Abstract
Background:
The aim of this study was to investigate the predictive value of a prognostic model based on the lymphocyte-to-monocyte ratio (LMR) before radioiodine treatment for the recurrence of papillary thyroid carcinoma (PTC).
Methods:
Clinicopathological data of 441 patients with papillary thyroid cancer were collected retrospectively. The Receiver operating characteristic (ROC) was used to determine the optimal cut-off value for predicting PTC recurrence by LMR before radioiodine treatment. Recurrence was the endpoint of the study, and survival was estimated by the Kaplan-Meier method, and any differences in survival were evaluated with a stratified log-rank test. Univariate and multifactorial analyses were performed using Cox proportional-hazards models to identify risk factors associated with PTC recurrence.
Results:
The ROC curve showed that the best cut-off value of LMR before radioiodine treatment to predict recurrence in patients with PTC was 6.61, with a sensitivity of 54.1%, a specificity of 73%, and an area under the curve of 0.628. The recurrence rate was significantly higher in the low LMR group (16%) than in the high LMR group (5%) (P = 0.001, χ2 = 12.005). Multifactorial analysis showed that LMR < 6.61 (P = 0.006; HR = 2.508) and risk stratification (high risk) (P = 0.000; HR = 5.076) before radioiodine treatment were independent risk factors predicting recurrence in patients with PTC. Patients with preoperative LMR < 6.61 and high risk stratification had the lowest recurrence-free survival rate and the shortest recurrence-free survival time.
Conclusions:
The LMR-based prognostic model before radioactive iodine treatment is valuable for early prediction of PTC recurrence and it can be used in clinical practice as a supplement to risk stratification and applied in combination to help screen out patients with poorer prognosis early.
Keywords
Introduction
Thyroid carcinoma is the most common malignant tumor in the endocrine system, and its incidence rate is increasing through the years in many countries. The incidence of thyroid cancer ranks fourth and fifth among all malignant tumors among women in China and the United States, respectively. 1 -3 It is estimated that there will be 52890 new cases in United States in 2020. 1 The rapid increase of thyroid cancer is mainly differentiated thyroid carcinoma (DTC), accounting for more than 90%. DTC includes papillary thyroid carcinoma (PTC), follicular thyroid carcinoma (FTC), and mixed carcinoma of the 2, among which papillary thyroid carcinoma (PTC) is the most common pathological subtype. After the standardized treatment of total thyroidectomy, radioactive iodine therapy and TSH suppression therapy, most patients with PTC have a good prognosis. 4 Despite a good prognosis, locoregional recurrence (LRR) in patients with PTC is still a persistent problem, with about 6.9%-18% of patients developing LRR after initial treatment. 5 -7 Tumor node metastasis (TNM) staging is the most commonly used staging method in clinical practice. Because the TNM staging does not fully assess the risk of recurrence in DTC, the American Thyroid Association (ATA) Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer classify patients as with low, moderate and high risk of recurrence in 2009, 8 and the 2015 version of the ATA thyroid cancer guidelines have been revised and supplemented on this basis. However, it has been reported that the local or distant recurrence rate of low-and medium-risk PTC patients is as high as 6%, 9 -11 so risk stratification alone is not enough to accurately predict the prognosis of patients. In recent years, it has been found that inflammation plays a key role in the initiation, promotion, and progression of cancers, and by mediating the interaction of immune responses, it affects the prognosis and therapeutic response of tumors. 12,13 Some scholars propose that the LMR can indirectly reflect the immune status of the host and serve as a predictor of the prognosis of malignant tumors. The literature shows that LMR before treatment has important value in predicting the prognosis of malignant tumors such as lung cancer, pancreatic cancer, colorectal cancer and breast cancer. 14 -18
Recently, it has been reported that preoperative low LMR is associated with poor overall survival and recurrence of thyroid cancer. 19,20 These studies of LMR have provided good evidence for it’s role in patients with Thyroid carcinoma. At present, there is no widely accepted LMR cutoff value, and there is no literature to combine this inflammatory marker with the existing risk stratification system to evaluate the prognosis of patients. Moreover, most of the literature is limited to the detection of preoperative LMR levels. Different methods of operation, lymph node dissection areas, and different surgical levels of surgeons, and whether the patient receive radioactive iodine therapy after the operation will have a certain impact on the prognosis of thyroid cancer. Therefore, we believe that the preoperative levels of LMR may not be the best choice to evaluate the prognosis of thyroid cancer. In our study, the clinicopathological data of 441 patients with PTC in our hospital were analyzed retrospectively, in order to explore the relationship between LMR before radioiodine therapy and clinicopathological features and prognosis of PTC patients.
