Abstract
Introduction
Liver cancer remains to be a global health challenge with staggeringly high morbidity and mortality. 1 There will be more than 1 million new cases of liver cancer each year by 2025. 2 The incidence of liver cancer ranks fourth in China among all types of cancer. Hepatocellular carcinoma (HCC) is the most common type of primary liver cancer, accounting for approximately 90% of all cases. 2 Although early stages of HCC can be treated with radiofrequency ablation or surgical resection,3,4 most patients with HCC presented to clinical attention at a relatively advanced stage with dismal prognosis.5,6 Thus, techniques like theranostics using a reliable and specific biomarker hold the promise of early detection and improved clinical outcome for HCC.
RecQ-mediated genome instability (RMI) protein 2 (RMI2), which is an important component of RMI complex, can form a stable subcomplex with RMI1 through an interaction similar to the OB folding domain in Replication Protein A (RPA). While RMI1 is present in most eukaryotes, RMI2 can only be found in plants and vertebrates, suggesting that RMI2 is essential in higher eukaryotes with complex genomes. 7 The OB-fold domains of RMI1 and RMI2 heterodimerize, likely bringing RMI2 to the BLM-Topo IIIα-RMI1-RMI2 (BTR) complex. In previous in vitro studies, the BTR complex was shown to resolve double Holliday junctions. The BTR complex was further shown to be unstable in vitro without the RMI2 component, and RMI2-deficient cells under replication stress could not efficiently recruit BLM (the mutated helicase in Bloom syndrome) to chromatin and nuclear foci. These data suggest that RMI2 holds key functions in the BTR complex maintenance of chromosome stability. RMI1 gene variants expose people to a higher risk of tumorigenesis. 8 The study reported by Xu et al 7 indicated that RMI2 gene mutations or genetic variations may also lead to genome instability and increased risk of tumorigenesis,9,10 such as lung cancer.11,12 The survival rate of patients with lung adenocarcinoma with RMI2 high expression was significantly lower than that with RMI2 low expression. However, studies with regard to the correlation of RMI2 and HCC are scarce. Herein, we explored the expression of RMI2 in HCC and its correlations with patient clinical features and outcomes.
Materials and Methods
Patient Population and Samples
Tissue samples from 330 HCC patients (268 men and 62 women with a median age of 56 years [range 30-85]) were collected from January 2006 to December 2012 at ZheJiang Provincial People's Hospital. All samples were fixed with formaldehyde, embedded in paraffin, and fabricated into tissue microarrays that contained 330 HCC samples and 190 adjacent normal tissues. The clinicopathologic data, including patient age, sex, alpha-fetoprotein (AFP), tumor size, location, number, lymph node/distant metastasis and Edmondson grade, were collected from the electronic medical records. Survival time was defined as the interval from surgery to death or the last follow-up. The study was approved by the Ethics Committee of ZheJiang Provincial People's Hospital (IRB:2021QT261). All patients signed an informed consent form for the advancement of clinical research.
Immunohistochemistry and Analysis
RMI2 expression was assessed by immunohistochemistry and Western blotting. Specifically, 4 mm thick sections were deparaffinized, dehydrated and rehydrated, and incubated with anti-RMI2 antibody (Delaware Avenue, CA, Santa Cruz Biotechnology Inc) mouse monoclonal antibody.
Two independent pathologists blind to the clinical data scored immunohistochemical slides according to the intensity and proportion of positive staining cells. Staining intensity standard: 0 = no staining, 1 = weak or light yellow staining, 2 = moderate or brownish yellow staining, 3 = strong or tan. The staining ratio scores are as follows: 0 points = cells stained less than 5%, 1 point = cells stained 6% to 26%, 2 points = cells stained 26% to 50%, 3 points = cells stained more than 50%. The final score, which was the result of positive cells multiplied by the staining intensity, was categorized as 0 to 4 points (low RMI2 expression) or ≥5 points (high RMI2 expression). Using RIPA buffer for protein extraction in tissues and perform Western blot analysis according to standard procedures. Primary antibodies were: RMI2 (Abcam, ab122685) the recommended dilution for IHC-P is 1:200, for WB is 1:1000, and GAPDH 1:10 000 (Abcam, EPR16891).
Statistical Analysis
The Student's
Expression of RMI2 mRNA in HCC and Non-Carcinomatous Liver Tissues.

Abbreviations: RMI2, RecQ-mediated genome instability protein 2; HCC, hepatocellular carcinoma.
