Abstract
Introduction
The androgen receptor (AR) has recently emerged as an appropriate surrogate indicator of prognosis and prediction in BC, a useful marker for the further refinement of BC subtype, and promising therapy in BC.1–3 The AR is a member of the sex nuclear steroid hormonal receptor family, which also includes estrogen receptor (ER) and progesterone receptor (PR). The AR also plays a critical role in metastatic development of cancer by promoting migration and invasion, through degradation of the extracellular matrix. 3 Perhaps surprisingly, the AR is the most widely expressed nuclear hormone receptor, more than ER or PR. The AR is accounted for 60% to 95% of the positive ER, 4 and 15% to 70% of the ER − ve BC.4–7 Overall, AR expression is varied in 56 to 90% of all BCs.4,8,9 The same to ER and PR, AR expression is correlated with a well-differentiated breast cancers, 10 and with more indolent BC. 11 Of interest, AR expression is also associated with better clinicopathological characteristics in the most aggressive BC types, such as enriched HER2 or triple-negative breast cancer (TNBC) subtype. 12 The high AR positivity has been correlated with a low risk (LR) of recurrence and death. Significant differences in AR expression have been identified in various molecular subtypes of BC with the different prognosis and clinical behavior, especially in TNBC.3,13 The association between AR expression and improved survival outcomes, especially in patients with hormone receptor (HR)-positive tumors, TNBC, and female BC were demonstrated. 14 Unexpectedly, including 30% to 50% of all ER + ve BCs show de novo and/or acquired resistance to traditional endocrine therapies.15–17 This treatment resistance could result from malignant cell adaptation from estrogen to androgen dependence. 18
Additionally, the ratio of nuclear AR to ER may critically have an influence on tumor biology and respond to endocrine therapy. A high AR:ER ratio may be a prediction of suboptimal response to ER-directed endocrine treatment. Additionally, high AR:ER ratio may also identify a BC subset that would respond more favorably to enzalutamide alone or combined with tamoxifen or aromatase inhibitors (AIs). 18 Interestingly, in TNBC, Lehmann et al. demonstrated that luminal androgen receptor (LAR) tumors are driven by AR signaling and AR represents a targeted therapy for this subtype. Importantly, AR status in TNBC patients represents a molecular marker for the selection of patients before anti-androgen therapy. 1
BC is a heterogeneous extraordinarily hormone-dependent tumor with different molecular subtypes that varies in the outcomes and therapy response. It is clear that the efficacy of systemic adjuvant chemotherapy or endocrine therapy, or first-line anti-HER2 regimen has been demonstrated in BC. Although in BC, the early detection, diagnosis, and treatment are improved; however, the survival rate is modest, mainly due to the unselective inclusion of patients within a broad risk category. Therefore, evaluating biological prognosis and prediction in BC, which are being developed, is still the most important target in BC. Among the prognostic parameter investigated, AR expression by immunohistochemistry (IHC) staining, received considerable attention as promising markers of prognosis and prediction. For the aim of improving the survival rate of patients with BC, making exact decisions on the adjuvant treatment after operating for BC is very important. Thus, this requires an essential demand for the application of the appropriate treatment protocols, which are used in Vietnam for managing patients with BC. 19 Therefore, to comprehensively decipher the role of AR in BC, the current study was investigated to define the AR expression and AR:ER ratio, and explored their correlation with the clinicopathological features, prognosis, and survival outcomes in the various subclasses of invasive BC.
Material and Methods
Patients and Sample Selection Criteria for Analysis
This is a retrospective cohort study of 522 patients with primary BC who were operated from 2011 to 2014, at the National Cancer Hospital, Vietnam. Participants were selected for the study using the method of convenience sampling. STROBE guidelines were applied. 20 Next, only treatment-natïve tumors, the primary infiltrating carcinoma of breasts were selected. The patients who presented with second, in situ carcinoma, another malignancy of BC (eg malignant phyllode tumor), or relapse BCs were excluded. The clinical information of patients was recorded, such as age, location of tumor, and date at initial diagnosis, therapy information, which were extracted from medical patient charts and records. All patients were operated onto removing the tumor by modified radical mastectomy, or conservative surgery, and they were combined with axillary lymph node dissection. Tumors were evaluated in their maximum diameter. Tumor and nodal samples were performed via histopathological tests.
After surgery, 468 of the cases were treated by adjuvant chemotherapy. Also, all positive HR cases were received by endocrine therapy. Among HER2-positive BCs, only 2 patients were able to pay for all expenses for target treatment by trastuzumab. All individual information was deleted or disguised, in order to make sure anonymity was present for the patient.
Histopathology
All specimens were received in the operating room and then transferred to the pathology department. Samples were fixed in 10% neutral formalin for 24 h. Nodal and tumor samples were obtained by routine pathological techniques, such as hematoxylin and eosin (H&E) staining. Experienced pathologists evaluated all histopathological features, such as tumor size, histopathological type, grade, nodal status, and peritumoral lymphvascular invasion (LVI). Histopathological types were classified according to the 2019 WHO classification. Histologic grades were assigned according to Elston and Ellis as grades I, II, and III. The Nottingham Prognostic Index (NPI) was divided into good, moderate, and poor NPI.
Immunohistochemistry and Fluoresence In Situ Hybridization (FISH)
All IHC staining was tested for formalin-fixed, paraffin-embedded tissue sections. The IHC method was performed by a Ventana automated machine, using ER, PR, HER2, Ki67, and AR markers in whole sections. All primary antibodies belonged to Ventana company with already used condition, such as the primary monoclonal mouse anti-human ER (Ventana-SP01), anti-PR (1E2) rabbit monoclonal primary antibody, monoclonal mouse anti-human c-erbB-2 oncoprotein, rabbit monoclonal (Ventana-4B5), confirm anti-Ki67 monoclonal rabbit antibody (Ventana-30-9), a rabbit immunoglobulin G1 monoclonal antihuman AR primary antibody (SP107, Lot No. 1226104B, Ventana), respectively. The 2011CAP/ASCO guidelines were used to assess ER and PR status. ER/PR positivity was altered to 1%. 21 HER2 IHC/FISH was evaluated by the 2007 CAP/ASCO guidelines. 22 A HER2 mark of 3 plus was considered positive HER2, or overexpression. Ninety six patients (19.2%) who exhibited an IHC HER2 score of 2 plus were tested by FISH to identify amplification of the HER2 gene 21.9% of HER2 gene amplification occurred by FISH. Cutpoint for Ki67 was proposed by the 2013 St. Gallen consensus meeting, Ki67 index was divided into 2 levels: low (≤20%) and high (>20%). 23
For AR, nuclear staining was evaluated by applying the same approach for HR score and considered at least one percent nuclear staining of any intensity (1 + to 3 + ) as a positive AR assay. The specific percentage of positive cells and their intensity of staining (1 + -3 + ) were also recorded for all cases. 21
To avoid the detective bias, the positive and negative controls were applied for IHC stain. IHC interpretation was analyzed independently by 2 experienced pathologists (blinders), using the international guidelines, and any disagreement was resolved by reinvestigation to reach a consensus.
AR:ER Ratio Calculation
AR and ER nuclear staining percentages were compared. Both AR and ER percent cell staining contribute to the AR:ER ratio. The ratio of nuclear AR to ER was calculated, and the AR:ER ratio cut-off point was 2.0. 18 This ratio was compared with clinicopathological features and survival outcome of the positive ER BCs.
