Abstract
Cancer is a major threat to human health today. Although the existing anticancer treatments have effectively improved the prognosis of some patients, there are still other patients who cannot benefit from these well-established strategies. Reprogramming of lipid metabolism is one of the typical features of cancers. Recent studies have revealed that key enzymes involved in lipid metabolism may be effective anticancer therapeutic targets, but the development of therapeutic lipid metabolism targets is still insufficient. ECHS1 (enoyl-CoA hydratase, short chain 1) is a key enzyme mediating the hydration process of mitochondrial fatty acid β-oxidation and has been observed to be abnormally expressed in a variety of cancers. Therefore, with ECHS1 and cancer as the main keywords, we searched the relevant studies of ECHS1 in the field of cancer in Pubmed, summarized the research status and functions of ECHS1 in different cancer contexts, and explored its potential regulatory mechanisms, with a view to finding new therapeutic targets for anti-metabolic therapy. By reviewing and summarizing the retrieved literatures, we found that ECHS1 regulates malignant biological behaviors such as cell proliferation, metastasis, apoptosis, autophagy, and drug resistance by remodeling lipid metabolism and regulating intercellular oncogenic signaling pathways. Not only that, ECHS1 exhibits early diagnostic and prognostic value in clear cell renal cell carcinoma, and small-molecule inhibitors that regulate ECHS1 also show therapeutic significance in preclinical studies. Taken together, we propose that ECHS1 has the potential to serve as a therapeutic target of lipid metabolism.
Keywords
Introduction
Cancer is a major public health problem that needs to be addressed rapidly and is a significant barrier to increasing human life expectancy. The global cancer burden as estimated by GLOBOCAN shows an estimated 18.1 million new cases and 10 million deaths in 2020. 1 Surgical procedure is the principal strategy for patients with early or intermediate stage cancers. 2 However, for patients with postoperative recurrence or advanced stages, chemotherapy combined with targeted therapy and/or immunotherapy is the primary treatment modality, the combined strategy has greatly improved the prognosis of some patients3–5 and even changed the treatment paradigm for some types of cancers.6–8 Nevertheless, there are still some patients who have difficulty benefiting from the existing treatment strategies.9,10 Therefore, the exploration of new anticancer therapeutic targets or combination therapeutic strategies remains the focus of current research. In recent years, metabolism reprogramming has been identified as an important step in tumorigenesis, 11 which includes alterations in carbohydrate, lipid and amino acid metabolism. 12 Among them, lipid metabolism reprogramming is one of the typical features of cancers. 13 In cancers, reprogramming of lipid metabolism is not only involved in the formation of cellular biofilms and the production of adenosine 5′-triphosphate (ATP), but importantly in the generation of biologically active lipid signaling molecules (also known as second messengers), such as fatty acids, ceramides, and sphingolipids, which are responsible for regulating complex signaling networks that modulate the malignant phenotypes of cancer cells. 14 This makes targeting key molecules of lipid metabolism a promising anticancer strategy. 15
T he reprogramming of lipid metabolism is mainly manifested by enhanced de novo synthesis of fatty acids (FAS) and β-oxidation of fatty acids (FAO). 16 The dysregulated FAS has been extensively studied for its involvement in the survival and viability of cancer cells,17–20 and inhibitors targeting metabolic enzymes in this process have shown promising therapeutic potential. For example, acetyl-CoA carboxylase (ACC), one of the rate-limiting enzymes in FAS, is overexpressed and exerts pro-carcinogenic effects in a variety of cancers,21–23 and its inhibitor ND-646 effectively inhibits adipogenesis and cell growth in preclinical models of non-small cell lung cancer. 24 Compared to FAS, FAO, as an important source of nicotinamide adenine dinucleotide (NADH), 1,5-dihydroflavin adenine dinucleotide (FADH2), nicotinamide adenine dinucleotide phosphate (NADPH) and ATP in cells, 25 and its importance in cancer development has only been gradually revealed in recent years. Carnitine palmitoyltransferase 1 (CPT1) is a rate-limiting enzyme of FAO that mediates the translocation of long-chain acyl-CoAs from the cytoplasm into the mitochondria, and its overexpression is associated with cancer progression. 26 The inhibitor of CPT1, etomoxir, effectively inhibits the cell proliferation of bladder, 27 cervical, 28 and prostate cancer 29 in vitro. Overall, essential lipid metabolism enzymes that played a key role in carcinogenesis and progression are effective therapeutic targets. 30 However, at present, research on therapeutic lipid metabolism targets and related drugs is still insufficient.
ECHS1 is an enzyme in FAO that facilitates the hydration process of short-chain enoyl-CoAs in the mitochondrial β-oxidation helix of fatty acids degradation. 31 Earlier studies on ECHS1 have focused on metabolism-related disorders such as Leigh syndrome.32–34 With the first discovery of ECHS1 down-regulation in hepatocellular carcinoma (HCC) by Kurokawa et al, 35 there is increasing evidence that ECHS1 is aberrantly expressed and exerts pro- or anti-cancer activity in different cancer contexts by regulating various malignant biological behaviors.36–42 Moreover, the expression level of ECHS1 is related to the early diagnosis and prognosis evaluation of cancer,43,44 and small molecule inhibitors that regulate ECHS1 also show therapeutic significance in preclinical studies,45,46 suggesting that ECHS1 may be an effective target for anti-metabolic therapy. However, the research content of ECHS1 in cancer is not yet comprehensive and more exploration is still needed. Therefore, this paper reviews the research status of ECHS1 in the field of oncology, expounds the biological functions of ECHS1 and its possible mechanism of action, and proposes potential research directions for the further exploration of ECHS1, provides evidence for ECHS1 as a candidate marker for early diagnosis and prognosis evaluation of tumors, and is expected to offer new targets for anti-metabolic therapy.
