Abstract
Introduction
Lung cancer was the leading cause of cancer death in both males and females and the second most commonly diagnosed cancer worldwide. 1 As 70% of lung cancers are present in advanced stages and are unresectable, small histological or cytological specimens are the primary diagnostic method for the majority of lung cancers. 2
Since the 1880s, percutaneous lung biopsy has been developed to be minimally invasive, highly accurate and commonly used in pulmonary lesions. 3 However, most studies about the small specimen obtained by computed tomography-guided percutaneous core needle biopsy (CT-guided PCNB) were focused on the distinction between malignant and benign lung lesions or between small cell lung cancers and non-small cell lung cancers.4–8 Nowadays driven by therapeutic development, more and more attention must be given to the diagnosis of histologic type and subtypes of lung cancers. Because different histologic type (such as adenocarcinoma [ADC] and squamous cell carcinoma [SQC]) of lung cancers may be sensitive to different therapies.2,9,10 For example, ADC is more responsive to pemetrexed than SQC; SQC is associated with life-threatening hemorrhage in patients treated with bevacizumab. Different subtypes of lung cancer (such as ADC in situ, minimally invasive ADC, and basaloid SQC) predict different prognosis.11–13
Given the increasing importance of accurate classification of lung cancer histologic type and subtypes, we evaluated the suitability of CT-guided PCNB specimens in typing and subtyping lung cancer based on the 2015 World Health Organization (WHO) classification of lung tumors. 14 The interobserver differences between pathologists with different experiences in diagnosing lung cancer histologic type and subtypes were also evaluated.
Material and Methods
Study Population
This retrospective study was approved by the institutional review board (approval number: 2021057) and informed consent of the patient was exempt. The reporting of this study conforms to strengthening the reporting of observational studies in epidemiology (STROBE) guidelines. 15 CT-guided PCNB and surgery for lung lesions performed from August 2007 to December 2015 were consecutively selected. Patients were considered eligible for this study if they had (1) CT-guided PCNB of lung lesions; (2) lung lesion surgery within 1 month after CT-guided PCNB; and (3) CT-guided PCNB and surgical specimen pathological sections were available for retrieve and re-diagnose. Patients were excluded if (1) lung lesion resection was not performed or was not performed within 1 month after biopsy; (2) patients received neoadjuvant chemotherapy or radiotherapy before surgery or CT-guided PCNB; (3) the bioptic or surgical specimen pathological sections were inadequate for reassessment. All patient details were de-identified.
Biopsy Procedure
All biopsy procedures were performed by an experienced radiologist or under the direct supervision of an experienced radiologist, who has engaged in CT-guided PCNB for at least 5 years. An 18-gauge coaxial automated cutting needle and a localization needle (Bard® Max-Core® biopsy needle; C.R. Bard, Inc.) were used for biopsy. All patients, except the ones who could not have contrast medium because of allergy or renal insufficiency, had contrast-enhanced chest CT images available for review before biopsy procedures.
The biopsy processes were shown in Figure 1. (1) Depending on the lesion location, patients were placed in prone, supine, or lateral positions. (2) Performing localization CT scans (a low-dose technique, lung care series: 50 mA; 120 kV; scanning field, 30-60 mm; slice thickness: 5 mm), to determine the appropriate puncture pathway and marking with a skin marker. (4) Repeat CT scan (biopsy single series: 50 mA; 120 kV; thickness, 4.5 mm; scanning field, 10 mm) was used to ensure the right localization of the skin marker. (5) After local anesthesia, a localization needle was inserted into the chest wall without puncturing the pleural, to indicate the needle entry route. Repeat CT scan to adjust the trajectory of localization needle. (6) Measuring lesion depth to determine the depth that the coaxial automated cutting needle required to be inserted. Then, the needle was directly advanced into the lung and fired to obtain a specimen (typically 2-3 specimens and 2 cm in length of each one). The dwell time of coaxial automated cutting needle in the lung was no more than 5s in each fire.