Materials and Methods
Patient Cohort
By consulting the electronic medical record system, the clinicopathological data of PTC patients in the first affiliated Hospital of Chongqing Medical University from January 2015 to December 2016 were collected retrospectively. Inclusion criteria were as follows: a. Postoperative pathology confirmed that the tumor was PTC, and there was no distant metastasis. b. Have complete clinicopathological data. c. All patients underwent radical thyroidectomy simultaneous neck lymph node dissection and radioiodine treatment. d. Age ≥ 18 years old. Exclusion criteria: a. Those who are not operated for the first time; b. Received chemotherapy or radiotherapy before operation; c. Clinical evidence showed a history of infectious diseases; d. Suffering from other systemic malignant tumors; e. Accompanied by Hashimoto’s thyroiditis or other autoimmune diseases; f. Other diseases that may affect peripheral blood cell count. Finally, a total of 441 patients with PTC were enrolled in the group. Blood cell detection was performed in all patients before operation and radioiodine therapy within 3 days. All patients received 100-150 mCi radioactive iodine therapy. In order to increase the level of thyrotropin in radioactive iodine therapy, patients began to stop taking levothyroxine 3 weeks before taking radioactive iodine. The response to the treatment was evaluated 6 months after the initial radioiodine therapy. According to the 2015 version of the ATA thyroid cancer guidelines, patients’ responses to radioiodine therapy were divided into 2 categories: Excellent response (ER) and non-Excellent response (non-ER), the latter including Biochemical incomplete response, Structural incomplete response and Indeterminate response. According to the 2015 version of the ATA thyroid cancer guidelines, intermediate risk of tumor recurrence was defined as 1 of the following: a. Microscopic invasion of tumor into the perithyroidal soft tissues; b. RAI-avid metastatic foci in the neck on the first posttreatment whole-body RAI scan; c. Aggressive histology (e.g., tall cell, hobnail variant, columnar cell carcinoma); d. Papillary thyroid cancer with vascular invasion; e. Clinical N1 or >5 pathologic N1 with all involved lymph nodes <3 cm in largest dimensional; f. Multifocal papillary microcarcinoma with ETE and BRAFV600E mutated (if known).And high risk of tumor recurrence was defined as 1 of following: a. Macroscopic invasion of tumor into the perithyroidal soft tissues (gross ETE); b. Incomplete tumor resection; c. Distant metastases; d. Postoperative serum thyroglobulin suggestive of distant metastases; e. Pathologic N1 with any metastatic lymph node ‡3 cm in largest dimension; f. Follicular thyroid cancer with extensive vascular invasion (> 4 foci of vascular invasion).
Follow-Up of Patients
The follow-up began after patients were treated with radioactive iodine, and the deadline for follow-up was July 31, 2020. The follow-up was conducted by the combination of inquiring electronic medical record system, outpatient service, and phone calls. The follow-up time was 44-71 months, and the median follow-up time was 56 months. Patients went to our hospital to review thyroid function, thyroglobulin (TG), thyroid autoantibodies (including TGAb and TPOAb) and thyroid ultrasound (TUS) once every 3 months within 1 year, and then every 6 months.
The primary endpoint for analysis was recurrence, if recurrence is suspected and further assessment is needed, additional diagnostic imaging examinations, including diagnostic dose 131I whole-body scan, cervicothoracic CT, whole-body bone scan, and 18F-FDG PET-CT, are performed to confirm LRR or distant metastasis. According to the 2015 ATA guidelines, disease recurrence is defined as biochemical recurrence (suppressed Tg >1 ng/mL, TSH-stimulated Tg > 10 ng/mL, or rising anti-Tg antibody levels in the absence of structural disease) and structural recurrence (either biopsy-proven or highly suspicious for disease with or without abnormal serum Tg). Recurrence-free survival was defined as the time interval between the date of receiving radioactive iodine treatment and the earliest detection of structural or biochemical evidence of recurrence, or the date of last follow up.
Statistics
SPSS 26.0 statistical software was used for data analysis. The parametric continuous data were expressed as mean ± standard deviation, and T-test was used for comparison between groups. The nonparametric data were presented as a median and interquartile range (IQR) (median and 25%-75%), and the Mann-Whitney U test was applied for comparison between groups. The categorical data were presented as counts and percentages, and the chi-square test was used for comparison between groups. Paired t test was used to compare the blood cell count of the same patient before thyroidectomy and radioiodine treatment. The best cut-off value of LMR was determined by the ROC curve, and its sensitivity and specificity in predicting recurrence in patients with PTC were analyzed. According to the best cut-off value, the patients were divided into the low LMR group and the high LMR group, and the differences in clinicopathological characteristics between the 2 groups were compared. The survival was estimated by the Kaplan-Meier method, and the differences in survival were evaluated with a stratified Log-rank test. The factors that may affect the recurrence of the patient with PTC were analyzed by Univariate and Multivariate analysis with the Cox proportional-hazards regression model. When P < 0.05, the difference was considered to be statistically significant. This retrospective study was approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (IRB No. 2021-065).
