Abstract
Introduction
Neoadjuvant chemoradiotherapy (nCRT), surgery, and adjuvant chemotherapy (CT) were standard treatment strategies for rectal cancers (RCs), especially locally advanced RCs.1,2 The implementation of nCRT before surgery could downsize tumor stage, reduce local recurrence rates, and improve organ preservation and quality of life for patients with low-RC.
Ultra-low subtype was recognized as an independent poor prognostic factor for RC patients. 3 The rate of sphincter preservation surgery for ultra-low RC was still < 50% due to high local recurrence and surgeon experience. 4 Nowadays, tumoral response to neoadjuvant treatment has been proven as a key factor in transforming the mutilating radical surgery to a sphincter preservation approach.5,6
In the GRECCAR prospective study, 4 the circumferential resection margin (CRM) showed a strongly unfavorable prognosis for ultra-low RC, the 10-year survival rates with a CRM of below 1 mm was less than the CRM greater than or equal to 1 mm (P < .001, 22.2% vs 68.9%). The significant volumetric tumoral response after neoadjuvant therapy was associated with tumor-free CRM, which was clearly confirmed as an essential prognostic factor for operative strategy selection and long-term overall survival in this study.
Approximately 10% to 40% of patients achieved pathological complete response (pCR) after nCRT shown in several research which were confirmed by pathology and surgery. 7 Although various retrospective analyses showed that an increased tumor response rate had a positive correlation with several clinical as well as pathologic factors such as T stage, MMR status, tumor location, interval time, or dose of radiotherapy, there were limited studies to assess the relationship between the predictive factors and pCR in ultra-low RC patients.
In view of the fact, that the tumor pathologic response was hard to assess before resection, it will benefit patients with RC to establish an effective predictive system to evaluate the tumor response, which would assist in offering more suitable clinical decisions for different patients. For example, ultra-low RC patients with pCR could adopt transanal endoscopic microsurgery (TEMS) or close observation to preserve sphincter function.
This study aims to determine the factors that provide independent predictive values of pCR for RC patients and ultra-low RC patients with neoadjuvant CRT (chemo-radiotherapy) and propose a clinical point score to evaluate the prediction of pCR for these patients.
Methods and Materials
Patient Selection
The inclusion criteria were as follows: RC patients confirmed by pathology with cT1-T4 and cN0-2 stage, who had accepted nCRT and surgery. Whereas patients with distant metastases or no surgery, as well as no detailed information on clinical characteristics and complete pathological information, were excluded.
The patients with RC were defined as 3 groups by the distance to the anal margin. They are a group of ultra-low (≤ 3 cm from the lower edge of the tumor to the anal margin), a group of low (between 3 and 5 cm), and a group of medium-high (between 5 and 10 cm and more than 10 cm).
Mismatch repair (MMR) status was defined by immunohistochemistry (IHC). Deficiency mismatch repair (dMMR) was defined as negative expression of human mutS homolog 6 (hMSH6), PMS2 genes, human mutL homolog 1 (hMLH1) or human mutS homolog 2 (hMSH2). Proficiency mismatch repair (pMMR) was defined as all the positive expressions of these 4 proteins. All resected specimens were assessed by 2 pathologists, and pCR was defined as the absence of any viable adenocarcinomatous cells in the resected sample using the American Joint Committee on Cancer (AJCC). 7 Circumferential tumor extent rate (CER) was calculated by the formula: maximal diameter of tumor/maximal diameter of total inner circumference × 100 8 (Figure 1).
