Abstract
Introduction
Oral cancer (OC) is a prevailing malignancy, with 377,713 new cases and 177,757 deaths worldwide in 2020. 1 Early detection of the malignancy is of paramount importance owing to the poor survival associated with advanced OC2–5 despite the advancements in treatment modalities over the past few decades, such as surgery, radiation, and chemotherapy.6,7 Therefore, attempts to reduce advanced OC and OC mortality via early detection of oral premalignancy (OPM) and OC through organized screening programs are of upmost priority.
The effectiveness of OC screening with visual inspection has been demonstrated in a randomized controlled trial 8 and in a population-based service screening program 9 in reducing late-stage OCs and mortality from OC, especially in the high-risk group.8–11 A significant reduction of mortality from OC by 24% and of incident stage III or worse OC by 21% among tobacco or alcohol users was reported in an Indian randomized controlled trial. 10 A similar finding was observed in the mass screening in Taiwan, with a reduction in mortality by 26% and advanced OC by 21%. 9 The trial in India provided screening every 3 years, while the program in Taiwan targeted cigaret and/or betel quid users with biennial screens. The differences in risky oral habits (betel quid chewing, cigaret smoking, and alcohol consumption), in addition to variations in participation rate, sensitivity of screening, and compliance to referral, hinder the screening scenario from being applied to other countries even if the natural history of OC is similar between the said contries. 12 The adoption of auxiliary methods of visual inspection, including autofluorescence, oral brush biopsy, and liquid-based cytology, may further increase the effectiveness of the screening program.13–17
Large-scale randomized controlled trials may help clarify how these variables affect the efficiency of OC screening, but the costs will be enormous and long follow-up periods will be required to achieve reasonable results. Moreover, trials are often designed to deal with 1 parameter and cannot examine multiple parameters simultaneously. Consequently, this approach is not practical in the real world, especially given the ethical concerns pertaining to those who are not screened. Another strategy is to construct an emulated cohort and use computer simulation to estimate the results, which is less expensive and more time-saving than trials. 18 With the application of available data and statistical synthesis of evidence, the model has been proven to be accurate in estimating parameters pertaining to different scenarios. 18 This strategy can help policymakers avoid ineffective trials or adjust screening programs, as reported in many cancer screenings, including breast,19–21 cervical,22,23 colorectal,12,24,25 and gastric cancer.26,27 The current study aimed to use computer simulation to evaluate the effectiveness of screening in reducing advanced OC and mortality from OC, taking into account variations in oral habits, interscreening intervals, compliance rates, and referral rates.
Materials and Methods
A Markov chain Monte Carlo algorithm 18 was used to simulate a prospective cohort under various scenarios of OC screening. The framework of the simulation model is shown in Figure 1. A hypothetical cohort of 8 million participants aged ≥30 years with oral habits of cigaret smoking and/or betel quid chewing was created and randomly assigned equally to the intervention and control arms. The cohort size in each arm was similar to that of the target population in Taiwan. The control arm followed the natural history of “OPM-oral cancer,” whereas the intervention arm was invited for OC screening. In the intervention arm, those who did not comply with screening (nonscreening group) had a natural history of OC. Those who complied with the screening (screen group) with positive results were referred to confirmatory diagnosis by the specialists, and those with OPM did or did not receive treatment; some eventually underwent malignant transformation to OC and died from the disease. Those with negative results were advised to participate in the screening at the suggested frequencies. OC was classified into different stages, and patients were treated accordingly, with some patients dying from the cancer, as per the 5-year survival data. The model was constructed using the above framework, and we simulated the effect of OC screening using a randomized controlled design with variable compliance rates, detection rates, referral rates, and intervention effects. We also examined the reduction in advanced OC, OC mortality, and number needed to screen (NNS) 28 under different screening policies with 1-year, 2-year, and 3-year frequencies.
