Abstract
Introduction
Oral squamous cell carcinoma (OSCC) is a serious problem in many countries of the world, which is expected to account for around 35 130 cases of incident cancer in the United States in 2019, and tongue squamous cell carcinoma (TSCC) is the most common type of OSCC, with an increasing incidence in many European countries.1,2 The incidence and mortality trends of oral and oropharyngeal cancers in China show an increasing trend. 3 It was estimated that 285 857 new cases and 132 698 deaths were associated to oral and oropharyngeal cancers in China between 2005 and 2013, and tongue cancers being one of the most frequently diagnosed and the leading causes of death among all oral and oropharyngeal cancers. 4 In the past decades, the poor survival of TSCC patients have not been obviously improved, mostly attributing to regional recurrence and the metastasis of cervical lymph node, bloodstream, and distant organs.5,6 Although tumor node metastasis (TNM) stage system is the primary basis in estimating the prognosis of TSCC, it is based fully on the anatomical range of the disease, which has limitations for survival analysis of TSCC patients. 7 Therefore, there is still a lack of more effective approaches to assess TSCC patient's prognosis.
It is known that systemic inflammation- and nutrition-related markers play important role in both the development and the spread of human cancers.8,9 For example, some studies demonstrated that white blood cell count (WBC) was a predictor for cancer. 10 WBCs are mainly composed of neutrophils, monocytes, and lymphocytes. Compelling evidence suggests that these leukocytes may be closely related to patients with different cancers. More recently, evidences have suggested that a high pretreatment neutrophil count (NC) and monocyte count (MC) levels in the blood are associated with poor outcomes in many cancer types,11–14 including tongue cancer. 15 In contrast, a higher preoperative lymphocyte count (LC) appears to be associated with favorable outcomes for several types of cancers.16,17 In addition, nutritional indexes, like red blood cell (RBC) and hemoglobin (HGB), have been also reported to be associated with cancer prognosis.9,18 For example, low RBC count was a risk factor of deep myometrial invasion in endometrioid endometrial carcinoma (EEC), 9 and preoperative anemia predicts poor survival outcomes in patients with endometrial cancer. 19 Preoperative low RBC count showed a negative association with tumor size in oral cancer. 20 The bloodstream is a major pathway for the dissemination of cancer cells, especially in patients with tongue cancer. Circulating platelets (PLT) play a role in vascular thrombosis, atherosclerosis, and inflammation. On the other hand, platelets mediate activation of coagulation and fibrinolytic systems in the causation and metastasis of cancers.21,22 Interestingly, tumor cell can induce platelet aggregation, which leads to tumor cell adhesion to vascular endothelium and facilitate tumor cell growth and invasion. 23 Increasing evidence indicates that elevated preoperative platelet count may be a predictive factor of poorer prognosis and unfavorable clinicopathological parameters for cancer patients. 24 platelet-to-lymphocyte ratio (PLR) is a known inflammatory marker of a derivative index of platelet and lymphocyte, which is considered as a potential marker of the diagnosis and prognosis of cancers in a variety of studies.25–28 Patients with high pretreatment blood PLR levels have poorer overall survival (OS) and disease-free survival (DFS) in various solid tumors.29,30 Although inflammation- and nutrition-related indexes have been studied as prognostic factors in head and neck cancers, there is an insufficient study on hematological prognostic markers in TSCC.
In the present study, we established a nomogram based on the evaluation of the value of age, tumor node metastasis (TNM) stage, pretreatment RBC count, leukocyte and platelet indices to predict the prognosis of patients with TSCC after surgery, thus providing an additional parameter to help guide the management of TSCC patients.
Methods and Materials
Study Population and Data Collection
We designed a retrospective observational study by retrieving data of 172 patients who underwent surgery with TSCC, from the Cancer Hospital of Shantou University Medical College from July 2008 to January 2019. Patients were enrolled in the analysis with the following criteria: (1) they were diagnosed as TSCC by histopathology; (2) they did not suffer from any cancer diseases before TSCC diagnosis or undergo any anti-cancer treatments; (3) patients with a history of chemoradiotherapy and neoadjuvant therapies were excluded; (4) they had complete baseline clinical information and complete follow-up data. This study was approved by the Hospital Ethics Committee in the Cancer Hospital of Shantou University Medical College in China (approval number 2020054) and verbal informed consents were attained from all participants for the retrospective study of their clinical data. All work was conformed to the principles of the Helsinki Declaration.