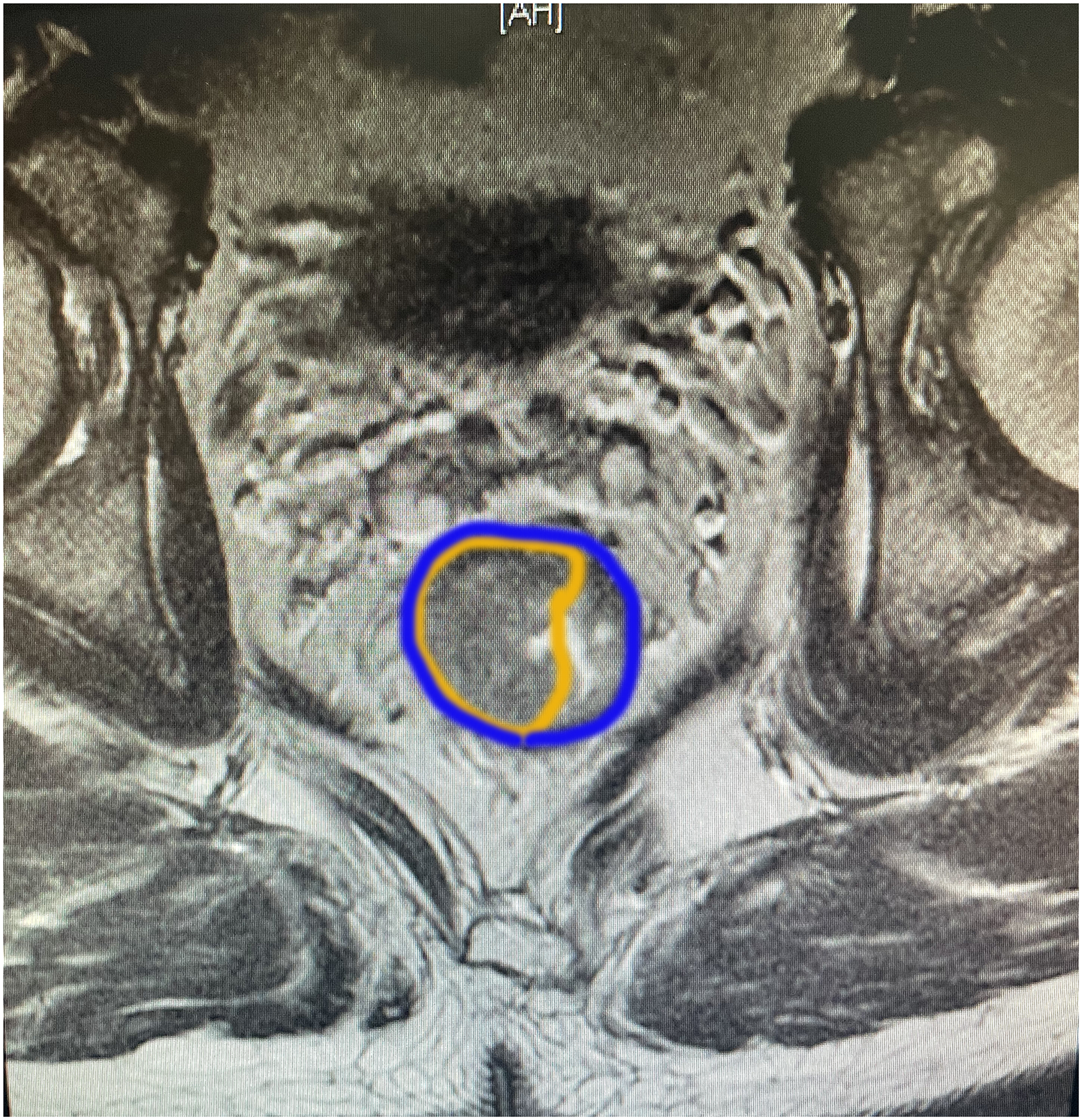
The graphical representation of CER in MRI.
A total of 402 patients were eligible from December 2015 to November 2020 at the Radiation Therapy Department, Changhai Hospital, Naval Military Medical University. The analyzed data included age, gender, cTNM stage (preoperative stage assessed by pelvis MRI), pathological information, MMR status, tumor location, interval time to surgery, type of radiotherapy, and system treatment records. The primary tumor pathological regression grade after nCRT was assessed by the Dworak regression scale.
Treatment Technique and Patients’ Follow-Up
All patients received neoadjuvant radiation therapy (RT) including a long-course treatment scheme of 40 to 50.4Gy delivered in 20 to 28 fractions over 4 to 6 weeks and short-course neoadjuvant RT (25-33 Gy delivered in 5-11 fractions). Neoadjuvant RT treatments with IMRT (intensity modulated radiation therapy) delivered by linear Accelerator (Varian Medical Systems, Palo Alto, CA), VMAT (Elekta Medical Systems, Synergy, VMAT), and PLDR (pulsed low dose rate radiotherapy).