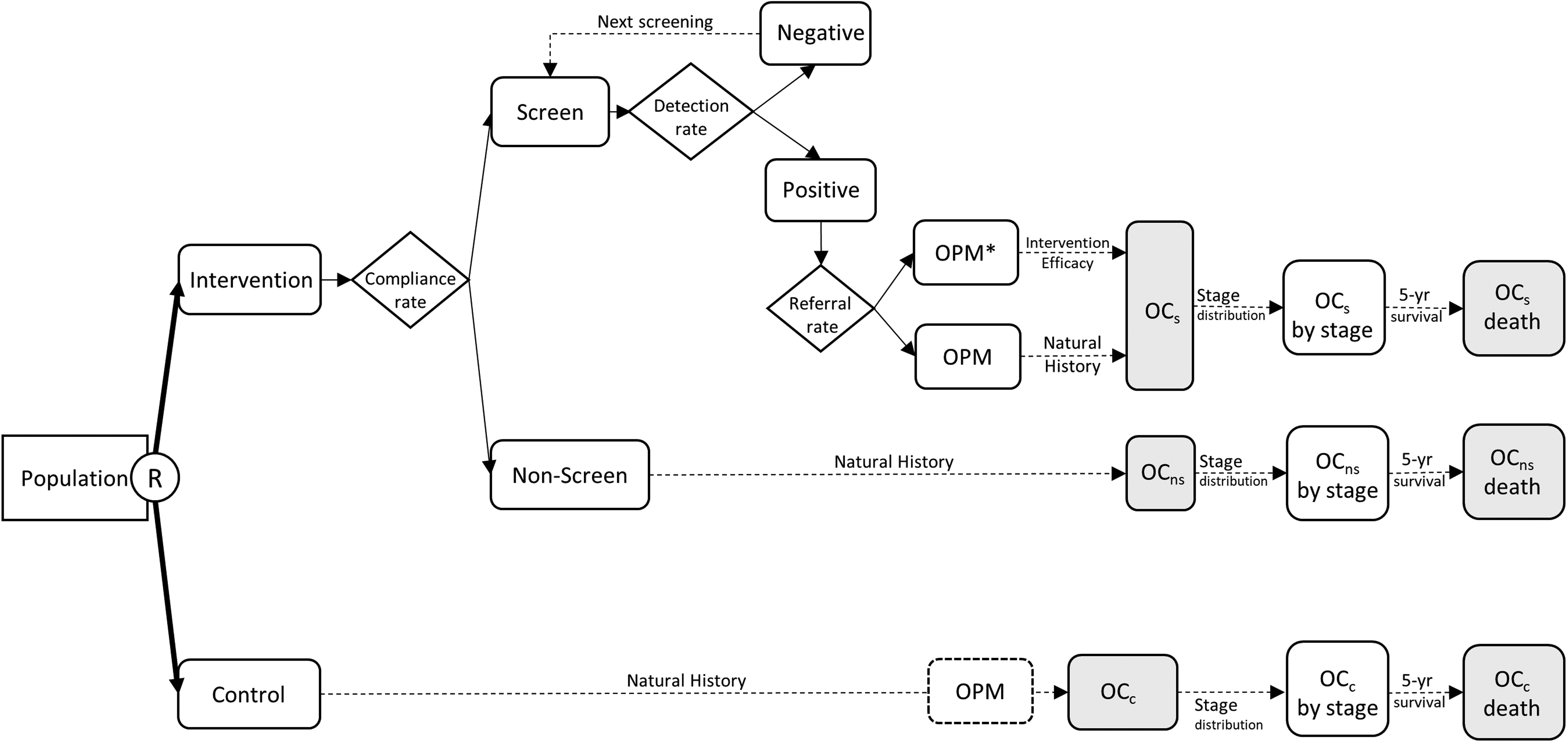
Framework of randomized controlled trial on oral cancer screening for the computer simulation model.
Temporal Natural History of Oral Cancer and Base-Case Parameter Acquisition
Table 1 shows the base-case parameters for model simulation with corresponding estimates retrieved from the relevant literature. To simulate the effect of different screening scenarios, the transition parameters of the temporal natural history in the absence of screening were required to represent the control arm without contamination. The estimates for the parameters were retrieved from the study by Shiu et al, 29 namely 0.0016 for the annual transition rate of leukoplakia, 0.0979 for the annual transition rate from leukoplakia to OC, 72.7% for the intervention efficacy in reducing malignant transformation, and other corresponding figures for different oral habits (A: alcohol drinking, B: betel quid chewing, and C: cigaret smoking).
Base-Case Parameters for the Simulation Model.

A, alcohol consumption; B, betel quid chewing; C, cigaret smoking; OPM, oral premalignancy; CIS, carcinoma-in-situ.
Based on the results of the OC screening program between 2004 and 2009 in Taiwan, 9 the sensitivity of screening was 88.29%, the compliance rate was 55.1%, the referral rate was 92.6%, and the OPM detection rate ranged from 0.00207 to 0.00991 according to different oral habits. The stage distributions of screen/nonscreen for the intervention arm from stage CIS (carcinoma-in-situ) to stage IV were assigned as 0.6/0.6, 26.1/21.1, 19.8/17.9, 12.5/13.3, and 41.0/47.1, respectively based on the results of Chuang et al 9 The corresponding figures for the control arm were assigned as 0, 14.08, 11.97, 24.65, and 49.30 based on the results of Sankaranarayanan et al 8 The 5-year survival rates for OC from stage CIS to stage IV were assigned as 76.7%, 77.4%, 68.3%, 54.7%, and 33.2% based on the data from Taiwan Cancer Registry for 2008 to 2012. 7 The reporting in this study conformed to STROBE guidelines. 30 The Research Ethics Committee of National Taiwan University Hospital approved this project and granted a waiver for informed consent (202002091 W) pursuant to the regulations of the Institutional Review Board on March 12, 2020.
