Abstract
Background
Colorectal cancer is associated with the third highest incidence rate among global cancers and is the fourth most common cause of cancer death worldwide.1,2 According to demographic trends, CRC is expected to increase its incidence by almost 80% per year, with more severe consequences in developing countries. 1 In China, more than 500 000 new CRC cases were reported and approximately 247 000 deaths in 2018. 3
CRC is a heterogeneous disease. The occurrence and development of CRC are related to many factors, including inflammation and nutritional status.4–6 Recently, some indicators related to the inflammatory process and nutritional status of tumors have been frequently used as tools for diagnosis and prognosis.6–8 Studies have shown that elevated neutrophil counts are one of the factors that cause poor prognosis for various cancers, including kidney cancer, lung cancer, and colorectal cancer.9–11 A high neutrophil–lymphocyte ratio (NLR) may be associated with poor prognosis in gastric and colorectal cancer patients.12–14 Body mass index (BMI) is important to assess human nutrition and is positively correlated with the risk of CRC.6,15,16 Obesity is a risk factor and an indicator of poor prognosis in CRC, which may be related to long-term chronic inflammation caused by obesity.5,6,16,17 Alternatively, weight loss and low BMI may be associated with shorter survival in patients diagnosed with CRC. 18
Previous studies have shown that the combined use of inflammatory markers and nutritional indicators can predict the prognosis of patients with CRC, such as NLR and nutritional status, 7 and C-reactive protein to albumin ratio (CAR). 19 Neutrophils are a representative inflammatory marker, and BMI is an indicator of nutritional status that is easy to obtain. Indeed, there is a relationship between nutrition and immunity. Hence, we hypothesized that the neutrophil–BMI ratio (NBR) may reflect the systemic immune inflammatory status of patients with CRC, and that abnormalities in preoperative NBR may predict the prognosis of these patients. Therefore, the purpose of this study was to evaluate the prognostic value of the preoperative neutrophil–BMI ratio in patients with CRC.
Materials and Methods
Study Population
The study was approved by the Ethics and Human Subject Committee of Guangxi Medical University Cancer Hospital. Based on the inclusion and exclusion criteria, approximately 2471 patients with CRC who underwent surgery at the Department of Gastrointestinal Surgery at the Guangxi Medical University Cancer Hospital between 2004 and 2019 were retrospectively reviewed. Cases were selected based on the following inclusion criteria: 1) histopathological diagnosis of CRC in patients, 2) patients who underwent primary tumor resection, 3) patients’ blood count and BMI data obtained preoperatively, and 4) patients underwent tumor resection after 4 weeks of neoadjuvant therapy. Cases were excluded based on the following exclusion criteria: 1) patients with a history of familial adenomatous polyposis or hereditary non-polyposis colon cancer; 2) patients with fever or other infections during blood collection; 3) patients with other malignant tumors, and 4) patients underwent tumor resection within 4 weeks of neoadjuvant therapy. The corresponding demographic and preoperative clinicopathologic characteristics were collected, including age, sex, body mass index (BMI), location, differentiation, TNM stage, CEA, neutrophil, KRAS phenotype, and microsatellite status. The blood samples of each patient were collected within 2 week before surgery. Neutrophil was measured as part of routine blood tests. Height (meters) and weight (kg) were measured by a nurse prior to surgery. The body mass index was calculated as weight (kg) divided by height (m) squared. NBR was calculated as follows: neutrophil count (1 × 109) / BMI (kg/㎡). Pathologic diagnosis was based on the eighth edition of the American Joint Committee on Cancer (AJCC) guidelines, Cancer Staging Manual. 20
Follow-up and Statistical Analyses
According to the National Comprehensive Cancer Network (NCCN) guidelines, patients were followed up by telephone and outpatient consultation, mainly based on informed patients. Survival time was estimated from the date of surgery to the date of the most recent follow-up or death. The follow-up period ended in December 2019.
