Abstract
Introduction
Polyneuropathy, organomegaly, endocrinopathy, M-protein, skin changes (POEMS) syndrome is a rare monoclonal plasma cell proliferation disorder. The patient suffered from multiple organ dysfunctions, including polyneuropathy, organomegaly, endocrinopathy, increased M protein, and skin lesions. 1 Current treatment of patients with POEMS includes anthracycline, immunomodulators, proteasome inhibitors, and autologous stem cell transplantation (ASCT) for patients in remission. Lenalidomide has immunomodulatory, anti-angiogenesis, and cytotoxic effects. It had achieved revolutionary results in the treatment of Bcell non-Hodgkin's lymphoma, and multiple myeloma (MM) and was often used in the treatment of POEMS syndrome. Its safety and efficacy were confirmed in multiple studies.2–6 As a proteasome inhibitor, bortezomib exerts antitumor effects by selectively inhibiting the ubiquitin-proteasome pathways.7,8 And bortezomib was mainly used in the treatment of MM, and it can improve the survival rate of patients. 9 In recent years, bortezomib has also been applied for the treatment of POEMS syndrome.10–12 ASCT can not only obviously improve the clinical symptoms of patients with POEMS syndrome but also has significant advantages in improving patients' overall survival (OS) and progression-free survival (PFS).13–15 The current treatment options are mainly derived from limited clinical trial data and nonclinical trial studies (cohort studies, case reports, and case series). It is unclear which one is the best first-line treatment for POEMS syndrome. Here, we report a case of patient diagnosed with POEMS syndrome and got complete response (CR) after being treated with ixazomib and ACST. In the present study, we conduct a meta-analysis containing 6 clinical trials and 14 nonclinical trials to summarize, compare and get favorable consequences for treatment recommendations.
Case Report
A male patient in his early 40s presented to the Department of Neurology due to weakness of his left leg and numbness of his left foot for more than 12 months. He had difficulty in walking and found pain and sensory disturbances in his legs. At the same time, both legs’ tendon reflexes were weakened. The patient performed laboratory tests, including blood counts, a biochemistry panel, coagulation function, anti-nuclear antibodies, cerebrospinal fluid analysis, and serum protein electrophoresis. These were noteworthy for thrombocytosis (platelets (PLT) 504 × 109/L, normal range: 100-300 × 109/L) and dyslipidemia (triglycerides 2.25 mmol/L, normal range: 0.56-1.70 mmol/L). Blood urea nitrogen, creatinine, calcium, albumin, and lactic dehydrogenase levels were normal. Immunofixation electrophoresis (IFE) found monoclonal immunoglobulin bands (IgGlambda immunoglobulin). Electromyography with nerve conduction studies showed a performance of peripheral neuropathy. He was diagnosed with multiple mononeuropathies and was treated with immunoglobulin (35 g × 5 d) and methylprednisolone (1000 mg × 5 d), and then the dose of methylprednisolone was adjusted to 1 mg/kg body weight per day. After a few weeks, the dose of methylprednisolone was gradually reduced, and the patient's symptoms gradually improved. Two months later, the patient developed bilateral upper limb weakness and was admitted to the Department of Neurology again. At that time, the dosage of methylprednisolone was 24 mg per day. Serumfree light chain showed kappa light chain 12.50 mg/L (3.30-19.40 mg/L), lambda light chain 232.50 mg/L (5.71-29.30 mg/L), and kappa/lambda 0.05 (0.26-1.65). Monoclonal immunoglobulin IgGlambda was tested by serum IFE, and M protein quantification was 0.85g/L. Prolactin level was increased to 14.09 ng/mL (2.64-13.13 ng/mL) and plasma vascular endothelial growth factor (VEGF) level was elevated at 1300.25pg/mL (0-146 pg/mL). The 18(fluorodeoxyglucose (FDG)-positron emission tomography (PET)/computed tomography (CT)) scan showed no significant bone destruction or lymph node enlargement. No significant abnormalities were observed in bone marrow morphology and immunological phenotype. The patient was diagnosed with POEMS syndrome. (Details of the patients’ diagnosis are shown in Supplementary Table 1).