Statistical Analysis
A battery of algorithms using the Markov chain Monte Carlo method was run with sampling from base-case parameters to construct the desired distribution of our hypothetical cohort. 18 Then, a computer simulation was performed using the constructed model to evaluate the reduction in advanced OC and OC mortality under various screening scenarios. Relative risk, risk difference, and NNS were reported with 95% confidence intervals (CI) for various simulated compliance and referral rates. The adequacy of the fitness of our model was validated by comparing the simulated numbers to the observed numbers using Pearson's chi-square test. Statistical significance was defined as a P-value <.05. All statistical analyses were performed using the SAS software (version 9.4; SAS Institute).
Results
Effects of Oral Cancer Screening Under Different Screening Frequencies
The incremental reduction in advanced OC was approximately 5%, with each reduction occurring in the interval between screenings (45%, 40%, and 35% for 1-year, 2-year, and 3-year screening frequencies, respectively). A similar trend was observed for OC mortality, with an average incremental reduction of 6.5% (36%, 29%, and 23% for 1-year, 2-year, and 3-year screening frequencies, respectively). The NNS per OC mortality reduction also decreased with a short interval between screenings; the NNS decreased from 1565 to 995 when the screening interval was reduced from a 3-year interval to 1-year interval, in a screening scenario similar to that in Taiwan, with a 55.1% attendance rate and 92.6% referral rate (Table 2).
Simulation for the Effectiveness on Reduction of Advanced Oral Cancer and Mortality at Different Screening Intervals*

Screening scenario similar to Taiwan with 55.1% attendance rate and 92.6% referral rate. Abbreviations: NNS, number needed to screen; adv OC, advanced oral cancer.
Sensitivity Analyses of the Relative Reduction Rate for Advanced Oral Cancer and Oral Cancer Mortality in a Screening Scenario Similar to Taiwan at a 2-Year Screening Frequency
The reduction in advanced OC increased from 31% to 34% under a 40% compliance rate to 37% to 41% under a 60% compliance rate, and the reduction in OC mortality increased from 19% to 22% under a 40% compliance rate to 25% to 30% under a 60% compliance rate, given that the referral rate shifted from 70% to 90%. The incremental reduction in the risk of advanced OC with 10% increase in the compliance rate was 3% to 4% whereas only a 1% to 2% incremental reduction was noted per 10% increase in the referral rate. The effectiveness of screening increased with a short screening frequency (Figure 2).

Sensitivity analyses of relative reduction rate for (A) advanced oral cancer and (B) oral cancer mortality.
Incremental Reduction Under Various Oral Habits
Accounting for different oral habits, our simulated model showed positive effects of screening high-risk populations, with a reduction in advanced OC ranged between 39% and 45% (Figure 3). Similarly, the relative mortality rate of OC ranged between 28% and 35%.

Relative reduction rate by oral habits for (A) advanced oral cancer and (B) oral cancer mortality.
Discussion
Evaluating the effect of screening programs for diseases with a long natural history via randomized controlled trials requires a long follow-up period and substantial costs, especially when using cause-specific mortality as the primary endpoint. 10 For health policy makers, evaluating the results of a study in the early stages is challenging, given the limited information available. We constructed a computerized 3-state Markov simulation model based on the transition parameters of OC natural history and distribution of tumors9,29 to predict the effects of OC screening programs in different screening scenarios. The model was then applied to empirical data from an OC screening program in Taiwan for validation (Supplement Table 1). Further analyses were conducted using the transition parameters of natural history with different oral habits 29 to predict the reduction in advanced OC and OC mortality under various scenarios.
Implication for the Oral Cancer Screening
Our prediction of the proportion of advanced cancer cases, cumulative incidence of advanced OC, and OC mortality in the intervention arm were consistent with the results reported for the OC screening program in Taiwan. 9 This finding verified the external validity of our decision model. A randomized controlled trial in India8,10,11 and a service screening program in Taiwan 9 demonstrated that visual inspection screening could reduce the incidence of advanced OC and OC mortality. As few countries have conducted OC screening due to cost-effectiveness considerations, our computer simulation model could predict the effect of screening under different screening scenarios, and the predicted results could provide useful information for designing and implementing OC screening programs.
