Abstract
Growing evidence suggests that aberration of the DNA repair pathway significantly contributes to tumorigenesis. Single-nucleotide polymorphisms in DNA repair-related genes such as WRN have been implicated in cancer risk. However, the results of published studies remain inconclusive. Therefore, we performed a meta-analysis of all available and relevant published studies to clarify the role of this polymorphism in cancer. We performed a computerized search of PubMed for publications on WRN Cys1367Arg (T>C) polymorphism and cancer risk and analyzed the genotype data. Odds ratios (ORs) with 95% confidence intervals (CIs) were estimated to assess the association. Sensitivity analysis, heterogeneity test, cumulative meta-analysis, and bias assessment were performed using STATA software 11.0. No association was found between WRN Cys1367Arg (T>C) polymorphism and cancer risk in all genetic models. When stratified by cancer type, results showed that this polymorphism increased the risk of breast cancer (2CC+CT vs 2TT+CT: perallele OR = 1.14, 95% CI = 1.03-1.26, Ptrend = .012; CC vs TT: OR = 1.43, 95% CI = 1.04-1.95, Pvalue = .026; CC+CT vs TT: OR = 1.14, 95% CI = 1.02-1.28, Pvalue = .027). In another analysis stratified by ethnicity, WRN Cys1367Arg (T>C) polymorphism was significantly associated with cancer susceptibility in Europeans (2CC+CT vs 2TT+CT: perallele OR = 1.09, 95% CI = 1.00-1.19, Ptrend = .042; CT vs TT: OR = 1.13, 95% CI = 1.01-1.27, Pvalue = .032; and CC+CT vs TT: OR = 1.13, 95% CI = 1.02-1.26, Pvalue = .025). Our study suggests that WRN Cys1367Arg (T>C) polymorphism is not associated with overall cancer risk, although subgroup analyses suggested an association with breast cancer and overall cancer specifically in European populations.
Keywords
Introduction
Cancer is a multifactorial disease that results from complex interactions between environmental and genetic factors. 1 Genetic variables ranging from single-nucleotide substitutions to major chromosomal aberrations contribute to cancer development. 2,3 Werner syndrome is a rare autosomal recessive disorder characterized by premature aging and cancer susceptibility. 4 –7 Werner syndrome is caused by deficiencies in the human gene WRN. WRN, a member of the RecombinaseQ (RecQ) family of helicases, is a major component of the DNA repair and replication machinery. This enzyme interacts with important DNA repair proteins such as DNA topoisomerase I, BLM helicase, poly(adenosine diphosphate-ribose) polymerase 1, and RAD 52. RecQ helicases are ubiquitous, highly conserved, and adenosine triphosphate-dependent enzymes that are involved in multiple DNA repair pathways; these enzymes protect the genome from illegitimate recombination during chromosome segregation in mitosis and maintain genomic stability. 8 Almost all cancers occur in the elderly population, thus, WRN gene polymorphisms might also be associated with age-related pathologies, including cancer risk. Over the past decade, 7 epidemiological studies 9 –15 evaluated the relationship between WRN Cys1367Arg polymorphism and human cancer risk through March 2014. However, the results of these studies are inconclusive. Therefore, we performed a meta-analysis to precisely characterize whether or not WRN Cys1367Arg polymorphism is associated with human cancer risk.
Materials and Methods
Identification and Eligibility of Relevant Studies
Articles were identified with a literature search using “WRN” and “polymorphism, variants” and “cancer, tumor” as keywords in PubMed (the last search update was March 23, 2014). The articles cited by the retrieved studies were also searched to find additional eligible studies. The current study was limited to articles written in the English language. The following inclusion criteria were used to select articles in our meta-analysis 1 : evaluation of WRN Cys1367Arg (T>C) polymorphism and cancer risk, 2 a case–control or case–cohort design, 3 available genotype frequencies in cancer cases and controls, and 4 randomly selected control group from the general population in the same geographical areas as the cases. The exclusion criteria are shown in Figure 1.