Considering this patient had significant neurological symptoms, while proteasome inhibitors ixazomib has fewer neurological side effects than bortezomib, we then treated the patient with 4 cycles of ICD (ixazomib 4 mg d 1, 8, and 15, cyclophosphamide 0.8 g d 2, and dexamethasone 20 mg d 1-2, 8-9, 15-16, and 22-23). Although there was no significant improvement in neurological symptoms and the M protein component could still be detected (1.68 g/L), other indicators including VEGF level and serum-free light chain returned to normal. He received ASCT with high-dose melphalan (200 mg/m2) as a preconditioning regimen. Efficacy assessments were carried out 2 months later. The weakness of limbs was improved significantly, and motor function showed further recovery. IgGlambda monoclonal protein disappeared and serum VEGF level was normal. Serum-free light chain showed kappa light chain 23.12 mg/L (3.30-19.40 mg/L), lambda light chain 27.17 mg/L (5.71-29.30 mg/L), and kappa/lambda 0.85 (0.26-1.65). The last follow-up date was December 6, 2021, and the patient could walk independently and did not have obvious symptoms of peripheral nerve damage. Both serum and urine IFE were negative and efficacy evaluation was CR. (Details of the patients’ posttreatment evaluation are shown in Supplementary Table 2 and diagnosis and treatment process are shown in Supplementary Figure 1). The reporting of this study conforms to CARE guidelines. 16 Written informed consents were obtained from the patient for the treatment and publication.
Materials and Methods
Search Strategy and Data Abstraction
We searched relevant articles in PubMed, Embase, and MEDLINE databases for the period up to July 2021. The search strategy included the keywords POEMS, Therapy, Drug Therapy, Biological Therapy, Combined Modality Therapy, Hematopoietic Stem Cell Transplantation, Immunotherapy, Molecular Targeted Therapy, Chemoradiotherapy, Salvage Therapy, Controlled Clinical Trial, Randomized Controlled Trial, etc. In addition, we checked all the references of eligible articles that our search retrieved to identify potentially eligible papers. The reporting of this study conforms to CARE guidelines. 16 Written informed consents were obtained from the patient for the treatment and publication. This study has been registered in Inplasy (Registration number: INPLASY202270061).
POEMS diagnosis is established according to the Mayo criteria, 17 and the major criteria for the syndrome are polyradiculoneuropathy, clonal plasma cell disorder, sclerotic bone lesions, elevated VEGF, and the presence of Castleman disease. Minor features include organomegaly, endocrinopathy, skin changes, papilledema, extravascular volume overload, and thrombocytosis. The diagnosis of POEMS syndrome is made with 3 of the major criteria, 2 of which must include polyradiculoneuropathy and clonal plasma cell disorder, and at least one of the minor criteria. 17 The CR is evaluated as follows: normal plasma VEGF level; negative M-spike in serum, urine immunofixation, and bone marrow; no FDG uptake in PET/CT; absence of ascites/effusion/edema; and diffusing capacity of carbon monoxide (DLCP)≥70% predicted. 17 Hematological remission was assessed according to the author's statements in the eligible individual manuscripts; if these were not clearly stated, we apply the Mayo criteria. Articles with fewer than 5 patients were considered to have insufficient sample size.
Result
The search strategy retrieved 221 articles. Of these articles, 6 were reviews, 47 were excluded due to language restrictions (articles in non-English), 119 were excluded due to incomplete POEMS diagnosis, 30 were excluded due to inadequate sample size (with fewer than 5 patients), and finally, 19 were eligible for inclusion in this systematic review. After examining all references to relevant checks and eligible articles, one more study was deemed suitable for inclusion. Overall, 20 articles were included in this pooled analysis. The steps mentioned above concerning the selection of studies are illustrated in Figure 1. (Specific retrieval steps are shown in Supplementary Table 3).

Stages of the search strategy.