Molecular Structure and Basic Function of ECHS1
FAO mainly occurs in mitochondria, and Acyl-CoA shortens 2 carbon atoms in each cycle to form Acetyl-CoA through 4 repeated enzymatic reactions of dehydrogenation, hydration, dehydrogenation, and thiolysis. 47 The generated Acetyl-CoA enters the tricarboxylic acid cycle and is completely oxidized and decomposed to generate 5′-triphosphate (ATP) (Figure 1). 47 Among them, ECHS1 is a key enzyme that mediates the hydration process of medium- and short-chain fatty acids (C4-C6), and catalyzes the conversion of trans-Δ2-enoyl-CoA thioesters to 3-hydroxyacyl-CoA thioesters (Figure 1). 31 The nuclear gene encoding ECHS1, approximately 11 kb in size, is located on human chromosome 10q26.2-q26.3 and expressed in high abundance in diverse human tissues (Figure 2). 48 The gene fragment consists of 8 exons, of which exon I and exon VIII, respectively, contain the 5′ and 3′ non-transcribed regions, and the 2 major transcription start sites of ECHS1 gene are located 62 and 63 bp upstream of the transcription start codon. 48 ECHS1 gene is transcribed to form a 1.4 kb sized mRNA that is translated into a protein precursor consisting of 290 amino acids in the cytoplasm. 48 The ECHS1 precursor protein then recognizes and binds to the import receptor Tom20 of the translocase of the outer membrane of mitochondria (TOM) complex with the assistance of the arylhydrocarbon receptor-interacting protein (AIP), and then translocated into the mitochondrial matrix via the channel protein Tom40 (Figure 3a).49,50