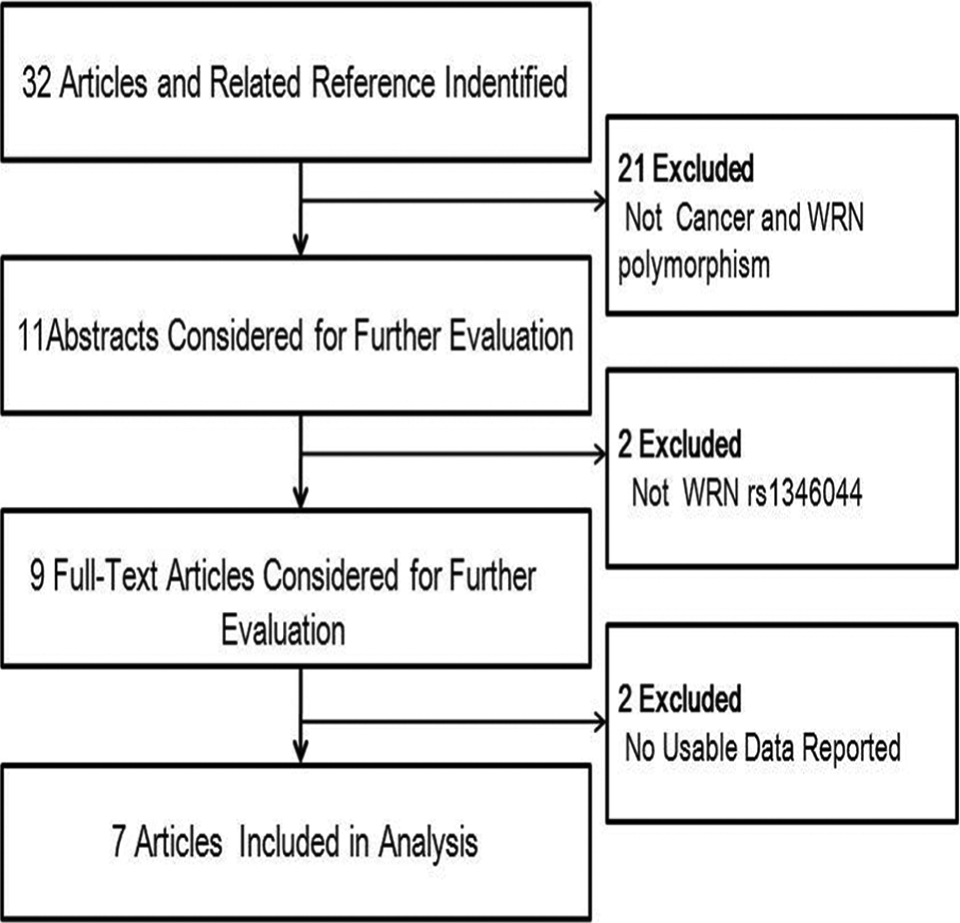
Studies identified with criteria of inclusion and exclusion.
Data Extraction
The data were extracted by 2 investigators independently. Any disagreements were resolved by discussion between the 2 investigators. The following data from each study were extracted: the first author’s name, publication year, country, ethnicity, cancer type, number of cases and controls, and related genotype numbers of cases and controls.
Statistical Analysis
The chi-square goodness-of-fit test was used to evaluate the Hardy-Weinberg equilibrium of the controls of each study. The strength of association between the polymorphism and cancer risk was measured by odds ratios (ORs) with 95% confidence intervals (CIs). The pooled ORs were performed for the following models 16 : additive model (2CC+CT vs 2TT+CT), homozygote comparison model (CC vs TT), heterozygote comparison model (CT vs TT), recessive model (CC vs CT+TT), and dominant model (CC+CT vs TT). Subgroup analyses were also carried out by ethnicity (Europeans and Asians) and cancer type (breast cancer). Z test was used to determine the statistical significance of the pooled ORs. P < .05 was considered to indicate statistical significance. The heterogeneity among studies was evaluated by the Q test. When P > .05, the fixed-effects model (the Mantel-Haenszed method) was used to calculate the pooled OR, otherwise, the random-effects model (the Der Simonian and Laird method) was used. 17,18 In addition, sensitivity analyses were performed by omitting 1 study at a time. Begg funnel plot and Egger test were used to evaluate the publication bias. All statistical analyses in this meta-analysis were conducted with STATA software (version 11; Stata Corporation, College Station, Texas).
