Abstract
Introduction
Biliary tract cancers (BTCs) are rare, aggressive, and heterogeneous malignancies arising from the bile duct system, including cholangiocarcinoma (CCA), gallbladder cancer (GBC), and ampulla of Vater cancer (AVC), and most patients are diagnosed with advanced disease. 1 In patients with advanced BTCs, systemic treatments are the only therapeutic option, and the combination of gemcitabine plus cisplatin (GP) has been the standard of treatment as first-line chemotherapy. However, the prognosis for these patients is poor, and median overall survival (OS) is less than 1 year.2,3
Recently, advances in whole-exome sequencing (WES) and next-generation sequencing (NGS) of multiple genes have defined the tumor biology of BTCs, and molecularly targeted therapy and immunotherapy including immune checkpoint inhibitors (ICIs) based on NGS findings have been developed.4-12 In addition, a high level of tumor mutational burden (TMB), the total No. of nonsynonymous mutations in the tumor coding regions, has been considered a predictive marker of response to immunotherapy or a prognostic marker in various tumor types.13-15 However, there has been little study on the role of high TMB in advanced BTCs.7,16-19
In current clinical practice, GP have been used as the standard first-line therapy in advanced BTCs. Tumors with high tumor mutational burden (TMB-H) tend to contain genomic instability; therefore, tumors with TMB-H are likely to have sensitivity to DNA-damaging agents, including platinum. 20 In the present study, we intended to evaluate the role of high TMB as a predictive biomarker to GP and a prognostic biomarker in advanced BTCs.
Patients and Methods
Patients
We analyzed all 119 advanced BTC patients who received GP as the first-line treatment were also tested for the TruSightTM Oncology 500 assay (Illumina Inc.) at Samsung Medical Center, Korea, between November 2019 and April 2021. Tumor samples used in NGS test were 60 biopsy and 59 resection samples, and the NGS test were conducted in before the start of systemic chemotherapy. The following clinicopathologic characteristics were collected for all 119 patients: age, sex, tumor site, initial disease status, pathology, chemotherapy, and survival. This study was approved by the Institutional Review Board (IRB No. 2021-05-170) at Samsung Medical Center and individual consent for this analysis was waived. Patients in the database were identified by patient No. only and the patient information kept as confidential information according to IRB protocol. This study was also conducted in accordance with the ethical principles of the Declaration of Helsinki and the Korea Good Clinical Practice guidelines. The reporting of this study conforms to REMARK guidelines. 21
TruSightTM Oncology 500 Assay
Forty (40) ng of DNA were quantified with the Qubit dsDNA HS Assay (Thermo Fisher Scientific) on the Qubit 2.0 Fluorometer (Thermo Fisher Scientific) and then sheared using a Covaris E220 Focused-ultrasonicator (Woburn) and the 8 microTUBE-50 Strip AFA Fiber V2 following the manufacturer's instructions. The treatment time was optimized for FFPE material. The treatment settings were as follows: peak incident power (W): 75; duty factor: 15%; cycles per burst: 500; treatment time (s): 360; temperature (°C): 7; water level: 6. For DNA library preparation and enrichment, the TruSightTM Oncology 500 Kit (Illumina) was used following the manufacturer's instructions. Post-enriched libraries were quantified, pooled, and sequenced on a NextSeq 500 (Illumina Inc.). The quality of the NextSeq 500 (Illumina) sequencing runs was assessed with the Illumina Sequencing Analysis Viewer (Illumina). Sequencing data were analyzed with the TruSight Oncology 500 Local App Version 1.3.0.39 (Illumina). The TruSightTM Oncology 500 is a comprehensive tumor profiling assay designed to identify known and emerging tumor biomarkers, including small variants, splice variants, and fusions. Importantly, the TruSightTM Oncology 500 measures TMB and microsatellite instability (MSI), features that are potential key biomarkers for immunotherapy. TMB was reported as mutations per megabase (Mb) sequenced, and high TMB was defined as more than 10 mutations per Mb (≥ 10 Mut/Mb).