Variables of Interest
Clinicopathological characteristics and treatment modalities were reviewed and collected from our hospital's digital medical record database. Patient features: (1) age at diagnosis and (2) young and elder groups. Tumor data: (1) tumor site, (2) tumor size, (3) tumor stage was based upon criteria by the American Joint Committee on Cancer (AJCC, seventh edition). Pathological features: (1) histopathological type (ductal NOS, lobular, mucin, and other), was classified according to 2019 WHO classification, (2) histologic grade of primary tumor were assigned according to Elston and Ellis, (3) lymph node metastasis, (4) the Nottingham Prognostic Index (NPI) was calculated for all BCs using the formula: NPI = 0.2˟tumor size (cm) + lymph node stage (1, 2, or 3) + histological grade (1, 2, or 3), (5) LVI, (6) tumor-infiltrating lymphocytes (TILs). IHC: (1) AR, (2) ER, (3) AR:ER, (4) PR, (5) HER2, (6) Ki67, (7) HR status was dichotomized into HR positive (ER + and/or PR + ) and negative (ER − and PR − ), (8) HER2 status was divided into HER2 positivity and negativity (any HR expression), (9) TNBC with AR expression was dichotomized into LAR + (AR presence) and LAR − (AR absence). Outcome: (1) overall survival (OS) and (2) disease-free survival (DFS).
The Combined Immunophenotypes of AR Expression
The AR expression of patients with enriched HER2, TNBC, or positive HR of the current study were combined into the different immunophenotypes, such as LAR- and LAR + groups (loss of all ER, PR, and HER2 plus AR − , or AR + ), AR-HER2 + and co-expression of AR and HER subclasses, and AR-HR + and AR + HR + groups (presence of ER and/or PR added in negative AR, or AR positivity) (Figure 1).

Flowchart for identification of immunophenotypic stratification of AR expression.
Definition of Breast Cancer Molecular Subtypes and Risk Category
All BC patients were classified into the different molecular subtypes and risk categories based on age, clinicopathological, and IHC data. According to St. Gallen 2013's criteria, molecular subtypes are composed of Luminal A (LUMA), Luminal B HER2 − (LUMBH − ), Luminal B HER2 + (LUMBH + ), HER2, and TNBC (Figure 1). 23 This approach used IHC criteria for its definition of ER and PR expression, the detection of HER2 overexpression and/or amplification, and Ki67 index, toidentify the molecular subtypes. According to St. Gallen 2007's criteria, BCs were divided into the different risk categories. Initially, patients were categorized into main 3 risk groups: LR, intermediate risk (IR) , and high risk (HR), based on the nodal status. After that, the modified versions were published in 2007 by adding more clinicopathological and IHC features to the 2005 stratification, and. 24 Main risk categories of BC patients were classified as LR (including negative node and all of the features such as pT≤2cm, histopathological grade 1, absent LVI, positive ER/or PR, Her2/neu neither IHC overexpression nor gene amplification, and patients aged≥ 35 years), IR (including OR negative node and at least one of the characteristics such as pT>2cm, grades 2 to 3, presence of LVI, negative ER/ or PR, Her2/neu either IHC overexpression or gene amplification, or patients aged< 35 years; either 1 to 3 metastasized nodes, and positive ER/ or PR, and Her2/neu neither IHC overexpression nor gene amplification), and HR is identified by OR 1 to 3 positive nodes and negative ER/or PR, Her2/neu either IHC overexpression or gene amplification EITHER 4 or more positive nodes.
Follow-up and Outcomes
OS was a period of OS was defined as the date of initial diagnosis to the dead day, due to BC, or the last available time before losing follow-up. Patients would be censored if they did not die of BC. Death documents were used to record the dead date of BC patients, such as certificates, which were issued by the commune government in Vietnam. The recurrence and dates were demonstrated by imaging analytic and/or morphological results. Patients would be censored until the dead date if they did not present any relapse. DFS was identified by a length of time which was measured as the date of BC surgery until the diagnosis of the recurrent BC or BC-specific death, including locoregional relapses and distant metastasis. Two hundred fifty three patients with BC were recorded for follow-up information, ranging from 12 to 92 months, a mean of 66.23 ± 18.22 months.
Ethical Considerations
All participants of the protocol’s this study was approved by the Scientific and Ethical Committee of CREDCA, Vietnam as number: 1520/QD-DDNCYSH, Dec 10, 2020. Written informed consent was applied to all patients before enrolling them in the study. Patients could withdraw from the study at any time without any threats or disadvantages and for no stated reasons.
Statistical Analysis
To determine the clinicopathological differences between the AR expression, and AR:ER ratio, the Pearson chi-square test, likelihood ratio, and Fisher's exact test were performed. Mann–Whitney U test was applied to access the percentage of ER or AR expression for AR:ER ratio. The Kaplan–Meier model was used to investigate the OS and DFS, according to the AR expression, and AR:ER ratio. Log-rank test was performed to compare survival curves. If a P value is less than 0.05, a difference will be considered statistically significanct. Univariate Cox regression analysis was applied to identify prognostic factors associated with DFS and OS. In multivariate analysis, Cox proportional hazards regression model was performed to calculate hazard ratios (HRs) and their associated 95% confidence interval (95% CI) to determine independent prognostic factors of OS and DFS including AR expression, AR:ER ratio, age group, Ki67 index, TILs, NPI, histological grade, risk group, pTNM, and lymph node metastasis. All of the analyses were conducted using the statistical software of SPSS version 19.0.
Results
Correlations of Some AR Immunophenotypes and Clinicopathological Features of Invasive BC
The current study was conducted on all 522 patients with infiltrating BC that had undergone operations. Table 1 shows the patients’ baseline clinicopathological features and correlations to some AR immunophenotypes. Among all 522 BC patients that were immunohistochemically stained for AR marker, the proportion of positive AR was 65.3%, AR:ER ratios were 90.3% and 9.7% (AR:ER<2 and ≥2, respectively), LAR + subtype was 45.4%, and AR-HER2 + and AR + HER2 + were 40.9% and 59.1%, in turn. Mann–Whitney U test was applied to access the percentage of ER or AR expression for AR:ER ratio. In regard to AR/ER<2, the ER percentage reached to 78.47 ± 1.25 (95% CI: 76.01-80.93), meanwhile, the percentage of AR was 27.43 ± 1.50 (95% CI: 24.48-30.37); conversely, in AR/ER≥2 group, ER quantity got 22.50 ± 2.21 (95% CI: 17.98-27.02), while, AR archived in 61.43 ± 4.36 (95% CI: 52.49-70.37) (P < .001 in both the Error Bar Plot) (Figure 3C and 3D, Figure 3a to d).

(A) OS of AR:ER ratio in invasive breast cancers (BCs). Chart B shows disease-free survival (DFS) of AR:ER ratio for infiltrating BCs. The log-rank test demonstrates that there was a powerful significant difference between the 2 survival curves for AR:ER ratio. (C) Percentage of the ER expression in group AR/ER<2 (n = 262) and AR/ER≥2 (n = 28), Mann–Whitney U test, P < .001. The error Bar plot D displays the expression of AR in percentage according to AR/ER<2 (n = 262) and AR/ER≥2 (n = 28) groups, Mann–Whitney U test, P < .001. Data are presented as mean ± standard error of mean. Photomicrographs indicated the positive AR and ER of tumor cell nuclei, AR:ER<2 and AR/ER≥2 (A: 70% of AR + ve, B: 80% of AR + ve, C: 20% of ER + ve, and D: all nuclei AR + ve; 400x).
Associations Between BC Baseline Clinicopathological Features With Some AR Immunophenotypes.

Fisher’s exact test.
Likelihood ratio.
Abbreviations: AR: androgen receptor; BC: breast cancer; LVI: lymphvascular invasion; NPI: Nottingham Prognostic Index; TILs: tumor-infiltrating lymphocytes; TNBC: triple-negative breast cancer.