Considering the correlation between the NBR and overall survival (OS) of patients, the optimal cutoff value was obtained according to the maximally selected rank statistics. The maximally selected rank statistics was a simple cut-point model that divided the data into two groups by dividing the value of each independent variable while calculating a standardized statistic, and then taking the maximum value, the corresponding division value was the best cut-off value. 21 The centrality of the differences in clinicopathological characteristics was evaluated using the chi-square test (categorical data) (utilizing NBR as a dichotomous variable) and Student's t-test (continuous variable). The Kaplan–Meier method was used to estimate the OS curves of H- and L-NBR patients, and the statistical differences were compared using the log-rank test. Univariate, multivariate, and subgroup survival analyses were performed using Cox regression models. A Cox regression model was utilized to establish a nomogram for predicting OS. The calibration curve was used to estimate the goodness of fit between the predicted and actual probabilities of the OS at 3 and 5 years to correct the nomogram. The receiver operating characteristic (ROC) curve and the area under the curve (AUC) were used to assess the survival prediction effect of the nomogram. A p value <.05 (two-sided) was considered statistically significant.
All statistical analyses were performed using R version 3.6.2 (https://www.r-project.org/). The optimum cutoff value for the neutrophil-BMI ratio was obtained using the ‘survminer’ software package. In addition, both univariate and multivariate ‘survival’ analyses were performed using the Kaplan–Meier software package (Version:3.1-8).
Results
Patient Baseline Characteristics
A total of 2471 patients with CRC were included in this study. Patients with CRC were divided into two groups according to the cutoff value for NBR: NBR >0.25 (n = 522) and NBR ≤0.25 (n = 1949). The relationship and distribution of the clinicopathological variables are shown in Table 1 and Figure 1. These results suggest that compared with the low NBR group, factors associated with patients in the high NBR group included female gender, low BMI, colon, right sided, poorly differentiated, T3 to 4 stage, M1 to 2 stage, high CEA, III-IV stage, high neutrophil, MSI, and no adjuvant chemotherapy (P < .05) (Table 1). There was no statistically significant difference between the two groups in KRAS, pN stage, age, and neoadjuvant therapy (P > .05) (Table 1). Box plots show the distribution between the clinicopathological variables and NBR in the included patients (Figure 1).

Box plots show the relationship between NBR and clinical features.
The relationship between main clinicopathological variables with NBR.
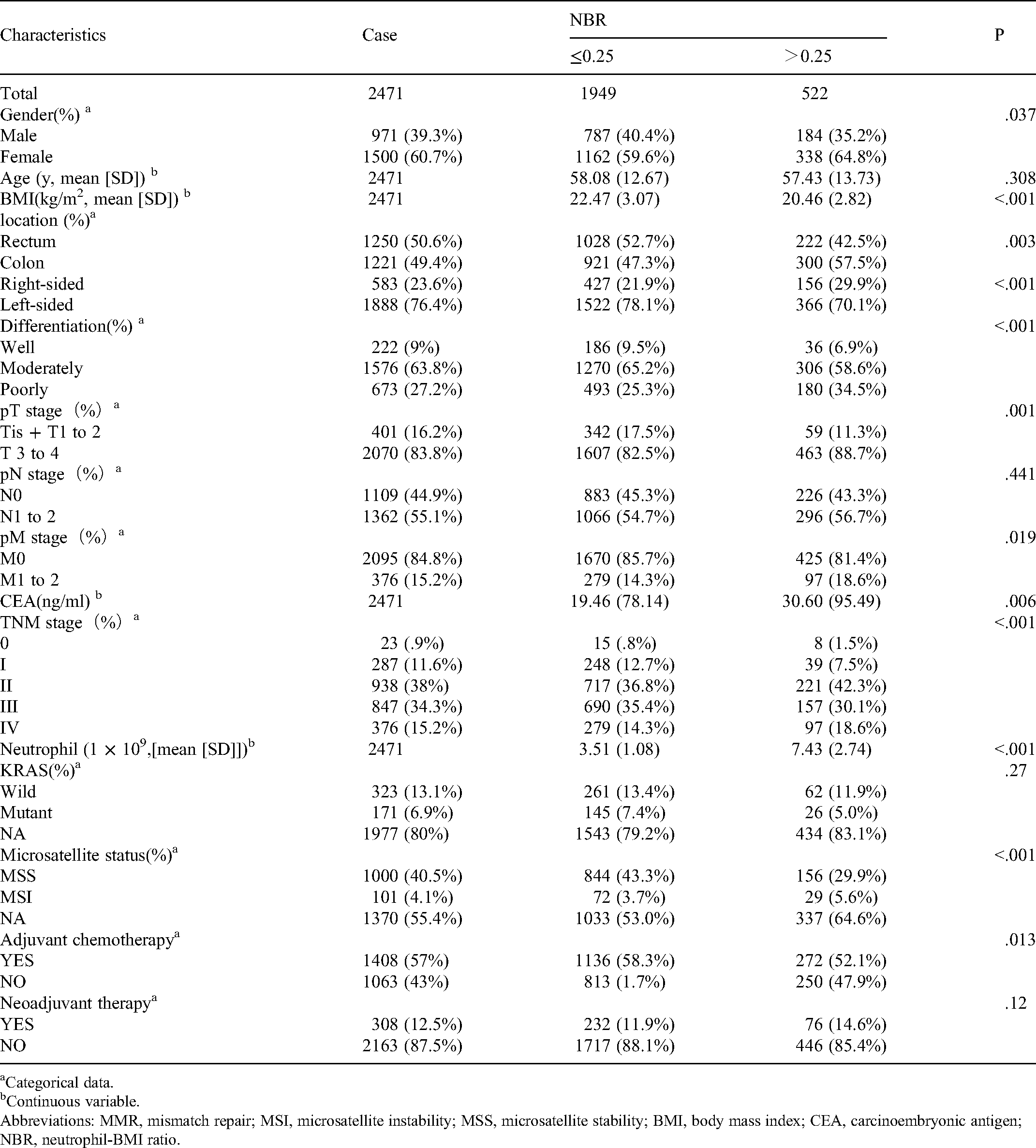
Categorical data.
Continuous variable.