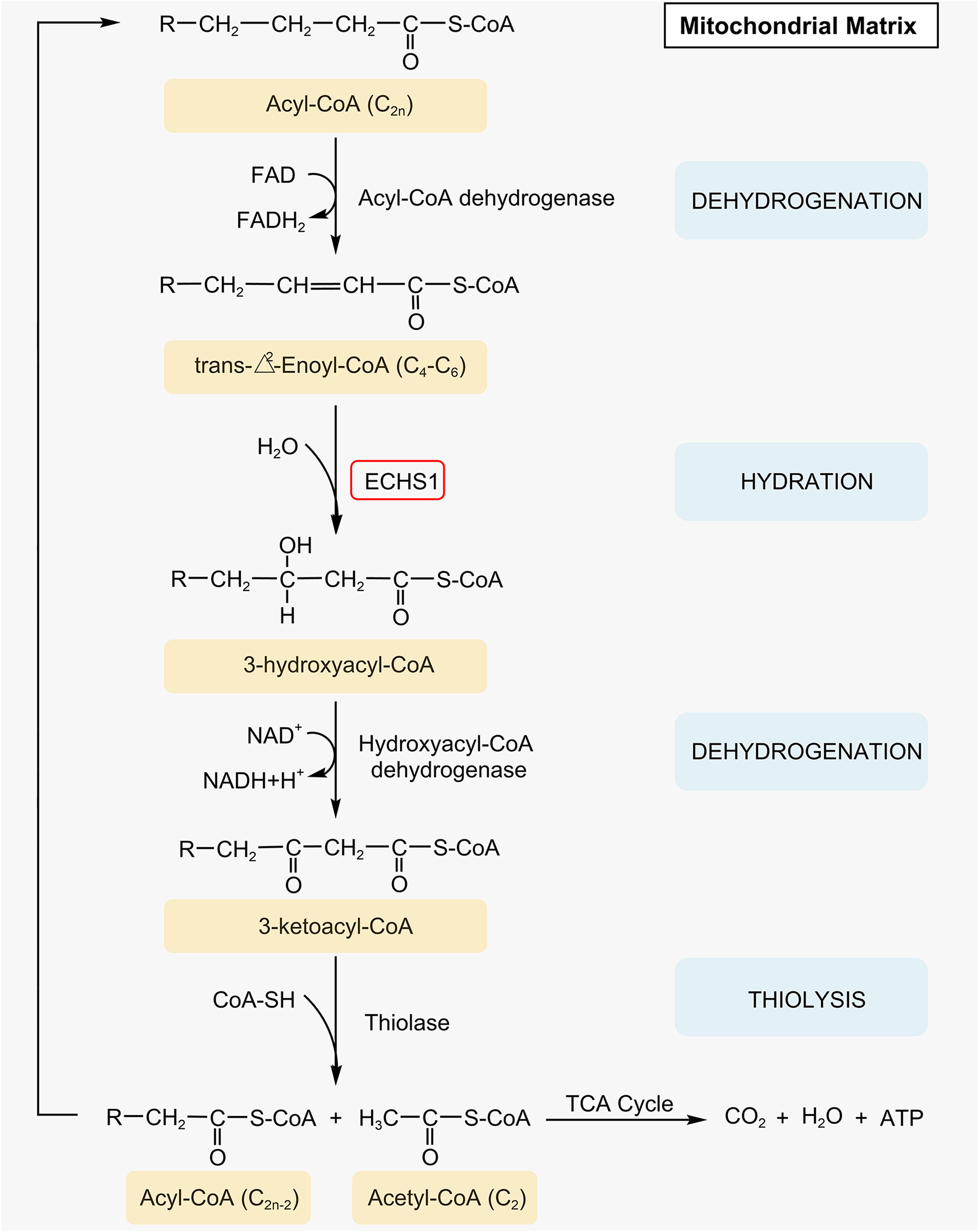
The process of mitochondrial FAO. Dehydrogenation: Acyl-CoA dehydrogenase dehydrogenates C-2 and C-3 of Acyl-CoA (C2n) and forms a double bond between them to generate trans-Δ2-enoyl-CoA. Hydration: ECHS1 hydrates the double bond between C-2 and C-3 on medium- and short-chain trans-Δ2-enoyl-CoA (C4-C6) to generate 3-hydroxyacyl-CoA. Dehydrogenation: 3-hydroxyacyl-CoA dehydrogenase dehydrogenates C-2 of 3-hydroxyacyl-CoA again to generate 3-ketoacyl-CoA. Thiolysis: Under the action of thiolase, HSCoA cleaves 3-ketoacyl-CoA and inserts a sulfhydryl group between its C-2 and C-3, resulting in a molecule of Acyl-CoA (C2n−2) shortened by 2 carbon atoms and a molecule of Acetyl-CoA (C2) containing 2 carbon atoms, the former continues the FAO cycle until the entire fatty acid chain is oxidatively decomposed into several acetyl CoA units, and the latter enters the tricarboxylic acid cycle to completely oxidize energy.
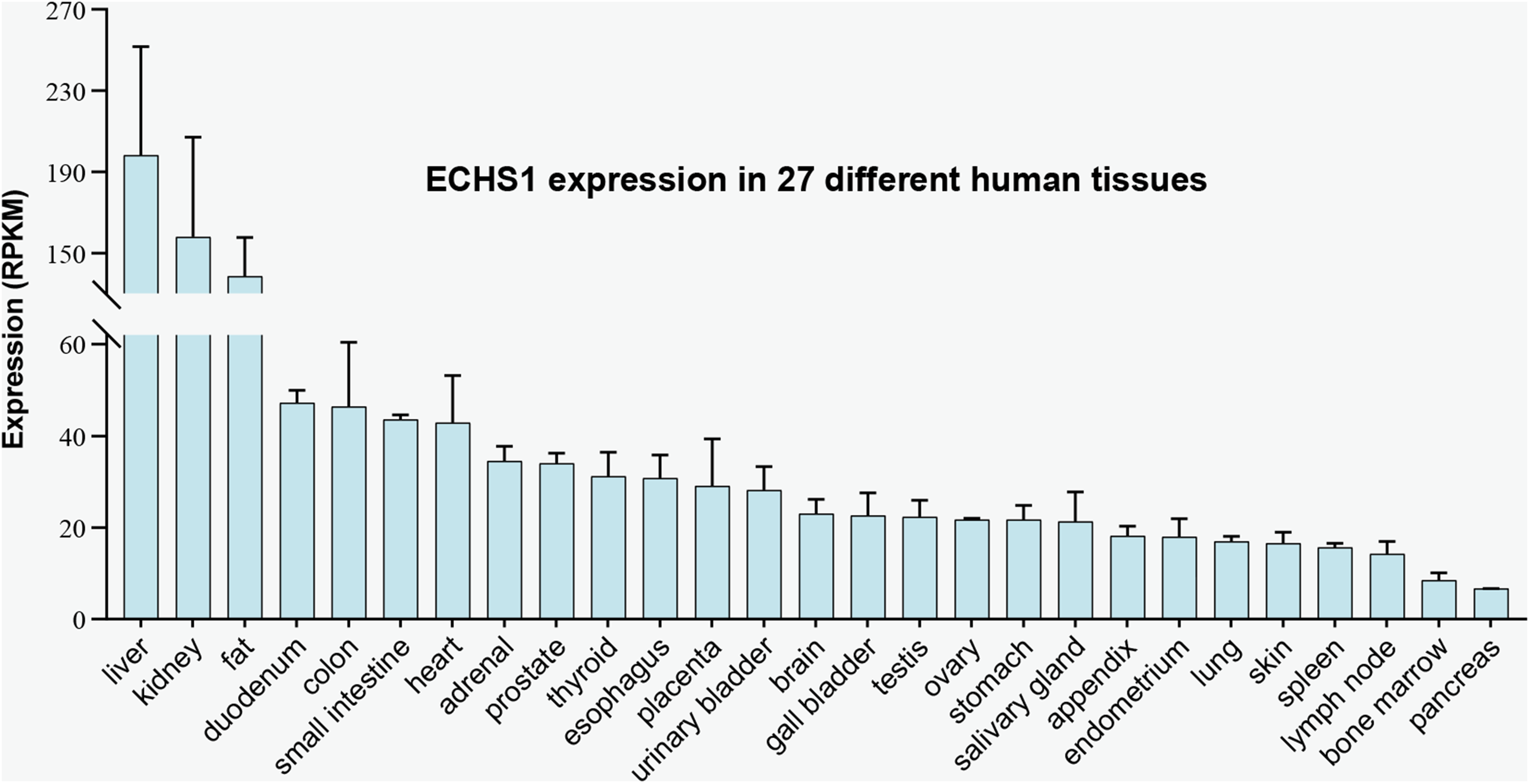
Expression of ECHS1 gene in human tissues. Reads per kilobase million (RPKM) values, derived from RNA-seq analysis of tissue samples from 95 human individuals representing 27 different tissues, were extracted from NCBI with Gene ID: 1892 (accessed on May 15, 2022).

