Abstract
Introduction
Oral squamous cell carcinoma (OSCC) is one of the leading causes of cancer-related mortality in Taiwan and in the world. 1 Oral cavity cancer is treated with a combination of different modalities. Advances in multidisciplinary approaches have significantly improved the treatment outcomes and resulted in the development of tailored therapeutic strategies. For example, adjuvant radiotherapy (RT) reduces the risk of tumor recurrence in patients with advanced features.2,3 Concurrent chemoradiotherapy (CCRT) after radical surgery significantly improves the treatment outcome of patients with a higher risk of recurrence.4,5 However, changes in adjuvant treatment over time and other controversies need to be further explored.
Several clinicopathological risk factors have been identified and incorporated into staging or risk classification systems.6,7 Perineural invasion (PNI) is a form of tumor spread that can be broadly defined as tumor cell invasion within the nerve or enclosing the nerve. PNI is mediated by neural cell adhesion molecules that are expressed on the surface of tumor cells.8–10 The incidence of PNI in OSCC ranges from 12% to 50%. PNI is a risk factor for locoregional recurrence and poor survival outcomes, and the presence of PNI is an indication of postoperative RT or CCRT. 11 Different PNI patterns are associated with different prognoses. The eighth edition of the American Joint Committee on Cancer (AJCC) Staging Manual proposed a subdivision of PNI into extratumoral and intratumoral PNI (EPNI and IPNI, respectively). 12 EPNI and IPNI affect prognosis, suggesting that the extent of PNI correlates with prognosis. Therefore, quantitative or subjective measurements may provide valuable information for outcome prediction.
The conventional method for identifying PNI, which is standard hematoxylin and eosin (H&E) staining, relies on the experience of the pathologist and the staining performance. A more objective and sensitive method should be used to improve the detection of PNI. S100 is a nerve system-specific cytoplasmic protein. Staining for S100 by immunohistochemistry (IHC) can identify various nerves and is a sensitive method for the detection of PNI. Kurtz et al 13 demonstrated that replacing standard H&E staining with S100 or using the 2 in combination increases the detection rate of PNI from 30% to 82% of oral cavity squamous cell carcinoma, and S100 staining improves the PNI detection rate in other cancers. For example, Conte et al 14 reported that failure to detect PNI by H&E can be overcome by S100 staining for colorectal cancer, thereby improving prognosis prediction. In this retrospective study, we performed a quantitative measurement of PNI using S100 IHC staining and evaluated its prognostic effect.
Materials and Methods
Ethical approval was granted by the institutional review board of an associated medical foundation (201800407B0, 2018/March/22, Taoyuan, Taiwan). The need for informed consent was waived owing to the retrospective nature of the study. The reporting of this retrospective study conforms to STROBE guidelines. 15 Clinical records of patients with OSCC who underwent curative treatment in the same hospital between July 2009 and November 2015 were reviewed. All records were retrieved following inclusion and exclusion criteria and de-identified and the identity of any person could not be ascertained in any way. The inclusion criteria were as follows: pathologically diagnosed OSCC, treated by radical surgery with or without adjuvant therapies, and the presence of PNI in pathologic samples. Patients with a previous cancer history, synchronous double cancers, evidence of metastatic disease, and those lacking clinical records were excluded. All pathological samples of selected patients were reviewed to confirm the presence and pattern of PNI. After confirmation, immunostaining for the S100 protein was performed, which indicated the presence of PNI (EPNI or IPNI, Figure 1). Immunostaining was performed using a sensitive avidin-biotin immunoperoxidase technique with the antibodies to the following antigen: S-100 protein (1:500, Leica, NCL-L-S100p). All PNI foci were recorded by one head and neck pathologist. The number of PNI foci in each slide and the average PNI (APNI) per slide were recorded. The methods of this study are reproducible since there are no special procedures to obtain data or materials.

Immunohistochemistry staining of S100 was performed using a sensitive avidin-biotin immunoperoxidase technique with the antibodies to the following antigen: S100 protein (1:500, Leica, NCL-L-S100p). (a) One intratumoral perineural invasion and (b) 2 extratumoral perineural invasion foci can be identified with assistance of S100 staining.
