Abstract
Colorectal adenoma is a precancerous lesion that may progress to colorectal cancer. Patients with colorectal adenoma had a 4-fold higher risk of developing colorectal malignancy than the rest of the population, with approximately 80% of colorectal cancer originating from colorectal adenoma. Therefore, preventing the occurrence and progression of colorectal adenoma is crucial in reducing the risk for colorectal cancer. The human intestinal microecology is a complex system consisting of numerous microbial communities with a sophisticated structure. Interactions among intestinal microorganisms play crucial roles in maintaining normal intestinal structure, digestion, absorption, metabolism, and other functions. The colorectal system is the largest microbial bank or fermentation system in the human body. Studies suggest that intestinal microecological imbalance, one of the most important environmental factors, may play an essential role in the occurrence and development of colorectal adenoma and colorectal cancer. Based on the complexity of studying the gut microbiota ecosystem, its specific role in the occurrence and development of colorectal adenoma is yet to be elucidated. In addition, further studies are expected to provide new insights regarding the prevention and treatment of colorectal adenoma. This article reviews the relationship and mechanism of the diversity of the gut microbiota, the relevant inflammatory response, immune regulation, and metabolic changes in the presence of colorectal adenomas.
Keywords
Introduction
Colorectal adenoma (CRA) generally refers to all kinds of protuberant lesions growing in the mucosal surface of the colon and rectum and protruding into the intestinal lumen. 1 It is a precancerous lesion of colorectal cancer (CRC). According to different pathological types, adenomas are further subdivided into villous, tubular villous, and tubular types. 2 Since the view of “adenoma-carcinoma” sequence of CRC was proposed, it has been gradually recognized by the public, and it is involved in the occurrence of tumors through different mechanisms.
When the adenoma volume is large or the pathology is villous or the proliferation is fast, the risk of cancer also increases. 3 It is generally believed that it will take 10 to 15 years for CRA to develop into CRC. 4 Therefore, identifying high-risk groups as early as possible is the most important form of intervention for reducing the incidence of CRC. In fact, the incidence and fatality rates of CRC in China have been increasing for the past few years, which are threatening public health and contributing to the increase of disease burden. 5
The intestinal microbiome is composed of the gut microbiota and its intestinal environment. The intestinal microbiome consists of at least 100 trillion bacteria, 10 times the total number of human cells, belonging to more than 1000 species and weighing an average of 2 kg. Human gut microbiota can be classified based on its source and divided into original and foreign flora, originally from the flora of normal intestinal flora. In addition to the dominant bacteria, intestinal microbe group includes viruses, fungi, and helix group, which are combined according to certain proportion, and restrict and depend on each other to maintain ecological balance.
Any factor that causes the dysregulation of the gut microbiota may lead to the change of species and quantity of the flora. 6 The fecal microflora profiles of patients with non-adenomatous colon polyps, colorectal adenomatous polyps, and colon cancer were significantly changed, and with the progression of the disease, their intestinal pH increased and the content of short-chain fatty acids (SCFAs) decreased, which was not a secondary change of colon cancer, but its initiatory factor. 7 Nowadays, many studies have illustrated that the number and species of bacteria in the cancer tissues or feces of patients with CRC are greatly altered.
In order to effectively reduce the incidence of CRC, researchers have been focusing more on the intestinal microecology of CRA, an increasingly trending research topic. Therefore, it is crucial to explore intestinal microecology for early clinical intervention and active prevention of cancer.
Composition of Gut Microbiota
A large number of microorganisms reside on the surface and inside the human body, collectively referred to as “microbiota.” Most of these microorganisms exist in the large intestine and small intestine, in which bacteria are an important part of the human body. Studies have shown that the number of bacteria surrounding and inside the body is 10 times the total number of human cells. 8 The intestinal bacteria can be divided into anaerobic bacteria, facultative anaerobic bacteria, and anaerobic bacteria according to their different degrees of oxygen demand. The majority of intestinal bacteria are anaerobic bacteria, represented by bifid bacterium, and accounting for more than 99% of the bacteria.
With the application of high-throughput sequencing technology in intestinal microecology, we can study changes in the gut microbiota's structure and composition in a more in-depth and comprehensive path. 9 Under normal circumstances, the gut microbiota structure, flora function, and interaction with the host constitute the intestinal microecology. The structure, species, and quantity of gut microbiota remain relatively stable, all of which play an important role in human health and the process of disease development. 10 The gut microbiota is related to gender, age, physiological and pathological states, living habits, psychological factors, and many other factors involved in the life processes of the organism.