A 63-year-old female with a 2.1 cm nodule in the right middle lung lobe. (A) The patient was placed in lateral position. (B) The localization needle inserted into the chest wall indicates the needle entry route. (C) After specimens were obtained, repeat CT scan showed little pulmonary hemorrhage along the needle track.
The biopsy specimens were fixed in 10% formalin solution immediately and were sent to the pathology department for hematoxylin and eosin staining. Immunohistochemical test were requested by pathologists. Immunohistochemical staining was used to aid the diagnosis of lung cancer types. After biopsy, the outpatients were asked to stay in the waiting room for at least 1 h with observation of the nurses; the inpatients were sent back to the ward with the residents' observation. Post biopsy CT scan was used to identify procedure-related complications. The extent of pneumothorax was measured on CT images as the maximum distance from visceral pleura to parietal pleura (mild: ≤ 2 cm; moderate: 2-4 cm; and severe: >4 cm). 16 Intrapulmonary hemorrhage was defined as haziness along the needle tracks or in adjacent air spaces (mild: bleeding presenting as ≤ 2 cm in width; severe: bleeding presenting as >2 cm in width). 17
Histologic Type and Subtypes Diagnose
Histologic type and subtypes of lung lesion were diagnosed based on the 2015 WHO classification of lung tumors. 14 All surgical specimens and corresponding biopsy specimen pathological sections were diagnosed on 3 levels. First, diagnose malignancy or benign of the lesion; second, diagnose histologic type of the lesion, such as SQC, ADC, and tuberculosis; and third, diagnose histologic subtype of ADC and SQC, such as papillary ADC or non-keratinizing SQC.
All surgical specimen slices were reassessed in consensus by 2 experienced pathologists (working more than 15 years). All biopsy specimen slices were reassessed in consensus by 2 junior pathologists working for 10 years (group A) and 2 senior pathologists working for 15 years (group B), respectively, without knowing the pathological results of surgery. In case of disagreement, the final diagnosis is determined by discussion. Surgical results were regarded as gold standards.
Statistical Analysis of Diagnostic Accuracy
The histologic results of CT-guided PCNB specimens consistent with surgical specimens were regarded as correct diagnoses. Diagnostic accuracy was calculated as the ratio of correctly diagnosed cases to total cases. Pearson chi-square test was used to compare the differences of diagnostic accuracy between pathologists in group A and group B. Statistical significance was considered for P< .05.
Results
There were 2016 patients who received CT-guided PCNB during the study period, of which 160 patients received CT-guided PCNB and lung surgery within 1 month were finally included. 114 were men (mean age: 61.53 years ± 8.94; age range: 36-81 years) and 46 were women (mean age: 60.17 years ± 9.83; age range: 39-78 years). Pneumothorax occurred in 33.75% (54 of 160) cases, of which 79.63% (43 of 54) were mild, 12.96% (7 of 54) were moderate, and 5.56% (3 of 54) were severe and required chest drain insertion. Intrapulmonary hemorrhage occurred in 55.00% (88 of 160) cases, of which 63.64% (56 of 88) were mild, 36.36% (32 of 88) were severe. 11.36% (10 of 88) patients were accompanied by hemoptysis and 4 patients treated were with tranexamic acid. No patient occurred needle track seeding, air embolism, or death because of the procedure. Nine CT-guided PCNB specimens had epidermal growth factor receptor (EGFR) mutation examination, of which 4 were wild type, 2 were 19-del, and 4 were 21-L858R. Ten surgical specimens had EGFR mutation examination, of which 4 were wild type, 3 were 19-del, 1 was 21-L858R, 1 was 20-ins, and 1 had both T790M and 19-del.
Diagnostic Accuracy on the First Level
In group A, 126 malignant lesions and 19 benign lesions were correctly diagnosed, while 13 malignant lesions were falsely diagnosed as benign, 2 benign lesions were falsely diagnosed as malignant. In group B, 131 malignant lesions and 20 benign lesions were correctly diagnosed, while 8 malignant lesions were falsely diagnosed as benign, 1 benign lesion was falsely diagnosed as malignant. The total diagnostic accuracy of group A and group B were 90.63% (145 of 160) and 94.38% (151 of 160), respectively. There was no significant difference in the diagnostic accuracy on the first level between the 2 groups (P = .20).