All patients underwent an extensive presurgical evaluation and complete staging work-up, including computed tomography (CT) or magnetic resonance imaging (MRI) and fluorine-18-fluoro-2-deoxy-d-glucose positron emission tomography (FDG-PET) scan. Radical surgery included tumor excision with ≥1 cm safety margins and neck dissection. Patients with the clinical node-negative disease received neck dissection at levels I to III. Neck dissection at levels I to V was performed in patients with the clinical node-positive disease. Staging results were recorded according to the eighth edition of AJCC Cancer Staging. 16 In cases with pathological risk factors, postoperative RT (60-66 Gy) was guided by the presence of pathological risk factors, such as pathological T4 criteria, node-positive disease, close resection margin, tumor invasion depth ≥10 mm, nerve or vessel invasion, and poorly differentiated histology. The target volume comprised the tumor bed area, regional neck lymphatics, and the possible nerve route from the skull base to the primary tumor. RT was delivered using intensity-modulated RT or volumetric-modulated arc therapy. Concurrent chemotherapy was offered to patients with major risk factors, such as nodal stage 2 or 3 (extranodal extension or multiple node metastases) and positive resection margins. Patients with 3 or more of the above-mentioned minor risk factors also received concurrent chemotherapy during RT. The regimen of concurrent chemotherapy was cisplatin, administered biweekly (50 mg/m2) or triweekly (100 mg/m2), with/without an additional oral 5-fluorouracil prodrug.17,18
After treatment, follow-up appointments were scheduled at least every 3 months and included physical examinations. Scope examination was optional for patients with moderate to severe trismus, or whenever pharyngeal examination was indicated. If the patient missed their appointment, a follow-up call was made. Another appointment was arranged or the patient's condition was noted if they had passed away or refused follow-up. Imaging studies (CT/MRI) were performed 3 months after completion of CCRT and every 6 to 12 months thereafter. Panendoscopy has performed annually. Outcomes included locoregional recurrence, distant metastasis, and death. Recurrence was confirmed by pathological examination or serial clinical/imaging findings if no tissue was available. The choice of salvage or palliative treatment was based on recurrent disease status and patient preference. Each death was reviewed in detail. Death is unrelated to recurrence or complications was not regarded as treatment failure. Time-to-event and survival were calculated from the date of diagnosis to the date of the corresponding event. For time-to-recurrence, the event was any recurrence, while for overall survival, the event was death. The endpoints were time-to-recurrence (TTR) and overall survival (OS).
Statistical Analyses
The t-test is used to compare the means of the 2 groups. The Kaplan–Meier method was used for survival analysis and the log-rank test was used to determine significance. Multivariate analysis was performed using a Cox regression model. Correlations between each variable and the endpoints were evaluated by univariate and multivariate analyses. Statistical analyses were performed using PASW statistics, version 18 (SPSS Inc., Chicago, IL, USA). P < .05 was considered statistically significant.
Results
Ninety-two patients were included in this study. The median age was 50.5 years old (range: 29-85). The mobile tongue was the most common subsite (66, 59.8%). All patients had PNI. Sixty-three patients (68.5%) had IPNI and 29 (31.5) patients had EPNI. In each slide, the APNI identified by S100 staining ranged from 0.25 to 33 (median: 2.8). Patients with EPNI had an APNI of 6.7, which was significantly higher than that in patients with IPNI (APNI, 3.8; P = .021, 2-tailed t-test). The patient characteristics are listed in Table 1.
Characteristics of All Patients.