Synthesis, translocation of ECHS1 and its functions in cancer. (a) The mRNA transcribed by ECHS1 gene in the nucleus is transported to the cytoplasm, then translated into the ECHS1 protein precursor. Later, ECHS1 protein recognizes and binds to the import receptor Tom20 of the TOM complex with the assistance of AIP and then translocated into the mitochondrial matrix via the channel protein Tom40. The functions of ECHS1 in cancer including (b) proliferation and metastasis, (c) cell cycle and apoptosis, (d) drug resistance, (e) autophagy, and (f) reprogramming of lipid metabolism.
ECHS1 exists as a 188 kDa homohexameric enzyme in mitochondria.51,52 In addition to FAO, ECHS1 also showed certain activities on the degradation of branched-chain amino acids, such as valine, leucine, and isoleucine.53,54 Based on these functions, in non-neoplastic diseases, ECHS1 deficiency not only reduces ATP production by inhibiting FAO activity to increase susceptibility of organ dysfunction, 32 but also induces the accumulation of toxic substrates by blocking branched-chain amino acid metabolism, such as methacrylate-CoAs and arylate-CoAs, which disrupts pyruvate dehydrogenase complexes and electron transport chains, 33 ultimately leading to patients presenting with Leigh syndrome typified by mental retardation or degeneration, cardiomyopathy, dystonia, epilepsy, and hyperlactatemia. 34 In cancers, aberrant expression of ECHS1 has been widely observed (Figure 4), which regulates the malignant biological behaviors through multiple mechanisms including remodeling fatty acid 44 and sphingolipid metabolism, 55 regulating intercellular signaling molecules such as AMPK, 56 Akt 36 and its downstream effectors GSK-3β 57 and mTOR, 43 and interacting with binding proteins.58,59

The expression levels of ECHS1 in human tumor or normal tissues were analyzed by Timer 2.0 (http://timer.cistrome.org/) 60 . Red color represents tumor tissue, blue color represents normal tissue, and purple color represents metastatic cancer tissue.
The Functions of ECHS1 in Tumors
As research progresses, there is increasing evidence that ECHS1 has demonstrated the potential to regulate proliferation, metastasis, apoptosis, cell cycle, drug resistance, autophagy, and tumor lipid metabolism (Figure 3b to f). Next, we will elaborate on the specific functions of ECHS1 in cancers and its potential mechanisms.
Lipid Metabolism Reprogramming
Fatty acid metabolism is an important component of lipid metabolism, converting nutrients into metabolic intermediates for membrane biosynthesis, energy storage, and the production of signaling molecules. 20 ECHS1, a key enzyme in the oxidative catabolic metabolism of fatty acids, plays an important role in the reprogramming of fatty acid metabolism. In HCC, ECHS1 downregulation directly inhibits FAS by suppressing the expression of fatty acid synthase (FASN), long-chain acyl-CoA synthetase, and ATP-citrate lyase in HepG2 and Huh7 cells. 56 However, the opposite conclusion of increased FAS is obtained in another study on clear cell renal cell carcinoma (ccRCC). 44 This study discovered that AMPK, ECHS1, and its transcription factor GATA3 are all down-regulated in ccRCC, which prevents fatty acid and branched-chain amino acid oxidative catabolism via the AMPK-GATA3-ECHS1 axis, resulting in substantial buildup of both. Interestingly, the accumulated fatty acids and branched-chain amino acids have an inhibitory effect on AMPK,61,62 thus forming a mutually inhibitory positive feedback loop between ECHS1 and AMPK, which further impairs catabolism of fatty acids and branched-chain amino acids. Moreover, the accumulated branched-chain amino acids act as physiological stimulators activating mTORC1, which inhibits adipose triglyceride lipase but up-regulates ACC and FASN, ultimately leading to the toxic accumulation of lipids and fatty acids.
It is well-known that there is a wide variety of lipids, including polar lipids (such as sphingolipid, phosphatidylinositol, and phosphatidylcholine) and non-polar or low-polar lipids (such as cholesteryl ester, triglyceride, and free fatty acid). In addition to fatty acid metabolism reprogramming, ECHS1 is also associated with the metabolic alterations of sphingolipids. Ceramide, a type of sphingolipid with central biological activity, plays an important role in the regulation of cancer growth and drug resistance. 63 Li et al found by immunoprecipitation assay that highly expressed ECHS1 binds to the tumor-promoting factor LIM and SH3 protein 1, increasing the glycosylation of ceramide, reprogramming sphingolipid metabolism, thus reducing ceramide levels and inducing drug resistance in colorectal cancer (CRC) cells. 55 In summary, ECHS1-mediated reprogramming of lipid metabolism is mainly reflected in both remodeling of fatty acid and sphingolipid metabolism, and the regulation of fatty acid metabolism reprogramming by ECHS1 may have cancer species specificity.