Diagnostic Accuracy on the Second Level
The surgical results of the 160 patients were shown in Table 1. There were 42 SQC, 72 ADC, 6 pulmonary sarcomatoid carcinomas (PSC), 11 pulmonary neuroendocrine tumors (PNET), 6 adenosquamous carcinomas (ADSQC), 1 primary pulmonary lymphoma, 1 metastasis, and 21 benign cases. On the second level, all of 6 ADSQC were falsely diagnosed as ADC, SQC, or benign. Respectively, 14.29% (6 of 42) (group A) and 16.67% (7 of 42) (group B) of SQC were falsely diagnosed as ADC, 2.78% (2 of 72) (group A) and 1.39% (1 of 72) of ADC (group B) were falsely diagnosed as SQC. On the second level, the total diagnostic accuracy of group A was 67.50% (108 of 160), in which the diagnostic accuracy of malignant lesions versus benign lesions was 72.66% (101 of 139) versus 33.33% (7 of 21) (P < .01), the diagnostic accuracy of ADC versus SQC was 84.72% (61 of 72) versus 69.05% (29 of 42) (P = .04). The total diagnostic accuracy of group B was 72.50% (116 of 160), in which the diagnostic accuracy of malignant lesions versus benign lesions was 76.98% (107 of 139) versus 42.86% (9 of 21) (P < .01), the diagnostic accuracy of ADC versus SQC was 90.28% (65 of 72) versus 71.43% (30 of 42) (P = .01). Figure 