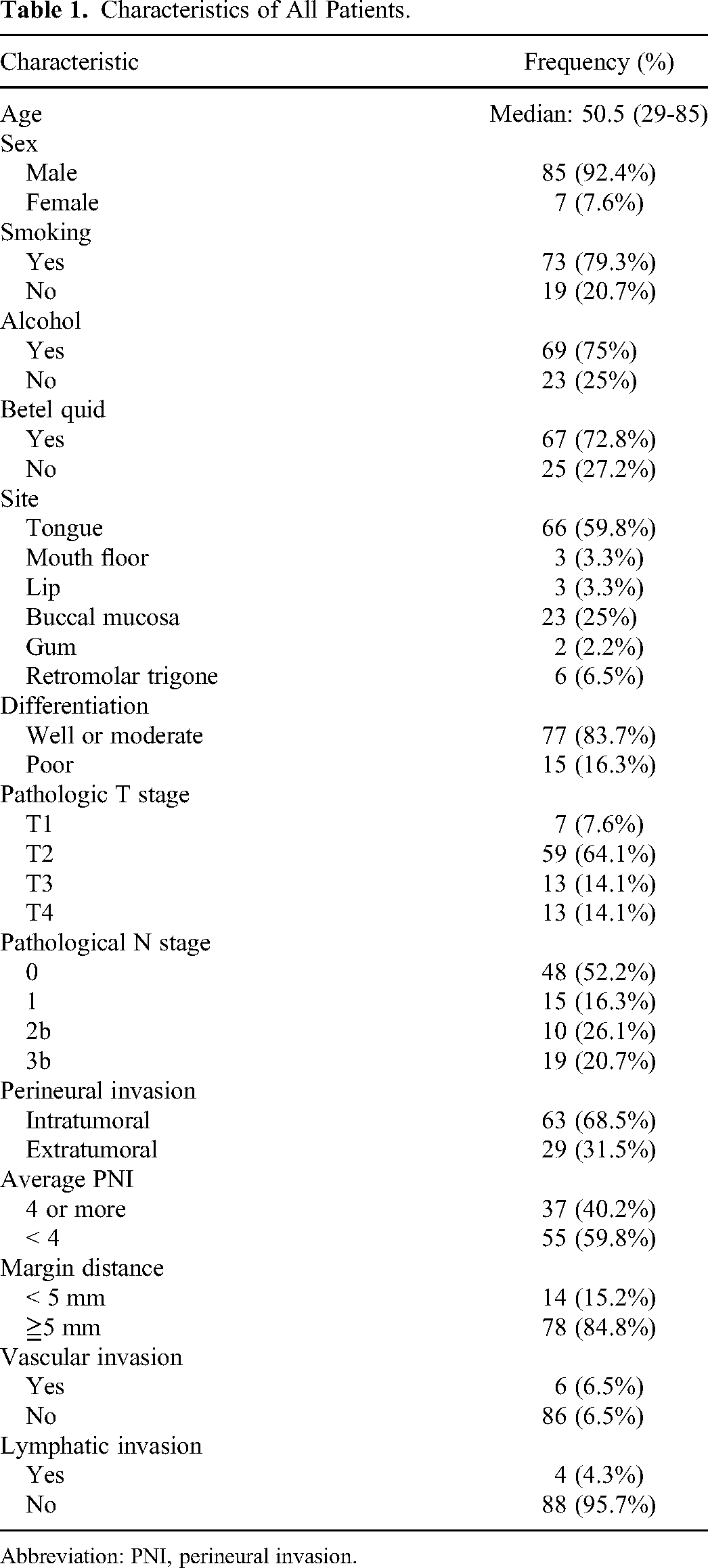
Abbreviation: PNI, perineural invasion.
The median follow-up was 59.5 months (range: 3-105 months). At the time of analysis, 71 patients were alive. Of the 21 patients who died, 12 died from cancer and the rest died from comorbidities or a second primary cancer. The 3-year OS rate was 82.5%. Pathological T4 stage and pathological N2-3 stage correlated with OS in the univariate analysis (P < .05). OS rates according to different characteristics are listed in Table 2. Pathological N2-3 stage was the only independent prognostic factor in the multivariate analysis (P < .05, Table 3).
Outcome by Patient Characteristic and Treatment Parameter.