Results
Characteristics of the Studies
A total of 32 articles relevant to the searched keywords were obtained through an extensive search. Seven case–control studies involving 5224 cases and 6168 controls fulfilled the inclusion criteria. The characteristics of the included studies are summarized in Table 1. All studies had a case–control design. The cases were patients with cancer, and the controls were matched to the cases. Three genotype distributions in the controls of the selected studies conformed to Hardy–Weinberg equilibrium.
Characteristics of Studies Included in Our Meta-analysis.

Abbreviation: NHL, non-Hodgkin lymphoma.
aRandomly selected from highly heterogeneous Brazilian population.
bCaucasian, African American, and others.
Quantitative Synthesis
Our meta-analysis involved 6168 control participants, including 2974 Europeans and 2684 Asians. The frequencies of C allele for Europeans and Asians were 0.346 (95% CI = 0.284-0.407) and 0.275 (95% CI = 0.259-0.291), respectively. The frequency of C allele was lower in the Asian populations than in the European populations.
We did not find any association between WRN Cys1367Arg (T>C) polymorphism and cancer risk in all genetic models (Table 2 and Figure 2; 2CC+CT vs 2TT+CT: perallele OR = 1.10, 95% CI = 0.94-1.28, P trend = .233; CC vs TT: OR = 1.10, 95% CI = 0.92-1.32, P value = .298; CT vs TT: OR = 1.11, 95% CI = 0.92-1.35, P value = .285; CC+CT vs TT: OR = 1.12, 95% CI = 0.93-1.36, P value = .243; and CC vs CT+TT: OR = 1.07, 95% CI = 0.90-1.28, P value = .429). When stratified by cancer type, results showed that WRN Cys1367Arg (T>C) polymorphism was associated with increased risk of breast cancer (Table 2 and Figure 3; 2CC+CT vs 2TT+CT: perallele OR = 1.14, 95% CI = 1.03-1.26, P trend = .012; CC vs TT: OR = 1.43, 95% CI = 1.04-1.95, P value = .026; CC+CT vs TT: OR = 1.14, 95% CI = 1.02-1.28, P value = .027). When stratified by ethnicity, results also showed a significant association between WRN Cys1367Arg (T>C) polymorphism and cancer risk in the different genetic models for Europeans (Table 2 and Figure 4; 2CC+CT vs 2TT+CT: perallele OR = 1.09, 95% CI = 1.00-1.19, P trend = .042; CT vs TT: OR = 1.13, 95% CI = 1.01-1.27, P value = .032; CC+CT vs TT: OR = 1.13, 95% CI = 1.02-1.26, P value = .025). However, no significant association was found between WRN Cys1367Arg (T>C) polymorphism and cancer risk in all genetic models for Asians (Table 2 and Figure 4).
Meta-analysis of the WRN Cys1367Arg (T>C) Polymorphism on Cancer Risk.