The relevant clinicopathological features and hematological data were collected for each patient at the time of diagnosis and before any clinical treatment. Specifically, demographic details such as age and gender, smoking behavior, drinking behavior, and pathologic factors such as pT and pN status, TNM stage, and tumor size were charted. In this study, the eighth edition of the American Joint Committee on Cancer (AJCC) TNM stage manual was adopted. The potential blood-based prognostic factors included WBC, RBC, HGB, PLT, NC, LC, MC, CRP, ALB. Lymphocyte-to-monocyte ratio (LMR), PLR, neutrophil-to-lymphocyte (NLR), C-reactive protein/albumin ratio (CAR), immune-inflammation score (SII), systemic inflammation score (SIS), and prognostic nutritional index (PNI) were further derived from these blood indices.31–33 LMR was calculated as the ratio of the absolute LC to MC. The PLR was calculated as the ratio of the absolute peripheral platelet count to LC, and the NLR was calculated as the ratio of the absolute count of NC to LC. CAR was the ratio of CRP to ALB ratio; SII was calculated by using a formula (SII = NE × LY/PLT); The SIS was defined as follows: patients with Alb level < 40 g/L and LMR < 4.44 were classified as 2; patients with either Alb level ≥ 40 g/L or LMR ≥ 4.44 were classified as 1, and patients with both Alb level ≥ 40 g/L and LMR ≥ 4.44 were classified 0. The PNI was calculated according to the formula: Alb (g/L) + 5 × lymphocyte count (×109/L). In this study, continuous variables were transformed into categorical variables by the X-tile, and then the best cut-off values were obtained.
Patients Follow-Up
The follow-up of patients’ survival data was acquired by retrieving medical records, email, and direct communication by mobile phone. All patients were followed up until death or June 2019. In this study, the terminal point was OS, which was defined from the time of receiving surgery to any form of death and the data were censored for patients who were alive at the date of the last follow-up of June 2019.
Statistical Analyses
Statistical analyses were performed by SPSS software, version 22.0 (SPSS Inc.) and R (version 3.6.1) for Windows. The OS of patients was calculated by the Kaplan–Meier method, and the differences between the survival curves were evaluated with the log-rank test. Univariate analysis was performed to evaluate the importance of potential prognostic factors. Variables with a significant level of P ≤ .05 (2 tailed) in univariate analysis were brought forward to a multivariate analysis by a backward stepwise procedure. The univariate and multivariate Cox regression models were used to calculate the hazard ratio (HR) and 95% confidence intervals (CI) to evaluate the impacts of prognostic variables for OS. A predictive nomogram model was established using all variables with a P-value of less than .05 in multivariate Cox regression model by the package of rms in R. In the predictive nomogram model, the prognostic factors of the 1-, 3-, and 5-year OS were analyzed. In addition, the associated calibration curves with bootstrapping (1000 bootstrap resamples) were used to evaluate the predictive ability of 1-, 3- and 5 years OS for the nomograms model. The predictive accuracy and discriminative ability of the prognostic factors were assessed by C-index and decision curve, and were compared with the TNM stage system by using the “survcomp” package. For subsequent analysis, patients were divided into high- and low-risk subgroups according to the optimal cut-off value of the nomogram model risk score. The difference of OS between 2 groups was analyzed by Kaplan–Meier survival analysis. All statistical tests were 2-sided, and P < .05 was considered statistically significant.
Results
Patient Characteristics
The median age for 172 eligible patients were enrolled in this study was 69 years (range: 25-88 years), of which 96 (55.8%) were males and 76 (44.2%) were females. The numbers of patients of I-II and III-IV stages were 121 (70.4%) and 51(20.6%), respectively. At the time of the last follow-up, the median OS was 65 months. In addition, the detailed patients’ demographic and clinical characteristics were summarized in Table 1.
Patient Demographics and Clinical Characteristics.

Abbreviations: T stage, tumor stage; N stage, lymph node stage; TNM stage, tumor node metastasis stage; WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; PLT, platelet; LC, lymphocyte count; MC, monocyte count; NC, neutrophil count; LMR, lymphocyte/monocyte ratio; PLR, platelet/lymphocyte ratio; NLR, neutrophil/lymphocyte ratio; CRP, C-reactive protein; ALB, albumin; CAR, C-reactive protein/albumin ratio; SII, immune-inflammation score; SIS, systemic inflammation score; PNI, prognostic nutritional index.
TNM stage was classified according to the AJCC eighth TNM stage system.