Formulas and Statistical Analysis
The logistic regression analyses were used to compare the different subgroups in univariate analysis. Multivariate analysis was performed to determine the independent predictive factors of pCR by using a logistic regression model. A risk score was derived from the final multivariate model. Factors with a P-value < .05 in the univariate analysis were entered as candidate variables into a multivariate analysis.
After the logistic regression model, the regression coefficients were rounded to the nearest decimal to facilitate the calculation in order to be easily applied in clinical practice. The receiver operating characteristic (ROC) curve was plotted using the IBM SPSS and the area of difference under the ROC curve (AUC) is used to assess the performance of the predictive model. Statistical significance was set at P < .05. Statistical analyses were performed using IBM SPSS version 24.0 (SPSS Inc., Armonk, NY).
Results
The Patients’ Clinical, Treatment, and Histopathological Characteristics
A total of 404 RC patients confirmed with pathology, who had undergone preoperative nCRT between December 2015 and November 2020 were enrolled in this retrospective study. Two patients were excluded because of the discontinued treatment. Most patients were male (292 cases, 72.6%) and the median age was 59 years (range: 24-81). All patients had Eastern Cooperative Oncology Group (ECOG) 0-1. 116 (28.9%) patients had a smoking history and 69 (17.2%) patients had a drinking history. Tumor staging was performed according to the eighth edition of the AJCC TNM classification. A total of 21 patients were classified as clinical stage I (cT1N0, cT2N0), 44 patients were classified as clinical stage II (cT3N0, cT4N0) while 337 patients were classified as stage III (cT1-4N1, cT1-4N2). The median distance of the tumor to the anal verge was 4 cm (range: 0-11 cm). The median length of the tumor was 4 cm (range: 1.3-12 cm), the median tumor thickness was 1 cm (range: 0.4-4 cm) and the median circumferential extent rate was 2/3 cycle (range: 1/4-one cycle). The median interval between the end of CRT and surgery was 8 weeks. The median CEA levels before and after preoperative CRT were 4.62 ng/mL (range: 0.64-675.72 ng/mL) and 2.59 ng/mL (range: 0.5-270.50 ng/mL), respectively. Similarly, the carbohydrate antigen (CA199) values before and after preoperative CRT were 14.19 U/mL (range: 2.17 -> 1200 U/mL) and 7.89 U/mL (range: 1.22 -> 1200 U/mL), respectively (Table 1).
Patients’ Clinical, Treatment, and Histopathological Characteristics.

Abbreviations: CEA, carcinoembryonic antigen; CER, circumferential tumor extent rate; MMR, mismatch repair; RT, radiotherapy; VAMT, Volumetric Modulated Arc Therapy; IMRT, intensity modulated radiation therapy; PLDR, pulsed low dose rate radiotherapy; TRG, tumor regression grade.
A total of 372 patients received concomitant CT: 5-Fu and leucovorin or capecitabine in 344 patients and with the addition of oxaliplatin in 28 patients. Besides that, in all 402 patients, 326 patients received neoadjuvant CT, which consisted of 5-FU/LV or capecitabine alone in 26 patients and FOLFOX/CAPEOX in 300 patients. According to the NCCN guidelines (version 3.2022), patients not receiving standard neoadjuvant CT were excluded. A total of 379 patients were treated with long-course RT with a total radiation dose of 50 to 50.4 Gy delivered in 25 to 28 fractions over 5 weeks, whereas 23 patients with short-course RT were carried out with a total dose of 25 Gy delivered in 5 fractions in 1 week. The median interval between CRT and surgery was 8 weeks.
Response to nCRT
A total of 402 patients were included, among which 75 (18.66%) patients had pCR after surgery. When histological types were assessed, 301 (74.9%) patients were found to be well/moderately differentiated and 42 (10.4%) patients were poor/undifferentiated. Tumor regression was graded (TRG) from 0 to 3. Among 402 patients, 75 (18.7%) patients had a complete response to CRT (TRG 0), and 260 patients (64.6%) had a good response (TRG 1 or 2). Poor response was observed in 67(16.7%) patients (TRG 3).