The composition of human gut microbiota is huge as the genome encoded by the gut microbiota is about 150 times that of the human genome itself. It is hence called “human second genome.” This human second genome not only participates in the decomposition, digestion, and absorption of amino acids, sugars, and fats but it also provides various nutrients needed for the host and promotes the growth and development of intestinal epithelium. Moreover, it also plays a role in inhibiting the reproduction of pathogenic bacteria, resisting the invasion of pathogens, and regulating the immune response of intestinal mucosa. 11 Eckburg et al12 found that the intestinal mucosa and fecal flora of healthy volunteers were mainly composed of bacteroides and firmicutes, which accounted for 90% of the intestinal microecosystem through the gene sequence of microflora ribosomal RNA. In addition, there are a few groups of bacteria such as actinomycetes, Proteobacteria, and clostridium. Gwen et al13 analyzed the gut microbiota of 3948 people around the world and revealed that 14 genera of gut microbiota widely existed in the human gut, constituting the core flora of human gut bacteria. At the same time, their study showed changes in the 14 genera of intestinal bacteria, especially Verrucomicrobia, Bacteroides, and Prevotella caused differences in gut microbiota composition among participating individuals.
Another study 14 also found that based on the relationship between the gut flora and the human body, bacteria can be roughly divided into 3 groups: harmful bacteria, such as Bacillus sphaericus and Enterococcus faecalis, beneficial bacteria, such as Bifid bacterium and Lactobacillus, and pathogenic bacteria under specific conditions, such as Escherichia coli. It is therefore important to study the mechanism of gut microbiota composition on the occurrence and development of adenoma.
Relationship Between Gut Microbiota Disorder and Colorectal Adenoma
Intestinal microflora abnormality includes microflora imbalance and microflora translocation. The imbalance of the flora refers to the change of the original gut microbiota, resulting in the decrease of probiotics and/or the increase of pathogenic bacteria. As for bacterial translocation, it can be divided into transverse translocation and longitudinal translocation. Transverse translocation refers to the transfer of bacteria from the original site to the surrounding area, such as upward transfer of bacteria from the lower digestive tract as well as the upward transfer of bacteria from the colon to the small intestine, causing intestinal infectious reactions. Longitudinal translocation refers to the transfer of bacteria from the original site to the deep intestinal mucosa or even the whole body. The factors affecting the dysbiosis of flora include changes in diet structure, intestinal infection, use of antibiotics, immune deficiency, and psychological stress. 15 For example, a long-term high-fat diet may cause dysregulation of host gut microbiota, destroying normal intestinal wall cells, activating oncogenic signaling pathway, and producing pro-tumor metabolites while inhibiting antitumor immune response, which may affect the occurrence and development of adenoma and CRC. Shen et al 16 determined the rectal mucosal flora of healthy people and CRA patients by molecular imprinting and cloning sequencing and found that the composition of gut microbiota of the 2 groups was significantly different. Here, CRA patients had an increased proportion and number of Proteobacteria and decreased abundance of Bacteroidetes. Scanlan et al 17 analyzed the intestinal microbial composition of patients with CRA and found decreased intestinal microbial diversity and dominant flora. On the other hand, the diversity of Clostridium leptum and Clostridium coccoides increased significantly.
In the mouse model of CRA (min/ + ) intestinal tumor, Fusobacterium nucleate also increased the diversity of tumor. Therefore, the enrichment of Fusobacterium nucleates in feces can be considered a potential risk factor for cancer development from intestinal adenoma. 18
A growing body of evidence suggested that microbial ecological imbalance was a key environmental factor in the development of CRC and its precancerous lesions. 19 Adenoma tissue is characterized by increased abundance and diversity of bacteria, especially Proteobacteria and Fusobacteria. 20 In the development of CRC, Bacillus and Fusobacterium nuclei have the functions of triggering mucosal inflammation, mediating oncogenic signaling pathways, and inhibiting host immune response,21 which are likely to lead to the occurrence of adenomas. Emergence of adenomas may in turn lead to the imbalance of microbiota, whereas local microbiota disorders can also counteract on adenomas and promote the occurrence of cancer. When the balance between adenoma and microbiota is disturbed, the relative abundance of potential opportunistic pathogens (such as Pseudomonas and Streptococcus) increases, which changes intestinal homeostasis, exhibits strong inflammatory cell infiltration, and directly or indirectly increases the risk of adenoma development. Fecal analyses in patients with CRA and healthy individuals reported no difference in fecal flora richness and α-diversity, but a significant difference was found in β-diversity. 22 This difference mainly manifested in the Proteobacteria taxa, and there was no significant relationship with the gender of the patients. Many studies have identified Fusobacterium as an important intestinal bacteria associated with CRA. A large number of R-DNA copy numbers of Fusobacterium and Fusobacterium nucleatum have been observed in the feces of patients with CRA. Their abundance gradually increased from healthy individuals to CRA and then to CRC. In conclusion, the abundance of Fusobacterium positively correlated with the degree of dysplasia of the polyp tissues. Even in small polyp tissues, the abundance of Fusobacterium was higher than in the normal mucosa without histological changes. 23
Through fecal samples of patients with polyps, Moore et al24 found the abundance of Bacteroides and Bifid bacterium positively correlated with the risk of colon polyps, while Lactobacillus and true bacteria had a protective effect on the intestinal tract. In addition, Lactococcus, which is generally considered as an intestinal symbiont with probiotic characteristics, can prevent the imbalance of gut microbiota, stimulate the immune defense mechanism of the host, and can reduce intestinal cell proliferation and increased interleukin-18 (IL-18) expression in human colon cancer. 25 In rat model, Abdulamir et al26 also found that Streptococcus bevies could increase the proliferation of intestinal cells and the production of IL-8 in rats to promote the progression of precancerous lesions.