Results
Baseline Characteristics
Of all patients, 64% (284) were female. The median age at diagnosis was 41 years (32-49), and the median size of tumor was 1.2 cm (0.8-2). The TNM stage distribution based on the 8th American joint cancer Committee (AJCC) staging system was stage I for 399 patients (91%), II for 34, Ⅲ for 7 and Ⅳ for 1. Among all patients, 378 cases (86%) had cervical lymph node metastasis, 70 cases (16%) had extra thyroid invasion, and 257 cases (58%) had multifocal tumors. The median absolute lymphocyte count (L) was 2.04 × 109/L (IQR 1.67-2.44), and that of absolute monocyte count (M) is 0.25 × 109/L (IQR 0.2-0.32). The median LMR was 8.07 (IQR 6.19-10.38). According to the 2015 version of the ATA thyroid cancer guidelines, 56 (13%) had low recurrence risk, while 257 (58%) and 128 (29%) had intermediate or high risk, respectively. Of the 429 patients, 325 (74%) responded excellent (ER) to the Initial radioiodine therapy. Up to now, 37 patients (8%) have relapsed, and the RFS rate of 441 PTC patients is 92%.
The Best Cutoff Value of LMR Before Radioactive Iodine Treatment and the Grouping of Patients
As shown in Figure 1, according to the ROC curve, the best cutoff value of LMR before radioactive iodine treatment for predicting recurrence of PTC patients was 6.61, and the area under the curve (AUC) was 0.628, with a sensitivity of 54.1% and specificity of 73%. According to the best cut-off value, 441 PTC patients were divided into the low LMR group (n = 129, 29%) and the high LMR group (n = 312, 71%).

This is the ROC curve for predicting the recurrence of PTC patients by the LMR level before receiving radioactive iodine treatment. According to the ROC curve, the best cutoff value of LMR for predicting recurrence of PTC patients was 6.61. The AUC was 0.628, with a sensitivity of 54.1% and specificity of 73%. ROC indicates receiver operating characteristic; PTC, papillary thyroid carcinoma; LMR, lymphocyte-to-monocyte ratio; AUC, area under the curve.
Relationship Between the LMR Before Radioiodine Therapy and Clinicopathological Features and Prognosis of Patients With PTC