Predictive Model of pCR for RC Patients
Based on univariate analysis, pCR was associated with CER (P < .001), CEA level before and after CRT (P = .032), and the interval between the end of CRT and surgery (P = .016). For the analysis of histopathological characteristics, pCR might be associated with histological type (P < .001), cytokeratin 5.2 (CAM5.2) status (P < .001), and TOPO II status (P = .048) (Table 2). Multivariate logistic regression analysis revealed that CER (≤ 2/3 cycle vs > 2/3 cycle, P < .001, OR = 4.834, 95% CI: 2.309-10.121), CEA level (both ≤ 5 vs pre > 5 and post ≤ 5 vs both > 5, P = .033, OR = 1.537, 95% CI: 1.035-2.281), and the interval between the end of CRT and surgery (P = .031, OR = 2.412, 95% CI: 1.086-5.358) were predictive factors for pCR (Table 3). The point values of the predictors were assigned by the regression coefficients and were rounded to the nearest decimal for easy calculation (Table 4).
Univariate Analysis of Predictors Associated With pCR.

Abbreviations: pCR, pathological complete response; CRT, chemoradiotherapy; CER, circumferential tumor extent rate; RT: radiotherapy; dMMR, deficiency mismatch repair; pMMR, proficiency mismatch repair.
a Univariable analysis with logistic regression; bold values: P < .05.
Multivariate Analysis of Predictors Associated With pCR.
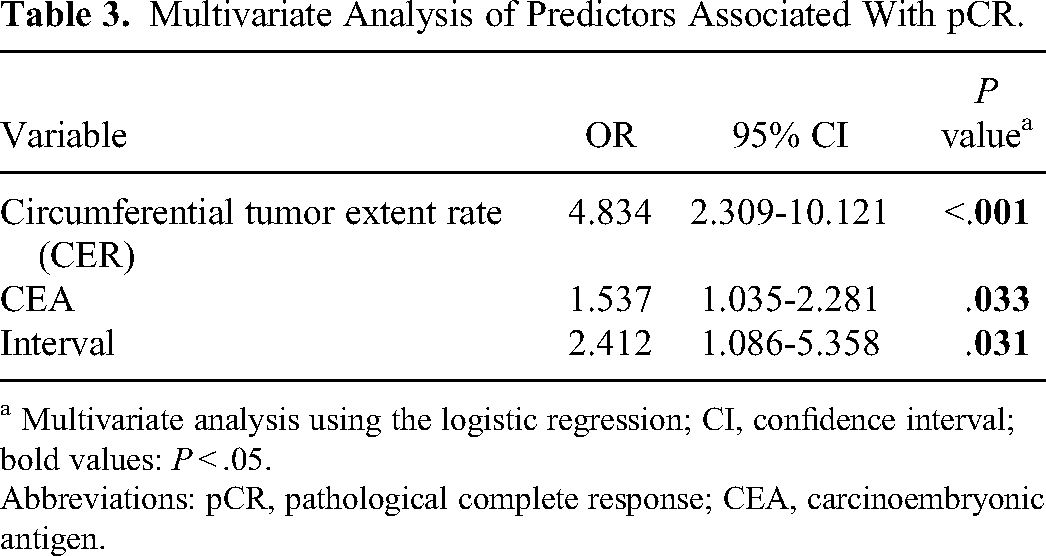
a Multivariate analysis using the logistic regression; CI, confidence interval; bold values: P < .05.
Abbreviations: pCR, pathological complete response; CEA, carcinoembryonic antigen.
Calculation of the Predictive Model Score.

Abbreviations: CRT, chemoradiotherapy; CER, circumferential tumor extent rate.