Abbreviations: MMR, mismatch repair; MSI, microsatellite instability; MSS, microsatellite stability; BMI, body mass index; CEA, carcinoembryonic antigen; NBR, neutrophil-BMI ratio.
Kaplan–Meier Curves of OS in Each TNM Stage
Figure 2 shows the relationship between the OS and TNM stage based on NBR in Kaplan–Meier curves. High NBR was associated with a significantly shorter OS compared to low NBR in all TNM stages, including all stages (P < .001) (Figure 2A), I–II stage (P = .0046) (Figure 2B), and III–IV stage (P < .0001) (Figure 2C). The outcomes showed that NBR could distinguish the prognosis of patients with CRC in the TNM stage, and a high NBR indicated a reduced OS.

Kaplan–Meier curves of OS of patients classified by NBR. A: Kaplan–Meier curves of OS in all patients with CRC. B: Kaplan–Meier curves of OS in I–II stage patients with CRC. C: Kaplan–Meier curves of OS in III–IV stage patients with CRC. A value of NBR above 0.25 is equivalent to the high level group.
Kaplan–Meier Curve for NBR Combined With Other Clinical Features
OS in relation to the NBR and combined with the main clinicopathological variables are shown in Figure 3. Age, sex, location, KRAS phenotype, and microsatellite status were found to be significant predictors of OS. In the NBR and age groups, analysis was performed by dividing the subjects into two groups based on age: > 60 and ≤ 60 combined with NBR. Older patients had a worse prognosis in both high and low NBR (P < .0001) (Figure 3A). In the NBR and gender groups, analysis was performed by dividing the subjects into two groups based on gender, combined with NBR. A high NBR was associated with a poor prognosis in females (P < .0001) (Figure 3B). In the NBR and location groups, analysis was performed by dividing the subjects into two groups based on the location of the CRC: left side and right side

Impact of NBR combined with other clinical features on prognosis. A: Kaplan–Meier curves of OS in NBR combined with age. B: Kaplan–Meier curves of OS in NBR combined with gender. C: Kaplan–Meier curves of OS in NBR combined with location. D: Kaplan–Meier curves of OS in NBR combined with KRAS status. E: Kaplan–Meier curves of OS in NBR combined with microsatellite status. The value of NBR above 0.25 is equivalent to the high-level group.
Univariate, Multivariate, and Subgroup Analyses of the NBR in Patients With CRC
In the univariate analysis, age (P = .019), left and right CRC (P = .022), differentiation (P < .001), TNM stage (P < .001), CEA (P < .001), neutrophil (P < .001), BMI (P = .001), neutrophil–BMI ratio (P < .001), and neo-adjuvant therapy (P < .001) were associated with OS (Figure 4A). In the multivariate analysis, age (P = .019), differentiation (P = .001), TNM stage (P < .001), CEA (P = .014), and neutrophil–BMI ratio (P < .001) were independent poor prognostic factors (Figure 4B). In the subgroup univariate analysis, all subgroups were identified with statistical significance, except for poor differentiation (P = .236) and neo-adjuvant therapy (P = .205) (Figure 5).