Cell Proliferation and Metastasis
Uncontrolled autonomous cell replication and unlimited proliferation are hallmarks of cancer, 64 a process that is inextricably linked to fatty acid metabolism. 65 Fatty acids are fundamental constituents for the synthesis of biofilm lipids and are important substrates of energy metabolism for FAO 66 which is thought to be a source of ATP that promotes the rapid growth of human cancers.67–69 Xu et al found in HCC cells that knockdown of ECHS1 inhibits cell proliferation by blocking the FAO process. 56 Their further experimental results showed that blocked FAO reduces the production of ATP and NADPH, and increases the NADP + /NADPH ratio, thereby activating AMPK. AMPK is involved in maintaining the balance between NADPH depletion and production responses, 70 and its activation inhibits cancer proliferation through multiple signaling pathways such as the TSC-mTOR axis.71,72 Thus, ECHS1 mediates the malignant proliferative phenotype of cancers through the regulation of FAO, which may be associated with the activation of the energy receptor AMPK.
Local invasion and metastasis are the basic characteristics of cancer cells. 64 FAO participates in the metastatic process of cancers through various mechanisms, including regulating the function of tumor stem cells to promote the metastatic phenotype, 73 and increasing ATP and NADPH levels to assist cells to overcome the oxidative stress and anoikis associated with detachment from the stroma. 74 The expression level of ECHS1 can directly affect the activity of FAO. 51 At this level, ECHS1 has some pro-metastatic potential. Li et al found that endogenous overexpression of ECHS1 significantly enhances the metastatic and invasive ability of HCT116 cells. 55 In addition, it has been found in cancers with high expression of ECHS1, such as ovarian cancer,42,75 breast cancer,38,76 and colon cancer,37,77 that cancer cells tend to spread to adipocyte-rich tissues. Which probably since lipids stored in adipocytes can derive large amounts of fatty acids that generate ATP via FAO to facilitate rapid growth and metastasis of cancer cells. Therefore, ECHS1 may promote cancer metastasis by mediating fatty acid metabolic heterogeneity via FAO from a metabolic perspective.
In addition, abnormal lipid metabolism can alter the Akt oncogenic signaling pathway in cancer cells. 13 Akt pathway is a potent signaling intermediary that transmits signals from upstream regulatory proteins (such as PTEN, PI3K, and receptor tyrosine kinase) to downstream effectors (such as GSK-3β, mTOR, and FOXO), thereby regulating cell survival, proliferation, migration, and metabolic processes. 78 Accumulating studies have revealed that ECHS1 has regulatory effects on Akt signaling networks. Specifically, knockdown of ECHS1 inhibits Akt phosphorylation to impair the proliferation and metastatic potential of HepG2 cells.36,79 Not only that, but down-regulated ECHS1 is also found in CRC 57 and gastric cancers 80 to attenuate the proliferation and metastatic ability of HCT-8 and BGC-823 cells by inhibiting phosphorylation of Akt and GSK-3β, respectively. Furthermore, in ccRCC, overexpression 43 or knockdown 44 of ECHS1, respectively, reduces or enhances cell proliferation and metastasis by inhibiting or activating mTOR. These findings suggest that regulation of Akt and its downstream effectors of GSK-3β and mTOR may be the main pathway that ECHS1 mediates cancer cell proliferation and metastasis. Currently, the specific regulatory mechanism between ECHS1 and Akt is unclear. Recent studies have found that accumulated fatty acids, such as palmitic acids 81 and punicic acids, 82 have inhibitory effects on the PI3K/Akt signaling pathway, suggesting that a fatty acid-dependent approach may be a potential mechanism for ECHS1 to regulate the Akt signaling pathway.
Cell Cycle and Apoptosis
In multicellular organisms, the maintenance of homeostasis within tissues depends on the orderly regulation of cell proliferation and death, which is achieved by coupling cell cycle processes and programmed cell death through the use and control of a shared set of factors, such as the cell cycle protein-dependent kinases (CDKs) and their inhibitors (CDKIs).83,84 The mammalian cell cycle is a highly organized and regulated process dependent on the regulation of CDKs to ensure the replication of genetic material and cell division. 85 Errors that occur at different stages of the cell cycle may induce apoptosis. 86 It was found that downregulation of ECHS1 enhances the cisplatin-induced apoptosis of HepG2 by suppressing the expression of cell cycle regulators cyclin D3 and CDK6 via inhibiting phosphorylation of Akt. 36 In addition, knockdown of ECHS1 in HCC cells represses phosphorylation of GSK-3β to suppress cyclin D3 and CDK6 while promoting the expression of CDKIs such as p16 and p21, thereby blocking the cell cycle in the G1 phase. 79 Thus, in addition to proliferation and metastasis, ECHS1 also mediates cell cycle and apoptosis through regulation of Akt and its downstream effectors.