The tumor maximum diameter.
The optimal cut-off values for all variables were calculated by X-tile and were showed in follows age (69 years), tumor size (4.20 cm), WBC (4.95 × 109/L), RBC (3.91 × 109/L), HGB (152.00 g/L), PLT (196.00 × 109/L), LC (1.59 × 109/L), MC (0.46 × 109/L), NC (2.85 × 109/L), LMR (3.40), PLR (177.40), NLR (2.85), CRP (3.26 mg/L), ALB (46.60 g/L), CAR (90.20), SII (20.40), PNI (47.70).
Univariate Analysis and Multivariate Cox Proportional Hazards Regression Analysis of OS
Univariate analysis showed that age (P = .049), T stage (P = .005), N stage (P = .002), TNM stage (P < .001), tumor size (P = .006), WBC (P = .024), RBC (P = .019), and PLT (P = .035), LC (P = .029), LMR (P = .041), PLR (P = .016) were associated with OS of TSCC patients. Subsequently, the prognostic factors significantly associated to OS in univariate analysis were brought into in the multivariate Cox proportional risk regression analysis. The results showed age (P = .029, HR = 2.549; 95% CI: 1.103-5.888), TNM stage (P < .001, HR = 4.090; 95% CI: 2.018-8.293), RBC (P = .047, HR = 0.453; 95% CI: 0.207-0.991), PLT (P = .002, HR = 0.276; 95% CI: 0.276-0.611), and PLR (P = .029, HR = 2.874;95% CI: 1.113-7.420) remained significant independent prognostic factors for OS (Table 2). According to multivariate Cox proportional hazards regression analysis, the forest plot further demonstrated that age, TNM stage, RBC, PLT, and PLR were significantly associated with OS of TSCC patients (Figure 1).

Forest plot revealed the hazard ratio and 95% confidence interval for OS based on the Cox proportional hazards regression analysis in TSCC patients.
Univariate and Multivariate Cox Proportional Hazards Regression Analysis for OS.

Abbreviations: T stage, tumor stage; N stage, lymph node stage; TNM stage, tumor node metastasis stage; WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; PLT, platelet; LC, lymphocyte count; MC, monocyte count; NC, neutrophil count; LMR, lymphocyte/monocyte ratio; PLR, platelet/lymphocyte ratio; NLR, neutrophil/lymphocyte ratio; CRP, C-reactive protein; ALB, albumin; CAR, C-reactive protein/albumin ratio; SII, immune-inflammation score; SIS, systemic inflammation score; PNI, prognostic nutritional index.
TNM stage was classified according to the AJCC eighth TNM stage system.
The tumor maximum diameter.
Nomogram for Prediction of OS
In this study, in order to predict OS, the independent prognostic factors of age, TNM stage, RBC, PLT, and PLR were used to establish a nomogram model for OS probability prediction (Figure 2). A weighted total score calculated from these variables was used to estimate 1-, 3-, and 5-year OS for TSCC patients with surgical treatment (Figure 2). The result showed a close relationship between age, TNM stage, RBC, PLT, and PLR for OS with a 5-year OS less than 15% (Figure 2). In these variables, each factor was assigned a number of risk points, which was obtained by drawing a straight line directly upward to the “points” axis from the corresponding value of the prognostic factor. The process was repeated for each prognostic factor. The points obtained for each covariate were summarized, and “Total Point” axis was located from the sum of the risk points. Finally, a vertical line was drawn directly down to the axis that determined the patient's probability of OS at 1-, 3-, and 5-year. In addition, the calibration plots for the overall survival probability of 1-, 3-, and 5-year revealed a good match between the prediction of OS in the nomogram model and the actual observation (Figure 3).

Nomogram model based on age, RBC, PLT, PLR, and TNM stage in the prediction of 1-, 3-, and 5-year OS probability in TSCC patients. The total points projected on the bottom scales show the probability of 1-, 3-, and 5-y survival.

The calibration plot (a)–(c) are used to estimate OS probability for the nomograms at 1-, 3-, 5-year survival rates.