In the study of 35 cases of adenomatous polyps, 27 APC mutation was also closely related to gut microbiota. The abundance of Clostridium mori was increased in the adenomatous group, while the abundance of Clostridium tenderis and Bifidobacterium pseudostrinalis was significantly decreased. These findings could be used to predict the transition from CRA to CRC. Furthermore, Sanapareddy et al28 found from biopsy samples that the number of bacteria was greater in individuals with adenoma than in individuals without adenoma, and the difference in distal gut microbiota between the 2 groups was more obvious than any other clinical parameters.
In the APCMin/+ mouse model of intestinal tumor genesis, Kostic et al29 found that Clostridium nucleates increased the diversity of tumor, selectively recruited tumor-infiltrating myeloid cells, and showed the same pro-inflammatory expression characteristics as human Clostridium positive CRC, thus promoting the progress of tumor. In addition, Shen et al's cross-sectional study 16 revealed that adherent bacteria may be an important participant in the development of CRA and CRC. Here, the number of Proteus increased and Bacteroides decreased in participants with adenoma. This change in composition and structure may contribute to the development of adenoma. Goedert et al 22 also identified the difference in the microbial composition of fecal samples between patients with CRA and healthy individuals, which may be due to the relative increase in the abundance of Proteus, which in turn accelerated the transformation of CRA to CRC. Therefore, the gut microbiota is fundamental in maintaining the intestinal microecological balance.
Relationship Between Intestinal Inflammatory Microenvironment and Colorectal Adenoma and Its Carcinogenic Mechanism
The imbalance of gut microbiota causes local inflammatory changes in the intestinal tract, whereas most cancers are caused by infection, chronic irritation, and inflammatory reactions. 30 Chronic inflammation stimulates tumor growth and development through the production of pro-inflammatory cytokines, 31 such as nuclear factor kappa-B (NF-κB) and signal transducer and activator of transcription 3 (STAT3). These factors will further stimulate the production of cytokines and chemokines, as well as lead to the recruitment of white blood cells32 and induction of cell proliferation, angiogenesis, lymph angiogenesis, and invasion of tumor cells. 33 Therefore, inflammatory changes in the microenvironment may be an important factor driving the transformation of adenoma into CRC. Many studies have shown that intestinal adenomas played an important role in the occurrence and development of CRC and that about 90% of colon cancers evolved from adenomas. 34 In 1863, Virchow proposed a connection between chronic inflammation and cancer. Chronic inflammation could lead to tissue damage and oxidative stress, and DNA damage would gradually accumulate in epithelial cells, leading to carcinogenesis of intestinal epithelial cells.
Chronic inflammation mediated by the dysregulation of gut microbiota is also associated with cytokines such as IL-6, IL-17, IL-23, and tumor necrosis factor-α (TNF-α). 35 Multiple studies have shown that gut microbiota can activate Toll-like receptors (TLRs) to promote the expressions of IL-1β and TNF-α as well as the production of COX-2, thereby mediating prostaglandin E2 synthesis and promoting inflammatory response and carcinogenesis. 36 Therefore, the increase in the expression levels of inflammatory cytokines has emerged as an important factor in the development of early CRC. 37 Moreover, anti-inflammatory agents such as aspirin and celecoxib are commonly used in the treatment of intestinal adenomas. These nonsteroidal anti-inflammatory drugs and cyctoxase-2 inhibitors can effectively slow down the development of intestinal adenomas, indicating that inflammation is a driving factor in colorectal carcinogenesis. Interestingly, the inflammatory process affects the whole transition process from adenomas to tumors, including initiation, progression, and metastasis. Common inflammatory factors, such as C-reactive protein (CRP), Interleukin (IL), and cyclooxygenase-2 (COX-2), are involved in the aforementioned process.
In one meta-analysis of observational studies, 38 studies related to the risk of CRP and CRA were searched using PubMed and Embase databases, and 14 case–control studies were summarized. When only advanced adenoma was considered, the odds ratio value increased significantly. Kim et al 39 also found that the elevated score of ultrasensitive CRP positively correlated with the prevalence of CRA. Subgroup analyses showed that women younger than 50 years old or premenopausal women were likely to have advanced adenoma or distal CRC.
One retrospective study 40 showed TNF-α, IL-1β, COX-2, and IL-4 were highly expressed in serrated adenomas. The expression levels of these factors varied in different types of adenomas, promoting the emergence of serrated structures in adenomas and affecting the development and progression of CRC. Also, Chen et al41 demonstrated that IL-22 may increase DNA damage and genomic instability, thereby driving tumorigenesis by accelerating cell metastasis from heterozygosis (APCMin/+) to homozygosis (APCMin/Min). This indicates that IL-22 may play an important role in promoting cancer.