Apoptosis is a systematic and ordered process of programmed cell death mainly mediated through intrinsic, extrinsic, and endoplasmic reticulum pathways.87,88 Mitochondria are the central organelles of the intrinsic apoptotic pathway. In response to internal stimuli such as irreparable genetic damage, hypoxia, extremely high cytoplasmic Ca2+ concentrations, and severe oxidative stress, the mitochondrial outer membrane permeabilization (MOMP) is altered, resulting in the release of multiple pro-apoptotic factors from the inner and outer mitochondrial membrane gaps into the cytoplasm, such as cytochrome C, apoptosis-inducing factor, and EndoG, thereby activating caspase-9 to trigger apoptosis. 89 It was found that knockdown of ECHS1 in SW480 cells promotes mitochondrial reactive oxygen species production and decreased mitochondrial membrane potential (MMP) to induce apoptosis by inhibiting ceramide glycosylation 55 which promotes apoptosis by affecting Bax integration through ceramide-rich macro-domains on the mitochondrial membrane90,91 and inducing MOMP. 92 Therefore, mitochondria may be a key organelle in the regulation of apoptosis by ECHS1 through metabolic pathways.
Moreover, interaction with binding proteins may be the main non-metabolic pathway of ECHS1 to mediate apoptosis. Yeast 2-hybrid and immunoprecipitation experiments conducted by Xiao et al suggested that ECHS1 is a novel binding protein for HBsAg and that both bind in the cytoplasm of HepG2 and promote apoptosis by blocking FAO to reduce MMP levels, inhibiting phosphorylation of the Akt/GSK-3β pathway, and regulating the Bcl-2 family. 58 In line with, another study identified by the Glutathione-S-Transferase pull-down experiment that ECHS1 is a binding protein for the apoptosis suppressor STAT3, 59 the first member of the STATs family 93 and that constitutively activated STAT3-induced changes in gene expression represent key molecular events in resistance to apoptosis, including upregulating the expression of the survival factors (such as Bcl-2, Bcl-xL, and Mcl-1) and the apoptosis inhibitor members (such as survivin and c-IAP2). 94 Further, the dual-luciferase reporter assay suggested that ECHS1 interacts with STAT3 and represses its transcriptional activity, phosphorylation, and the expression of its downstream target genes such as BCL2. Taken together, HBsAg and STAT3 may be pro-apoptotic binding proteins of ECHS1.
Autophagy
Autophagy plays a 2-sided role in cancer development. In the early stages, autophagy inhibits cancer formation by alleviating cellular stress, and after cancer establishment, autophagy promotes the transformation of intracellular components through keen nutrient-sensing properties, providing cells with macromolecular precursors and energy in response to nutrient deprivation95–97. In addition to metabolism reprogramming, autophagy is one of the main ways in which cancer cells respond to adverse conditions including ischemia, hypoxia, and therapeutic stress. 98 In the presence of restricted ATP availability or essential nutrient deficiency, autophagy is usually initiated with activation of the intercellular energy receptor AMPK. 99 Xu et al suggested that knockdown of ECHS1 reduces ATP production by blocking FAO to activate AMPK and thus upregulating the expression of the autophagy-related biomarker LC3-II in HepG2 and Huh7 cells. 56 Currently, although accumulated saturated and unsaturated fatty acids have been reported to directly induce autophagy, such as palmitate and oleate, 99 their mechanism of action is unclear. ECHS1 may be a regulator of fatty acid-triggered autophagy, and its potential mechanism may be related to the activation of AMPK by FAO blockade.
Drug Resistance
Systemic treatment of cancer inevitably induces drug resistance, which is a major cause of treatment failure and recurrence in cancer. 100 Drug-induced resistance is an acquired autonomous phenotypic variant that dynamically regulates the expression of drug resistance driver genes through cellular plasticity to reduce the sensitivity of cancer cells to anticancer drugs and reprograms intrinsic metabolic patterns to maintain cell survival and metastasis under drug stress. 101 FAO has now been shown to be involved in the generation of drug resistance in cancer cells.102,103 Hu et al analyzed 9 HCC cell lines and found that the expression level of ECHS1 is significantly correlated with doxorubicin resistance. It was further hypothesized that downregulation of ECHS1 expression may increase drug resistance in HCC cells by inhibiting the NADH production of FAO and reducing free radical formation, thereby attenuating the DNA damaging effects of doxorubicin. 104 Furthermore, glycosylated ceramides induce the development of cancer drug resistance by regulating drug transport, reducing apoptosis, and promoting cell proliferation.105–108 Li et al found that overexpression of ECHS1 promotes ceramide glycosylation, which in turn increases resistance to oxaliplatin/5-fluorouracil in HCT116 cells. 55 In summary, inconsistent results were obtained for the regulation of cancer resistance by ECHS1 in HCC and CRC, which may be related to different cancer types, drug species, and mechanisms of action, and still need to be further explored.
Study of ECHS1 in Various Cancers
Based on the multiple functions mediated by ECHS1 under different mechanisms, dysregulated ECHS1 exerts pro- or anti-cancer activity in different types of cancer backgrounds. The functions and mechanisms played by ECHS1 in HCC, ccRCC, and other types of cancers will be described in detail below (Table 1).
Functional Characteristics of ECHS1 in Various Human Cancers.

Hepatocellular Carcinoma
About 75% to 85% of primary liver cancers are HCC, 109 which is the sixth most common malignancy in the world and has the third-highest mortality rate. 1 The liver is the central organ of lipid metabolism with a rich system of enzymes and participates the oxidative breakdown of fatty acids and the synthesis of fatty acids and cholesterol as well as phospholipids. In 2003, Kurokawa et al first reported that ECHS1 is downregulation in HCC independent of hepatitis virus infection status compared with the normal liver and HCC tissue samples from 68 hepatitis B virus- or hepatitis C virus-negative patients at mRNA level. 35 However, another integrated microarray analysis that pooled gene expression data from 16 HCC tissues and 12 controls identified ECHS1 as the most significantly upregulated differentially expressed gene in HCC and was further validated by reverse transcription-PCR in HepG2 cells. 110 Therefore, the expression of ECHS1 in HCC remains unclear based on the available findings (Table 2).