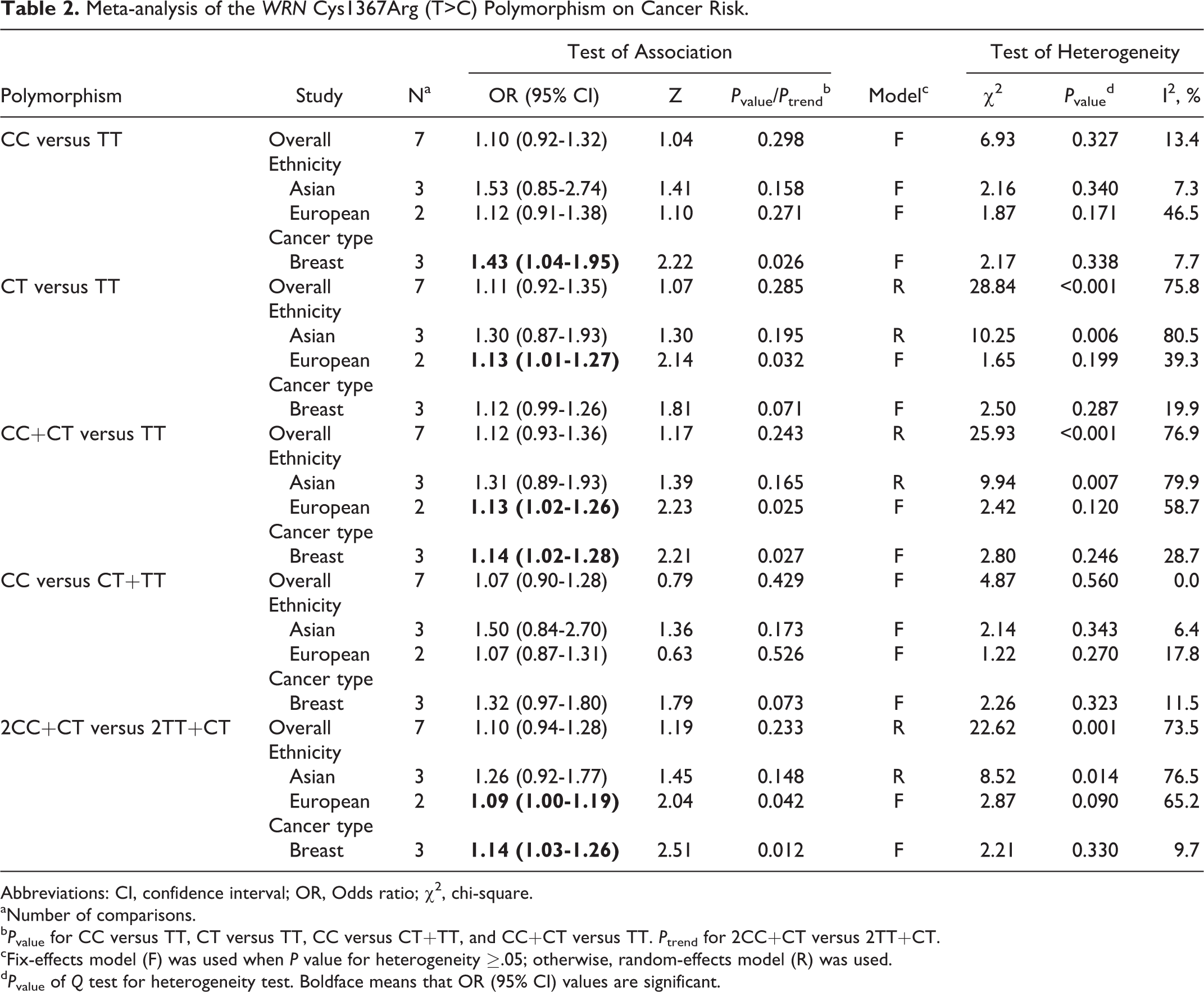
Abbreviations: CI, confidence interval; OR, Odds ratio; χ2, chi-square.
aNumber of comparisons.
b P value for CC versus TT, CT versus TT, CC versus CT+TT, and CC+CT versus TT. P trend for 2CC+CT versus 2TT+CT.
cFix-effects model (F) was used when P value for heterogeneity ≥.05; otherwise, random-effects model (R) was used.
d P value of Q test for heterogeneity test. Boldface means that OR (95% CI) values are significant.