The total score for the 3 predictive factors could thus range from 0 to 4. The median score of the prognostic factors score in all patients was 3.0 points. If the scores were divided into 2 groups, patients with scores of 3 to 4 showed a higher rate of pCR than those with scores of 0 to 2 (27% vs 6.7%, P < .001). The sensitivity was 85.3% (64/75) with 47.1% (173/327) for specificity, 27% (64/237) for positive predictive value, 93.3% (154/165) for negative predictive value, and 54.2% (218/402) for overall accuracy. Furthermore, if the scores were divided into 2 groups, 0 to 3 versus 3.5 to 4, only 20 of the 75 patients with a score of 0 to 3 achieved a pCR, whereas 55 patients with scores of 3.5 to 4 achieved a pCR (30.6% vs 9.1%, P < .001). The sensitivity was 73.3% (55/75) with 61.8% (202/327) for specificity, 30.6% (55/180) for positive predictive value, 91.0% (202/222) for negative predictive value, and 63.9% (257/402) for overall accuracy (Table 5). The AUC of the predictive model was 0.709 (95% CI: 0.649-0.769), which was significantly higher than the 3 clinical factors, circumferential tumor extent rate (CER) (0.646, 95% CI: 0.584-0.709), interval time (0.563, 95% CI: 0.495-0.631) and CEA level (0.586, 95% CI: 0.518-0.655) (Figure 2). In addition, the variance inflation factor (VIF) of the 3 predictive factors were 1.017, 1.015, and 1.003, respectively. Tenfold cross-validation (mean AUC = 0.989) further demonstrated the reliability and high predictive value of the nomogram.

The area under the curve (AUC) of the prognostic model in all rectal cancer patients.
The Predictive Model for Assessment of pathological complete response (pCR).

Ultra-Low RC Patients
A total of 135 patients of ultra-low RC were further analyzed. Based on univariate analysis, pCR was associated with CER (P = .012), CEA level before and after CRT (P = .032), and hemoglobin before CRT (P = .047). For the analysis of histopathological characteristics, pCR may be associated with mismatch repair status (P = .008) and CAM5.2 status (P < .001) (Table 6). Multivariate logistic regression analysis revealed that both CER (≤ 2/3 cycle vs > 2/3 cycle, P = .003, OR = 7.203, 95% CI: 1.934-26.823) and MMR status (pMMR vs dMMR, P = .016, OR = 0.173, 95% CI: 0.041-0.720) were predictive factors for pCR (Table 7). The point values of the predictors were assigned by the regression coefficients and were rounded to the nearest decimal to facilitate the calculation in ultra-low RC patients (Table 8). The AUC of the predictive model was 0.653 (95% CI: 0.474-0.832) (Figure 3) and the predicted efficiency was decreased in a way.

The area under the curve (AUC) of the prognostic model in ultra-low rectal cancer patients.
Univariate Analysis of Predictors Associated With pCR in Ultra-Low Patients.

Abbreviations: pCR, pathological complete response; CRT, chemoradiotherapy; CER, circumferential tumor extent rate; RT: radiotherapy; dMMR, deficiency mismatch repair; pMMR, proficiency mismatch repair.
a Univariable analysis with logistic regression; bold values: P < .05.
Multivariate Analysis of Predictors Associated With pCR in Ultra-Low Patients.

Abbreviations: pCR, pathological complete response; MMR: mismatch repair.
a Multivariate analysis using the logistic regression; CI, confidence interval; bold values: P < .05.
Calculation of the Predictive Model Score in Ultra-Low Patients.

Abbreviations: MMR: mismatch repair; dMMR, deficiency mismatch repair; pMMR, proficiency mismatch repair.
Discussion
The role of pCR as a prognostic factor for RC patients has been widely reported in the last decades,9–13 and an average pCR rate of 22% (range 10%-30%) was observed in LARC patients with CRT before surgery. A review of 7 prospective studies including 2165 RC cases showed that the patients who received total neoadjuvant therapy (TNT) were associated with higher pCR rates of 29.9% compared with standard CRT of 14.9%. 14
Although the relationship between the OS and pCR was controversial in several studies, RC patients with pCR had a significant survival benefit, lower local relapse, and fewer metastases compared to non-pCR patients. In a meta-analysis of 16 studies, 15 complete responders had a 75% reduced risk of distant metastases and local recurrence rates compared with those nonresponders.