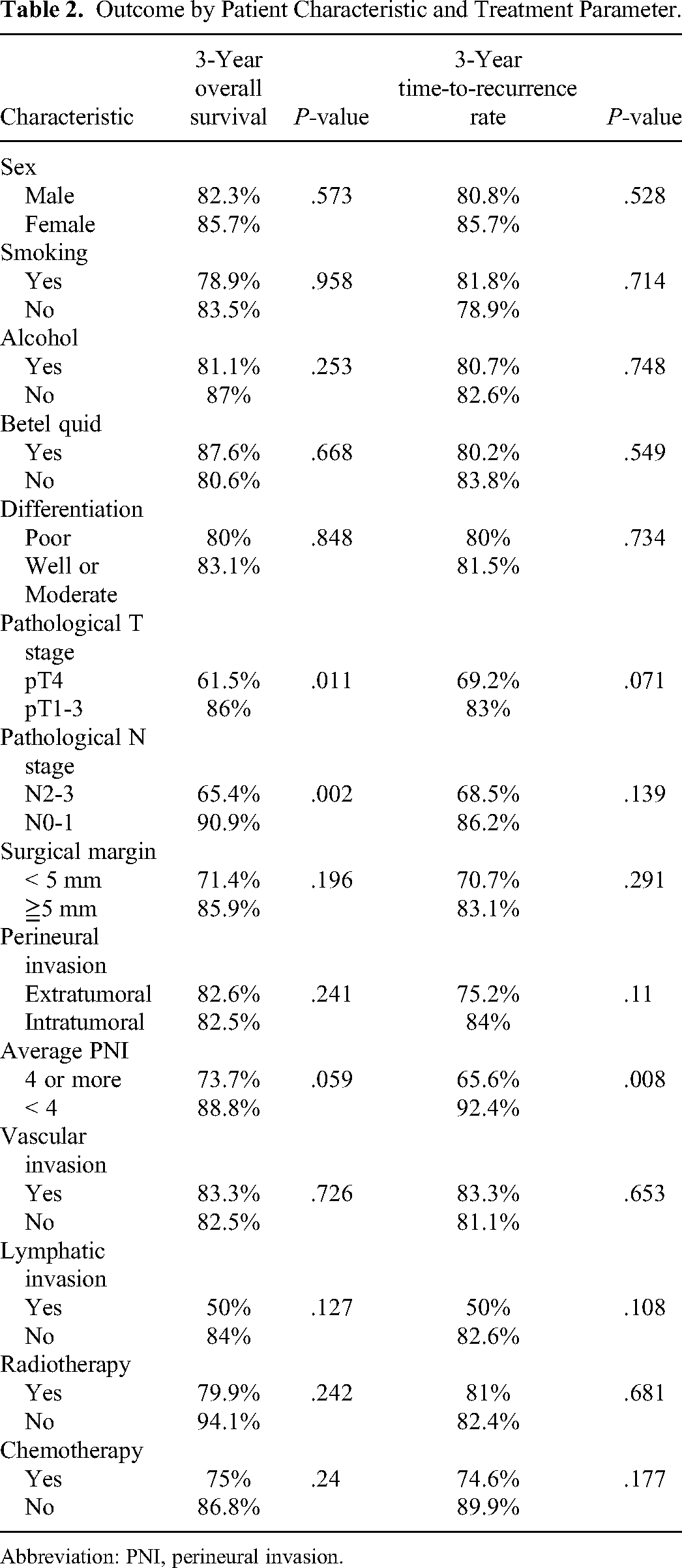
Abbreviation: PNI, perineural invasion.
Multivariate Analysis of Risk Factors for 3-Year Overall Survival and Disease-Free Survival.