Statistical differences were significantly observed between the AR expression with some clinicopathological parameters such as age group, Ki67 index, HER2 (P < .05); even the strong correlation revealed significantly the statistical differences with histological grade, presence of metastasized lymph nodes, NPI, LVI, TILs, pathological Tumor Node Metastasis (pTNM), molecular subtypes, HR status, and risk category (P < .001). In the abovementioned clinicopathological factors, women with the positive AR BC had a trend to be accounted for a higher proportion in the good characteristics than the poor features; in the good NPI, presence of AR was observed in 30.2%, higher than negative AR (10.5%) (P < .001). Similarly, in stage I BCs, AR positivity was more common than absent AR (30.8 vs 16.0%, respectively) (P < .001). These statistical differences continued to be maintained in the all AR immunophenotypes such as AR:ER, LAR − versus LAR + , and AR-HER2 + versus AR + HER2 + with factors as axillary lymph node status, risk subclasses. For other clinicopathological variables, among of the AR immunophenotypes, a statistically significant difference was seen between AR:ER with Ki67 index, molecular subtypes, and HER2. While in AR-HER2 + versus AR + HER2 + subgroups, there was a statistically significant difference for histological grade, NPI, LVI, pTNM. Interestingly, in ER+ve BCs, the larger AR:ER ratio, AR is strong positive and ER percentage is low, BC patient displayed the worse prognosis; BCs with AR:ER≥2 exhibiting poor clinicopathological features were demonstrated a higher rate than AR:ER<2 BCs; in BC patients with more 3 positive lymph nodes, AR:ER≥2 was observed in the larger frequency than AR:ER<2 (14.3 vs 7.3%, respectively) (P = .043 < .05). Similar to BCs with more 3 positive lymph nodes, in HR group, AR:ER≥2 was noted with a higher proportion than AR:ER<2 (32.1% vs 10.3%, respectively) (P < .004). An opposite trend was observed in the poor prognostic BCs (TNBC and HER2+), AR was the stronger positive, the prognosis of breast cancer was better; considering BC risk stratification, the frequency of LAR- was the more common than LAR+ (54.2 vs 28.6%, respectively) (P = .007), parallely AR-HER2+ percentage was observed in the higher frequency than AR+HER2− (70.8 vs 28.7%, respectively) in high risk BCs (P < .001) (Figure 5A to D).

(A) Overall survival of AR − HER2 + and AR + HER2 + immunogroups in invasive breast cancers (BCs). Diagram B shows disease-free survival (DFS) of 2 these immunophenotypes for positive HER2 BCs. The log-rank test illustrates that there was a closely significant difference between the survival curves of OS and DFS for AR-HER2 + and AR + HER2 + immunophenotypes, as well. Microscopical pictures were stained by IHC stain with AR and HER2 makers (A: AR − ve, B: AR positivity, C and D: HER2 overexpression; 400x).
On the contrary, no difference was noted for the expression of the all AR immunophenotypes with the remaining parameters such as young and older, tumor site, neoplastic size, and morphological type (P > .05).
Survival Estimates
Overall, the mean OS of the patients with operated BC was 79.76 ± 1.44 months, in which the negative and positive AR were 66.97 ± 2.82 and 84.81 ± 1.40 months, respectively. For AR:ER ratio, the mean OS of AR:ER<2 group displayed the higher (87.35 ± 1.37 months); on the contrary, AR:ER≥2 category demonstrated the lower (63.61 ± 8.2 months). Concerning the LAR subtypes in TNBC, LAR + revealed in 79.6 ± 3.63 months of the mean OS, meanwhile, LAR − was counted in the lower mean survival interval (59.45 ± 4.09 months, respectively). In regard to the AR expression combined with HER2 positivity, AR + HER2 + showed a higher OS than AR-HER2 + (83.14 ± 2.79 vs 62.09 ± 5.40 months, respectively). OS investigation of the AR expression related to positive HR state (ER and/or PR positivity) in BC, AR-HR+ immunophenotype accounted in the higher proportion than AR+ HR+ (85.86 ± 2.07 vs 84.57 ± 1.67months, respectively). In general, AR:ER<2 group illustrated the longest OS, on the contrary, patients with LAR- exhibited the worst.
OS curves according to AR expression are summarized in Figure 2A, a strongly noticeable correlation was demonstrated (P < .001). Patients who were positive for AR protein demonstrated a better prognosis with an OS rate of 82.7%. BC patients with AR negativity got a lower OS rate (56.0%). Figure 3A shows that a closed difference was statistically significant between OS curves, according to AR:ER immunophenotype (P < .001). AR:ER<2 group was clearly observed in higher than the AR:ER≥2 category in the OS proportion (87.9 vs 25.0%, respectively). The OS curves, according to LAR categories of TNBC, are shown in Figure 4A. BC patients with the LAR+ category revealed the better prognosis, with an OS rate of 82.5%; by the contrast, LAR- group exhibited in the lower OS rate (42.1%), a statistical significance was observed in these differences (P = .001). Regarding the AR + HER2 + group compared to the AR-HER2 + phenotype, Figure 5A showed that the OS of patients with AR + HER2 + was twice higher than the remaining BC phenotypes, 82.1 versus 41.1%, respectively; this adverse trend was illustrated in the statistical significance (P = .001). In contrast, observing the AR-HR + immunophenotype with the AR + HR + BCs, the demonstrated difference was not statistically significant (P = .138 > .05), and the OS rates of patients with AR-HR + were higher than the BCs with AR + HR + , it was 96.2% and 82.4%, respectively (Figure 6A). When investigating the AR expression according to ER status, in the ER + ve BC group, the OS of the patients with AR − ve was higher than that of the positive AR (95.2 vs 81.7%) (Figure 7A). However, the difference was not statistically significant (P = .20). Meanwhile, in ER + ve BCs, OS of patients with AR + was twice higher than AR − (85.5% vs 41.9%) (P < .0001) (Figure 7D). In the group of more 50 year-old, the OS of breast cancer patients with AR+ve were significantly higher than the AR−ve group (P = .0011) (Figure 8A).
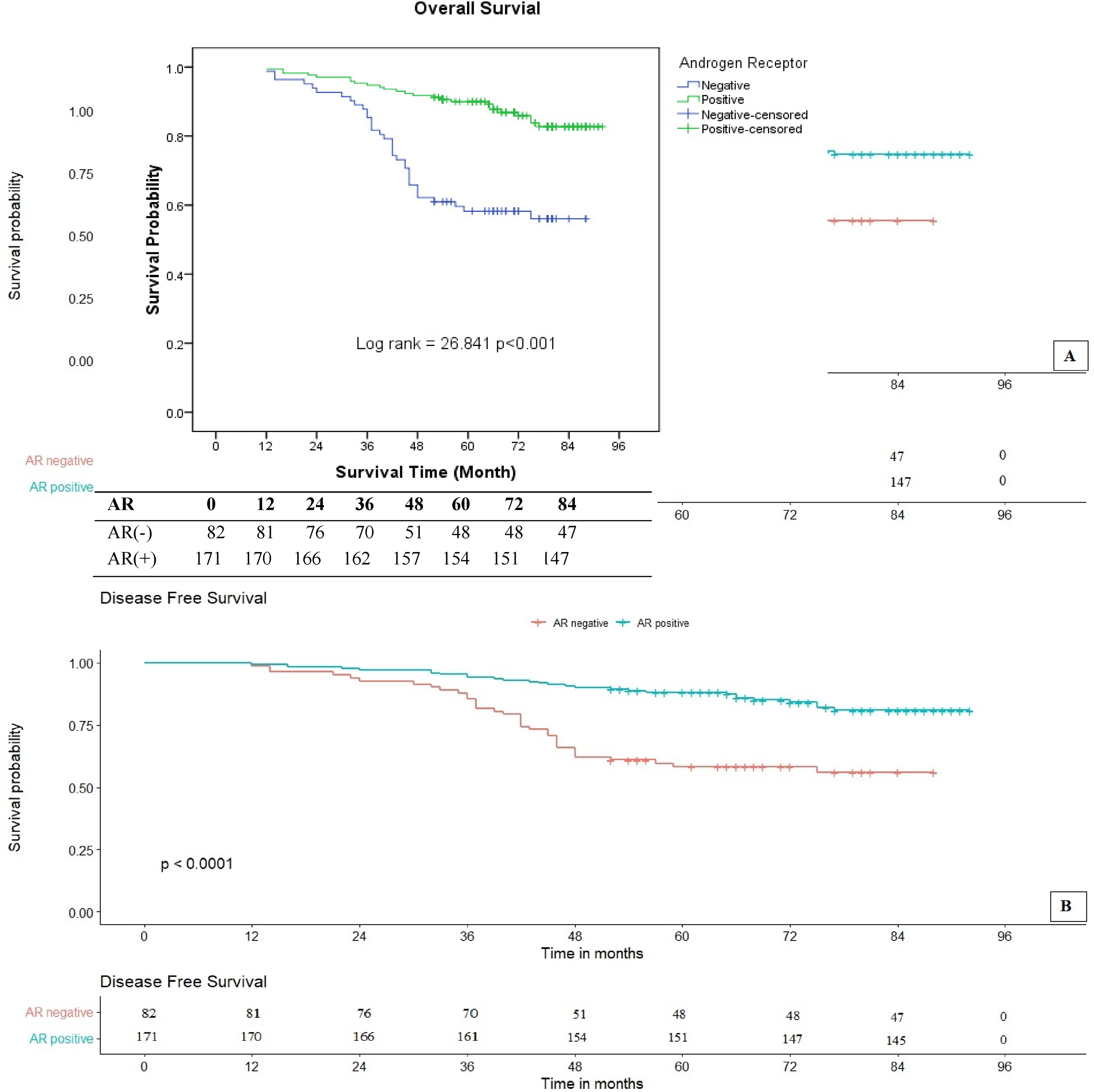
(A) Overall survival of AR expression for invasive BCs. (B) DFS of infiltrating BCs with AR status. The log-rank test indicates that there was a strongly significant difference between the 2 survival curves with AR expression.