Celecoxib is a new-generation compound with a unique mechanism of action that specifically inhibits COX-2. Inflammatory stimuli induce the production of COX-2, which in turn leads to the synthesis and accumulation of inflammatory prostanoids, especially prostaglandin E2, causing inflammation, edema, and pain. In addition, a prospective study 42 ,43 reported that 89.5% of patients completed colonoscopy follow-up in the first year, 75.7% of patients completed colonoscopy in the third year, and by year 3, the estimated cumulative incidence of finding one or more adenomas among patients receiving placebo was 60.7%. The cumulative incidence of one or more adenomas was 43.2% and 37.5% in patients taking 200 and 400 mg of celecoxib daily, respectively. These findings suggested that COX-2 is a significant independent factor for the development of advanced CRA and that celecoxib can effectively prevent CRA. By analyzing serum samples of patients with CRC, Huang et al44 found that the elevated circulating levels of CCL20 and growth-related gene oncogene products were associated with an increased risk of adenoma, suggesting the role of inflammatory factor-related pathways in the occurrence of early CRC.
The occurrence and development of many human tumors are accompanied by chronic inflammatory reactions. Inflammation can release cytokines, produce free radicals, damage DNA, and promote angiogenesis, cell proliferation, and eventually tumor progression. Colorectal adenomas are closely related to chronic inflammation. When the gut microbiota is balanced, it can secrete a variety of enzymes to prevent the invasion of pathogenic bacteria and ensure the normal function of the immune system. When the gut microbiota is out of balance, it often leads to the reduction of beneficial bacteria; the proliferation of pathogenic bacteria; the increase of endotoxins and other intestinal G-bacteria cell wall outer membrane's secretions; and the activation of the inflammatory cascade in the body, resulting in increased secretion of TNF-α, IL-6, and other inflammatory factors, triggering inflammatory reactions. For example, IL-6 can activate transcription factors and induce the generation of Bcl-xl and Bcl-2 antiapoptotic factors, leading to abnormal accumulation of T cells in the intestinal mucosa, further aggravating inflammatory response and accelerating the transformation of adenoma into CRC. 45
Other researchers believe that myeloid differentiation factor 88 (My D88) mediates the production of inflammatory cytokines such as IL-23 when opportunistic pathogens invade and destroy the intestinal barrier, IL-23 can also activate inflammatory factors such as IL-6, IL-22, IL-17A, and these inflammatory factors can induce Th17 immune cell differentiation.46,47 Through a series of inflammatory cascades, signaling pathways such as NF-κB and transcription factor 3 (STAT3) are eventually activated, thereby promoting tumor cell proliferation. 48 In addition, during intestinal tumorigenesis, the gut microbiota also upregulates the expression of IL-17C in colon epithelial cells through TLR/My D88 signal transduction, and IL-17C promotes tumorigenesis by inducing the expression of Bcl-2 and Bcl-xl. COX-2, an enzyme that induces inflammatory processes, is generally unexpressed or underexpressed in tissue cells and highly expressed in tumor cells. When the gut microbiota is out of balance, it can stimulate a large increase in COX-2, accelerate the tissue inflammatory response, and then promote the development of CRC. Recent studies have found that abnormal activation of Wnt/β-catenin signaling pathway leads to significant accumulation of β-catenin in the nucleus, affects the immune function of the body, activates the production of inflammatory mediators, participates in a variety of inflammatory reactions, induces tumor angiogenesis and colorectal epithelial cell proliferation, and leads to CRA. 49
Relationship Between Intestinal Immune Microenvironment and Colorectal Adenoma and Its Carcinogenic Mechanism
A large number of studies have shown that gut microbiota plays an important role in the immune microenvironment and interacts with the intestinal immune system, participating in the development and differentiation of immune cells, and regulating the signaling pathways of immune function. 50 Most cancer types, including CRC, develop from a long period of precancerous lesions, in which host immunity is a key factor in stabilizing the latter. However, precancerous tissues can escape from host immune surveillance in various ways and induce immunosuppression directly or indirectly, promoting the transformation of precancerous lesions into tumors. The transition from adenoma to CRC would undergo 3 immune stages, namely, elimination, equalization, and escape. During the elimination stage, the precancerous lesions can theoretically be eradicated, and their cells’ early regression can be induced. However, the early lesions are usually small in size, and it is difficult to prove that immune cells can eliminate them, and it is also difficult to observe them clinically. During the homeostasis stage, some precancerous cells retain their presence by evading immune surveillance through a variety of means. During the escape stage, precancerous cells have acquired the ability to evade immune surveillance, so that they are not attacked by the immune system and not restricted by growth, and eventually develop into malignant tumors. 51 Maglietta et al52 found 429 immune pathways through gene set enrichment analysis in different shape and size of tumors and showed obvious differential regulation. Compared with normal mucosa, most of the immune pathways were upregulated or downregulated in colorectal adenomas. The relationship between different immune pathways and lesion size could only be observed in adenomas. Furthermore, the antibodies of various immune cells were detected. The number of T lymphocytes and macrophages was significantly higher in polyploidy lesions than in non-polyploidy lesions, and the number was significantly different based on the size of adenomas. Also, the larger the adenoma, the more immune cells were in the stoma. These findings indicated that immunological differences may influence the developmental trajectory of a specific lesion toward cancer.