This meta-analysis included 6 clinical trials and 14 retrospective studies, 936 patients (28 cases were relapsed or refractory patients, and the rest were newly diagnosed patients). There were 2 clinical trials with 196 patients and 6 retrospective studies with 440 patients whose treatment plan was ASCT. Three clinical trials with 64 patients and 3 retrospective studies with 132 patients whose treatment plan was lenalidomide plus dexamethasone. And only 1 retrospective study with 17 patients whose treatment plans were bortezomib. The overall complete hematologic remission (CRH) and partial hematologic remission (PRH) was 33.0% (95% confidence interval [CI]: 0.19-0.49) and 42.0% (95% CI: 0.100.79) in clinical trials (Figure 2A and B); 44.0% (95% CI: 0.360.53) and 34.0% (95% CI: 0.230.47) in nonclinical trials (Figure 3A and B).

The hematologic response in clinical trials. (A) Complete hematologic remission (CRH) of clinical trials. (B) Partial hematological remission (PRH) of clinical trials. Logit transformation and random intercept logistic regression models. The red squares represent the remission rates and the horizontal lines reflect the 95% confidence interval (CI). The dark diamond corresponds to the 95% CIs for the pooled mutation rates.
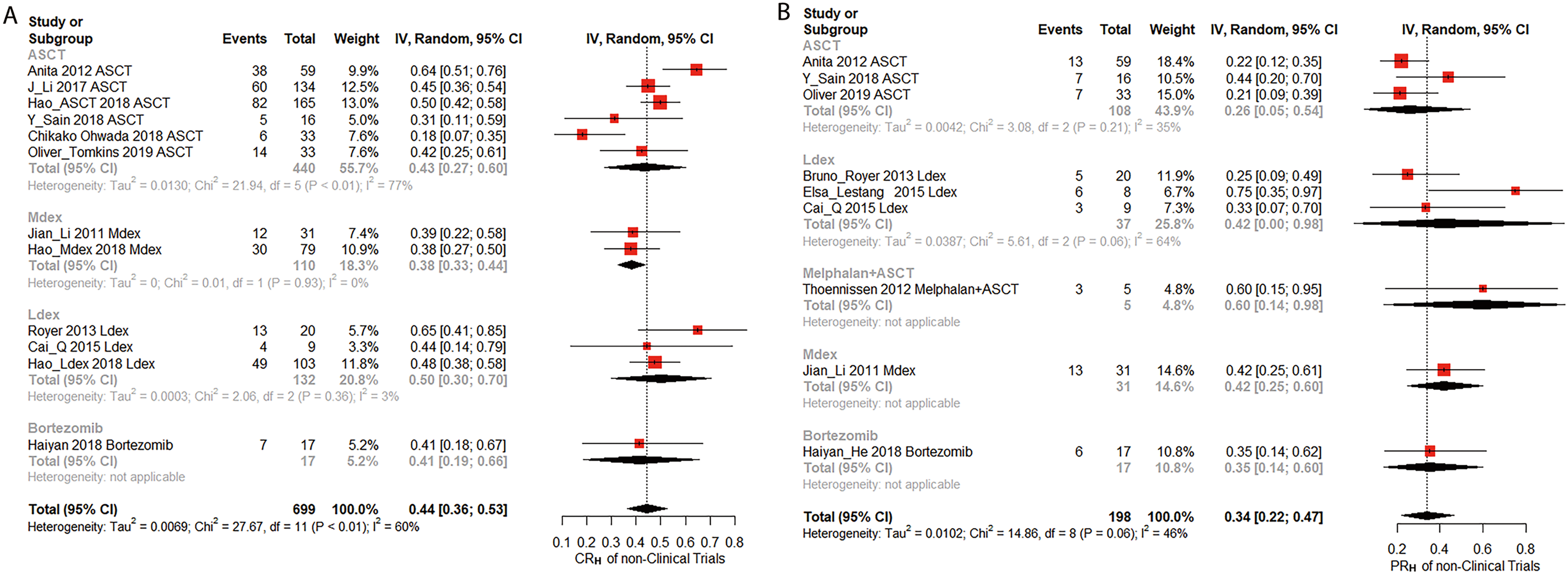
The hematologic response in nonclinical trials. (A) Complete hematologic remission (CRH) of nonclinical trials. (B) Partial hematological remission (PRH) of nonclinical trials. Logit transformation and random intercept logistic regression models. The red squares represent the remission rates and the horizontal lines reflect the 95% confidence interval (CI). The dark diamond corresponds to the 95% CIs for the pooled mutation rates.