Graph A shows the OS of LAR subgroups in TNBC. Meanwhile, chart B shows relative disease-free survival (DFS) of LAR subclasses in TNBC. The log-rank test exhibits that there was a closely significant difference between the 2 survival curves for LAR + and LAR − as well.

(A) Overall survival (OS) of AR positivity and negativity in positive HR invasive breast cancers (BCs). A significant difference between these 2 survival curves were demonstrated. (B) The log-rank test displayed that there was not a significant difference between these disease-free survival (DFS) curves of combination of AR expression and HR positivity for infiltrating BCs as well.
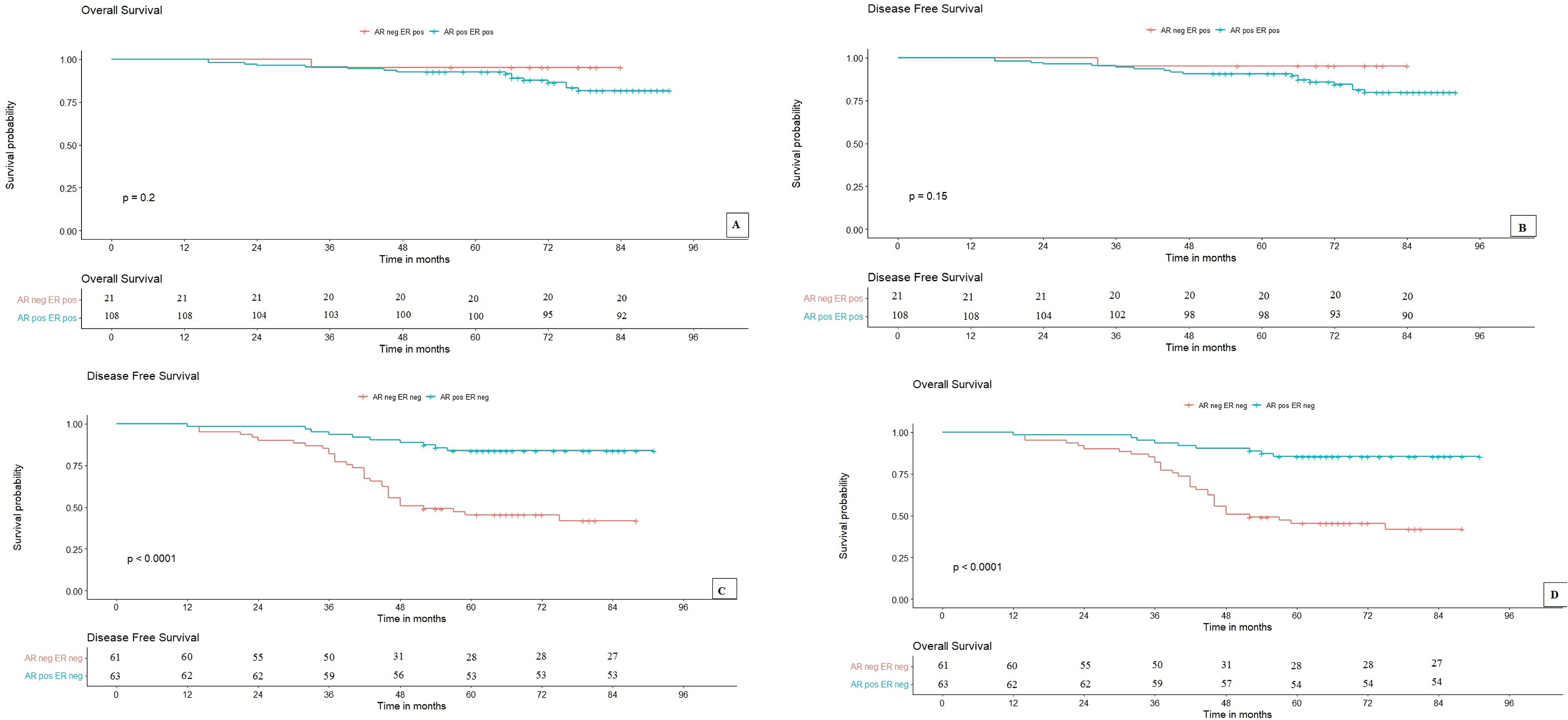
(A) and (B) Overall survival (OS) and DFS of AR expression for invasive BCs, respectively. The log-rank tests illustrated that there were not a significant difference between these OS and DFS curves of combination of AR expression and ER positivity for infiltrating breast cancers (BCs). On the contrary, DFS and OS of AR expression for invasive BCs without ER expression (C and D, respectively), the log-rank tests indicate that there were the strongly significant differences between these survival curves with AR expression in invasive BCs with ER absence.