Assessment of Performance Between Prognostic Model and Conventional Staging Systems
The assessment of the predictive accuracy and the discrimination performance between the prognostic model and traditional staging systems were done using C-index and decision curve analysis. As shown in Table 3, the C-index for the prognostic model was 0.794 (95% CI: 0.729-0.860), which was higher than that of TNM staging 0.685 (95% CI: 0.605-0.765, P = .040). The results of time-dependent C-index curve analysis for the predictive accuracy of the prognostic model and TNM stage showed that the model was higher than that of the TNM stage alone (Figure 4). In addition, the analysis of the decision curves for 1-, 3-, and 5-year OS (Figure 5) showed that the model seemed to have higher overall benefit than the traditional TNM stage systems alone in the majority of the range of threshold probabilities. Taken together, these results indicate the prognostic model had a better net benefit and predictive accuracy to predict survival outcomes when compared to the traditional TNM stage systems.

Comparison of predictive accuracy between prognostic model, RBC, PLT, PLR, age, and TNM stage using time-dependent C-index curve analysis.
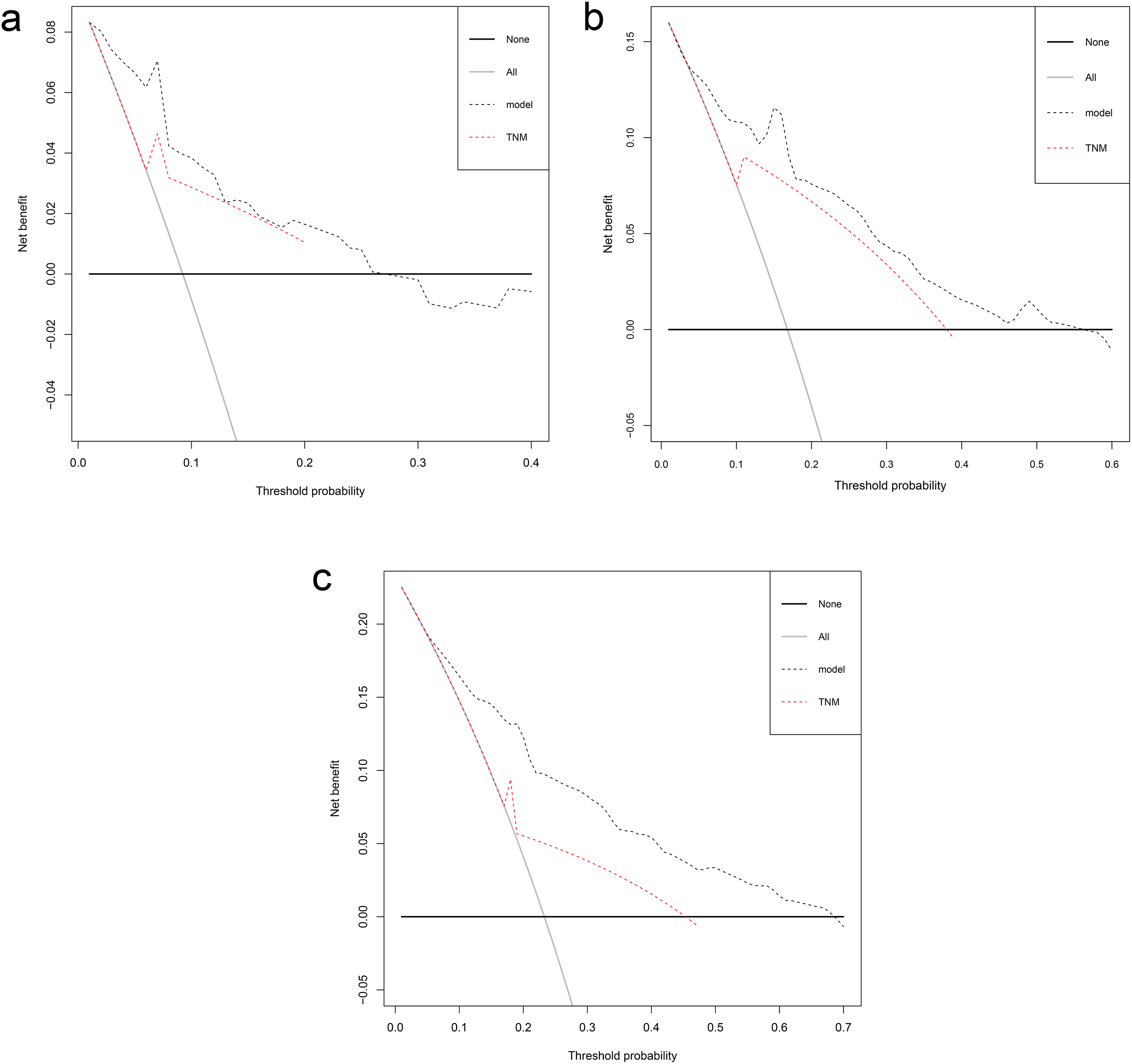
Decision curve analysis the predictive accuracy of model in TSCC patients. The decision curve of 1- (a), 3- (b), and 5- (c) year OS. The y-axis represents the net benefit, which is calculated by summing the benefits (true positive results) and subtracting the harms (false positive results). The horizontal line represents the assumption that no deaths happen.