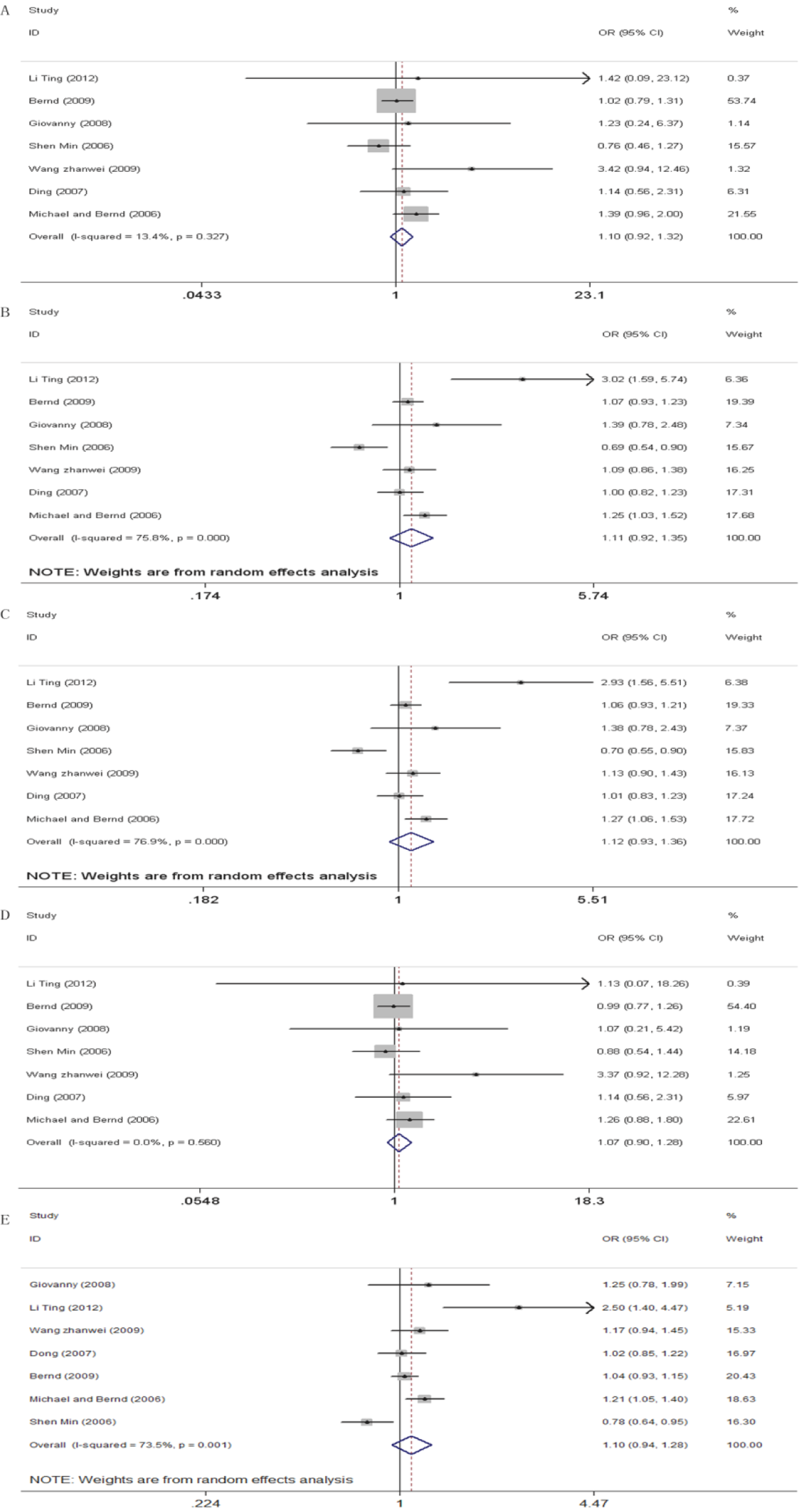
Forest plot showing the association between WRNCys1367Arg (T>C) polymorphism and cancer risk. A, CC versus TT, (B) CT versus TT, (C) CC+CT versus TT, (D) CC versus CT+TT, and (E) 2CC+CT versus 2TT+CT.