Studies have shown that host immunity failed to produce a satisfactory antitumor immune response in most patients with CRC, which indicated that there is a mechanism of immune escape in CRC.
53
There are different subsets of T regulatory cells (Tregs), orchestrating critical roles in the regulation of antitumor immunity in CRC. Among them, forkhead box protein-3 ( + ) regulatory T cells (
Many recent studies have investigated the changes in the neutrophil-to-lymphocyte ratio (NLR) and other hematological components in some chronic diseases and tumors. High-density neutrophils can promote the growth and metastasis of tumors. Neutrophils can produce cytokines, reactive oxygen species, proteases, and other substances that contribute to the body's inflammatory response in different microenvironments. Neutrophils also inhibit the activity of lymphocytes, leading to a decreased number of lymphocytes and diminished body's immunity and differentiation ability of intestinal mucosa cells, which can lead to the imbalance of epithelial cell proliferation and apoptosis.
Lymphocytes play an important role in the immune system and play a key role in the monitoring and defense of tumor immunity. Lymphocytes can weaken tumor immune monitoring by inducing cytotoxicity and further reducing the host's antitumor immunity.62,63 Emir et al 64 found that preoperative NLR and preoperative platelet-to-lymphocyte ratio (PLR) could be used as markers to monitor the progression of adenomatous polyps to aggressive tumors, indicating that the higher NLR and PLR values, the greater the carcinogenesis risk.
The intestinal immune environment is comprised of a large number of lymphocytes and myeloid-derived immune cells, which play an important role in the maintenance of intestinal local immune balance and systemic immune balance. 65 When the gut microbiota is unbalanced, the immune homeostasis of the human body will also be disturbed, and these changes in the immune environment may be related to the occurrence of CRC. Enterotoxin-producing Bacteroides Fragilis (ETBF) can cause intestinal epithelial exfoliation and γ-dependent e-mucin lysis by releasing toxins. 66 In addition to acting on the epithelial barrier, ETBF can also act on immune cells and molecules. ETBF increases IL-6 and TNF-α by activating TLRs, further activates STAT3 and NF-κB, inhibits antitumor immunity, and promotes tumorigenesis. 49
Fusobacterium nucleatum (Fn) is highly enriched in CRC, which can induce local inflammation and increase IL-6, IL-8, and IL-12, as well as transform growth factor-β, TNF-α, and other inflammatory cytokines, leading to the occurrence of colorectal tumors. Fn and its components allow TLRs on tumor-infiltrating bone marrow cells to participate in and activate the production of inflammatory cytokines mediated by MyD88, most notably the production of IL-23. IL-23 promotes the development of CRC by inducing the production of IL-17A, IL-6, and IL-22. 67 Fn regulates E-cadherin/β-Catenin by promoting tumor immune escape and inhibiting natural killer cells or T cell antitumor defense to support the development and metastasis of CRC. 68 Studies have also reported that Fn in CRC tissue will metastasize with cancer cell metastasis. 69
Enterococcus faecalis (EF) activates mucosal macrophages to produce bystander effect. When untransformed primary epithelial cells are exposed to EF-polarized macrophages, intestinal epithelial cells develop chromosomal instability (CIN) and then transform into cancer cells.
TNF-α acts as a diffusion medium of macrophages-induced bystander effect. This inflammatory cytokine is triggered by Enterobacter faecalis and leads to the proliferation of colon epithelial cells through the antiapoptotic effect of neurite guiding factor netrin-1. 70 EF can induce the formation of superoxide in intestinal epithelial cells, then enhance the expression of COX-2 in macrophages, and promote the formation of CIN in intestinal epithelial cells, leading to the injury of colon epithelial cells.
Relationship Between Intestinal Metabolites and Colorectal Adenoma and Its Carcinogenic Mechanism
Recent studies have shown that the metabolites of the gut microbiota are closely related to the occurrence of CRA. Intestinal microflora is an important part of host digestion and nutrient absorption, which can decompose the indigestible substances into nutrients. Different metabolites are produced by different bacteria groups, mainly including SCFAs, colic acids, choline metabolites, phenols, benzoyl and phenyl derivatives, indole derivatives, vitamins, polyamines, and lipids. 71 Ammonia, amines, phenols, and hydrogen sulfide may participate in the occurrence and development of CRC through chronic inflammation and DNA damage in tissue cells. 72 In addition, intestinal microorganisms can transform DE-binding primary bile acids (BAs) into secondary BAs. 73 These secondary BAs, such as lithotomic acid and deoxycholic acid (DCA), have cytotoxic and genotoxic effects. 74 They have the same function as pro-inflammatory bacteria, such as Mogibacterium and Sutterella. They can lead to DNA damage and inflammation, promoting adenoma development and eventually malignant transformation.
By investigating the interactions and influence of intestinal metabolites on the host and gut microbiota, Kim et al75 found that the levels of several bioactive lipids (including polyunsaturated fatty acids, secondary BAs, and sphingolipids) were higher in adenoma patients than in the control group. Most of these metabolites showed consistent changes in patients with CRA, indicating that these changes may represent an early event of carcinogenesis.