Results
RMI2 is predominantly located at the cytoplasm of HCC cells. RMI2 positivity was found in 33 (17.4%) in the control specimens, which was significantly lower than that in the HCC tissues (17.4% vs 64.6%,

Immunostaining of RMI2 in liver tissue near tumor and cancerous liver tissue. A1-A3 Magnification: × 40 A1-A3: tissue. Immunohistochemical staining of negative expression of RMI2; B1-B3: tissue. Immunohistochemical staining of moderate expression of RMI2; C1-C3: tissue. Immunohistochemical staining of strong expression of RMI2. Magnification: × 40 (A1-C1), 100 (A2-C2), and × 400 (A3-C3).

RMI2 expression in HCC patients.
The Relationship Between RMI2 Expression and Pathological Data of HCC
The correlation between RMI2 expression and patient pathological parameters were tabulated in Table 2. The rate of RMI2 expression was significantly correlated with tumor size, number and microvascular invasion (
Relationship Between RMI2 Expression and Clinicopathological Parameters of HCC Patients.
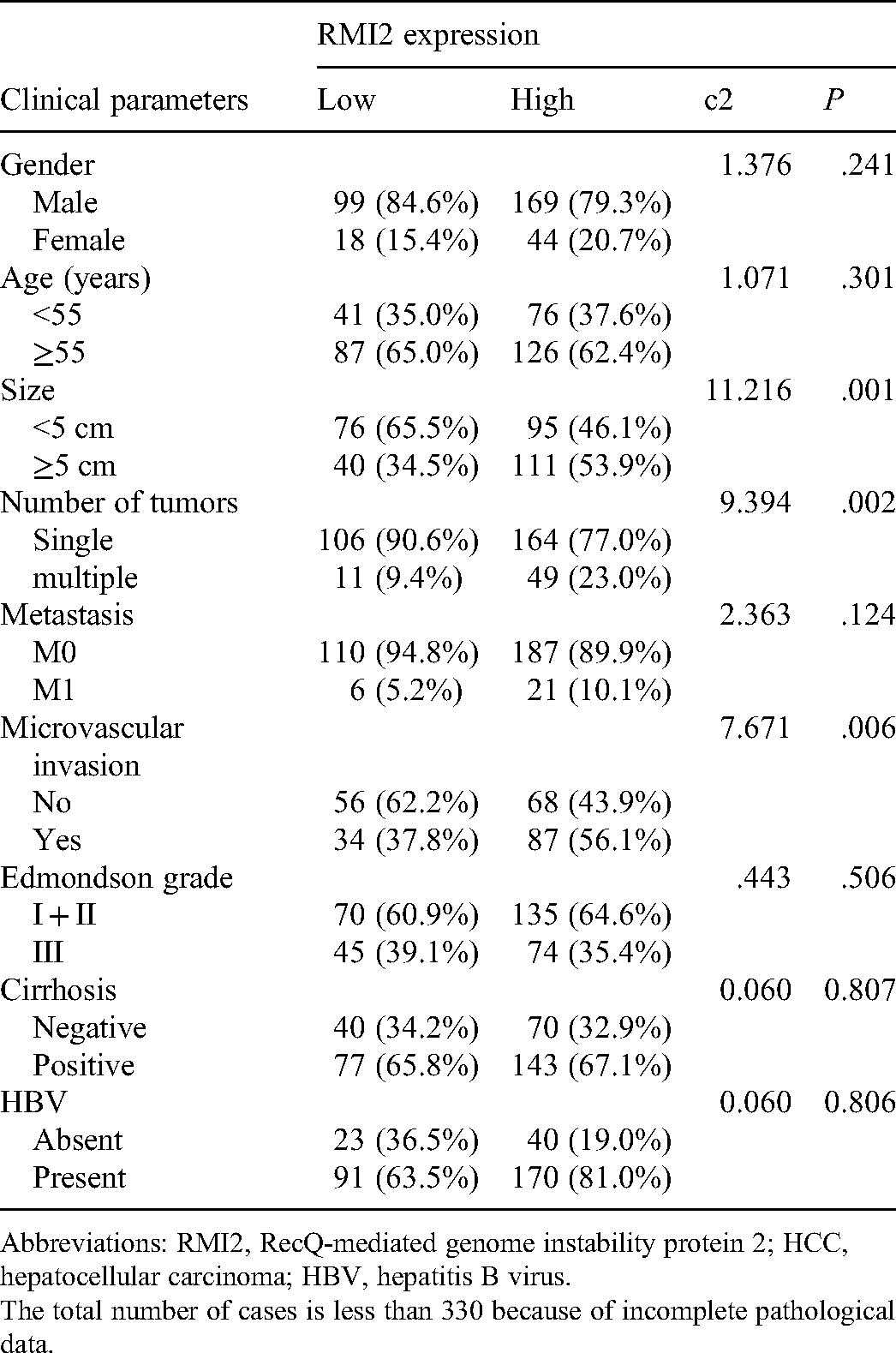
Abbreviations: RMI2, RecQ-mediated genome instability protein 2; HCC, hepatocellular carcinoma; HBV, hepatitis B virus.