Univariate and multivariate analysis of clinicopathological variables for OS in 2471 patients with CRC. A: Univariate analysis of clinicopathological variables. B: Multivariate analysis of clinicopathological variables.

Subgroup univariate analysis of NBR in patients with CRC.
Development of a Nomogram Based on NBR in Female Patients With CRC
Considering that in the subgroup univariate analysis, female patients with high NBR had worse prognosis, further study is needed in this field. These patients (n = 1500) were devided into a training set (n = 1050) and a validation set (n = 450). Three independent poor prognostic factors were identified by Cox regression model, including TNM stage (P < .001), CEA (P = .016), and neutrophil–BMI ratio (P = .006) (Supplementary Figure 1). A nomogram was constructed based on the results from the Cox regression model to evaluate OS in female patients with CRC combined with high NBR, as shown in Supplementary Figure 2. After the summation of the points against each factor, the OS of 3 and 5 years could be predicted. In the training set, a nomogram for predicting the prognosis of female patients with CRC, the AUC was .79, which was more associated with prognosis than the TNM stage (AUC = 0.76) (Supplementary Figure 3A). The relationship between the two AUC rates over time is shown in Supplementary Figure 3B. Compared to the TNM stage, the nomogram had the strongest OS prediction effect in 3 years. In addition, the nomogram also had a good ability to predict prognosis in the validation set (Supplementary Figure 3C and D). The AUC value of this nomogram was 0.769, which was more associated with prognosis than the TNM stage (AUC = 0.73). In the internal validation, calibration curves demonstrated good agreement between the predicted probability and the actual probability of the OS at 3 and 5 years in the training set and the validation set (Supplementary Figure 4A and 4B). In addition, the Cox regression model demonstrated that NBR could distinguish the prognosis of female patients with CRC in the training set and validation set, and the high-risk group was associated with a worse long-term prognosis (Supplementary Figure 5A and 5B).
Discussion
A growing number of studies have demonstrated that systemic inflammatory factors and nutritional status may be used as indicators to assess the prognosis of human malignant tumors.4,5,22 However, it remains unclear how NBR can be used to predict the prognosis of patients with CRC. In this study, we analyzed the relationship between NBR and clinicopathological variables on prognosis, and obtained significant results. First, according to the correlation between the NBR and the OS of patients, the optimal cutoff value for NBR was 0.25. Second, we demonstrated that high NBR was associated with clinicopathological variables, including female sex, low BMI, colon, right sided, poor differentiation, T3 to 4 stage, M1 to 2 stage, high CEA, III-IV stage, high neutrophil, MSI, and adjuvant chemotherapy. Third, based on the TNM stage and subgroup analysis of clinical features, the Kaplan–Meier curve showed that patients with colorectal cancer in the high NBR group had a worse prognosis than patients in the low NBR group. Fourth, in the multivariate analyses of OS, the results indicated that NBR was a novel independent predictor of poor prognosis in patients with CRC. Fifth, we successfully established a nomogram based on NBR to predict the prognosis of female patients with CRC.
In the tumor microenvironment, neutrophils have a tumor-promoting impact and play a significant role in all stages of tumor progression. A large number of in vivo and in vitro functional studies have indicated that tumor cell migration, invasion, and metastasis were highly associated with tumors stimulating neutrophil secretion of cytokines and chemokines to mediate inflammatory cell recruitment, tumor cell angiogenesis, and immunosuppression. 23 An increase in the number of neutrophils was observed in the peripheral blood and tumor tissues of patients with different tumors, and high levels of neutrophils were associated with poor prognosis, which has also been confirmed in CRC.23,24 In contrast, the relationship between BMI and prognosis was U-shaped, and underweight (too low BMI) and overweight (too high BMI) were associated with low overall survival and high distant metastasis in patients with CRC. 25 However, other studies have shown a J-shaped relationship between BMI and CRC prognosis, and people with high BMI may have higher survival rates than those with low BMI.26,27 Therefore, low BMI is a significant marker of poor prognosis; however, further research is needed to elucidate the relationship between high BMI and prognosis. These findings confirmed that cancer survival depends not only on tumors, but also on host-related factors, especially inflammation and nutrition.23,24,28 Interestingly, a study on the surgical treatment of patients with low weight before surgery (BMI <20 kg / m2) showed that BMI was significantly negatively correlated with preoperative NLR. 29 Considering the results obtained in our study, our NBR measurements could reflect both inflammation and nutrition, with high NBR representing an enhanced systemic inflammatory response and/or malnutrition in patients with cancer.