Kendall’s Tau-b correlation analysis showed that the LMR before radioiodine therapy was negatively correlated with age (r = −0.097, P = 0.003), multifocality (r = −0.096, P = 0.014), TNM stage (r = −0.089, P = 0.022), response to Initial radioiodine therapy (r = −0.128, P = 0.001), recurrence (r = −0.101, P = 0.01), and positively correlated with recurrence-free survival time (r = 0.167, P = 0.000).
The clinicopathological features of the 2 groups are shown in Table 1. Compared with the high LMR group, the patients in the low LMR group are older (42 years vs. 40 years, P = 0.029), and are more likely to have multifocal tumors (66% vs. 55%; P = 0.037, χ2 = 4.348), with higher risk stratification (37% vs. 26%; P = 0.037, χ2 = 6.59). However, there were no significant differences in gender, tumor diameter, extra thyroid invasion, lymph node metastasis, unilateral and bilateral, T stage and TNM stage between the 2 groups (P > 0.05).At the same time, Figures 2 and 3 show that the recurrence-free survival time of the low LMR group was significantly shorter than that of the high LMR group (P = 0.001).
Comparison of Clinicopathological Features and Prognosis of PTC Patients Between the High LMR Group and the Low LMR Group.
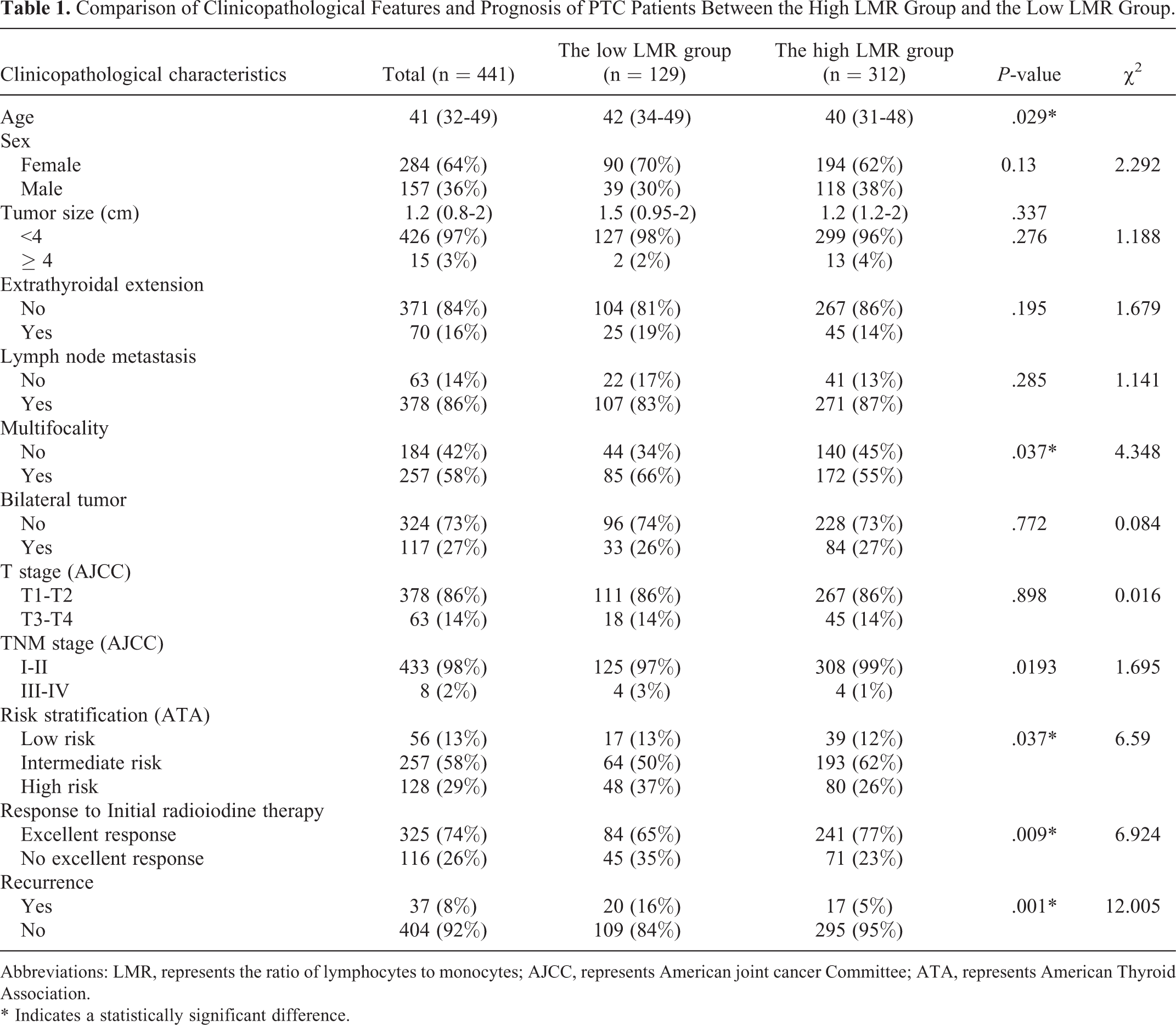
Abbreviations: LMR, represents the ratio of lymphocytes to monocytes; AJCC, represents American joint cancer Committee; ATA, represents American Thyroid Association.
* Indicates a statistically significant difference.

The best cutoff value of LMR before radioactive iodine treatment for predicting recurrence of PTC patients was 6.61. According to the best cut-off value, 441 PTC patients were divided into 2 groups (LMR > 6.61 and LMR < 6.61) and the recurrence-free survival rates of the 2 groups were compared. The results showed that patients with LMR < 6.61 had a lower recurrence-free survival rate than those with LMR > 6.61 (P = 0.001). LMR indicates lymphocyte-to-monocyte ratio.
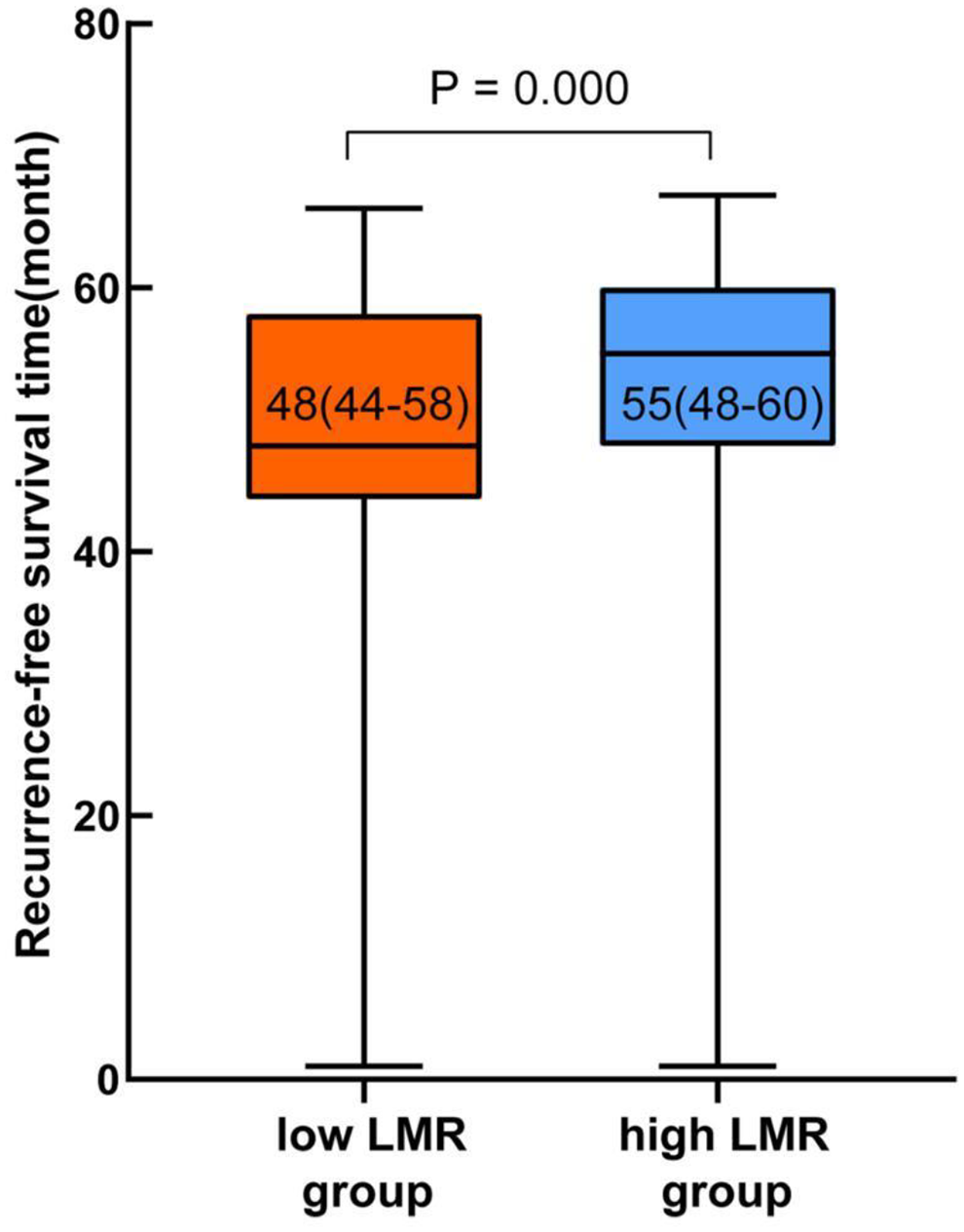
This is a comparison of the recurrence-free survival time between the 2 groups. The median recurrence-free survival time was 48 (44-58) months in the low LMR group and 55 (48-60) months in the high LMR group. The recurrence-free survival time of the low LMR group was significantly lower than that of the high LMR group (P = 0.000). LMR indicates lymphocyte-to-monocyte ratio.
Factors Affecting Recurrence in Patients With PTC