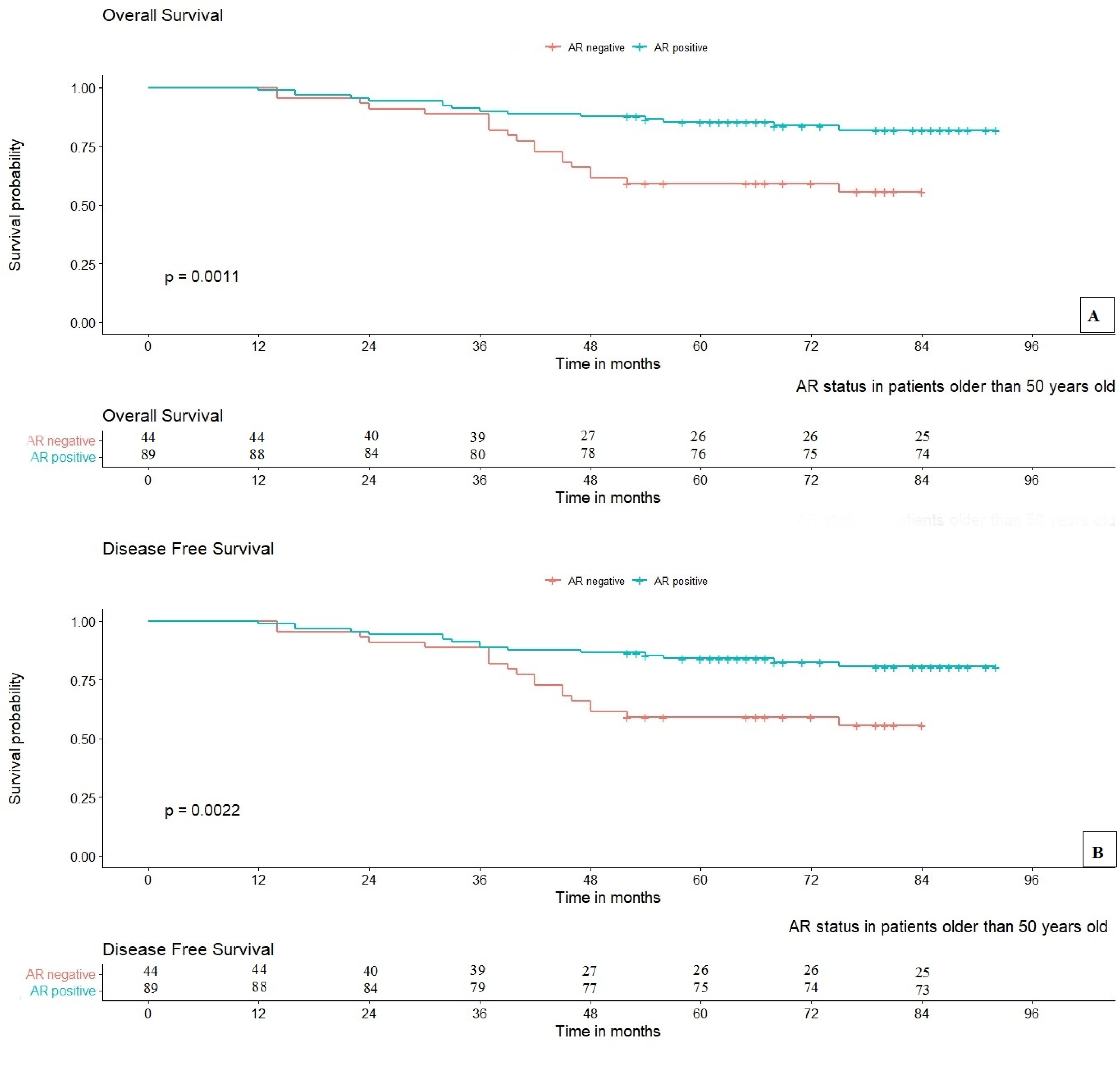
(A) OS of older 50 years old BC patients with AR expression. Chart B shows DFS of older 50 years old patients with the AR expression in invasive BCs. The log-rank test demonstrates that there was a powerful significant difference between the two survival curves for AR status in invasive BC patients older than 50 years old.
Totally, mean DFS of the invasive BC patients were 79.17 ± 1.46 months. Mean DFS for positive AR was higher than AR absence, with 83.94 ± 1.47 and 66.96 ± 2.82 months, respectively. With respect to AR:ER, the mean DFS of AR:ER<2 was longer than the AR:ER≥2 BC (86.47 ± 1.48 and 63.61 ± 8.20 months, respectively). For AR expression of TNBC, the mean DFS of LAR + group showed the longer period than LAR − (79.52 ± 3.63 and 60.05 ± 4.16 months, in turn). According to the combined AR and HER2 + immunophenotypes, AR + HER2 + was 82.11 ± 2.90 months, and by contrast, the AR-HER2 + category displayed the lower DFS (62.09 ± 5.40 months), similarly to whose OS. Meanwhile, BC patients with AR-HR+ showed the longer DFS, similarly to whose OS, than AR+HR+, with 85.89 ± 2.07 and 84.57 ± 1.67 months.
Evaluating DFS curves to different immunophenotypes are shown in Figures 2B, 3B, 4B, 5B, 6B, 7B, 7C, and 8B. A significant difference was observed in the DFS rate of AR expression, AR:ER ratio, LAR subtypes, AR expression of more 50-year-old patients, and the combined AR expression and HER2 + (P < .05), or ER − ve (P < .001), meanwhile correlation of AR positivity/negativity and HR + groups, and the combined AR expression and ER +ve group with DFS were not significant (P > .05). The DFS rate of the BC patients with the AR positivity were higher than AR−ve (80.9% vs 56.0% similarly to whose OS) (Figure 2B) (P < .001). Figure 3B showed that the DFS proportion of AR:ER<2 was counted on 86.2%, higher than AR:ER≥2 category (25.0%, the same to whose OS) (P < .001). The DFS curves of AR expression in TNBC were shown in Figure 4B, and DFS of women with LAR + BC were longer than LAR − (82.5%, similarly to OS ones vs 43.2%) (P = .001). Interestingly, BC patients with AR+HER2+ also revealed the longer DFS than AR-HER2+ (79.7% vs 41.1% similarly to whose OS) in Figure 5B (P = .003). Nevertheless, the DFS curves according to the combined AR presence/lost and positive HR categories were shown in Figure 6B, patients who were in both of these categories were demonstrated to archive the equivalent survival time to whose OS, although the difference was not observed the statistically significances (P = .102). Similar to OS, in ER + ve BC, DFS of patients without AR expression was higher than AR + (95.2% vs 79.8%, respectively) and no statistically significant difference was observed (P = .150). In contrast, for ER − ve BC, the rate of DFS in patients with AR + was also twice higher than AR − ve group (83.9% vs 41.9%, respectively) (P< 0.001) (Figure 7B and C, respectively). Figure 8B also demonstrated that the DFS of breast cancer patients with AR+ were significantly higher than BC with AR−ve in the more 50 year-old group (P = .0022).
Immunophenotypes of AR Expression Have an Independent Prognostic Significance for Favorable Clinical Outcome
To identify the significantly independent prognosis of clinicopathological variables and expression of IHC markers according to OS, DFS, multivariate models were performed. The most characteristics belonging to Cox univariate model were used in the multivariate analysis, including AR status, AR:ER ratio, age group, LAR subtypes, TILs, NPI, histological grade, Ki67 index, axillary lymph node state, risk categories, and pTNM (Table 2). Notwithstanding, Table 3 showed that AR:ER ratio was only the correlation with OS, and DFS of patients with the invasive BC. The remaining variables had no significant independent impact on OS and DFS, which were not demonstrated as an independent prognostic indicator. AR:ER ratio was the unique factor and is considered to be a parameter of the independent prognosis of both the OS (HR = 0.153, P = 0.005) and DFS (HR = 0.192, P = .010) (P < .05) in the positive ER population.
Estimated Hazard Ratios (HRs) for OS and DFS Univariate Analysis.

Abbreviations: AR: androgen receptor; ER: estrogen receptor; DFS: disease-free survival; LAR: luminal androgen receptor; NPI: Nottingham Prognostic Index; OS: overall survival; pTNM: TILs: tumor-infiltrating lymphocytes.
Estimated Hazard Ratios (HRs) for OS and DFS Multivariate Analysis.