Furthermore, a study 76 with a small sample size assessed the level of sphingolipids in human CRAs and found that the content of pro-apoptotic and antiproliferative ceramide increased while that of antiapoptotic and pro-proliferative sphingosine-1-phosphate decreased in tubular adenomas with low malignancy. On the contrary, in tubular villous adenoma with high dysplasia degree, the level of ceramide decreased while the concentration of sphingosine-1-phosphate increased. It has been demonstrated that different histological types of adenomas have different changes in sphingolipid metabolism, and adenomas with lower ceramide level and higher sphingosine-1-phosphate concentration tend to increase the risk of CRC. By using 16S R-RNA sequencing, Hale et al77 found that the increase in metabolites, such as sugar, protein, fat, and BA in adenoma tissue, may promote the growth of bile-resistant microorganisms, such as Bibliophile and Desulfovibrio. On the other side, the generated inflammatory products can induce the occurrence of adenoma and the generation of tumor.
Short-chain fatty acids are derived from the food in the colon and rectum through bacterial fermentation. They are mainly produced by Roseburia and Lachnospiraceae. These bacterial metabolic end products can enter the blood through intestinal epithelial cells and participate in the host's immune response through blood circulation. 78 Studies have confirmed that SCFAs have the functions of inhibiting the proliferation of tumor cell, promoting apoptosis, and maintaining T cell homeostasis. They can also stimulate the expression of anti-inflammatory molecules in dendritic cells and macrophages and induce the production of Treg and IL-10. Butyrate is a ligand of G protein-coupled receptor and an inhibitor of histone deacetylase. It is a key signal molecule that affects the immune response of the body. It can participate in the energy metabolism of the body and regulate the host immune response. 79 Butyrate can also protect intestinal epithelial cells, reduce DNA damage, delay the proliferation rate of tumor cells, protect the integrity of intestinal wall, reduce intestinal inflammation, reduce the occurrence of CRC and other diseases, 80 and promote Treg differentiation by participating in the regulation of spleen and lymph nodes. 81 The absence of both factors can contribute to the development of colon tumors. 82 The anticancer properties of conjugated linoleic acid (CLA) and conjugated linoleic acid (CLNA) in vivo and in vitro have been widely recognized in previous research. Degen et al83 found that CLA and CLNA had a stronger inhibitory effect on the growth of lt-97 colon adenoma cells in a human cell culture model of colon cancer. Lactic acid bacteria or Bifidobacteria can reduce the production of toxic metabolites in the intestine by reducing the dehydroxylation of primary BAs and reducing the concentration of DCA in feces.
Bile acid is one of the common regulatory factors of intestinal microbiota, which can absorb lipids, cholesterol, and fat-soluble vitamins. However, BA contains lithocholic acid and DCA, which have potential carcinogenic effects. Most studies demonstrated that BA can induce CRA by accumulating in the intestinal tract and affecting the composition of the gut microbiota. 84 Other microorganisms in the intestine can further reduce the production of BAs and accelerate the metabolism of BAs by promoting the depolymerization, dehydrogenation, and dehydroxylation of distal small-molecule BAs. In vitro experiments have shown that secondary BAs can induce DNA damage in epithelial cells, induce oxidative stress, activate NF-κB signaling pathway, promote the production of inflammatory factors, and induce inflammatory response. 85
Relationship Between Helicobacter pylori and Colorectal Adenoma and Its Carcinogenic Mechanism
Helicobacter pylori (H. pylori) has been a recent hot topic in clinical research. The mechanism of H. pylori infection causing CRA remains unclear. In 2011, a study in the United States found no correlation between H. pylori infection and CRA. 86 However, some studies have suggested that H. pylori infection correlated with the occurrence of CRA. H. pylori infection causes intestinal endocrine abnormalities and subsequently induces the expression of plasma gastrin 17 and COX-2, thereby nourishing intestinal mucosa, stimulating intestinal mucosal proliferation, and promoting the growth of polyps. Selgrad et al87 found that the prevalence of large intestine polyps was 63.4% in the H. pylori infection group, and H. pylori infection mostly occurred in patients with hyperplastic polyps and low-grade intraepithelial neoplasia adenoma. Therefore, H. pylori is a risk factor for the development of CRC. 88 H. pylori can also cause systemic immune response and intestinal mucosal damage by releasing antigens and lead to the formation of CRA.
A recent large-scale western study based on a national database showed that the association between H. pylori-positive gastritis and colonic tumors became stronger as the size and number of CRAs increased. H. pylori infection was found to be an independent risk factor for CRA in a model of transformation from benign polyps to malignancies. 84 A retrospective cohort study in Korea also found89 that H. pylori infection rate was higher in adenomas than in nonadenomas. It is suggested that H. pylori infection is an independent risk factor for colon adenoma, especially in the case of advanced or multiple adenomas. This notion was also demonstrated by the Zhang et al90 team. The positive rate of H. pylori was higher in patients with CRA than in the control group. Multivariate logistic regression analysis also demonstrated that positive H. pylori is an independent risk factor for CRA. In addition, positive H. pylori positively correlated with tumor size and high-grade intraepithelial neoplasia in patients with CRA. Cytotoxin-associated gene A (CagA) may enhance the carcinogenicity of H. pylori in CRA.