Univariate analysis showed that age ≥ 55 years (P = 0.008; HR = 2.745), bilateral tumors (P = 0.006; HR = 2.472), Multifocality (P = 0.031; HR = 2.286), TNM stage (III-IV) (P = 0.000; HR = 8.714), risk stratification (high risk) (P = 0.000; HR = 4.888), LMR < 6.61(P = 0.001; HR = 2.958) were associated with the recurrence of patients with PTC. Multivariate analysis showed that risk stratification (high risk) (P = 0.000; HR = 5.076) and LMR < 6.616 (P = 0.006; HR = 2.508) were independent risk factors for predicting recurrence of patients (Table 2).
Cox Proportional Hazards Model for the Recurrence in Patients With PTC.

Abbreviation: LMR, represents the ratio of lymphocytes to monocytes.
* Indicates a statistically significant difference.
In order to evaluate the value of LMR before radioiodine therapy combined with risk stratification in predicting the recurrence of patients with PTC, patients with low or moderate risk of recurrence and LMR > 6.61 were defined as Score = 0, high risk of recurrence or LMR < 6.61 were defined as Score = 1, and those with high risk of recurrence and LMR < 6.61 were defined as Score = 2. The results showed that the recurrence-free survival rate of patients with a score of 2 was significantly lower than that with a score of 0 (P = 0.000) or 1 (P = 0.001) (Figure 4), and the recurrence-free survival time of patients with a score 2 was significantly shorter than that with a score of 0 (P = 0.000) or 1 (P = 0.005) (Figure 5). Figure 6 compares the combined application of LMR and risk stratification with risk stratification alone for prognostic assessment of PTC and shows that patients with preoperative LMR <6.61 and high risk stratification had the lowest recurrence-free survival rate.

In order to evaluate the value of LMR before radioiodine therapy combined with risk stratification in predicting the recurrence of patients with PTC, patients with low or intermediate risk of recurrence and LMR > 6.61 were defined as Score = 0, high risk of recurrence or LMR < 6.61 were defined as Score = 1, and those with high risk of recurrence and LMR < 6.61 were defined as score = 2. This is a comparison of the recurrence-free survival rates of the 3 groups of patients with different scores. The results showed that patients with higher scores among the 3 groups had a lower recurrence-free survival rate. LMR indicates lymphocyte-to-monocyte ratio.