The Treatment Outcomes
All patients were evaluated radiologically for clinical outcomes of objective response rate (ORR), progression-free survival (PFS), and OS to GP as the first-line therapy, according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 through computed tomography (CT) or magnetic resonance imaging (MRI).
Statistics
Descriptive statistics were reported as proportions and medians. Data are presented as the number (%) for categorical variables. Correlations between TMB status and clinicopathologic features were analyzed by Fisher’s exact test. Response categories were assessed according to RECIST 1.1. Survival analyses were performed using the Kaplan–Meier method, and differences were analyzed by log-rank test. Median values with corresponding 95% confidence intervals (95%CIs) were reported. PFS was defined as the time from the start of GP until the date of disease progression or death from any cause. OS was defined as the time from the start of GP until death from any cause. All P-values were two-sided, and statistical significance was set at P < .05. Statistical analysis was performed using IBM SPSS Statistics 25 (Armonk, NY, USA).
Results
Patient Characteristics
One hundred nineteen patients were included in this study. Twenty-two of these patients were categorized as tumors with TMB-H. Among the 22 patients with TMB-H, 6 with intrahepatic cholangiocarcinoma (IHCCC), 9 extrahepatic cholangiocarcinoma (EHCCC), 5 GBC, and 2 AoV cancers were included. Table 1 presents the clinical characteristics between tumors with and without TMB-H. There were no significant differences for clinical characteristics between patients with and without TMB-H, except microsatellites instability (MSI).
Patient characteristics.

Abbreviations: AVC: ampulla of Vater cancer; EHCCA: extrahepatic cholangiocarcinoma; GBC: gallbladder cancer; IHCCC: intrahepatic cholangiocarcinoma; TMB-H: high tumor mutational burden.
Relationship Between the Status of Tumor Mutational Burden and the Effect of Gemcitabine Plus Cisplatin
We compared the efficacy and the PFS to GP according to the status of TMB. The ORR to GP was 40.9% (9 of 22, 95% CI: 20.7-63.7) in patients with TMB-H and 24.8% (24/97, 95% CI: 16.5-34.5) in non-TMB-H. The DCR to GP was 81.8% (18/22, 95% CI 59.7-94.8) in patients with TMB-H and 74.3% (72/97, 95% CI: 64.4-85.6) in non-TMB-L. There were no significant differences in ORR and DCR between patients with TMB-H and non-TMB (P = .126 and p = .454, respectively) (Table 2).
Objective response rate (ORR) to chemotherapy.

Abbreviation: TMB-H: high tumor mutational burden
The median PFS to GP was 5.6 months (95% CI: 3.1-8.1 months) in patients with TMB-H and 6.9 months (95% CI: 4.8-8.9 months) with non-TMB (P = .599) (Figure 1). The mean OS was 24.8 months (95% CI: 18.6-31.0 months) in patients with TMB-H and 23.5 months (95% CI: 17.6-29.3 months) in non-TMB (P = .430) (Figure 2).

Kaplan–Meier curves of progression-free survival (PFS) to gemcitabine plus cisplatin (GP) according to tumor mutational burden (TMB).

Kaplan–Meier curves of overall survival (OS) according to tumor mutational burden (TMB).
Relationship Between the Status of Tmor Mutationa lBurden and the Effect of Immunotherapy
In 32 patients with ICIs as second-line therapy, we evaluated the relation between the status of TMB and the effect of ICIs. Of five patients with TMB-H, three (60%, 3 of 5) achieved tumor response to ICIs (Table 3). In 27 patients with non-TMB-H, only three (11.1%, 3 of 27) achieved a PR to ICIs. There were significant differences in the efficacy of ICIs between patients with TMB-H and non-TMB (P = .034). The median PFS to ICIs was 7.4 months (95% CI: 5.0-9.9 months) in patients with TMB-H and 2.2 months (95% CI: 1.0-3.4 months) in non-TMB (P = .025) (Figure 3).