The total number of cases is less than 330 because of incomplete pathological data.
Survival Analysis
The median survival time of patients with high RMI2 expression was significantly shorter than that with low RMI2 protein expression (27.2 ± 1.4 months vs 34.5 ± 1.7 months,

Kaplan-Meier survival curves of hepatocellular carcinoma (HCC) patients.
Multivariate Analysis of the Correlation Between Clinicopathological Parameters and Survival Time of Patients With HCC patients.
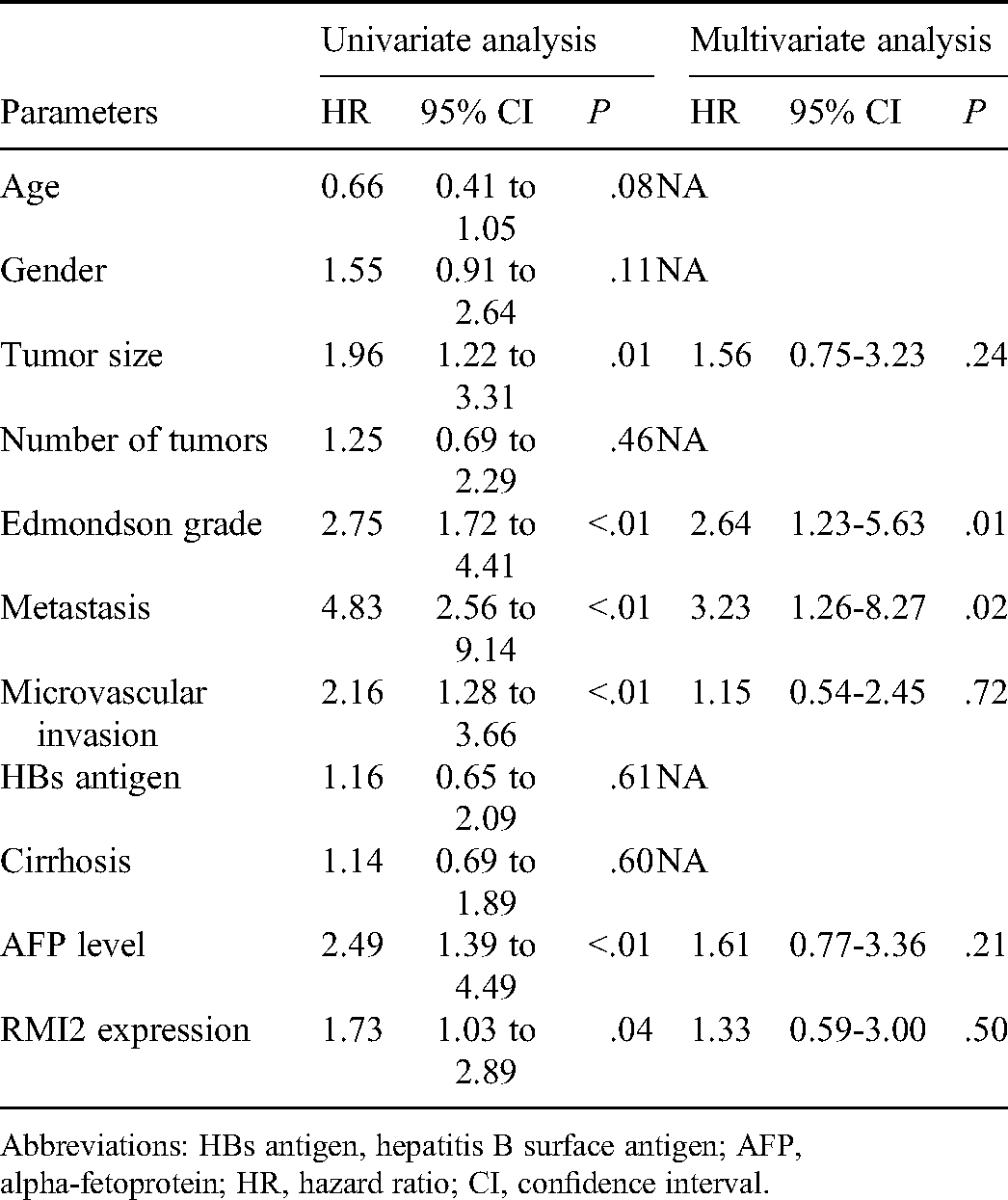
Abbreviations: HBs antigen, hepatitis B surface antigen; AFP, alpha-fetoprotein; HR, hazard ratio; CI, confidence interval.