A long-term study demonstrated a 95% of 5-year overall survival rate and a 92% of 5-year disease-free survival for pCR patients, 77% of whom achieved a longer OS exceeding 10 years before the end date of the study. 16
In our study, the pCR rate was 18.66% (75/402) in all enrolled patients and 22.22% in patients with ultra-low RCs (30/135). Achieving pCR was even more important for organ preservation in patients with ultra-low RCs. According to the limited prospective evidence, there was insufficient evidence to substantiate that the clinical and pathology factors are associated with pCR to CRT in ultra-low RC. Therefore, patients with ultra-low RC were further analyzed in our study.
In recent studies,17–21 clinical characteristics of cTNM stage, tumor size, tumor location, the interval between neoadjuvant therapy and surgery, pathological factors of tumor differentiation, and mucinous histopathology were widely investigated to predict tumor response to CRT for RC. Moreover, the new predicting models of molecular biomarkers such as p53, p21, Ki-67, and epidermal growth factor receptor (EGFR) have been proven to be correlated with pCR for LARC, which strengthens the predicting efficacy of tumor response for preoperative CRT. 22 A genotype of PGS-LARC model 23 comprised of 15 genes for predicting pCR had been established with whole exome sequencing, which was superior to other clinical features on the sensitivity and specificity of pCR prediction.
The 402 RC tumor samples had been allocated in the training cohorts, and constructed new predictive models comprising clinical and pathological factors for pCR prediction after nCRT, with satisfactory AUC, accuracy, sensitivity, and specificity. The AUCs of the new models in the training cohorts were 70.9% and 65.3%, which were all higher than the respective clinical factors (CER, interval time, CEA level and CER, mismatch repair status) in AUC, accuracy, sensitivity, and specificity for all RC and ultra-low RC cases, respectively. The clinical implication of this study is that it could provide probability for clinical decision-making in formulating a more personalized treatment strategy.
Serum carcinoembryonic antigen (CEA) was widely used as a biomarker for the diagnosis, assessment of treatment efficacy, and prognosis prediction of colo-RC. Several studies have shown that the pre-CRT serum level of CEA ≤ 5 ng/mL was related to pCR and positive tumor regression.24–28 Cheong et al 29 demonstrated that both pre- and post-CRT CEA levels < 5 ng/mL and the change of CEA levels were associated with pCR (P < .001) and good response (OR = 5.07; 95% CI: 1.92-14.83, P = .002). Therefore, in our study, we evaluated the serum CEA levels of both pre- and post-CRT. The median CEA values before and after preoperative CRT were 4.62 ng/mL (range: 0.64-675.72 ng/mL) and 2.59 ng/mL (range: 0.5-270.50 ng/mL), respectively. We also analyzed the relationship between serum CEA and treatment response to CRT. Multivariate logistic regression analysis revealed that CEA level (both ≤ 5 vs pre > 5 and post ≤ 5 vs both > 5, P = .033, OR = 1.537, 95% CI: 1.035-2.281) was a predictive factor for pCR on the treatment response to CRT in patients with LARC.
The most commonly reported clinical factors related to pCR included cTNM stage, tumor size, circumferential extent rate (CER), and the tumor distance to the anal margin. It was also observed in our study that CER less than 2/3 was a predictor of pCR in univariate analysis (P < .001). A limited circumferential extent rate of less than 50% or 60% was considered as a significant predictor for patients with pCR in several studies.30,31 CER had a positive correlation with tumor size, tumor invasion depth, nodal involvement, and TNM stage. 8 Although there was no relationship between cTNM stage and pCR in our study, CER was easy to evaluate by MRI imaging as an independent predictor with pCR preoperative CRT.
There is sufficient evidence to demonstrate that the longer interval between neoadjuvant and surgery is not associated with an increased cumulative rate of pCR.32–34 A phase III randomized trial showed that the delayed surgery interval of 11 weeks did not increase the rate of pCR after surgical resection compared to the 7-week group. Moreover, a longer interval may be associated with higher morbidity and more challenged mesorectal excision. 35
A recent meta-analysis 36 including 7 randomized trials showed a positive trend of pCR percentage over the weeks of interval before surgery was established until 16 weeks, which suggests that at least 10 weeks should be considered to achieve 95% of pCR cases. In our study, the relationship between interval and response to CRT was evaluated. Multivariate logistic regression analysis revealed that an interval of ≥ 8 weeks before surgery (P = .031, OR = 2.412, 95% CI: 1.086-5.358) was a predictive factor for pCR on the treatment response to CRT in patients with LARC.