The TNM stage is widely acknowledged as the best index for predicting the prognosis of colorectal cancer. However, the prognosis of different patients with the same TNM stage is often different, suggesting that different factors must be evaluated to predict prognosis. 30 Therefore, we compared the difference in the survival curve of NBR among different TNM stages. Patients with high NBR have a worse prognosis in TNM stage I–II, TNM stage III–IV, and all TNM stages. These results indicate that the NBR can be utilized as a supplement to the TNM stage. In addition, according to subgroup analysis of clinical features, the Kaplan–Meier curve also showed that high NBR has a worse prognosis for patients presenting some clinical factors, including age >60 years, female sex, right side, wild-type KRAS, and MSS. In recent years, the influence of molecular subtypes of CRC on prognosis has attracted much attention. The microsatellite status and KRAS phenotype are two such molecular subtypes. 31 Microsatellite instability (MSI) is caused by the deletion or insertion of small fragments in short tandem repeats in tumor DNA, and genetic changes in mismatch repair (MMR) genes (MLH1, PSM2, MSH2, and MSH6) or EpCAM play an important role.4,32 Patients with CRC can be divided into prognostic subgroups by measuring their MSI status and predicting the prognosis for the adjuvant 5-fluorouracil (5FU) and immunotherapy.33,34 The current investigation demonstrates that MSI has a better prognosis than MSS.4,33,34 KRAS is one factor of the GTPase RAS superfamily and plays a key role in the MAPK signaling pathway; 35 indeed, when it is abnormally activated, it can result in tumors formation and progression. 35 In the treatment of metastatic CRC with cetuximab, the left-sided metastatic CRC of the wildtype KRAS has a longer OS and better prognosis than right-sided colorectal cancer,4,36 which may be related to the differences in embryology, biology, and anatomy between the left and right sides.4,37 KRAS mutation can negatively predict the response to these inhibitors, such as cetuximab and panitumumab, 38 and anti-VEGFR (vascular endothelial growth factor) therapy has been proven to be superior to other therapies. 39 Interestingly, our study shows that the OS of high NBR is worse than low NBR in KRAS state and the OS of wild-type KRAS is worse than that of mutant KRAS in the high NBR. High NBR with KRAS state have worse OS may be related to high-level inflammation promoting KRAS-driven tumorigenesis. 40 In addtion, the results show that wild-type KRAS has a worse prognosis in the high NBR, however, previous studies have shown that the prognosis of mutant KRAS CRC patients is worse than that of wild-type KRAS CRC patients in the microenvironment of high levels of inflammation, 40 which does not exclude a bias related to the inclusion of too few CRC patients with mutant KRAS. Combining existing microsatellite status and KRAS status studies with the results of this study, NBR can stratify microsatellite status and KRAS status to provide patients with a more adequate treatment.
Regarding demographics, male and elderly individuals have consistently been demonstrated to be highly associated with the incidence of CRC.4,41 Interestingly, our study showed that female patients with CRC had a worse prognosis in Kaplan–Meier analysis and subgroup univariate analysis than male patients with CRC. Therefore, we constructed a predictive nomogram based on NBR to predict the prognosis of female patients with CRC, and the results showed that the AUC value of this nomogram was higher than that of the TNM stage, and it has a stronger ability to predict the prognosis of female patients with CRC. Previous studies have shown that female patients with CRC have a genetic performance of MSI-H, and the prognosis of the tumor in the right-sided colon was worse than that of male patients with CRC.4,37,41,42 In addition, right-sided CRC was more likely to cause long-term chronic inflammation and malnutrition than left-sided CRC, resulting in poor prognosis for cancer patients.4,31,37,41 Our NBR-based nomogram could be used as a simple and easy-to-use system, which could be helpful in female patients with CRC in decision-making regarding treatment and risk adjustment monitoring.