2 showed hematoxylin and eosin stains of SQC and ADC. There was no significant difference between the two groups in the total diagnostic accuracy on the second level (P = .33), ADC (P = .31), and SQC (P = .81).
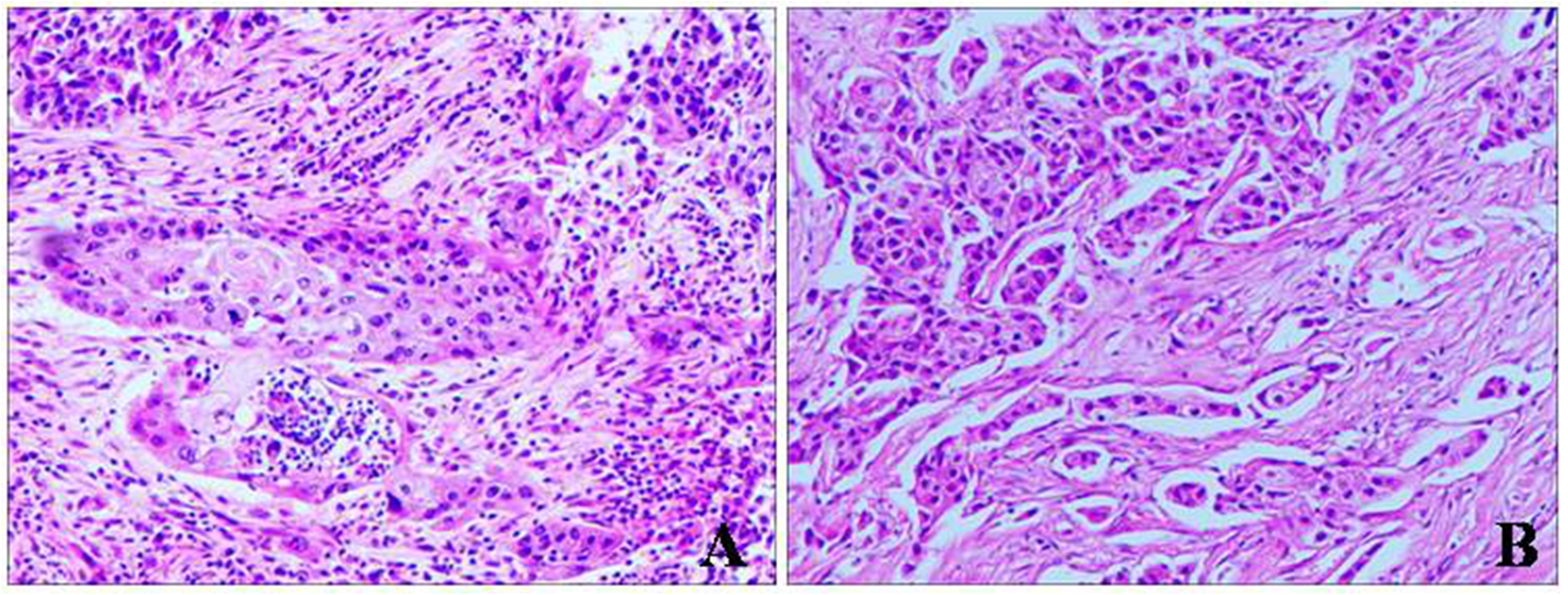
Hematoxylin and eosin staining for specimens obtained by CT-guided PCNB (original magnification × 100). (A) Lung squamous cell carcinoma (SQC) and (B) lung adenocarcinoma (ADC). Note: CT-guided PCNB: computed tomography-guided percutaneous core needle biopsy.
The Diagnostic Accuracy of Typing and Subtyping Lung Lesions in CT-Guided PCNB Specimens by Group A and Group B Pathologists.
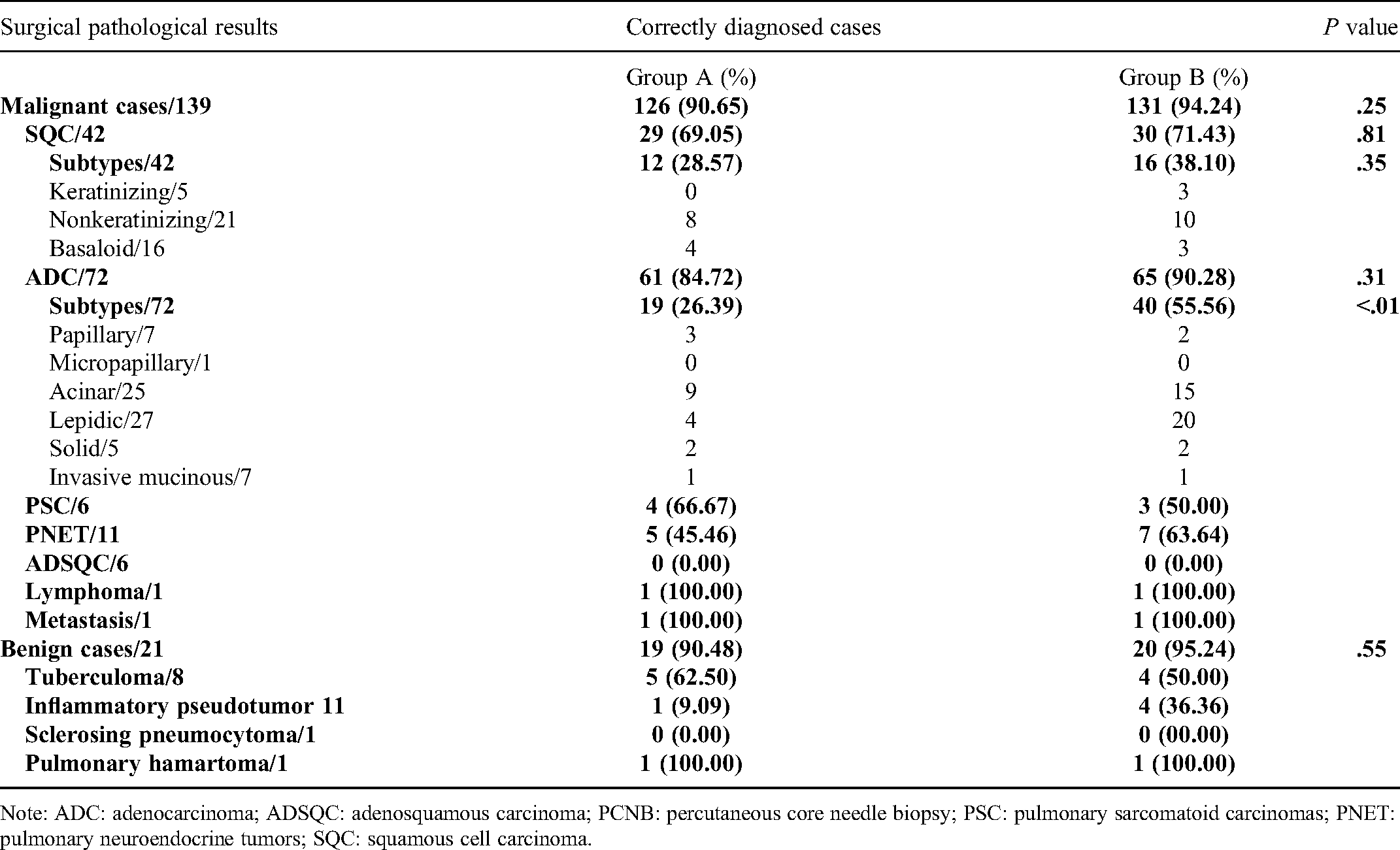
Note: ADC: adenocarcinoma; ADSQC: adenosquamous carcinoma; PCNB: percutaneous core needle biopsy; PSC: pulmonary sarcomatoid carcinomas; PNET: pulmonary neuroendocrine tumors; SQC: squamous cell carcinoma.
Diagnostic Accuracy on the Third Level
As shown in Table 1, on the third level, the total diagnostic accuracy of group A was 27.19% (31 of 114). Of which the diagnostic accuracy of histologic subtype of ADC versus SQC was 26.39% (19 of 72) versus 28.57% (12/42) (P = .80). The total diagnostic accuracy of group B was 49.12% (56 of 114). Of