Discussion
There are currently no standard treatments for patients with POEMS syndrome. At present, published studies mainly focused on case reports, case series, and retrospective analysis. Due to the diversities of patients in different studies, insufficient sample size, and large errors, it is impossible to guide clinical treatment based on the available evidence. Therefore, we meta-analyzed the main current POEMS treatments and summarized them into 3 principal treatments (ASCT, immunomodulators, and proteasome inhibitors) and compared the advantages and disadvantages.
ASCT plays an important role in the treatment of POEMS. The safety and efficacy of ASCT have been demonstrated.14,18,19 We found 8 articles about the ASCT treatment of POEMS. According to our analyzed results, the CRH rate of patients who received ASCT treatment in clinical trials and nonclinical trials were 25.0% (95% CI: 0.000.93) and 43.0% (95% CI: 0.270.60) (Figure 2A, Figure 3A); while the PRH rate were 35.0% (95% CI: 0.001.00) and 26.0% (95% CI: 0.050.54), respectively (Figure 2B, Figure 3B). Due to lack of data, we didn't analyze the serum VEGF response, neurologic response, and the improvement of other important symptoms of POEMS syndrome. Our meta-analysis had relatively high heterogeneity, which may be related to the absence of risk stratification, different interventions for patients, and different enrolled patients (newly diagnosed or relapsed).
Immunomodulators and proteasome inhibitors are also given a higher position in treating POEMS syndrome.5,11,20,21 Lenalidomide has obvious antiVEGF and anti-plasma cell effects. It can quickly reduce the patient's hematological and neurological indicators and relieve the patient's clinical symptoms.5,22 However, lenalidomide is mainly excreted in urine and is toxic to the kidneys. Therefore patients with renal impairment should be cautious or avoid using lenalidomide. 23 In our meta-analysis, the CRH rate of individuals who received lenalidomide treatment was 45% (95% CI: 0.001.00) in clinical trials (Figure 2A), the CRH rate of persons receiving lenalidomide combined with dexamethasone (LDex) and proteasome inhibitors bortezomib treatment in nonclinical trials were 50.0% (95% CI: 0.300.70) and 41.0% (95% CI, 0.190.66), respectively (Figure 3A). The PRH rate of patients receiving LDex treatment and bortezomib were 42.0% (95% CI: 0.000.98) and 35% (95% CI: 0.140.60) in retrospective research, respectively (Figure 3B). From the above data, it can be found that compared with ASCT, there were no significant differences in the hematological response rate of patients treated with immunomodulators or proteasome inhibitors.
Among the 23 patients reported by Nakaseko, 5-year OS and PFS were 81% and 59.8%, respectively. 24 The latest study conducted by Yu et al. showed that among patients who received ASCT treatment, the 5-year OS rates for the 3 periods before 2010, 2010 to 2014, and 2015 to 2019 were 70.0%, 85.4%, and 92.2%, respectively. 18 In our meta-analysis, the 5year PFS rate was 74.0% (95%CI: 0.630.84) (Figure 4A), the 5year OS rate was 87.0% (95% CI: 0.730.98) of persons who received ASCT treatment (Figure 4B). Due to lack of data, we cannot make statistics on OS and PFS of patients treated with immunomodulators or proteasome inhibitors. Li et al. reported a phase II trial of 12 cycles of lenalidomide and dexamethasone in 41 patients. After a median follow-up of 34 months, the estimated 3-year OS and PFS were 90% and 75%, respectively. 5 However, Li et al. have performed a retrospective review of nearly 350 patients receiving melphalan and dexamethasone, ASCT, or lenalidomide and dexamethasone and concluded that the highest response rates were seen with ASCT. Patients receiving lenalidomide and dexamethasone had the shortest PFS and a trend for a shorted OS.