Forest plot showing the association between WRNCys1367Arg (T>C) polymorphism and breast cancer. A, CC versus TT, (B) CC+CT versus TT, and (C) 2CC+CT versus 2TT+CT.

Forest plot showing the association between WRNCys1367Arg (T>C) polymorphism and cancer risk in European and Asian population. A, CC versus TT, (B) CT versus TT, (C) CC+CT versus TT, (D) CC versus CT+TT, and (E) 2CC+CT versus 2TT+CT.
Publications Bias
Begger funnel plot and Egger test were used to evaluate the publication bias of the included studies. As shown in Figure 5, the graphical funnel plots were asymmetrical in all comparison models. Egger test was performed to provide statistical evidence of funnel plot symmetry. Results also did not show any evidence of publication bias (2CC+CT vs 2TT+CT: t = 0.96, P = .382; CC vs TT: t = 0.84, P = .439; CT vs TT: t = 0.88, P = .417; CC+CT vs TT: t = 0.90, P = .411; and CC vs CT+TT: t = 1.04, P = .345).
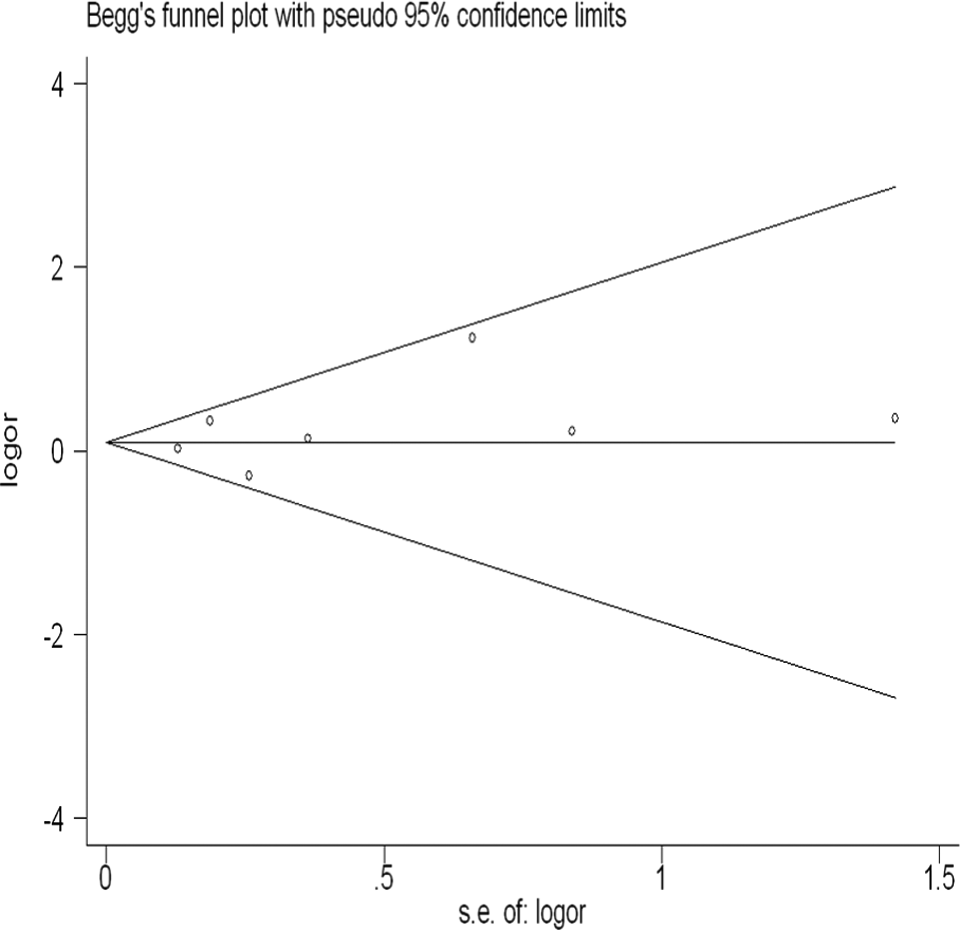
Begg funnel plot for publication bias test (CC vs TT). Each point represents a separate study for the indicated association. Log (OR): nature logarithm of OR. Horizontal line represents size of effect. OR indicates odds ratio.