The C-Index of RBC, PLT, PLR, TNM Stage and Prognostic Model for Prediction of OS in TSCC.

Abbreviations: C-index, concordance index; CI, confidence interval; RBC, red blood cell; PLT, platelet; PLR, platelet/lymphocyte ratio; TNM stage, tumor node metastasis stage; TSCC, tongue squamous cell carcinoma; OS, overall survival.
P-values are calculated based on normal approximation using function rcorrp.cens in Hmisc package.
Risk Stratification Based on the Nomogram Risk Score
Based on optimum cutoff values of the risk score determined by the X-tile program, we subdivided patients into low- and high-risk subgroups. Subsequently, the stratified subgroups were analyzed by Kaplan–Meier survival analysis to assess their OS, and the result indicated that the high-risk group had shorter OS than the low-risk group (23% vs 49%, P < .0001), and the median OS of patients was less than 8 years (Figure 6). This stratification demonstrated that the prognostic model could effectively separate those patients into the 2 risk subgroups with significant differences in OS.

Kaplan–Meier analyses of OS according to the prognostic nomogram model risk score in subgroups of TSCC patients.
Discussion
In recent years, with the characteristic of increasing incidence and the poor prognosis, an increasing number of studies have focused on TSCC. Clinically, it is a dilemma to identify those cases of TSCC who will suffer an aggressive behavior with a poor prognosis and would benefit from surgical resection and/or neck dissection. Therefore, there is an urgent need to find an effective and low-cost method to predict outcomes and help guide clinical treatments. In the current study, we successfully established a prognostic model to predict OS for TSCC by combining clinicopathological features and pretreatment inflammation- and nutrition-related indicators based on the survival analysis approach, including age, TNM stage, RBC, PLT, and PLR. Our model showed better prognostic accuracy and discriminative ability in surgical patients with TSCC when compared with the conventional TNM staging system. The prognostic model successfully divided those patients into high- and low-risk subgroups with significant differences for OS.
Cancer patients frequently present with anemia that may result from the tumor progression, which includes direct and indirect effects of the tumor that lead to decreased red cell production or increased red cell destruction. 34 In tongue cancer, preoperative RBC level was found to be associated to tumor size and histopathological grading.20,35 RBC is an important nutrition-related prognostic factor for cancers and decreased RBC has been found to associate with poor survival in breast cancer and colorectal cancer.36,37 In the present study, we also revealed that a reduced preoperative RBC level was an independent prognostic factor in TSCC.
Chronic inflammation has been identified as an accelerator in cancer development through cytotoxic reaction, abnormal tissue repair, cell proliferative responses, invasion, and metastasis.38,39 Platelets have been considered to mediate activation of coagulation and fibrinolytic systems. Tumor-induced platelets activation and coagulation not only increase the risk of thrombosis, but also are conductive to tumor progression by promoting critical processes such as angiogenesis and metastasis. 21 In addition, as one of the inflammation factors, platelets regulate the immune system to attune cancer-associated inflammation milieu by changing the activation status of the endothelium and by recruiting leukocytes to primary and metastatic tumor sites. 40 Therefore, some researchers support the point of view that the role of platelet-leukocyte interactions may play an important role in the development and progression of cancer. 41 PLR, as another inflammation-associated marker, was included in our model. According to the literatures, an elevated PLR has been identified as a prognostic biomarker in various malignancies, including gastric cancer, 28 tongue cancer, 42 lung cancer, 31 colorectal cancer, 43 and other solid cancers. 29 These studies showed that an increased PLR was correlated to a poor OS or DFS, which was also confirmed in our study. We found that a high PLR could serve as an independent prognostic factor to predict OS in surgical patients with TSCC. On the other hand, it is a fact that PLT or PLR level is easily affected by medication, chemotherapy, and acute inflammation. In the current study, the PLT or PLR level was all evaluated at baseline, and thus could reduce the impact of the relevant aspects. We believed that PLT and PLR might be associated with the clinical outcomes of tongue cancer patients, but the clinical application of such markers for long-term outcome of TSCC patients still needs to be verified repeatedly.