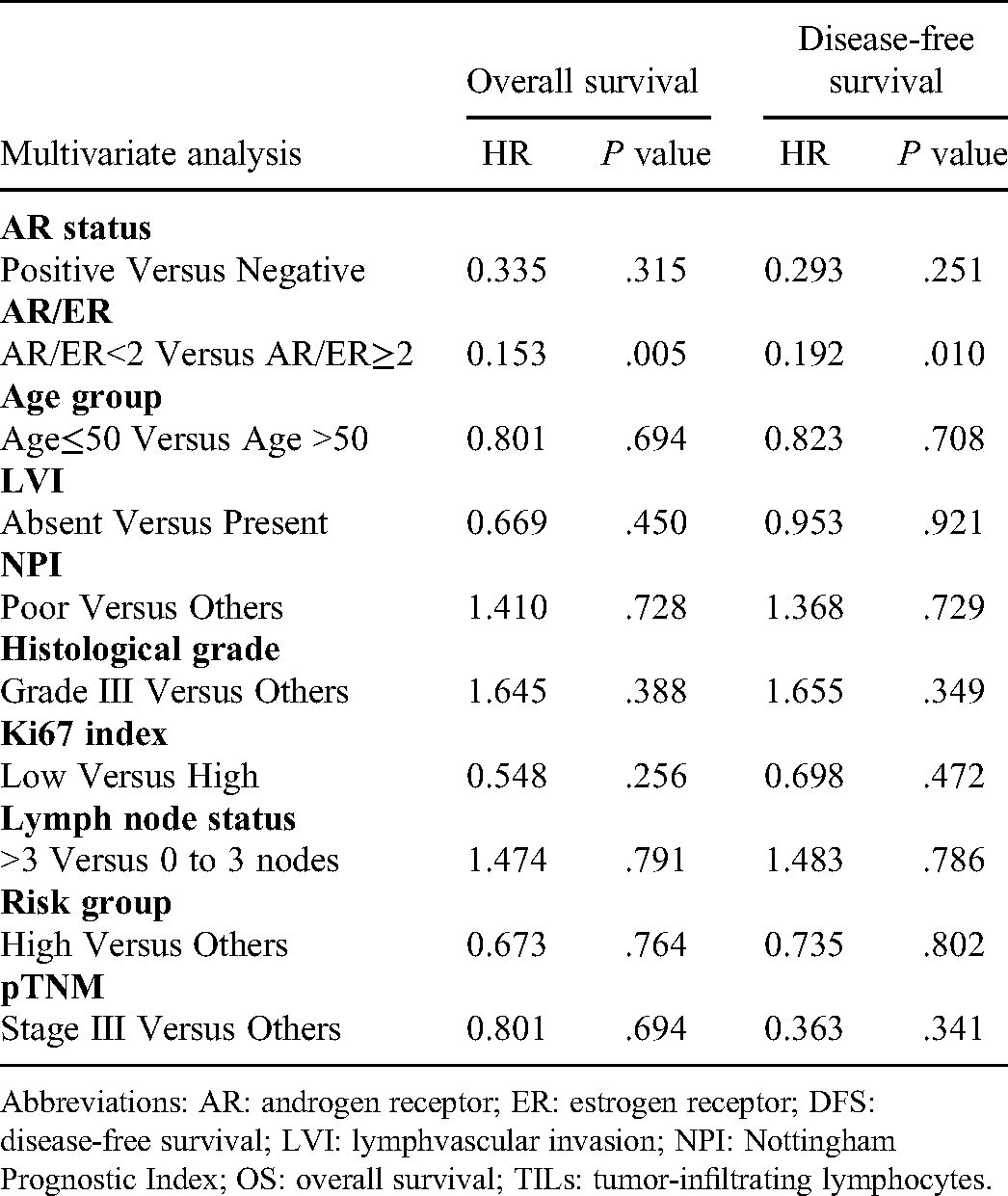
Abbreviations: AR: androgen receptor; ER: estrogen receptor; DFS: disease-free survival; LVI: lymphvascular invasion; NPI: Nottingham Prognostic Index; OS: overall survival; TILs: tumor-infiltrating lymphocytes.
Discussion
The AR has recently emerged as a useful marker for the more favorable prognosis and better outcomes among women with ER + ve BC and the further refinement of BC subtype.1,2,11 Additionally, determination of AR status may provide novel opportunities for promising targeted therapy in BC.7,11,25,26 Like any IHC markers, determination of the positive AR threshold is very important. The cut-off values of AR were investigated. Among of the studies using IHC, the relationship between AR expression and OS showed no significant differences according to the cut-off values of 1%, 5%, and 10% (P = .435). Thus, these different cut-off values of AR expression had no effect on the survival outcomes. 14 A prognostic index incorporating scores for AR positivity (>1%), small tumor size, and lack of lymph node involvement has been used to predict good prognoses in ER + ve BCs. 27 The 2011CAP/ASCO guidelines defined the value of AR positivity as considering at least one percent nuclear staining of any intensity. The present study applied this guideline.
In Vietnam, first, we have evaluated the AR expression to provide additional useful information for the treatment and prognosis of BC. The results of the present work demonstrated that the presence of AR was strongly correlated with better clinicopathological parameters, such as older patients, low histological grade, less axillary lymph node metastasis, and LVI, low NPI, BCs with HR positivity, aberrant Her2/neu, low proliferative activity, early stage BC, and common in LUMA subgroup and LR level compared to negative AR. According to the our 2 previous cohorts, LUMA and LR category belong to 2 BC groups with the best prognosis.19,28 Some previous studies also illustrated that AR expression has been reported to be the highest in luminal-type BCs, with LUMA expressing AR more frequent than LUMB.4,29,30 According to the research of Anand et al., their results demonstrated AR expression is associated with lower axillary nodal burden and early AJCC stage. 9 Interestingly, the study found an inverse correlation between the AR expression and tumor size, lymph node status, and histological grade. AR − ve BCs were significantly associated with larger tumor size, higher NPI, and more frequent tumor cell necrosis. 7 Generally, the majority of the AR + ve tumors were related to good prognostic factors, including lower histological grade, smaller tumor size, and negative nodal metastasis.4,31,32 The results are constant with our findings. An overwhelming number of publications demonstrate that the AR is a favorable prognostic marker, regardless of the histopathological subtype, and suggest that in most, AR expression BCs are inversely proportional to tumor size, aggressiveness, histological grade, and directly proportional to DFS, progression-free survival (PFS), and OS, suggesting that AR activation is favorable in BC.6,30,33 In the overall populations of this study, the OS and DFS rates in BCs with negative AR were the same and significantly lower than those in BCs with AR + ve (P < .001). In some cohorts, in cases of AR expression, OS and DFS were significantly longer compared with BCs without AR expression.8,14,26 Following the other investigators, AR + ve BCs (all types) had better OS and DFS, and AR positivity was also associated with lower risk of recurrence. 34 Similar to several other studies, AR expression correlated with better DFS and OS in both luminal subtype and TNBC. 13 Constantly, AR expression in both LUMA and/or LUMB is associated with a reduced risk of relapse and longer survival.27,29 In Hu et al.’s study, AR was significantly demonstrated into DFS (not OS) and its prognosis, in multivariate analyses. 11 Similarly, Castellano et al. showed that AR expression is an independent prognostic factor of a better outcome in patients with ER + ve BCs, in particular for the subset of LUMB. 27 In multivariate model of the present study has demonstrated that expression of AR is not an independent prognostic indicator of OS as well as DFS in the infiltrating BCs. The current finding is not constant to Hu’s study, Castellano’s study, and Kensler’s cohort also suggests that AR may have the value as a prognostic biomarker in ER−ve BCs, it is the independence of other patient, tumor, and treatment characteristics. 26 However, it still exists the controversial claims about the value of AR on breast cancer with luminal types. In some studies, AR was identified as an independent prognostic biomarker in BCs with positive HR, by contrast, AR was shown to be independent from the hormonal receptor expression, in other studies.8,18
Considering the significance of AR status in the invasive BCs, the authors recommended that the AR is considered as one of the three prognostic markers to classify into triple-positive BCs (ER, HER2, and AR + ve) or triple-negative BCs (ER, HER2, and AR − ve). Since PR is an ER target gene, PR is most likely to align with the pattern of ER expression and hence was logical to exclude it from the list of prognostic markers. 5 These results were reproduced in other studies, which were conducted in different patient cohorts around the world.31,34–37 The present results showed a higher positive rate of ER than PR (55.6 vs 52.9%, respectively). Furthermore, only 4.7% of BCs expressed with single PR (ER absence) in this study, and the ER proportion of nuclear staining was often higher than that of PR. Owing to the above reasons, maybe the further refinement of BC subtypes based on 3 ER, HER2, and AR markers can be a very useful suggestion in the prognosis and treatment of BC. However, further investigation should be performed.
