensity score matching. A. 5-year CSS was 71.9% in radiotherapy subgroup and 75.1%(HR = 1.013, 95% CI = 0.714-1.438, p = 0.940); B. 5-year CSS was 75.6% in chemotherapy subgroup and 68.5% in non-chemotherapy subgroup(HR = 0.616, 95% CI = 0.430-0.884, p = 0.008); C. 5-year CSS was 52.5% in radiotherapy-alone subgroup and 71.9% in non-adjuvant treatment subgroup(HR = 1.604, 95% CI = 0.575-4.471, p = 0.360); D. 5-year CSS was 72.9% in chemoradiotherapy subgroup and 79.5% in chemotherapy-alone subgroup (HR = 1.365, 95% CI = 0.859-2.172, p = 0.185).
Univariate and Multivariate Analysis
To explore an optimization model of whether radiotherapy could benefit MILN GC patients, analyses of univariate and multivariate cox regression were conducted in this study (Table 2). Univariate analysis of potential prognostic factors revealed that the patient’s diagnosed year (2010, 2011, 2012, 2013, 2014, 2015), sex, age (<60 vs. ≥60 years old), race (white, black and others), marital status (unmarried, married and unknown), insurance situation, Lauren classification (intestinal, diffuse and unknown), grade (G1-G2, G3-G4 and unknown), tumor location (esophagogastric junction vs. stomach), primary tumor invasion (T3 vs. T4), tumor size (<5 cm, 5-10 cm, ≥10 cm and unknown), chemotherapy (yes or no), radiotherapy (yes or no) and gastrectomy (distal, total, proximal and Gastrectomy, Nos) were regarded as covariates. Both before and after PSM, the variables with p < 0.01 in the univariate survival analysis were further analyzed in multivariate survival analysis, and the variable of radiotherapy was always included in the survival analysis.
Univariate and Multivariate Cox Regression Analysis for Cancer-Specific Survival After Psm.

The univariate survival analysis revealed that race (p < 0.05), primary tumor invasion (p < 0.05), tumor size (p < 0.05), Lauren classification (p < 0.05), chemotherapy (p < 0.05), and radiotherapy (p < 0.05) were associated with the CSS of the MILN GC patients who underwent gastrectomy, but radiotherapy did not improve survival at all(Table 2).
After PSM, the multivariate survival analysis showed that the patients who had received chemotherapy generally had better CSS (HR = 0.32, 95% CI = 0.15-0.68). Furthermore, patients with T4 tumors were likely to have worse CSS than those with T3 tumors (HR = 1.691, CI = 1.01-2.84). Meanwhile, tumor size also influenced patient prognosis; specifically, patients with bigger tumors (d≥10 cm) had worse CSS (e.g. HR = 2.48, CI = 1.46-4.22). Furthermore, compared with the race of white, black and other ethnic backgrounds were likely to have better CSS (HR = 0.47, CI = 0.18 -1.18 and HR = 0.39, CI = 0.20-0.76, respectively)(Table 2).
Meanwhile, survival analysis was also used to analyze the cohort before PSM, generally, the results were essentially the same, superior CSS remained associated with patients who had received chemotherapy, smaller tumor, less invasive tumors and intestinal-type classification.(Table S1). However, no predominant statistical significance was found for radiotherapy before or after PSM(Table 2& Table S1).
Prognostic Nomogram Construction and Calibration
The constructed nomogram (Figure 4) can assign survival probability by adding up the scores identified on the points scale for each variable. The total scores projected to the bottom scales indicate the probability of 3- and 5-year CSS survival. The C-index for CSS was 0.699 (95% CI, 0.638-0.76) (Figure 5A-B). For validation, the nomogram was tested by 3000 bootstraps resamples and show that the median actual survival corresponded closely CSS to the predicted survival. The area under curve(AUC) values of the ROC predicted the 3-, and 5-year CSS of the nomogram to be 0.708 and 0.761, indicating good agreements between prediction and practical observation(Figure 5C-D).

Nomograms predicting 3- and 5-year cancer-specific survival (CSS). The nomogram is used by adding up the points identified on the points scale for each variable. The total points projected on the bottom scales indicate the probability of 3- and 5-year survival.

Validation of the nomogram for predicting 3- and 5-year cancer-specific survival(CSS) for lymph node metastasis-indolent locally advanced gastric cancer after gastrectomy. (A-B). Calibration plot. The x-axis represents the nomogram-predicted survival, and the y-axis represents actual survival measured by Kaplan-Meier analysis. The C-index for CSS was 0.699 (95% CI, 0.638-0.76). (C-D). Discrimination plot. The area under curve(AUC) values of the receiver operating characteristic (ROC) predicted the 3-, and 5-year CSS of the nomogram to be 0.708 and 0.761.
In addition, to evaluate the predicting probability of our nomogram further, patients were stratified into 3 incremental risk level groups: low risk(0-12), medium risk(12-15) and high risk(>15)) which indicate different prognosis depending on X-tile program (Figure 6). A significant difference can be observed between different risk groups. The stratification strategy and result were summarized in supplementary figure (Supplementary Figure 1).