There are several potential limitations of our study. First, this was a retrospective study conducted in a single center, and there may be a selection bias. Indeed, our results may not be applicable to populations in other regions and should be confirmed in future multicenter and multi-sample studies. Second, incomplete data may affect the accuracy of the analysis. In addition, this study was able to verify the feasibility of preoperative NBR to predict the prognosis of CRC; however, there is still a large gap between our results and clinical application of our findings. Therefore, further screening of inflammation and nutrition-related markers to assess the prognosis of patients with CRC is urgently needed.
Conclusion
In this study, we systematically analyzed the relationship between neutrophils and BMI, and confirmed NBR as a novel inflammation and nutritional marker in patients with CRC. For the first time, preoperative NBR has been demonstrated to be a viable and promising biomarker for predicting the prognosis of CRC.
Supplemental Material
sj-tif-1-tct-10.1177_15330338211064077 - Supplemental material for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer
Supplemental material, sj-tif-1-tct-10.1177_15330338211064077 for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer by Weishun Xie, Xiaoliang Huang, Chunyin Wei, Xianwei Mo, Haiming Ru, Lihua Zhang, Lianying Ge, Weizhong Tang and Jungang Liu in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-2-tct-10.1177_15330338211064077 - Supplemental material for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer
Supplemental material, sj-tif-2-tct-10.1177_15330338211064077 for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer by Weishun Xie, Xiaoliang Huang, Chunyin Wei, Xianwei Mo, Haiming Ru, Lihua Zhang, Lianying Ge, Weizhong Tang and Jungang Liu in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-3-tct-10.1177_15330338211064077 - Supplemental material for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer
Supplemental material, sj-tif-3-tct-10.1177_15330338211064077 for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer by Weishun Xie, Xiaoliang Huang, Chunyin Wei, Xianwei Mo, Haiming Ru, Lihua Zhang, Lianying Ge, Weizhong Tang and Jungang Liu in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-4-tct-10.1177_15330338211064077 - Supplemental material for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer
Supplemental material, sj-tif-4-tct-10.1177_15330338211064077 for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer by Weishun Xie, Xiaoliang Huang, Chunyin Wei, Xianwei Mo, Haiming Ru, Lihua Zhang, Lianying Ge, Weizhong Tang and Jungang Liu in Technology in Cancer Research & Treatment
Supplemental Material
sj-tif-5-tct-10.1177_15330338211064077 - Supplemental material for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer
Supplemental material, sj-tif-5-tct-10.1177_15330338211064077 for Preoperative Neutrophil-BMI Ratio As a Promising New Marker for Predicting Tumor Outcomes in Colorectal Cancer by Weishun Xie, Xiaoliang Huang, Chunyin Wei, Xianwei Mo, Haiming Ru, Lihua Zhang, Lianying Ge, Weizhong Tang and Jungang Liu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
The authors thank Lixian Liao, Dingyu Liang, Jinlian Huang, Yuqing Deng, and Lihaopin Huang for their assistance with data collection. We would like to thank Editage (www.editage.cn) for English language editing.
Author Contributions
Conceived and designed the experiments: WT, WX, XH, JL; Performed the data collection: WX, JL, XH, CW, XM, HR, LG, LZ and WT; Analyzed the data: XH, JL, WX; Contributed reagents/materials/analysis tools: WX, JL, XH, CW, XM, HR, LG, LZ, WT; and contributed to the writing of the manuscript: XH, JL, WT, WX, LG; All authors reviewed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the 2019 Guangxi University High-level Innovation Team and the Project of Outstanding Scholars Program, and Guangxi Science and Technology Project (2019AC03004); the Guangxi Clinical Research Center for Colorectal Cancer (Guike: AD19245197); the “Future Academic Star” Project of Guangxi Medical University (WLXSZX21128); the Guangxi Medical and Health Appropriate Technology Development and Promotion Application Project (S2021016); the Natural Science Foundation of Guangxi (2021JJA140081); and the project of improving the basic ability of scientific research of young and middle-aged teachers of Guangxi Zhuang Autonomous Region Education Department (2021KY0086).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