which the diagnostic accuracy of histologic subtype of ADC versus SQC was 55.56% (40 of 72) versus 38.10% (16/42) (P = .17). The diagnostic accuracy of ADC subtypes in group B was significantly higher than group A (P < .01). No significant difference was found between the 2 groups in the diagnostic accuracy of SQC subtypes (P = .36). Figures 3 and 4 showed hematoxylin and eosin stains of different subtypes of ADC and SQC.
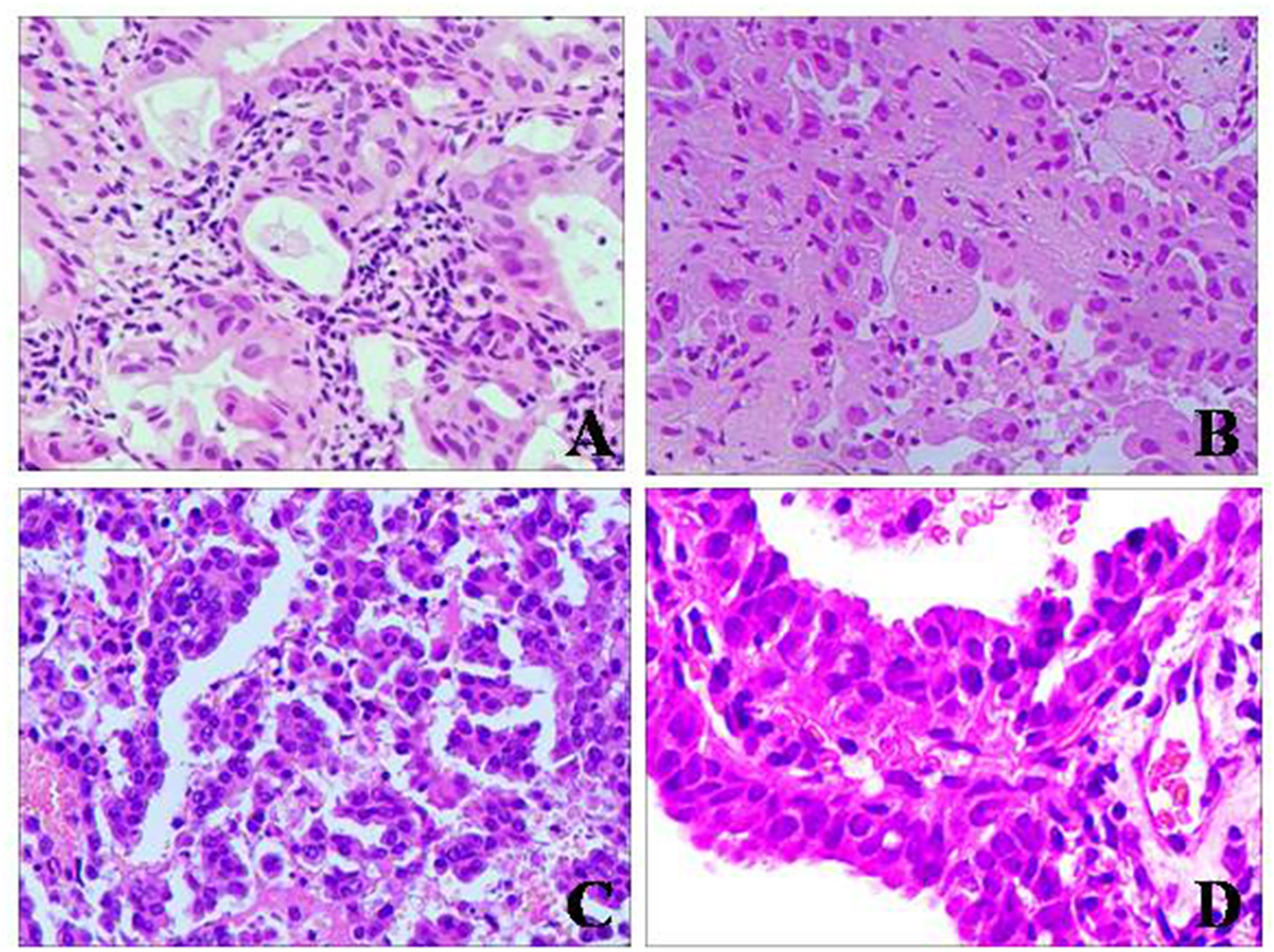
Hematoxylin and eosin staining for specimens obtained by CT-guided PCNB for lung adenocarcinoma (ADC) subtype diagnoses (original magnification × 200). (A) Acinar pattern, (B) lepidic pattern, (C) micropapillary pattern, and (D) papillary pattern. Note: PCNB: percutaneous core needle biopsy.

Hematoxylin and eosin staining for specimens obtained by CT-guided PCNB for lung squamous cell carcinoma (SQC) subtype diagnoses (original magnification × 100). (A) Keratinizing pattern, (B) nonkeratinizing pattern, and (C) basaloid pattern. Note: CT-guided PCNB: computed tomography-guided percutaneous core needle biopsy.
Discussion
In this study, we found that small specimens were suitable for lung cancer type diagnosis but not enough for subtype diagnosis. Pathologists with different working experiences showed no difference in diagnosing lung cancer types, while more experienced pathologists were better at diagnosing ADC subtypes. To our knowledge, this is the first study to analyze the diagnostic accuracy of small specimens obtained by CT-guided PCNB compared with surgical specimens on 3 levels (malignant and benign diagnosis, histologic types diagnosis, and histologic subtypes diagnosis) and compare the diagnostic differences between pathologists with different working experiences.
Studies had reported the diagnostic accuracy of small cytological and/or histological biopsy specimens in diagnosing lung cancer types, with diagnostic accuracy varied from 54% to 74.73%.18–20 In these studies, cytological specimens were also evaluated and compared with both surgery and endoscopic biopsies results. That may influence the result of diagnostic accuracy for lung cancer type, as small biopsy specimens may not accurately diagnose the histological types of lung cancers because of the heterogeneity of lung cancer.21,22 In our study, all the pathological results of small biopsy specimens were compared with corresponding surgery results to make the study results more reliable.