Patients were stratified into 3 incremental risk level groups (low risk, median risk and high risk) to evaluate the efficacy of the nomogram. Significant difference can be observed between different risk groups.
Discussion
In contrast to NCCN guidelines that recommended that pT3-4Nx GC patients undergo R0 resection should receive chemoradiotherapy, 12 our research indicated that only chemotherapy contributed to superior prognosis rather than radiotherapy for MILN GC in Western practices. Thus these results suggested radiotherapy could be substracted for this subgroup of patients. The survival analysis showed the CSS in radiotherapy subgroup was similar to that in non-radiotherapy subgroup, while CSS in chemotherapy subgroup was significantly better than that in non-chemotherapy subgroup. Further analyzing showed prognosis of radiotherapy-alone subgroup is not significantly different from that in non-adjuvant treatment subgroup, and the survival of chemoradiotherapy subgroup is similar to that in chemotherapy-alone subgroup. These results of the survival analysis indicated that the variate of radiotherapy could not improve survival. Furthermore, the Cox regression analysis confirmed that radiotherapy could not independently affect survival. Consistently, when the chemotherapy situation was totally the same between radiotherapy subgroup and non-radiotherapy subgroup by PSM, the survival of radiotherapy subgroup was inferior to that in non-radiotherapy subgroup.
In consistent, recently, the CRITICS trial, the first trial to directly compare postoperative chemoradiotherapy with perioperative chemotherapy in patients with resectable GC, showed that postoperative chemoradiotherapy did not improve survival compared with resectable GC treated with adequate preoperative chemotherapy and surgery. 27 After preoperative chemotherapy, 372 (95%) of 393 patients in the chemotherapy subgroup and 369 (93%) of 395 patients in the chemoradiotherapy subgroup proceeded to surgery. With a median follow-up of 61.4 months (IQR 43.3-82.8), mOS was 43 months (95% CI 31-57) in the chemotherapy subgroup and 37 months (30-48) in the chemoradiotherapy subgroup (HR 1.01, 95% CI 0.84 -1.22; p = 0.90). This result supported our hypothesis and finding in our study that the survival benefit of chemoradiotherapy in Western practices may be attributed primarily to chemotherapy rather than to radiotherapy. The recent meta-analysis also showed that for advanced GC, radiochemotherapy displayed similar OS in comparison to chemotherapy alone. 28 From the perspective of biological characteristics, this result could also be explained. Stage T4aN0M0 GC, in which the tumor cells have infiltrated the serosa, is prone to cell exfoliation in the abdominal and thus is likely to recur. Although radiotherapy cannot remove these detached GC cells and potential micro-hematogenous metastasis, systematic chemotherapy may work to some extent due to the difference in the mechanism of action.
To explore our hypothesis that chemoradiotherapy conferred a better prognosis compared with surgery-alone in previous experiences might be attributed to chemotherapy rather than radiotherapy even in Western practices, we investigated it firstly in MILN GC based on previous research. The study that analyzed the relapse patterns and therapeutic effects of D1 and D2 dissections found that the addition of postoperative chemoradiotherapy had a major impact on local recurrence in resectable gastric cancer with D1 dissection (2% vs 8%; p = 0.001), while there was no difference in patients undergoing D2 dissection. 29 So the survival benefits associated with adjuvant chemoradiotherapy may be a compensation for inadequate LN dissection, such as that in D1/D0 dissection. However, in Western practices, most patients only underwent D1/D0 dissection, so we decided to investigate on MILN GC patients. In addition, the defining of MILN GC as stage T3-4N0M0 patients with more than 15 LNs examined based on the following reasons. Overall, the more invasive the tumor depth is, the more extensive the LN involving is. 30,31 However, due to tumour heterogeneity, some T3-4 patients may have no LN metastasis, while some T1a-1b patients may confront extensive LN metastasis. Of course, the stage T3-4N0 GC are distinguished from the subgroup of biologically LN metastasis-active GC (T1a-1bN+) and could be conformed to our study demanding.
Also, the association between the extent of lymphadenectomy and survival benefit of radiotherapy is an interesting point worthing in-depth discussing to comprehend this research. Dikken et al. retrospectively compared survival and recurrence patterns to evaluate more intensified postoperative chemoradiotherapy than those from the Dutch Gastric Cancer Group Trial (DGCT), which randomly assigned patients between D1 and D2 lymphadenectomy. 29 Survival and recurrence patterns of 91 patients with adenocarcinoma of the stomach who had received surgery followed by radiotherapy combined with fluorouracil and leucovorin (n = 5), capecitabine (n = 39), or capecitabine and cisplatin (n = 47) were analyzed and compared with the survival and recurrence patterns of 694 patients from the DGCT (D1, n = 369; D2, n = 325). The results revealed that the addition of postoperative chemoradiotherapy had a major impact on local recurrence in resectable gastric cancer with D1 LN dissection, while there was no difference in patients undergoing D2 dissection. Consistently, patients in the ARTIST 19 and ARTIST- II trial, 17 most of who underwent D2 LN dissection, did not benefit from the addition of radiotherapy. In addition, in the view of fundamental research, some vital researches have indicated epibiotic cancer cells in LNs resulting from the inadequate LN dissection of positive LNs would be active hubs for systemic tumor cell spreading and thus more prone to facilitating recurrence. 32,33 Thus, radiotherapy could be an important complementary measure to reduce the potential cancer cells in LNs. Therefore, the benefit associated with chemoradiotherapy may be compensation for inadequate LN dissection (D1/D0) for nonspecific GC. While for LAGC patients who undergo standard radical D2 lymphadenectomy, which has been confirmed to achieve the maximum oncology efficacy for LAGC, 34,35 or who are characterized as being MILN, the prognosis benefit of radiotherapy would be negative. And in our study, the MILN GC patients who mainly underwent D1/D0 LN dissection might be equal to the LN dissection effect of nonspecific GC patients mainly underwent D2 LN dissection. From this perspective, our results are reasonable and might be generalized.