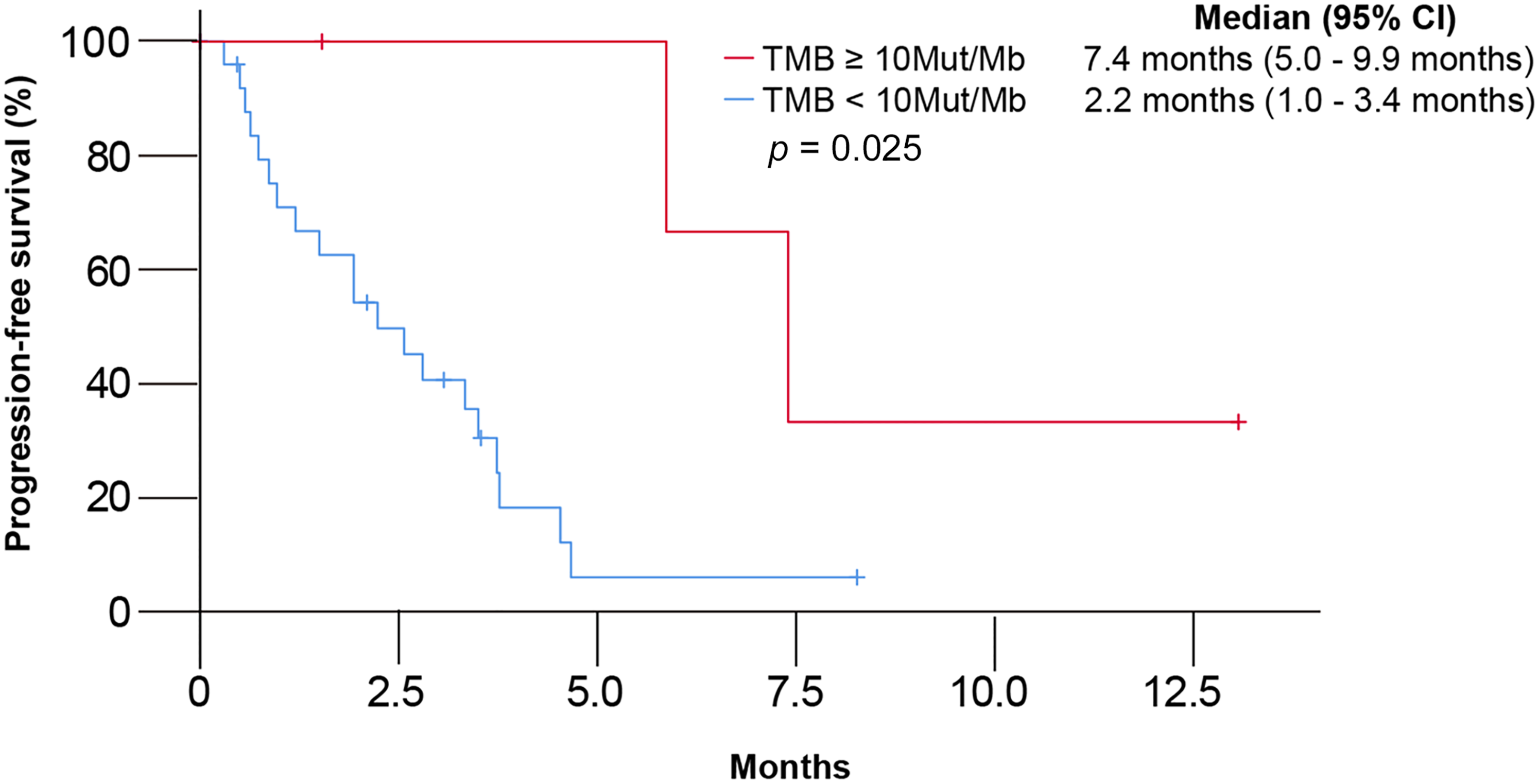
Kaplan–Meier curves of progression-free survival (PFS) to immune checkpoint inhibitors (ICIs) according to tumor mutational burden (TMB).
Objective response rate (ORR) to immunotherapy.