The Controversial Expression of ECHS1 in Hepatocellular Carcinoma.

Currently, the pro- or anti-cancer role played by ECHS1 in HCC is still controversial (Table 1). It was found that downregulated ECHS1 may inhibit the cell cycle proteins of cyclin D3 and CDK6 by attenuating phosphorylation of the Akt/GSK-3β pathway, thereby impairing the proliferation and metastasis of HCC cells,36,79 which inversely reveals the pro-cancer role of ECHS1 in HCC. Interestingly, ECHS1 may exert cancer suppressive potential in response to stimulation by binding proteins or chemotherapy drugs. Specifically, ECHS1 binding to the HBsAg or STAT3 promotes apoptosis by decreasing MMP levels, inhibiting phosphorylation of the Akt/GSK-3β pathway, and regulating the Bcl-2 family.58,59 In addition, reduced ECHS1 enhances the resistance of HCC cells to doxorubicin, and the underlying mechanism may be related to FAO blockade. 104 Collectively, contextual relevance is needed to clarify the pro- or anti-cancer role of ECHS1 in the different contexts of HCC.
Clear Cell Renal Cell Carcinoma
Metabolic abnormalities are an important factor in the pathogenesis of renal cell carcinoma. 111 ccRCC is the most common type of renal cell carcinoma, and aberrant fatty acid metabolism and pathological accumulation of intracellular lipid droplets are its basic features. 112 ECHS1 expression is downregulated in ccRCC and plays a role as a tumor suppressor (Table 1). Qu et al investigated 367 pairs of ccRCC tissues and corresponding normal kidney tissues and determined that ECHS1 expression is downregulated in ccRCC at the mRNA and protein levels. 44 Experimentally, downregulation of ECHS1 blocked oxidative catabolism of fatty acids and branched-chain amino acids via the AMPK-GATA3-ECHS1 axis, which further activated mTORC1 to upregulate ACC and FASN to enhance FAS and inhibit adipose triglyceride lipase to induced cytotoxic lipids accumulation. 44 Overexpression of ECHS1 mediates proliferation and metastasis inhibition in ACHN cells by inhibiting phosphorylation of mTOR. 43 In terms of clinical significance, the expression level of ECHS1 well distinguishes ccRCC tissues in general and in stage I from adjacent normal tissues. 43 In addition, Cox regression analysis and survival analysis suggested that ECHS1 is an independent prognostic factor for progression-free survival (PFS) and overall survival (OS) in ccRCC, respectively, and that ECHS1 downregulation is associated with poorer PFS and OS. 44 This evidence suggests that ECHS1 functions as a suppressor in the development of ccRCC and has the potential as a biomarker for early diagnosis and prognosis.
Other Types of Cancers
Immunohistochemistry technique and/or proteomic analysis showed that ECHS1 is commonly highly expressed in CRC, 37 breast cancer, 38 gastric cancer, 39 lung cancer, 40 prostate cancer, 41 and ovarian cancer 42 (Table 1). Downregulation of ECHS1 attenuates the proliferation and metastatic potential of CRC HCT-8 cells and gastric cancer BGC-823 cells by inhibiting the Akt/GSK-3β pathway.57,80 Moreover, ECHS1 is downregulated as a target of miR-548a-3p in breast cancer MCF-7 and ZR-75-30 cells, which in turn mediates apoptosis resistance and proliferation inhibition. 38 Importantly, overexpression of ECHS1 regulates sphingolipid metabolism to inhibit autophagy, apoptosis, and promote chemotherapeutic drug resistance. 55 Clinically, high expression of ECHS1 is associated with poor tissue differentiation, distant lymph node metastasis, later TNM stage, and poorer OS in CRC. 37 Another study similarly suggested that the expression level of ECHS1 was negatively correlated with OS and disease-free survival in CRC. 55 Taken together, from basic to clinical, these results suggest that ECHS1 plays a pro-cancer role in a variety of cancers and is expected to be a biomarker for cancer prognosis assessment.
ECHS1 as a Therapeutic Target
Metabolism reprogramming confers a survival advantage to cancer cells but also provides therapeutic opportunities. 113 The available researches results provide a molecular basis for the treatment of targeting ECHS1. ECHS1 mediates activation of PI3K/Akt/mTOR signaling pathways in diverse cancers, indicating that targeting ECHS1 in combination with mTOR inhibitors may have synergistic anticancer effects.44,55,114 It is worth mentioning that small molecule drugs regulating ECHS1 have also demonstrated good anticancer effects in preclinical studies. 4-amino-5-(4-chlorophenyl)-7-(t-butyl)pyrazolo[3,4-d]pyrimidine (PP2) enhances the pro-apoptotic effect on breast cancer cells by suppressing the expression of ECHS1. 45 Moreover, Remoldin reverses the fatty acids metabolism reprogramming and lipids accumulation in CRC and breast cancer by downregulating ECHS1 to inhibit fatty acids prolongation and FAO in mitochondria. 46 In addition, metabolic alterations in cancer cells may increase their drug resistance, and studying the role of metabolism reprogramming in treatment-resistant states may also lead to a new therapeutic window. 115 As previously described, ECHS1 is involved in doxorubicin resistance of HCC cells and oxaliplatin/5-fluorouracil resistance of CRC cells, suggesting that targeting ECHS1 has the potential to overcome chemotherapeutic drug resistance.55,104 In summary, these results provide a research basis for the development of ECHS1-targeted drugs, making it promising as a new target for anticancer therapy.