A total of 33.6% (135/402) patients with ultra-low RC had achieved 22.22% pCR, which is better than the overall pCR rate of 18.66% (75/402) in our study. Das et al 30 analyzed 562 RC patients and found that lower tumor distance to the anal verge was a significant predictor of greater rates of pCR and downstaging.
The analysis of pCR in ultra-low RC patients was rarely reported in previous studies as an independent cohort. Univariate analysis revealed that both tumor circumferential extent rate (CER) (P = .003) and MMR status (P = .016) were independent predictors for pCR in patients of ultra-low RC in our study. Microsatellite instability (MSI) is a genome phenotypic abnormality that shows the defects in mismatch repair (MMR) system, in meanwhile, MSI exists in 15% of colorectal patients. 37 RC patients with MSI-high (MSI-H)/dMMR had been reported as a positive prognostic factor compared to MSI-low (MSI-L), microsatellite stability (MSS) or pMMR in several studies.38,39 On the other hand, both pMMR and MSS were found to be significantly related to pCR in RC patients who accepted nCRT. 40 The relationship between pCR and MMR or MSI status in RC is still contradictory. MMR status had not been found to correlate with pCR for all cases in our study, while dMMR was found to have higher expression in the complete response group for ultra-low RC.
Some prospective trials supported that nearly 40% of colorectal cancers with dMMR responded to immunotherapy of the programmed death-1 (PD-1) blocker. 41 Now the immunotherapy combined with conventional CT was under investigation to prove its effectiveness in metastatic colorectal cancer with dMM. 2 Many clinical trials were designed to evaluate the effectiveness of immunotherapy in the neoadjuvant setting for dMMR RCs. To our knowledge, this is the first study of the pCR predictive model in ultra-low RC after nCRT. Mismatch repair (MMR) status was found as a predictive factor for pCR in ultra-low rectal patients. These outcomes may provide significant evidence in increasing pCR rate, preserving organ function, and designing to evaluate the effectiveness of immunotherapy in the neoadjuvant setting for ultra-low RC in future prospective studies. Moreover, the watch and wait strategies for different groups of patients with RC could be classified with reference.
The combination of clinical features, pathological characteristics, imaging information features, molecular markers, and gene expression profiling could predict the efficacy of neoadjuvant therapy more accurately and guide the individualized clinical strategies for RC patients.
We proposed a new prediction model including clinical features, pathological characteristics, imaging information features, and molecular markers for different groups of RC patients. It will be beneficial for RC patients to evaluate the tumor response to nCRT with such an established effective predictive system.
The clinical significance of the predictive model was to facilitate the calculation and provide a new predictive system for ultra-low RC. Whereas some limitations in our study to establish the system. Firstly, it was a retrospective study in a single institution. Secondly, the incomplete pathological information for partial pCR patients before treatment may cause statistical bias. Lastly, large patient samples as well as longer follow-ups were needed. The applicability warrants prospective study for further validation.
Conclusion
In conclusion, we developed a prediction model to evaluate the trend of tumor response to nCRT for both RC patients and ultra-low RC patients.
Footnotes
Abbreviations
Acknowledgments
None.
Authors’ Contributions
SQ, LG, and TD have made equal contributions to the study and drafted the manuscript. XY made contribution to the statistical analysis. ZK made contributions to data collection. ZHJ is the principal investigator (PI) of this study and made contributions to the study design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This retrospective study obtained approval from the independent Ethics Committee of Changhai Hospital on August 30, 2019, The ethics number is CHEC2019-123 and all patients had signed informed consents.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ministry of Science and Technology of China (2022YFC2503701, FW-ZXKT2022102502004), Shanghai Health Commission Leading Talent Program (2022LJ019), National Natural Science Foundation of China (82272742), Changhai Hospital Guhai Program (GH145-33) and Youth Starup Fund (2020QNB09).