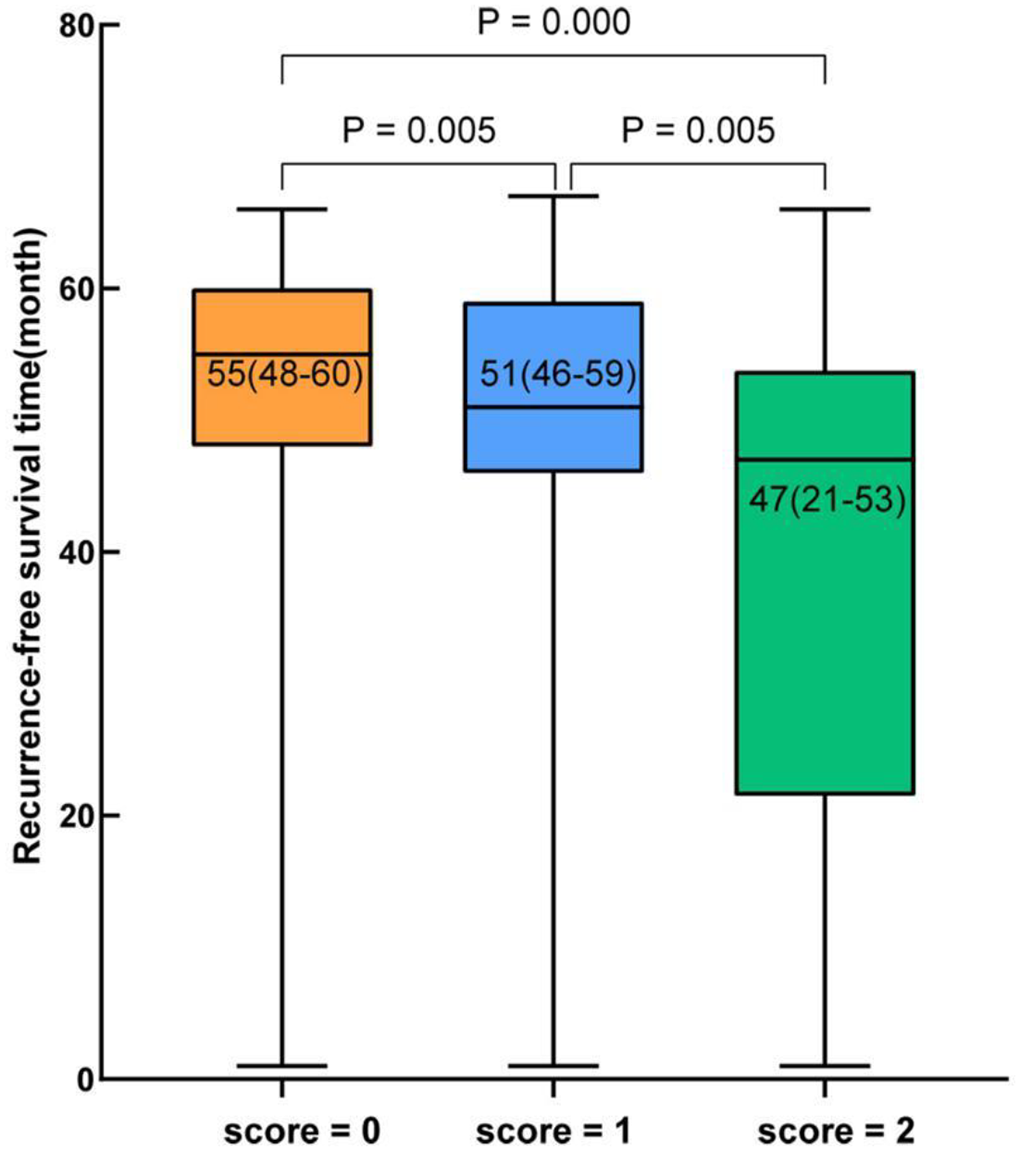
Patients with low or intermediate risk of recurrence and LMR >6.61 were defined as score = 0, high risk of recurrence or LMR < 6.61 were defined as score = 1, and those with high risk of recurrence and LMR < 6.61 were defined as score = 2. This is a comparison of the recurrence-free survival time between the 3 groups. The median recurrence-free survival time was 55 (48-60) months in the Score = 0 group and 51 (46-59) months in the Score = 1 group and 47 (21-53) months in the Score = 2 group. Patients with higher scores have shorter recurrence-free survival times. LMR indicates lymphocyte-to-monocyte ratio.

The dashed line indicates the recurrence-free survival curves of patients with different risk stratification, and the solid line indicates the recurrence-free survival curves of patients with different scores when combined LMR with risk stratification. The patients with a score of 2 had the worst prognosis, so the combined application of LMR and risk stratification is beneficial for early detection of patients with a poor prognosis.
Comparison of Peripheral Blood Lymphocytes, Monocytes and LMR in Patients With PTC Before Thyroidectomy and Radioiodine Therapy
Table 3 compares the peripheral blood LMR levels of the same patient before and after receiving radioactive iodine treatment. Paired t-test showed that the lymphocyte count (P = 0.000) and LMR (P = 0.000) before radioiodine treatment were significantly higher than those before thyroidectomy, while the monocyte count was significantly lower than that before thyroidectomy (P = 0.000).