Considering all these factors, the extent of LN dissection, biological LN status and primary tumor invasion should be taken into account when discussing and determining radiotherapy for GC. Furthermore, from the perspective of histology, the patients with intestinal-type GC are more likely to benefit from chemoradiotherapy than those with diffuse-type GC in the subgroup analysis of the INT0116 and ARTIST trials. 10,19 Also, It has been acknowledged that therapeutic strategies in a multidisciplinary discussion for GC should be determined by individual patient characteristics. 28 Thus, identifying patients could benefit from radiotherapy or not is critical to subtract unnecessary treatment without undermining treatment effect. Thus, well-designed trials to determined tailored treatment for specific subgroups are in urgent. 36
The present study has several limitations that should be noted. This study was a retrospective study and the patients’ characteristics were unbalanced between groups. To compensate for this inherent limitation, we performed PSM with the variates that were unbalanced between groups and could independently affect the prognosis of MILN GC to improve the balance of baseline data. Notably, the significance of the difference in chemotherapy situation between radiotherapy subgroup and non-radiotherapy subgroup was changed from 0.001 to 1.000. Also, although we could identify whether the patients received chemotherapy or not, the SEER database did not provide information on the chemotherapy regimes, durations or the relationship with the surgery. Besides, MILN is a relevant concept, and the pathology result of LN-negative may not guarantee MILN nature and the number of LNs examined after gastrectomy might affect the detection of LN-metastasis status. 37 -40 Thus, to compensate for this limitation, in our study, only patients with more than 15 LN examined were enrolled since this feature was defined as a surrogate for the evaluation of LN dissection. 41,42
Conclusions
MILN GC could not benefit from radiotherapy, which may only work as compensation for the poor surgical outcomes in LAGC patients with potential LN metastasis in Western practices. Chemoradiotherapy confers superior prognosis to MILN GC patients compared with surgery alone might only be attributed to chemotherapy rather than radiotherapy. This finding suggested that, contrary to NCCN guidelines, radiotherapy could be subtracted to reduce the side effects and treatment burden of radiotherapy for MILN GC patients. However, determining conclusions should be further verified in well-designed randomized trials.
Supplemental Material
Supplemental Material, Fig_S1 - Distinguish the Role of Radiotherapy From Chemoradiotherapy for Gastric Cancer With Behavior of Metastasis-Indolent in Lymph Node
Supplemental Material, Fig_S1 for Distinguish the Role of Radiotherapy From Chemoradiotherapy for Gastric Cancer With Behavior of Metastasis-Indolent in Lymph Node by Yunfei Zhi, Zhousheng Lin, Jinyuan Ma, Weiming Mou and Xinhua Chen in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Table_S1_ - Distinguish the Role of Radiotherapy From Chemoradiotherapy for Gastric Cancer With Behavior of Metastasis-Indolent in Lymph Node
Supplemental Material, Table_S1_ for Distinguish the Role of Radiotherapy From Chemoradiotherapy for Gastric Cancer With Behavior of Metastasis-Indolent in Lymph Node by Yunfei Zhi, Zhousheng Lin, Jinyuan Ma, Weiming Mou and Xinhua Chen in Technology in Cancer Research & Treatment
Footnotes
Acknowledgments
We thank Prof. Jiang Yu, from Department of General Surgery and Prof. Liang Zhao from Department of Pathology, for their professional guidance and useful comments which have greatly improved the manuscript.
We also would like to thank the staff members of the National Cancer Institute and their colleagues across the United States and at Information management Services, Inc., who have been involved with the Surveillance, Epidemiology and End Results (SEER) Program. Finally, we would like to thank anonymous reviewers who gave valuable suggestion that has helped to improve the quality of the manuscript.
Author Contribution
Yunfei Zhi and Zhousheng Lin are authors contributed equally to the work.
Data Availability
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The SEER was public-use data: informed consent was waived. And our study was deemed exempt from institutional review board approval by NanFang Hospital, Southern Medical University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the College Students' Innovative Entrepreneurial Training Plan Program of Southern Medical University, Guangzhou. (Grant No.201912121290 and X202012121295)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