In respect to relationship between positive ER and/or PR and AR in BCs. AR is expressed in 70% to 95% of ER + ve BCs.4,7,8,37–39 Nevertheless, only 31.8% to 49.0% of the ER − ve BCs expressed the AR.8,37 Patients with concordant AR and ER expression had the better prognoses than those of discordant AR and ER (P = .02). 40 Estrogen status seems to play a major role in determining the prognostic significance of AR since the same studies are not supportive for any association between AR positivity and better outcomes in ER − ve tumors.11,41 In the majority of studies, AR expression in ER + ve BCs have been associated with favorable characteristics such as older age at diagnosis, lower tumor grade, lower Ki67 positivity, smaller tumor size, and less necrosis.7,11,27,41–44 The results of this work showed that AR + ve is often associated with HR + tumors such as LUMA and LUMBH-, LUMBH + , as well as BCs with present ER and/or PR. In contrast, the loss of AR is more common in basal-like or HER2 subtypes, and it also displays the high prevalence in absent ER and/or PR and HER2 + BCs (P < .001). Evaluating the patients with HR + ve tumors, in a study, those with AR expression had a significantly prolonged DFS (P = .040). However, the OS data showed no significant differences (P = .140). 14 The present work has demonstrated that in patients with HR + ve BC, the OS and DFS prevalence of AR positivity is lower than AR − ve. However, the difference has not been shown to be statistically significant. Although the results of our study are not the same as those of Kim's study, the concordance of 2 cohorts found that there is no association between survival, especially OS, and AR status in HR + ve BCs.
Additionally, regarding the AR:ER ratio, the previous cohorts showed that its value could be used as a highly specific and sensitive prognostic tool for local relapse or progression to invasive subtypes.18,45 Its cut-off value of 2.0 was significantly different between the 2 groups (failed vs nonfailed tamoxifen), which seems to be able to affect the prognosis and to predict the response to anti-estrogen therapy in BC.18,45 The AR:ER ratio was different among the subgroups, indicating the unfavorable prognostic role of AR and AR:ER in this subset of patients. 9 The current findings indicated that as opposed to the low AR:ER ratio (<2), the higher AR:ER ratio (≥2) was strong and significant correlated with the worse clinicopathological features of BC patients, such as more three metastasized lymph nodes, high risk category, high nuclear proliferative activity, less common in LUMA, and positive HER2 (<2). Similarly, in Cochrane's cohort, women with the higher AR:ER ratio are more likely to have positive lymph nodes. 18 According to the previous research, the AR:ER ratio was the most significant marker of poor survival (tamoxifen failure, P <.0001; DFS, P < .0001; and DSS, P < .0001). 18 Overall, AR expression in ER + ve BCs is associated with good prognostic indicators including improved OS and DFS, lower grade, smaller tumor size, and lack of lymph node involvement at diagnosis.27,42,43 Notably, the present work illustrated the OS and DFS of patients with the low AR:ER reached significantly to the longer survival interval compared to the high AR:ER (nearly 3.5-fold) (P < .001). In some of these studies, multivariate analyses have demonstrated that AR is an independent predictor in ER + BC, positively associated with favorable outcomes (time to relapse, DFS as well as OS).11,27,41–43 The AR:ER ratio continued to be an independent predictor of failure for DFS and DSS. 18 This confirmation is constant with our findings that the AR:ER ratio is an independent indicator for predicting the OS and DFS of BC patients in both univariate and multivariate analyses.
Endocrine therapies that target estrogen and ER signaling pathways play a critical role in the treatment of the majority of positive HR BC patients. Unfortunately, up to 30% of patients show resistance to tamoxifen either de novo or acquired resistance. 15 It may be the most relevant to understand the relationship between ER and AR signaling pathways when discussing its role in anti-estrogen therapy. In patients with previously treated ER + ve BCs, increased androgen production is considered to be a potential indicator for resistance to anti-estrogen therapy and a possible pathway for the growth and survival of their tumors. 46 The AR:ER ratio determines the impact of both AR and ER in the regulation of BC cell proliferation.1,42,47 When the expression level of AR is higher than that of ER, the AR binds to the estrogen response elements (EREs), thereby exerting inhibitory effects on cell growth. In contrast, if the ER is more abundant than the AR, then ER binds to androgen response elements (AREs), leading to cell proliferation.48–51 In Cochrane et al. when the authors added ER + ve percentage to the model, the risk went down to 2.87-fold, which was demonstrated that although ER + ve percentage explained some of the increased risks from a higher ratio, actually the AR:ER ratio has an independent effect on risk for failure above ER + ve percentage. 18 These provocative results suggest that the ratio of nuclear AR to ER may be a new, independent predictor of response to traditional E2/ER-directed endocrine therapies. In our study, the high AR:ER group, which shows the worse prognostic features, the mean percentage of AR expression reached to a level of 2.7-fold higher than the average percent ER positivity. In contrast, in the low AR:ER group (the group with good prognostic characteristics), the moderate percentage of positive ER was nearly 3 times higher than the percentage of AR. Peters et al. showed that AR levels are significantly associated with disease outcome in ER-positive BC, a 3.0-fold increased risk of relapse and a 4.6-fold increased risk of cancer-related death for patients with lower than median percentage of AR positivity, and that AR is a direct repressor of ER signaling in BC cells. 42 These findings suggest that, at least in ER + ve BC, AR is an independent predictor of the outcome. Simultaneously, the findings help to explain, in BCs with low ER + ve, a high percentage of AR will result in the stronger antagonist competition between ER and AR, leading to resistance to endocrine therapy, thus patients in this group have a worse prognosis. To further delineate this relationship, Peter et al. looked at AR:ER ratio. The results showed that a high ratio of AR:ER, rather than absolute AR level, may be more predictive of tamoxifen failure. 42 This is suitable with the current findings. These above results suggest that in ER + ve patients, it is necessary to further IHC stain with AR is able to determine the expression level of both ER and AR, in particular, it is necessary to determine the ratio of AR:ER. This information is useful for the prognosis and prediction of response to hormone therapy in the invasive BCs. Since clinical trials have shown promising results, research should focus on the identification of novel biomarkers to stratify patient subgroups that will benefit the most from AR-targeted therapies. 25
In TNBCs, researchers are increasingly recognizing that TNBC is a heterogeneous disease that encompasses distinct intrinsic molecular subtypes. Most ER − ve tumors (HER2 and TNBC) are considered as aggressive, but it appears that AR reactivity can be used to prognosticate these tumors. 7 The present findings showed that in patients with ER − ve BC, it is clear that AR expression improved OS as well as DFS. Studies that have attempted to divide TNBC into 6 or 4 molecular subtypes, each characterized by differential response to therapy and potentially new therapeutic targets, among of them which are AR + ve BCs named the luminal androgen receptor (LAR: 12%).1,52 Generally, LAR + tends to be less proliferation and poor response to chemotherapy in both the neoadjuvant and adjuvant setting, but also associated with higher OS.3,18,53,54 TNBCs generally have a low and various frequencies of AR expression compared with other BC subtypes, with as few as 10% to 53% of TNBC patients expressing AR (LAR + ).4,7,10,12,13,29,31,41,43,55–57 LAR + was correlated with the menstrual status, histological grade and lymph node metastasis. 31 In detail, a meta-analysis by Wang et al. LAR + was significantly associated with postmenopausal status, low tumor grade, and with a HR of nodal involvement. 12 Similarly, in some studies, LAR + is a favorable prognostic factor, and associated with the good tumor characteristics such as lower grade, lower mitotic score, a lower clinical stage.10,11,13,31,32,58 As opposed to LAR + , in LAR − , both Rakha and Sutton and colleagues have shown the association with an increased risk for recurrence and distant metastasis in lymph node-positive BCs.59,60 Simultaneously, a retrospective study illustrated that LAR − has a higher proportion of positive lymph node and poor prognosis, whereas LAR + is significantly associated with a prolonged DFS and OS. 55 The current findings showed a stronger correlation between LAR + with no lymph node metastasis and a moderate-risk level which were higher than ones of LAR-. Similarly, regarding the survival interval of LAR + and LAR − , this study has demonstrated that OS, DFS of LAR + are almost twice longer than ones of LAR − in invasive BCs. These results are the same as He's cohort and the previous researches.13,14,31,55 Multivariate analysis showed that tumor size, lymph node metastasis, and AR expression were correlated with 5-year OS of TNBC patients. 31 Although this work exhibited a significant association of AR expression in TNBC with DFS and OS. Due to the limited number of patients, LAR+ and LAR- groups were not sufficient to perform the multivariate analysis for identification of the independent prognostic value’s this immunophenotype.