Comparison of Peripheral Blood Lymphocytes, Monocytes and LMR in Patients With PTC Before Thyroidectomy and Radioiodine Therapy.

Abbreviations: L, lymphocytes; M, monocytes; LMR, the ratio of lymphocytes to monocytes.
* Indicates a statistically significant difference.
Discussion
In the 19th century, Rudolf Virchow first proposed a possible link between inflammation and cancer. 21 Later studies 22 -24 have found that inflammation is involved in all aspects of tumorigenesis, especially cell proliferation, angiogenesis, surrounding tissue invasion and distant metastasis. Lymphocytes are the key immune cells in humoral immunity and cellular anti-tumor immune response. 25
Lymphocytes in peripheral blood can accumulate to tumor sites and differentiate into tumor-infiltrating lymphocytes (TIL) under certain stimulation. 26 TILs include T lymphocytes, B lymphocytes, and natural killer cells, of which T lymphocytes account for the majority (about 75%).CD8+T lymphocytes account for the largest proportion of TIL subsets, which can induce tumor cell apoptosis through the interaction between CD95 L molecules (the Fas ligand) and CD95 molecules (the Fas receptors) in the tumor cell surface. At the same time, CD8+T lymphocytes can directly kill tumor cells by secreting perforin and granzyme.CD4+ helper T cells (Th), including Th1, Th2, and regulatory T cells, assist CD8+T lymphocyte-mediated cytotoxicity by secreting cytokines and regulating transcription factors. 24,27,28 Inflammation can trigger the activity of monocytes from bone marrow to peripheral blood, 29 and excess circulating blood monocytes can differentiate into tumor-associated macrophages (TAM) after being recruited into tumor tissue by chemotaxis. 30 Thyroid cancer cells can recruit monocytes from the peripheral circulation to differentiate into TAMs, by releasing chemokine 2 (CCL2). 31 In addition, TAMs itself produce CCL2 and promote monocytes to recruit to the tumor site. 32 TAMs includes M1 and M2 phenotypes, in which M2 macrophages promote tumor progression by inhibiting inflammation, removing fragments, promoting angiogenesis, rebuilding tissue, and repairing damage through specific cytokines. 33 In addition, TAMs have a certain immunosuppressive effect, which can up-regulate the expression of PD-L1, thus forming a local immunosuppressive microenvironment, which is convenient for tumor cell immune escape. 34 The study by Guo et al 35 found that monocytes and the LMR are significantly connected with the number of immunity cells in CRC tissue, and proposed that the number of monocytes and LMR can be used to ascertain the immunological status in the tumor microenvironment. Therefore, the LMR which combined with lymphocyte count and monocyte count can indirectly reflect the immune function of the host.
In recent years, some studies have discussed the value of preoperative LMR in evaluating the prognosis of thyroid cancer, but there are few related studies, and the results are still controversial. Yokota et al 19 have found that preoperative low LMR is a risk factor for poor prognosis of thyroid cancer. Compared with patients with high LMR, patients with low preoperative LMR have a lower overall survival rate and PTC-free survival rate. However, some scholars 36 have found that the increase of LMR is a prognostic factor for advanced TNM, and the logistic regression model shows that LMR ≥ 5.45 is an independent risk factor for advanced TNM. The prognosis of PTC is easily affected by the surgical approach or the physician level of experience. Using preoperative LMR to evaluate the prognosis of PTC may be interfered with by other factors. Therefore, our study evaluated the prognostic value of LMR before radioiodine therapy in patients with PTC. Because most patients with PTC have a good prognosis and low mortality, but local recurrence is more common, so this study takes disease recurrence as the end event, and the factors that predict recurrence are used as an alternative index to predict the death of PTC patients.