Methodological Consideration and Application
The unique feature of the current study is the application of a 3-state Markov model based on the transition parameters of natural history in conjunction with the survival rate for OC. The advantage of this model is the incorporation of the effects of the 3 risky oral habits on the annual incidence rate of leukoplakia and annual malignant transformation rate to estimate the effect of different screening scenarios to save the cost and time required for randomized control trials. For example, by retrieving reference data from the literature (oral habits of patients with OC: 14.5% with cigaret smoking alone, and 58.8% with both cigaret smoking and alcohol drinking 31 ) and application of our constructed simulation model, a 34% reduction (relative risk [RR] = 0.66, 95% CI:0.58-0.74) in OC mortality for smokers but nondrinkers and a 35% reduction (RR = 0.65, 95% CI:0.54-0.79) for smokers and drinkers could be anticipated after a 6-year follow-up period in a screening scenario similar to Taiwan's screening program. Considering that there are more than 377 thousand new OC cases and 177 thousand deaths annually, 1 if more than half of the smokers worldwide attend OC screening, at least 45,000 OC deaths annually could be prevented at the 6-year follow-up.
In Taiwan, indigenous people over 18 years of age have a higher rate of risky oral habits and are more frequently affected by OCs than the nonindigenous population. However, only 32% of this group attended the biennial OC screening program with a much lower referral rate of 77%.9,32 Among those with risky oral habits, the proportions of betel quid chewing and cigaret smoking, betel quid chewing alone, and cigaret smoking alone accounted for 58%, 30%, and 12%, respectively. With the application of our OC screening simulation model, a 29% reduction in advanced OC (RR = 0.71, 95% CI = 0.63-0.80) and a 17% reduction in OC mortality (RR = 0.83, 95% CI = 0.72-0.96) at the 6-year follow-up was predicted, given that all indigenous Taiwanese participated in the screening program. These 2 examples demonstrate that our constructed simulation model can predict the effect of OC screening; researchers, policymakers, and authorities interested in this issue can use these data.
In recent years, studies have shown that a novel tool, fluorescence visualization, could improve the detection of potentially malignant oral disorders in population-based screening.13–16 The sensitivity (94.4% vs 83.3%) and specificity (96.2% vs 95.1%) of this method were also higher than those of conventional oral visual examination. 13 With the application of our constructed model, we can extract parameters from the studies and simulate the RR of advanced OC and OC mortality at the 6-year follow-up. The simulation shows that the relative rate of advanced OC is quite similar for either cigaret smokers and betel quid chewers (fluorescence visualization/conventional oral examination: 0.60/0.61) or cigaret smokers and alcohol drinkers (0.56/0.56), while the corresponding figures for OC mortality were 0.71/0.72 and 0.65/0.66, respectively (Supplemental Table 2). These results suggest that fluorescence visualization has better performance in detecting oral premalignancy than conventional methods, yet the difference with respect to reducing advanced OC and OC mortality may not be as significant even in high-risk populations. These simulated data provide valuable information for health policymakers and researchers interested in designing and conducting large-scale trials to evaluate novel tools for OC screening.
Limitations
The current study has several limitations. First, the transition parameters of natural history were retrieved from hospital-based studies rather than community-based studies. Since there was no comparable literature, we used these surrogate sources of data instead. Second, the differences in risky oral habits in India (tobacco and/or alcohol) and Taiwan (cigaret and/or betel quid) may lead to discrepancies in understanding the carcinogenesis of OC, thus reducing the accuracy of the estimates of our model. This difference could account for the higher reduction in mortality with the screening of low-risk group (cigaret smoking alone, 34%) than with the screening high-risk group (alcohol drinking and betel quid chewing, 28%) based on Taiwan's scenario, despite the proper validation of our simulated model.
Conclusion
Our computer simulation model demonstrated that the effectiveness of OC screening varied under various screening parameter scenarios. The predicted results provide health policymakers with evidence-based guidelines to emulate studies and launch screening programs to save lives.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221147771 - Supplemental material for The Effectiveness of Population Mass Screening to Oral Cancer: A Simulation Study
Supplemental material, sj-docx-1-tct-10.1177_15330338221147771 for The Effectiveness of Population Mass Screening to Oral Cancer: A Simulation Study by Chiu-Wen Su, William Wang-Yu Su, Sam Li-Sheng Chen, Tony Hsiu-Hsi Chen, Tsui-Hsia Hsu, Mu-Kuan Chen and Amy Ming-Fang Yen in Technology in Cancer Research & Treatment
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Health Promotion Administration, Ministry of Health and Welfare of the Taiwanese government under the program of “Breast cancer, oral cancer, and colorectal cancer screening data monitoring and evaluation research program, Year 2021” (grant number A1091116).
Ethical Approval
The Research Ethics Committee of National Taiwan University Hospital approved this project and granted a waiver for informed consent (202002091 W) pursuant to the regulations of the Institutional Review Board on March 12, 2020.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