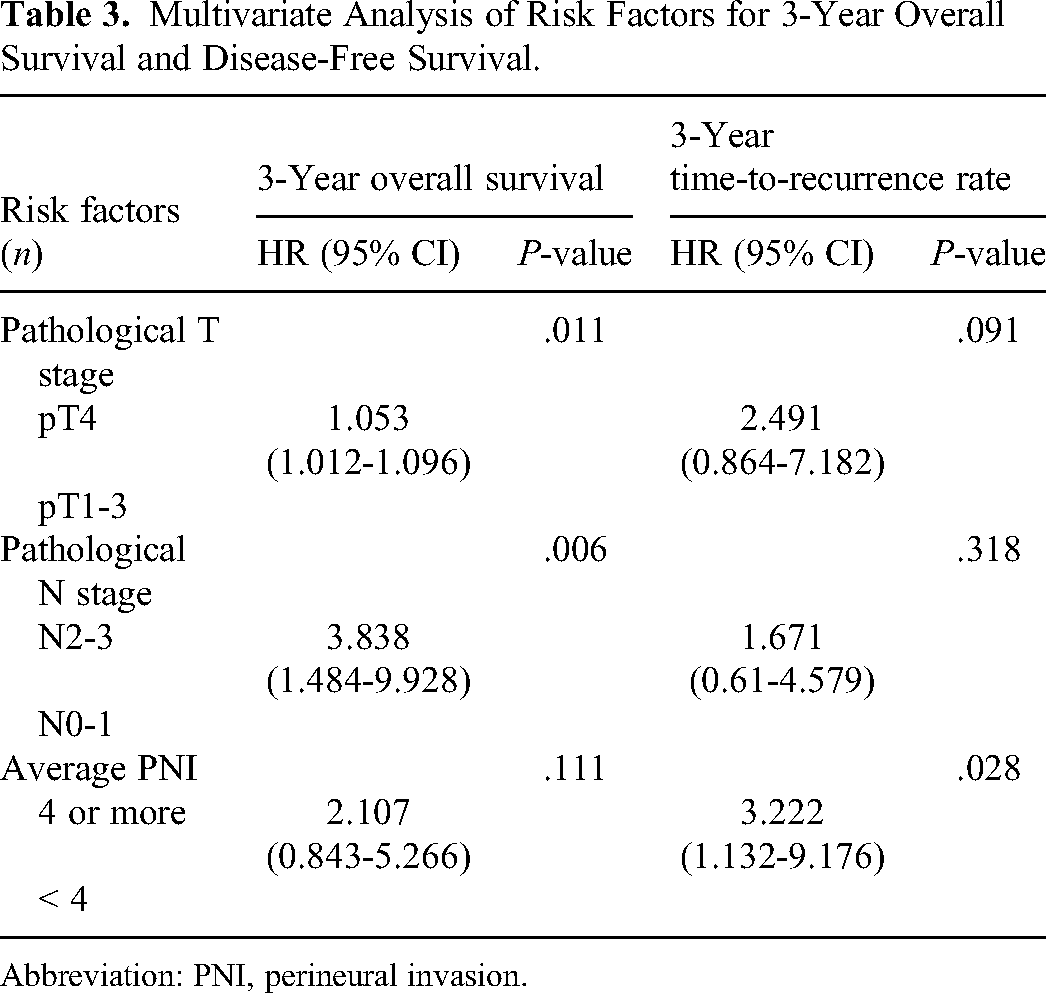
Abbreviation: PNI, perineural invasion.
The 3-year TTR rate was 81.2%. Tumor recurrence was observed in 22 patients. Nodal recurrence was the most common type of recurrence (n = 17), followed by local recurrence (n = 10) and distant metastasis (n = 9). APNI ≥4 was the only prognostic factor significantly correlated with TTR in univariate (Table 2) and multivariate analyses (P < .05, Table 3). The 3-year TTR rates were 92.4% and 65.6% for diseases with APNI < 4 and ≥ 4, respectively, (P = .008, Figure 2) TTR.