Discussion and Outlook
Metabolism reprogramming is a hallmark of cancer and targeting metabolism is a potential therapeutic strategy. 116 The earliest described alteration in cancer metabolism was the Warburg effect, discovered by the German biochemist Otto Warburg, whereby normal cells produce energy primarily through mitochondrial oxidative phosphorylation, whereas cancer cells prefer the glycolytic pathway even under aerobic conditions. 117 This process supports cancer growth through the biosynthesis of metabolites. 118 However, drugs targeting aerobic glycolysis have never been successfully developed clinically, especially the use of 2-deoxyglucose, which, despite its ability to inhibit glycolysis, has proven to have limited efficacy and adverse side effects.119,120 As research progressed, it became clear that in cells undergoing malignant phenotypic transformation, lipid metabolism is often dramatically altered to meet the plasma membrane synthesis and energy production required for rapid proliferation. 121 Based on this, lipid metabolism reprogramming takes the stage of cancer metabolism. Currently, small molecule inhibitors targeting the reprogrammed lipid metabolism process have shown therapeutic promises, such as SB-204990 (ATP-citrate lyase) 122 and TVB-2640 (FASN), 123 and some of them have even entered clinical trials. Nevertheless, metabolic targets targeting the reprogramming of lipid metabolism, especially the FAO process, are still underdeveloped.
In this paper, we propose that ECHS1 could be a new metabolic therapeutic target, which is a mitochondrial enzyme that mediates the short-chained enoyl-CoAs hydration process in FAO. Through a review of previous literature, we found that small-molecule inhibitors with modulatory effects on ECHS1 have demonstrated promising anti-cancer effects in preclinical studies, such as PP2 45 and Remoldin, 46 suggesting that ECHS1 has potential as a therapeutic lipid metabolism target. However, based on the plasticity of cancer metabolism, the metabolic complexity of different cell types in the tumor microenvironment (TME), and the ill-defined metabolic crosstalk between various cellular components, targeting a single metabolic enzyme or specific metabolic pathway may not fully exploit the potential of metabolism in cancer treatment. 124 Iwamoto et al reversed the reprogramming of FAO induced by anti-angiogenic drugs through applying CPT1 inhibitors, which significantly enhances the anti-cancer effects of anti-angiogenic drugs, and proposed that combined targeting of lipid metabolism and anti-angiogenesis is an effective anti-cancer strategy. 125 This suggests that a combination therapy with chemotherapy, targeted therapy, or immunotherapy as the mainstay and anti-lipid metabolism therapy targeting ECHS1 as a supplement may be a meaningful research content in the future.
Currently, ECHS1 is aberrantly expressed in a variety of cancers and mediates the malignant biological behaviors of cancer cells by remodeling lipid metabolism and regulating intercellular oncogenic signaling pathways. Meanwhile, it also showed the value of early diagnosis and prognostic evaluation, suggesting that ECHS1 has the potential to become a candidate marker for early diagnosis and prognostic evaluation of cancers. However, there are still some limitations. First, there is a lack of high-grade evidence to clarify the expression levels of ECHS1 in different cancers, especially HCC. Second, more in-depth basic research is lacking to illustrate the specific mechanism by which ECHS1 regulates the intercellular oncogenic signaling pathway. Further exploration of related directions may help us better grasp the anti-lipid metabolism therapy targeting ECHS1. In addition, more comprehensive studies to elucidate the full functions of ECHS1 in cancers are lacking. Recent studies have found that inflammatory factors may be involved in inducing changes in lipid metabolism in cancer cells, 126 suggesting that the interaction between inflammatory cytokines and ECHS1 in cancer may be a promising research direction in the future. Furthermore, existing cancer-related studies in ECHS1 have mainly focused on cancer cells, while studies in other cellular components of the sophisticated TME remain vacant, especially in immune cells. Recent studies have found that regulatory T cells, 127 as well as tumor-associated macrophages, 128 can drive tumor immunosuppression by reshaping their own lipid metabolism. Therefore, in order to sufficiently exploit the full potential of various well-established anticancer therapies and realize the clinical application of anti-lipid metabolism therapy, it will be inevitable to further explore the related lipid metabolism molecules including ECHS1 in other cellular components of TME in the future.
Conclusion
As mentioned above, ECHS1, which is abnormally expressed in cancers, exerts pro- or anti-cancer activities in different cancer backgrounds by regulating a variety of malignant biological behaviors, and shows potential as a candidate marker for early diagnosis and prognostic evaluation as well as a target for anti-lipid metabolism therapy, which has space and value for in-depth research. Although there are still many challenges, we believe that anti-lipid metabolism therapy targeting ECHS1 is promising as a new anticancer approach in the future.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Project of Department of Science and Technology Sichuan Province (2020JDTD0036).