Discussion
Hepatocellular carcinoma (HCC) is the sixth most common cancer in the world. 13 The prognosis of advanced HCC is still unsatisfactory, and treatment options are limited. 14 Surgical resection and liver transplantation are the two main treatment modalities for HCC, but the clinical outcomes remain unsatisfactory. A reliable and specific biomarker of HCC has the potential to assist in early detection of HCC. Several biomarkers, such as AFP and des-gamma-carboxy prothrombin (DCP), have already been approved for the early detection of HCC. 15 However, the sensitivity of AFP for the diagnosis of HCC is only 60% even if a low-level cut-off (10-20 ng/mL) is used. 16 Furthermore, AFP is normal in 15% to 30% of patients with even advanced HCC. 17 DCP is usually used in combination with AFP and serves as a supplementary indicator. It is still unclear whether the combination of AFP with DCP provides better clinical performance than AFP alone.18-20 Oncogene and tumor suppressor gene mutations are involved in the tumorigenesis and progression of HCC. For instance, common carcinogens like HBV and HCV can cause the activation of oncogenes and the inactivation of tumor suppressor genes. The accumulation of genomic alterations will finally lead to the tumorigenesis.21 The identification of these genes, the understanding of their functions and true mechanisms provide specific targets for early theranostic technique.22
RMI2 is a eukaryotic family of OB3, oligo-nucleotide-binding proteins. It is a dispensable component of the RMI complex and plays a vital part in the producing of homologous recombination intermediates in order to control DNA-crossover-formation in cells.23 RMI2 can interact with cyclin B2 (CCNB2) and cell division cycle-associated protein 3 (CDCA3), which function also as regulatory proteins and interact with other proteins at some vital phases in the cell cycle that play a role in tumorigenesis, thus causing the tumorigenesis of malignant tumors. 11 However, the correlation between the expression of RMI2 and HCC has not been previously reported.
Tumor size, lymph node involvement, and distant metastasis determine HCC prognosis. Our study result indicated that RMI2 was an independent prognosticator with its overexpression portended poor prognosis. Further analysis of the average survival period showed that the survival period of HCC patients with up-regulated RMI2 expression was significantly shortened. Thus, RIM2 can serve as an effective biomarker for predicting the progression of HCC.
Herein, the relative abundance of RMI2 protein in 330 cases of HCC tissues was quantitatively evaluated by immunostaining. Correlation analysis between clinicopathological characteristics and RMI2 staining showed that the proportion of stained cells in early and mid-stage HCC tissues were higher than those in controls, suggesting RMI2 has the potential as a marker for early diagnosis of HCC. Moreover, the positively stained tissues were featured by the morphology of multiple large-sized tumor cells and microvascular invasion. Kaplan–Meier analysis was further applied to prove that the abundance of RIM2 can be an independent predictor of HCC prognosis. Analysis of the survival period of HCC patients demonstrated that the overall survival time of patients with RMI2 overexpression was significantly reduced, indicating the potential of RIM2 to be a predictor for HCC prognosis.
Our study provides a novel tumor-specific target for HCC prognosis prediction. The clinical significance of RMI2 was proved in clinical samples, promising its reliability. Prospective multicenter studies are needed in the future to conclusively demonstrate the prognostic significance of RMI2. However, the value of our study might be discounted by the inherent defect of methodology due to the limited volume of collected samples and limited visualization under microscope.
Supplemental Material
sj-pdf-1-tct-10.1177_15330338211045496 - Supplemental material for The Clinical Significance of RMI2 in Hepatocellular Carcinoma
Supplemental material, sj-pdf-1-tct-10.1177_15330338211045496 for The Clinical Significance of RMI2 in Hepatocellular Carcinoma by Bin Zheng, MS, Heng Wang, MS, Jin-xue Wang, MS, Zheng-hong Liu, MS, Pu Zhang, MD, Dahong Zhang and MD in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338211045496 - Supplemental material for The Clinical Significance of RMI2 in Hepatocellular Carcinoma
Supplemental material, sj-docx-2-tct-10.1177_15330338211045496 for The Clinical Significance of RMI2 in Hepatocellular Carcinoma by Bin Zheng, MS, Heng Wang, MS, Jin-xue Wang, MS, Zheng-hong Liu, MS, Pu Zhang, MD, Dahong Zhang and MD in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
WJX and LZH analyzed and interpreted the patient data regarding the bladder cancer. WH performed the histological examination of the tissue. ZB was a major contributor in writing the manuscript and ZDH, ZP edited the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the Zhejiang Province Bureau of Health (no. 2021KY042), Zhejiang Provincial Natural Science Foundation (no. LQ21H160041).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