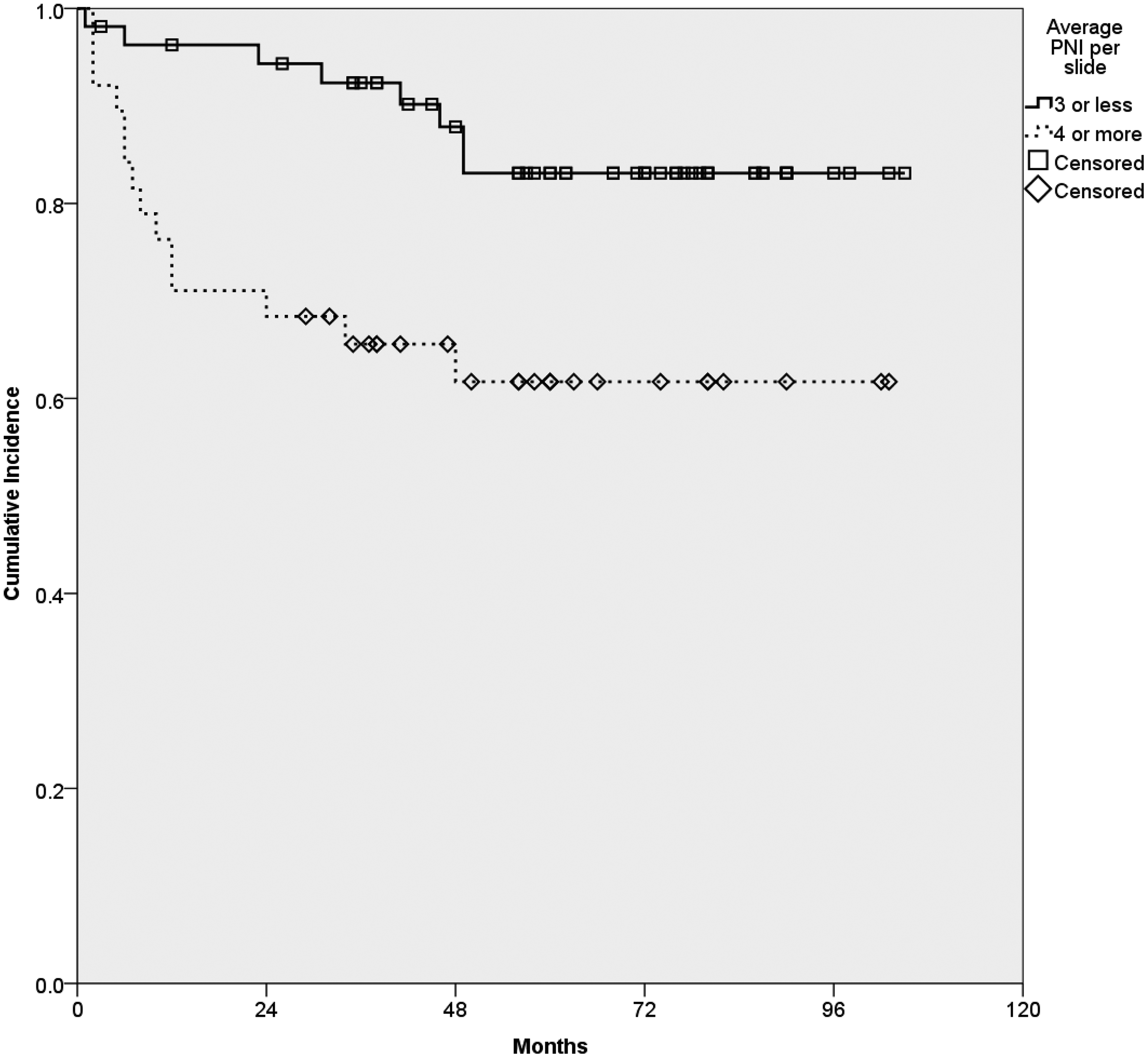
Kaplan–Meier curves of time-to-recurrence according to average perineural focus per slide.
Discussion
In the present study, we showed that quantification of PNI by S100 IHC staining could be a useful prognostic factor for OSCC with PNI. A higher APNI per slide correlated with poorer TTR, and PNI ≥ 4 per slide was identified as the cut-off point.
Studies have shown different correlations between PNI and prognosis.19,20 The presence of PNI is a crude description of disease status. Considerable differences in the methodology of data collection and risk factor identification may have led to inconsistent study results.21,22 The lack of detailed PNI assessment may further confound the results of analyses. To clarify the pattern of PNI, Miller et al 12 proposed a classification according to the extent of PNI. The extent of PNI can be assessed by measuring the distance from each focus of invasion to the tumor edge. The results demonstrated that disease-free survival was longer in patients with IPNI than in those with EPNI. The eighth edition of the AJCC Staging Manual proposed dividing PNI into 2 groups, namely, IPNI and EPNI.23,24
PNI quantification can provide a detailed assessment of PNI. The amount of PNI correlates with survival outcomes in many malignancies. Wu et al 25 reported that the identification of more than 3 PNI foci after radical prostatectomy for prostate cancer is associated with a higher rate of biochemical recurrence. In oral cavity cancer, Wei et al 26 demonstrated that the number of PNI foci is a novel parameter for PNI quantification in early OSCC. A higher number of PNI foci identified in pathologic samples are associated with decreased disease-specific survival. Aivazian et al 27 showed that multifocal PNI is associated with a worse prognosis irrespective of postoperative RT. These studies support that quantification of PNI extent to predict prognosis is feasible.