Long-term outcomes of patients who received autologous stem cell transplantation (ASCT) treatment. (A) 5-year progression-free survival (PFS) of nonclinical trials. (B) 5-year overall survival (OS) results of nonclinical trials. Logit transformation and random intercept logistic regression models. The red squares represent the remission rates and the horizontal lines reflect the 95% confidence interval (CI). The dark diamond corresponds to the 95% CIs for the pooled mutation rates.
Furthermore, proteasome inhibitors have also been increasingly applied to treat POEMS syndrome and have achieved satisfactory results.10,11,25 A retrospective study conducted by He et al., reported 20 patients who received a bortezomibbased regimen, the hematological response rate was 76.5%, VEGF response rate was 88.2% and no obvious neurological toxicity was found. 11 And a study confirmed that bortezomib is effective and safe for patients with renal failure. 26 In addition, case reports that in our statistical analysis, 9 patients were treated with bortezomib-containing treatment and all achieved CRH.10,26–30 However, bortezomib still has the possibility of aggravating the patient's neurological symptoms. Ixazomib is a secondgeneration proteasome inhibitor that is mainly used to treat MM. 31 Compared with bortezomib, ixazomib has a shorter dissociation half-life from 26S proteasome, has better tissue permeability, and can be taken orally. 31 A controlled clinical study conducted in China showed that ixazomib is beneficial to patient's PFS and has less additional toxicity. 32 At present, there is no report on the treatment of POEMS syndrome with ixazomib. Because ixazomib is convenient to administer and has fewer neurotoxic effects, we chose ixazomib sequential ASCT treatment for this patient, achieving CR. That provides positive clinical evidence for ixazomib in combination with ASCT in the treatment of POEMS syndrome.
Conclusion
ASCT, immunomodulators, and proteasome inhibitors all have reasonable response rates in treating POEMS syndrome patients, and ASCT-based therapy is the standard of care. Based on the above data, we speculate that immunomodulators and proteasome inhibitors-based therapy can be used in combination with this modality as induction and alone in patients who are not suitable for ASCT. In addition, ixazomib combined with ASCT therapy may be a safe and effective method for patients with POEMS syndrome.
Supplemental Material
sj-tif-1-tct-10.1177_15330338221123634 - Supplemental material for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis
Supplemental material, sj-tif-1-tct-10.1177_15330338221123634 for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis by Ji Ma, Xiaoyue Wang, Hui Lu, Zhihao Liang and Liang Wang in Technology in Cancer Research & Treatment
Supplemental Material
sj-doc-2-tct-10.1177_15330338221123634 - Supplemental material for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis
Supplemental material, sj-doc-2-tct-10.1177_15330338221123634 for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis by Ji Ma, Xiaoyue Wang, Hui Lu, Zhihao Liang and Liang Wang in Technology in Cancer Research & Treatment
Supplemental Material
sj-doc-3-tct-10.1177_15330338221123634 - Supplemental material for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis
Supplemental material, sj-doc-3-tct-10.1177_15330338221123634 for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis by Ji Ma, Xiaoyue Wang, Hui Lu, Zhihao Liang and Liang Wang in Technology in Cancer Research & Treatment
Supplemental Material
sj-csv-4-tct-10.1177_15330338221123634 - Supplemental material for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis
Supplemental material, sj-csv-4-tct-10.1177_15330338221123634 for Ixazomib Combined With Autologous Stem Cell Transplantation for POEMS Syndrome: A Case Report and Meta-Analysis by Ji Ma, Xiaoyue Wang, Hui Lu, Zhihao Liang and Liang Wang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Due to the nature of this article, no ethics statement was needed. Written informed consent was obtained from the patient for the publication of this case report.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