A meta-analysis 91 showed that H. pylori infection is associated with an increased risk of CRA in Caucasian and Asian populations. In addition, case–control studies and cross-sectional studies have also demonstrated H. pylori infection to be a risk factor for CRA.
The possible mechanism of H. pylori infection causing the increase of gastrin level is that H. pylori infection leads to the atrophy of gastric mucosa, reducing the secretion of gastric acid, and the urease of H. pylori hydrolyzes urea to produce ammonia. Ammonia can neutralize gastric acid and reduce the content of gastric acid. Gastrin cells in gastric antrum increase the level of gastrin through negative feedback regulation. Gastrin, a gastrointestinal hormone mainly secreted by G cells of the antrum and duodenum, is important for regulating digestive tract function and maintaining its structural integrity. G-17 is only secreted by G cells in the antrum and can be used as an important indicator to reflect the situation of gastric mucosal injury. Studies have shown that the growth of intestinal mucosal cells is related to the level of G-17, and the increase in serum G-17 level also increases the risk of colorectal adenoma. 92 Not only can gastrin directly create a suitable microenvironment to promote the occurrence of intestinal adenomas but it can also indirectly promote the formation of intestinal adenomas through the proliferation signal mediated by gastrin receptor. 93 At present, it is generally believed that the increase in gastrin secretion caused by H. pylori infection is related to colorectal adenoma.
Relevant studies have found that H. pylori can colonize both gastric mucosa and colorectal adenomas. And, in colorectal adenoma, H. pylori is “granular,” which is a special spherical state of H. pylori. 94 Other studies have found that H. pylori implanted in gastric mucosa can increase the risk of colorectal polyps. H. pylori residing in the colon can promote the occurrence of colorectal adenoma by chemotactic inflammatory factors. 95 H. pylori can secrete a variety of virulence factors, which can act on the mucosa and maintain the continuous infection of H. pylori, resulting in tissue damage and chronic inflammatory reaction. 96 CagA exists in the highly virulent strain of H. pylori, and its expression product CagA protein is an important virulence factor for the pathogenic effect of H. pylori. The pro-inflammatory factors released by CagA protein may promote the proliferation of colorectal epithelial cells, leading to colorectal adenomas in CagA + H. pylori-infected people. 97
The mechanism through which H. pylori infection destroys the balance of the gut microbiota may be that long-term H. pylori infection leads to the reduction of gastric acid secretion, and the produced urease will decompose urea to produce ammonia, which will increase the pH value of the intestine, thus destroying the steady state of the gut microbiota, causing its imbalance and making some pathogenic bacteria invade colon mucous membrane cells and cause an inflammatory reaction. This in turn will lead to the occurrence of colonic adenoma. 98 Therefore, H pylori eradication might have an impact on the prevention of colorectal neoplasm formation.99,100
Based on our review, the diversity of the gut microbiota associated inflammatory responses, immune regulation, and metabolic changes have played an important role in the development and progression of colorectal adenomas (Figure 1).