However, this favorable prognostic significance of AR is not uniform across the literature in all BC types. The discordant findings among certain studies still exist. 59 Hu and colleagues have shown AR expression to be associated with 83% of the increased overall mortality among of women with ER − ve and TNBCs compared with those with AR − ve tumors. 11 Similarly, Park and co-investigators have shown a trend toward poorer outcomes in AR + ve and ER − ve BCs. 10 Additionally, in other studies, AR does not predict survival in TNBCs.11,43,56 Both Qu and Wang's studies found no association between AR status and OS.12,34
The different ethnic composition of cohorts may be affected by AR prognostic value. Population-specific differences between cohorts were more apparent upon evaluating the prognostic role of AR. Bhattarai et al. found that LAR + displayed better OS than LAR − in the United States and Nigerian cohorts among all patients, as well as among patients with adjuvant chemotherapy; by contrast, BC with LAR + showed poorer OS than LAR − in the Ireland, Norway, and India cohorts. 59 AR + ve correlated with 5-year survival in TNBC, but not in non-TNBC. 31 This result may be constant with the relationship between geographical factors and TNBCs.
TNBC is commonly considered more sensitive to chemotherapy compared to others, however, the molecular tumor features associated with LAR + may result in an inferior responsive phenotype to chemotherapy in the neoadjuvant setting,3,13,54 and AR positivity correlated with a lower pathological complete response (pCR) rate.3,13,61 Masuda et al. found that patients with the LAR + had one of the lowest pCR rate, but OS was better, constantly to the other cohorts.3,13,53,54 These findings reflect the impact of AR in BC, like HR + ve BC, often responds poorly to neoadjuvant systematic therapy—not achieving pCR; notwithstanding, LAR + was shown to be associated with better clinicopathological features and longer survival than both OS and DFS or PFS in most of the above studies, suggesting that AR could be a good marker for the prognosis, prediction of response to treatment, as well as new promising targeted therapy regimens in TNBC, one with few treatment options, in the future. From previous to now, studies have explored the valuable prognostic role of AR in TNBC to better understand androgen action in TNBC, identify actionable factors that drive outcomes, and determine if testing for AR status should become part of routine clinical practice for TNBCs. Laboratory studies have shown inhibitory roles of androgen analogues on ER − ve/AR + ve cell lines. Similar approaches can be used for ER − ve/AR + ve BC as an adjunctive therapy. 7
In respect to AR expression in enriched HER2 BCs, in some previous, the AR expression was accounted in approximately 30% to 60% of HER2 + BCs.4,56 Evenly, AR is enriched in ER − /HER2 + BCs with 77%, compared to 30% in HER2 − , 56 constantly to the other cohorts.7,62 The current results show that BC with AR + HER + are closely associated with good clinical features such as common in BCs with low histology (I, II), little or no lymph node metastasis, good NPI, absent LVI, early stage BCs, LR category compared to AR-HER2 + immunophenotypes. In some series, survival appears to be unaffected by AR status, whereas the others suggest more favorable morphology when HER2 and AR are coexpressed.43,56,63 Wang et al. found that cases demonstrating AR positivity had improved PFS and OS. Their cohort revealed a significantly increased 5-year OS rate in patients with AR + HER2 + , compared to AR-HER2 + (65.3 vs 36.2%, P < .001), and AR + ve status was significantly and independently associated with improved survival (PFS, OS), in multivariable analysis. 64 Similarly, the results of this study show that OS in the BC with AR + HER2 + is twice higher than the AR-HER2 + group (P = .001). The DFS of the AR + HER2 + group was also nearly twice higher than that of the AR-HER2 + BCs (P= .003). HER2 + ve tumors are more likely to express AR than other ER − ve subtypes,7,41,56 suggesting an association between HER2 and AR in these cancers, although this is not observed in all studies.9,10 The results of our study are similar to Wang’s cohort, although the limited number of patients with HER2+ combined in AR expression were not sufficient to perform the multivariate analysis for identification of the independent prognostic value’s this immunophenotype.
AR expression is considered to predict the benefit from first-line trastuzumab treatment in patients with enriched HER2 BCs. AR positivity was significantly associated with a better response to first-line trastuzumab treatment. 64 Additionally, this has immense therapeutic value as inhibition of the AR or HER2 pathways, alone or in combination, may result in additional therapies for ERBB2-type tumors. 7 So AR status should evaluate on patients with HER2 + ve types, or HER2-enriched metastatic or recurrent BC to obtain comprehensive information and as a complement for predicting the efficacy of trastuzumab according to recommendation of Wang's cohort. 64
Up to now, studies have explored the valuable prognostic role of AR in the infiltrating BCs, especially, enriched HER2 and TNBC to better understand androgen action in these 2 BC groups, identify actionable factors that drive outcomes, and determine if testing for AR status should become part of routine clinical practice for TNBCs, and HER2 positivity. However, this favorable prognostic significance of AR is not uniform across the literature. The discordant findings among certain studies may still exist. To some extent, the prognostic discrepant results mentioned above can be attributed by multiple factors, including the use of tissue microarrays versus whole-tissue sections; use of different AR protein detection techniques (radioligand binding, IHC) or reverse phase protein array; source and sensitivity of the anti-AR antibodies used for IHC staining, the methodology of detection, the cut-off values used to define AR positivity, and use of different thresholds to define AR positivity; measurement of AR mRNA via different platforms in transcriptome profiling studies; differences in clinical parameters such as various sample sizes, differences in the ethnic composition of cohorts, prior treatment regimens or adjuvant therapy, length of follow-up and histopathological type.51,53,59,61 Evenly, in TNBCs, this discrepancy among studies could be due to the overlap between AR and molecular apocrine signatures. Molecular apocrine tumors are a distinct subset of TNBCs characterized by AR expression and AR pathway activation. 61
Taken together, interestingly, the authors observed the AR expression may be an appropriate surrogate marker of prognosis and prediction in BC. Therefore, testing AR status at initial diagnosis could lead to a better selection of patients who are likely to benefit from a more aggressive neoadjuvant treatment. Routine AR evaluation by IHC staining in overall BCs, especially to TNBC, enriched HER2 BC could provide further insight in this direction. It is also essential to determine the AR:ER ratio in the positive ER BCs to provide the important prognostic information and prediction of response to endocrine therapy. The findings show that further investigation to elucidate the AR implications of the promising therapy in TNBC, enriched HER2 BCs, or patients with a resistance to anti-estrogen therapy is required.
Limitations of the Study
At present, some limitations still remained in the current study. Not all patients were followed up, especially in the group of patients with TNBC and HER2 + BCs. There are some reasons to explain this including the fact that the patient database was not being systematically managed on the computer, and moreover, Vietnamese patients often tend to change their phone numbers. Therefore, it is a challenge to keep in contact with them when they completed their treatment. Continued follow-up and analysis of all patients are planned to confirm the more prognostic value of all combined immunophenotypes in BCs.
Conclusions
The AR expression was adversely correlated to clinicopathological characteristics, and revealed a distinct OS and DFS. The current finding demonstrated that AR:ER ratio is an independent prognostic indicator for prolonged DFS and OS in patients with positive ER BC. We recommend that AR status should be routinely tested in overall BCs, especially to the patients with HER2-enriched, or TNBC for providing valuable information for treatment, prognosis, and prediction of Vietnamese patients with BCs.
Footnotes
Author Contributions
H.T.P, C.V.N, and K.H.P equally major contributed to this work.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