Most studies reported a single blood-based indicator served as an independent prognostic factor for survival prediction of patients with tongue cancer, such as CRP, 44 NLR,45,46 and LMR. 47 However, the prognostic value of the detection of a single marker seems to be limited for tongue cancer. Ozturk et al 48 found that multiple blood-based indicators including NLR, PLR, and N × PLR improved prognostic efficacy, which were related to local recurrence and poor survival of patients with early-stage tongue cancer. On the other hand, recent publications indicated that nomogram based on patients’ demographics and clinicopathological parameters, such as age, gender, and depth of tumor invasion, might also have predictive ability for the prognosis of TSCC patients.49–52 In this study, to increase the prognostic accuracy, many potential blood-based indicators and patients’ demographics and clinicopathological features were included and evaluated together to establish a multivariate prognostic nomogram model for OS prediction. Moreover, we found that our prognostic nomogram based on integrating age, TNM stage, RBC, PLT, and PLR was more accurate in predicting OS than that of the conventional TNM stage system alone. Similarly, Lu et al 53 also established a nomogram model based on the combination of patients’ clinicopathological parameters with an inflammation-related biomarker to predicting OS for patients with TSCC. But the indicators they included in the nomogram were age, lymph node density, and SII, which were different from ours. Furthermore, in the study from Lu et al SII serves as a significant independent prognostic factor for OS and DFS of patients with TSCC. This result was inconsistent with our findings. We noted that SII was not a significant prognostic factor for OS in the current study (Table 2). This may be due to the different sources of patients included. Although their data were validated in an independent cohort, the total number of patients they included was similar to ours (170 cases vs 172 cases). Thus, whether SII is a marker for predicting the survival of patients with TSCC needs to be verified in more extensive studies.
In general, our model might be used as a potential tool for clinicians to select and plan treatment strategies for TSCC patients, and our model offers a convenient and low-cost method to predict outcomes for surgical patients with TSCC, as the blood-based indicators are easy to obtain from routine admission laboratory tests. However, some limitations of our study should be taken into account. Firstly, this is a retrospective study, and thus, the retrospective character of this study cannot completely exclude all potential biases. Secondly, patients’ data used to identify the prognostic factors were obtained from a single cancer center. In the future, it is necessary to obtain a large-scale sample from other research institutions to conduct multicenter validation of the results. Moreover, the increased value for outcome prediction, even if confirmed, may not be of clinical benefit. Because the discovery, validation, and clinical application of a prognostic model is a complex and long process. Nonetheless, our study may have an enlightening effect on exploiting serum inflammation- and nutrition-related markers for prognostic prediction of TSCC patients in the future. Although the above-mentioned shortcomings existed, our prognostic model might serve as a useful tool for clinicians to estimate individualized outcomes and help make treatment strategies for TSCC patients.
Conclusions
In summary, we established a multiparametric prognostic model derived from clinicopathological features and pretreatment blood-based inflammation- and nutrition-related indicators that presented favorable performance when compared to the traditional TNM stage for OS prediction in surgical patients with TSCC. Therefore, it is appealing to imagine that in the case of TSCC, if further validation in multicenter and large-scale samples could be completed, this convenient, low-cost, and simple tool may help clinicians in consulting patients and estimating prognosis for TSCC patients.
Footnotes
Abbreviations
Acknowledgments
The authors thank the reviewers for their critical comments.
Author Contributions
LFW designed the study, analyzed and interpreted the data, and wrote the manuscript; HPG designed the study, provided patient samples and clinical data, and wrote the manuscript; XCH analyzed and interpreted the clinical data; YWL, YL, and TYD collected patient samples and clinical data, and analyzed the data; CTL and LYC collected patient samples and clinical data; YHP, YWX, and HPG conceptualized and designed the study, supervised the project, and revised the paper. All authors have approved the final version and agreed to publish the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the Hospital Ethics Committee in the Cancer Hospital of Shantou University Medical College (Guangdong, Shantou, China, number: 2020054).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Natural Science Foundation of China (81972801), the Guangdong Basic and Applied Basic Research Foundation (2019A1515011873), the Guangdong Esophageal Cancer Institute Science and Technology Program (Q201906), the Medical Project of Science and Technology Planning of Shantou (200605115266724 and 190413105262902); and the 2020 Li Ka Shing Foundation Cross-Disciplinary Research Grant (2020LKSFG01B).