For pathologists with different working experiences, no significant difference was found in diagnosing lung cancer types. Steinfort et al 23 had reported that moderate interobserver agreement was seen in the interpretation of endobronchial ultrasound-guided transbronchial needle aspiration specimens for determination of non-small cell lung cancer types, which was similar to our study. While we found that the diagnostic accuracy of histologic types of malignant lesions was higher than benign lesions. The difference may come from the fact that pathologists pretended to use descriptive diagnosis when no typical benign histological structure was found and the false-negative result on the first level may also contribute to it. So, for a benign biopsy result, they should have the procedure reviewed to ensure the results are reliable.
There is inconsistency in the diagnostic accuracy of SQC and ADC in small preoperative specimens. Some studies showed that the diagnostic accuracy of SQC in small preoperative specimens was higher than ADC18,24 and other studies showed the opposite results.19,25 In our study, the diagnostic accuracy of ADC was higher than SQC in small histological specimens (90.28% vs 71.43%). We speculate this may be related to the application of immunohistochemistry and improved understanding of ADC. Diagnostic differences between small biopsy specimens and surgical specimens may come from a variety of reasons: lack of material, heterogeneity of tumor, and tissue necrosis. 19
Recent studies had analyzed the utility of core biopsy for histologic subtype diagnosis for lung ADC and found the overall concordance of biopsy samples with predominant subtype from surgical specimens were varied from 40% to 77%.26–29 In these studies, all included cases were already known as ADC, the pathologists just need to diagnose the subtypes. In our study, the pathologists not only need to diagnose the histological subtypes but also need to diagnose the histologic types at first. The diagnostic mode in our study was more consistent with a clinical process that a pathologist could not know the histologic type of a specimen before diagnosis. If the diagnosis of ADC were already known, the diagnostic accuracy of ADC subtype in our study was raised from 51.39% to 67.69% for experienced pathologists (which was comparable to previous studies) and raised from 26.39% to 37.93% for less experienced pathologists. The low diagnostic accuracy of lung cancer subtype may come from 2 reasons: one is the heterogeneity of lung cancer, a tumor may present different pathological subtypes; 22 another one is the pathologists' lack of understanding of the new WHO classification of lung tumor. So, for subtype diagnosis, discussion with experienced pathologists is recommended.
CT-guided PCNB was good to distinguish malignant pulmonary lesions from benign lesions, while the false-negative results brought by small biopsy specimens are inevitable. In our study for different experienced pathologists, respectively, 12 (7.5%) and 8 (5.0%), positive cases were falsely diagnosed as negative and no significant difference was found in pathologists with different working experiences. Studies showed that older age, insufficient tissue obtained, increased size of lesion, fewer adjustments of the needle, needle tip not in the lesion, and the occurrence of a pneumothorax were factors that associated with false-negative results.30–32 Doctors should paid more attentions to these factors when biopsy to decrease the false-negative rate.
There are several limitations in our studies. First, this is a retrospective study and all patients came from one single institution. Second, the study sample size is a little small and we hope to continue to study this problem in our future study after more samples were collected. Third, due to the insufficient understanding of molecular diagnosis for lung cancer before 2015 of our institution, seldom cases had molecular examinations that the value of small specimens for molecular diagnosis was not evaluated sufficiently.
In conclusion, the small specimens obtained by CT-guided PCNB were suitable for the diagnosis of lung cancer histologic types, which may contribute to the selection of suitable treatment strategies for the unresectable lung cancers. While for the diagnosis of lung cancer histologic subtypes, the small specimens were not sufficient enough and more experienced pathologists were better at subtyping ADC. So, for subtype diagnosis, discussion with experienced pathologists is recommended.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This retrospective study was approved by the Medical Ethics Committee, Zhongnan Hospital of Wuhan University (approval number: 2021057), and informed consent was not required.