Abbreviation: TMB-H: high tumor mutational burden.
Discussion
The present study demonstrated that the incidence of TMB-H (≥ 10 Muts/Mb) in advanced BTC was 18.5% (18 of 119). There were no significant differences between TMB status and clinical outcomes to GP, including ORR (P = .126), disease control rate (DCR) (p = .454), and median PFS (P = .599).
Additionally, the OS was not different between the two groups. However, in subgroup analysis for 32 patients with ICI as second-line therapy, there were significant differences in the efficacy of ICIs between patients with TMB-H and non-TMB (P = .034). These findings suggested that TMB-H might be a novel biomarker to select ICIs in BTC but was not sufficient as a biomarker for GP use, the standard first-line regimen in BTC.
The role of TMB status was controversial as a predictive or prognostic biomarker to cytotoxic chemotherapy. One study reported that TMB was not a useful predictor of ORR and PFS in patients receiving cytotoxic chemotherapy for various tumor types. 22 In contrast, another study showed that a high TMB was a favorable prognostic factor. 23 That study evaluated the relationship between the TMB status and OS in 1415 immunotherapy-naive patients with various advanced malignancies, including only 13 BTC patients. These previous studies included few patients with advanced BTCs. The present study demonstrated the role of TMB-H as a predictive and prognostic biomarker in advanced BTCs of a relatively large sample size.
There is little information on the effect of TMB-H to ICIs in advanced BTCs.17-19 A previous study reported three patients with TMB-H who received ICIs, and all these patients showed tumor response. 24 Except this study, no additional study has reported relationships between the status of TMB and ICIs. Herein, we analyzed the correlation between the TMB status and the efficacy to ICIs. There were significant differences for the efficacy and median PFS of ICIs between patients with TMB-H and non-TMB-H (P = .034 and P = .025, respectively). This finding suggested that a high TMB was a novel biomarker to predict the tumor response to ICIs in advanced BTC patients.
Although the role of TMB in BTC patients are insufficient, previous studies have demonstrated TMB was reliable predictors of the response to ICI. Also, tumors with TMB-H tend to contain genomic instability and could hypothetically make a tumor more sensitive to DNA-damaging chemotherapy, including platinum. Therefore, we reviewed a new perspective which was a prognostic and predictive roles of TMB in advanced BTCs. There were no significant differences between TMB status and clinical outcomes to GP. However, our results showed significant differences between TMB status and clinical outcomes to ICIs. Based on these results, ICIs to treat advanced BTCs with TMB-H might be reasonable after failure of GP. Simultaneously, we found that 3 of 27 patients with non-TMB-H (11.1%) had a PR to ICIs. This finding revealed that the TMB status alone was not sufficient as a predictive marker to select patients who might benefit from ICIs. Further studies are needed to evaluate single or combination biomarker research, such as PD-L1, MSI, tumor microenvironment, and other predictors
Our study has several limitations. First, this study had a small sample size and was retrospective in nature. Because of the small sample size and retrospective nature of our study, our results should be confirmed in prospective study. Second, only Asian patients with BTC were analyzed in the study, limiting the generalizability because of differences in molecular profiles and clinical features between Western and Eastern patients with BTC. Third, the results of the NGS test before the start of GP of first line therapy were used in this analysis. The status of TMB might be changed by the effect of GP. The finding between the TMB status and efficacy of ICIs in the present study should be interpreted with caution. This study was for relatively small sample size. However, as you already know, the biliary tract cancers were rare orphan disease. Especially, the acquisition of tumor sample in biliary tract cancers is very difficult work. This study tried to conduct the molecular study in biliary tract cancer.
Conclusions
This study revealed that TMB-H in advanced BTC did not have a prognostic role or a predictive role in the standard first-line treatment. However, TMB-H might be a predictive biomarker for ICIs in advanced BTC.
Footnotes
Acknowledgments
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HR20C0025 (SK, KK). .
Ethical Statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by Institutional Review Board (IRB No. 2021-05-170) at Samsung Medical Center and individual consent for this analysis was waived.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