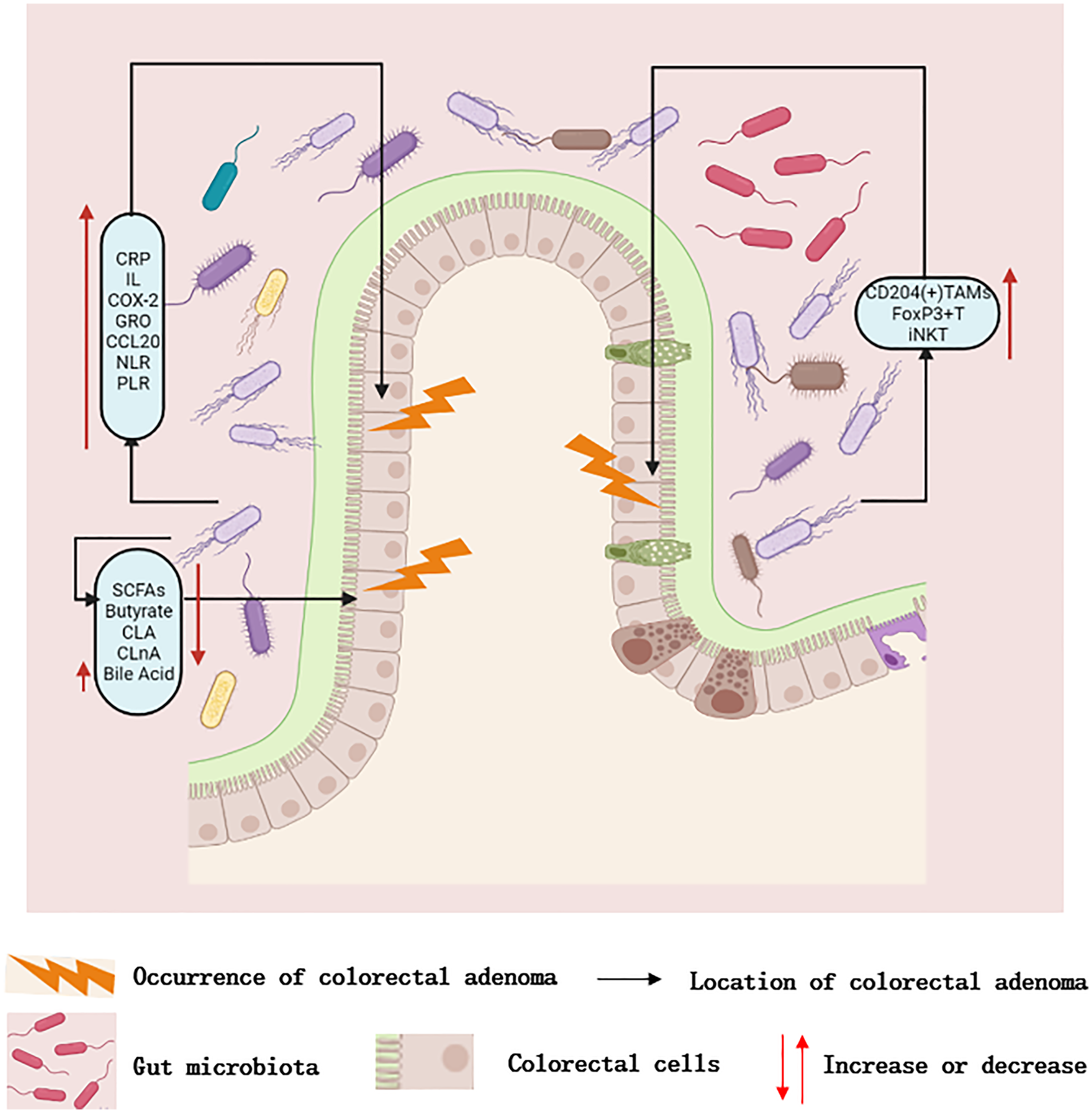
Dysregulation of gut microbiota can promote carcinogenesis through changes in microbial diversity, chronic inflammation, immune disorders, and metabolic products. (1) Dysbiosis of gut microbiota causes local inflammatory changes in the intestine, and the increase in CRP, IL, COX-2, and other inflammatory substances promotes the occurrence of adenomas. (2) The imbalance of gut microbiota leads to the disorder of immune microenvironment, and the increase in CD204 ( + ) TAM, Foxp3 + T regulatory cells, and INKT, which promotes the occurrence of adenoma. (3) The imbalance of gut microbiota leads to changes in metabolic products. The decrease in SCFAs, butyrate, CLA, and CLNA, as well as the increase in bile acids, can promote the occurrence of adenoma.
Conclusion
Current literature has confirmed that the gut microbiota is closely related to the occurrence and development of CRA. Studies have also shown that the intestinal microenvironment plays an important role in maintaining homeostasis. Intestinal dysbacteriosis promotes the occurrence and development of CRA by triggering chronic inflammation, mediating immune response, and producing metabolites. Although intestinal microecological imbalance has attracted more and more attention as a factor affecting the development of CRAs and tumors, host-bacterial interactions have not been fully elucidated.
In the study on the association between intestinal microorganisms and the occurrence of CRA and CRC, it is unclear whether mucosal or fecal samples are more appropriate to be investigated. Microecological imbalance induces mucosal inflammatory response and the production of active metabolites, which is the initial stage of colonic mucosal adenoma formation. Further experimental animal models are needed to further clarify the mechanism between intestinal microorganisms and CRA.
Overall, with the continuous advancement of microbiome research, the exact relationship between host intestinal microorganisms and CRA will become increasingly clear, which will provide new insights and approaches for the prevention, diagnosis, and treatment of CRC.
Footnotes
Abbreviations
Authors’ Note
NNL generated the ideas and designed the structure of manuscript. ZPZ organized all figures and wrote the manuscript. NL BA drafted the article or revised it critically for important intellectual content. NNL and MYS revised and approved the final manuscript. All authors read and approved the final manuscript. All the data obtained and/or analyzed during the current study were available from the corresponding authors on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Shanghai Science and Technology Committee (19401972200), the Key projects of the National Natural Science Foundation of China (81830120), and the Key projects of the National Natural Science Foundation of China (82030118). Natural Science Foundation of Shanghai (19ZR1458500); Three-year Plan of Action for the Development of Traditional Chinese Medicine in Shanghai (ZY(2018-2020)-CCCX-2003-02);Science and technology innovation action plan, the Major Project of Shanghai Municipal Science and Technology Commission (Grant number: 19401972300); “major scientific and technological innovation project,” Key R & D plan of Shandong Province (Grant number: 2021CXGC010509); The fourth batch of excellent talents in traditional Chinese medicine, the State Administration of traditional Chinese medicine (Grant number: 2017-124).