To precisely measure the amount of PNI, sensitive methods for the detection of nerves in and around the tumor are necessary. IHC staining for S100, a nervous system-specific cytoplasmic protein, can detect the location of nerves, which provides a sensitive method of PNI identification. Conte et al 14 showed that S100 staining could detect PNI that was undetected by H&E staining in colorectal cancer. In 151 patients, the incidence of PNI increased from 30.5% to 40.4%. Berlingeri-Ramos et al 28 also demonstrated that S100 staining improves the detection rate of PNI, especially for small nerves. These studies support the use of S100 IHC staining to increase the PNI detection rate.
Several mechanisms underlying PNI were recently proposed. Some head and neck cancers secrete neurotrophic factors, which can cause interactions between cancer cells and Schwann cells or neurons, such as de-differentiation and cancer cell recruiting.29,30 Cell migration, changes in motility, and mesenchymal transition also affect PNI in different malignancies.31,32 Amit et al 33 reported an association between the reprogramming of neurons and cancer growth in head and neck cancer. These pathophysiological theories may provide new treatment strategies to reduce the risk of recurrence. In this study, we did not identify a specific recurrence pattern correlated with PNI that would provide information regarding the molecular basis of PNI. A better understanding of the molecular mechanism underlying the development of PNI in OSCC may help clarify its prognostic effects and lead to the development of more effective therapeutic strategies in the future.34,35
While quantifying perineural invasion shows its prognostic effect on tumor control, some well-known tumor characteristics, such as T and N stages, may not be identified as independent prognostic factors. This current study selected patients with oral cavity cancer with perineural invasion who were treated using various modalities. Adjuvant treatment was performed for patients with a higher risk of recurrence. It is possible that adjuvant therapy improves treatment outcomes and offsets the negative prognostic effects of pathological T and N stages, as they are indications for adjuvant therapy. Lin et al 7 demonstrated that treatment outcomes could be similar with proper treatment arranged, except for cancers with extranodal extension or positive resection margins. On the other hand, new prognostic factors can still be discovered under current treatment guidelines. Schmitd et al 36 demonstrated new insights into the tumor–nerve relationship and its correlation with treatment outcomes under the current standard of care for oral cancer. In that study, tumor stage did not correlate with treatment outcomes either. Therefore, it is reasonable to conclude that some prognostic factors do not correlate with inferior treatment outcomes under properly designed treatment. Meanwhile, other factors can still be discovered.
The present study had several limitations including its retrospective design and the sole inclusion of patients living in a betel quid chewing endemic area. External validation is necessary to verify the clinical significance of PNI quantification. Furthermore, a prospective observational study or clinical trial should be performed to provide more reliable information.
Conclusions
In patients with OSCC with PNI, a greater extent of PNI identified by S100 staining indicated poorer TTR regardless of stage and other prognostic factors. Quantification of PNI by S100 IHC may be used for prognosis prediction. Prospective studies are warranted to verify the prognostic effect of PNI quantification, which may lead to improved treatment outcomes.
Footnotes
Acknowledgments
The authors would like to acknowledge the cancer center and cancer registry group of Chang Gung Memorial Hospital for their support of data retrieving.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was granted by the institutional review board of an associated medical foundation (201800407B0, 2018/March/22, Taoyuan, Taiwan).
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by grants CMRPG 3I0011 from the Chang Gung Memorial Hospital in Taiwan.
Human and Animal Rights
This retrospective study was a part of the clinical study approved by institutional review board No. 201800407B0 from Chang Gung Medical Foundation Institutional Review Board.