Sensitivity Analyses
To test the influence of each study to the pooled ORs, sensitivity analyses were performed by omitting 1 study at a time. The results were not significantly altered when any study was deleted. These results indicate that our meta-analysis is stable (data not shown).
Discussion
Recent studies have focused on the relationship between aberration of the DNA repair pathway and cancer risk. WRN, a member of human RecQ, monitors genome integrity and controls cellular response to genotoxic stress. 8 WRN, together with other related factors, helps recruit the suitable DNA repair factors to the lesion site. In addition, the WRN complex can directly activate the apoptotic pathway during an excessive damage.
Single-nucleotide polymorphisms (SNPs) are the most common sources of human genetic variation, which contributes to cancer susceptibility. 19 Although the relationship between WRN Cys1367Arg (T>C) polymorphism and cancer risk has attracted increasing attention and has been extensively explored, the results of previous studies are conflicting and heterogeneous. For instance, Li et al 9 found that WRN Cys1367Arg (T>C) polymorphism contributes to esophageal tumorigenesis, whereas Shen et al 11 showed that WRN Cys1367Arg (T>C) polymorphism is associated with decreased risk of non-Hodgkin lymphoma. In the present meta-analysis, we analyzed pooled data from case–control studies to determine the role of WRN Cys1367Arg (T>C) polymorphism in cancer susceptibility. Overall, we did not find any association between WRN Cys1367Arg (T>C) polymorphism and cancer risk in all genetic models.
Aside from prolonged estrogen exposure (indicated by early menarche and late menopause), interaction with some genetic factors also greatly contributes to breast cancer development. 20 Cheng found that BRCA1452-1079 directly binds to WRN C-terminal region and subsequently lightly affects its functions. The most common polymorphisms of WRN Cys1367Arg are located in the binding domain of BRCA1. 21,22 Thus, we suspected that interaction with genetic factors increases the susceptibility of an individual to breast cancer. Our results supported this hypothesis and showed that WRN Cys1367Arg polymorphism increases breast cancer risk.
In the subgroup analysis by ethnicity, our meta-analysis showed that Europeans were associated with an increased cancer risk. Smaller sample size of the Asian populations compared to the European populations may have contributed to the nonsignificant findings in Asians. In addition, differences in socioeconomic, lifestyle factors, and biological factors also contribute to racial/ethnic differences in cancer outcomes. For example, Sridhar 23 controlled for socioeconomic factors and found that black and white men have similar prostate cancer outcomes. The differences of common DNA variations in different ethnicities also play important roles in cancer development. 3 Consistent with this result, our data showed that the frequency of C allele was lower in the Asian populations than in the European populations.
This meta-analysis has some limitations. First, lack of original data in the included studies limited further evaluation of gene–gene and gene–environment interactions. Second, only studies written in English were included, which may have affected the accuracy and universality of the conclusion. 24 Third, our results were based on unadjusted estimates. A precise analysis is warranted if individual data such as sex and age are available. Regardless of these potential limitations, this meta-analysis has several strengths that should be acknowledged. First, sufficient data were extracted from well-selected studies, and the results were statistically more powerful than any single study. Second, no publication bias was found in our meta-analysis. In summary, our study suggests that WRN Cys1367Arg (T>C) polymorphism is associated with breast cancer risk and cancer susceptibility in Europeans. However, large-scale well-designed epidemiological studies with ethnically diverse populations and functional evaluations must be conducted to confirm our findings.
Footnotes
Abbreviations
Authors’ Note
Bo Wang, Guifang Li, and Fei Sun contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