The results showed that the recurrence rate of the low LMR group (16%) was significantly higher than that of the high LMR group (5%), and the median recurrence-free survival time of the low LMR group (48 months) was significantly shorter than that of the high LMR group (55 months). Our study found that LMR before radioiodine therapy was negatively correlated with age, multifocality, TNM stage, response to Initial radioiodine therapy and recurrence, and positively correlated with recurrence-free survival time in patients with PTC. Compared with the high LMR group, the PTC patients in the low LMR group were older at diagnosis, and more likely to have multifocal tumors, later clinical stages, and higher risk stratification. These results suggest that patients with lower LMR before radioiodine therapy have higher tumor invasiveness and higher risk of recurrence. A recent paper reported that the pre-RAI LMR below 7.05 was an independent prognostic markers for worse RFS in the enrolled PTC patients. 37 The optimal cut-off value of LMR before radioiodine treatment found in this study is 6.61, which is roughly the same as the previous literature. 37 This suggests that LMR as a hematological marker is reproducible in different populations, and it also strengthens the argument that LMR is a prognostic factor for papillary thyroid cancer. In univariate and multivariate analysis, low LMR was a significant risk factor for recurrence, which was consistent with the conclusion of Yokota et al. 19,20,38 The progression of cancer is related to the impaired function of the immune system. 39 When the number of circulating lymphocytes decreases relatively, it may mean that the number of lymphocytes in the paracancerous tissue decreases, reducing its mediated anti-tumor immune response, thus forming a tumor microenvironment suitable for cancer cell proliferation and metastasis. 40 When the lymphocyte count decreased and / or the monocyte count increased, the peripheral blood LMR decreased, which may indicate that the host’s anti-tumor immune status is insufficient and the tumor burden is increased. Whether ER can be obtained after the first radioiodine treatment is related to the patient’s next treatment and follow-up plan. In this study, the number of ER patients in the low LMR group (76, 60%) was significantly lower than that in the high LMR group (227, 73%), indicating that patients with low LMR had a poor response to initial radioiodine therapy. Therefore, we believe that measuring the levels of LMR before radioactive iodine treatment can both monitor the early response to treatment, but also help restratify patient for management. However, the ROC curve in this study showed that the sensitivity of the prognostic model based on pre-radioiodine treatment LMR to predict postoperative recurrence in PTC patients was 54.1%, the specificity was 73%, and the AUC was 0.628, which indicates that the value of pre-operative LMR alone in predicting PTC recurrence is limited and needs to be applied in combination with risk stratification in clinical practice to improve the predictive efficacy.
Our research has some advantages compared with previous studies. First of all, our sample size is relatively large, which makes the conclusion of this study more accurate. Secondly, we compared the LMR of the same patient before thyroidectomy and before initial radioactive iodine treatment. Because the confounding effect of postoperative complications of which most occur within the first 10 to 14 days post surgery. It has been suggested that the 21 to 56 days postoperative period represents an optimal period for remeasuring the systemic inflammatory response (SIR). 41 The interval between the 2 LMR tests is 42 days (IQR 36-45). Our results showed that the lymphocyte count and LMR before radioiodine treatment were significantly higher than those before thyroidectomy, while the monocyte count was significantly lower than that before thyroidectomy. Another study by Ahn et al 20 investigating the changes in LMR at approximately the 29th day (IQR 16-51.5) after surgery and found that there was no significant difference between the baseline and follow-up LMR. This is different from our findings, indicating the LMR before radioiodine therapy was significantly higher when compared with the postoperative level at the time point. Considering that the presence of cancer may be related to systemic inflammation, the increase in LMR after total thyroidectomy may reflect a significant reduction in tumor burden. This suggests that changes in the levels of LMR derived from the blood may reflect the changes in the patient’s systemic or local inflammatory state to some extent after surgical treatment, which validates the conclusion of this study from another point of view. There is no relevant report about the value of the dynamic changes of LMR in the prognosis of PTC. Our study suggest for the first time that monitoring the dynamic changes of LMR may reflect the efficacy of treatment and predict prognosis.
In addition, this study combined the LMR before radioiodine treatment with risk stratification to predict the recurrence of PTC patients. The results showed that the recurrence-free survival rate and recurrence-free survival time of PTC patients with low LMR and high-risk stratification were significantly lower than those of other groups (P < 0.05), which indicates that the combined application of LMR and risk of recurrence stratification facilitates the early detection of patients with poorer prognosis, thus helping clinicians to develop more individualized treatment and follow-up strategies for this group of patients to improve their prognosis as much as possible. In addition, since all patients undergo hematological testing prior to radioiodine treatment, this test is relatively convenient and inexpensive, and using LMR as a prognostic marker will not impose any additional financial burden on patients. Moreover, the use of LMR before radioactive iodine treatment to evaluate the prognosis is not easily affected by the surgical approach or the physician level of experience, which is more advantageous than preoperative LMR.
However, this study also has some limitations. First, there may be some risk of selection bias in this study due to the limitations of retrospective analysis. Secondly, the prognosis of most patients with PTC is usually good, but the follow-up time in our study is relatively short. By the end of follow-up in this study, some patients who will relapse in the future may not have been monitored, which limits us to make more accurate conclusions on the prognosis of these patients in this study. The exact mechanisms by which these blood-derived markers affect tumor prognosis is unclear, but they are believed to reflect the interaction between local immune response in tumor microenvironment and SIR. The purpose of this study is not intended to provide a clear understanding of why the LMR is prognostic, but to provide a way to better understand the interaction between inflammation and tumor in the future. Thus, we do not believe that this limitation significantly detracts from the current results. In the follow-up study, we intend to reduce the experimental deviation and improve the accuracy of LMR as a prognostic marker of PTC by multicenter, large sample, longer follow-up period, and dynamic monitoring of LMR level.
In conclusion, the prognostic model based on LMR before radioactive iodine treatment is valuable for predicting PTC recurrence and has the advantages of convenience and economy, but its predictive efficacy is limited when applied alone, and it can be used as a supplement to risk stratification in clinical practice and applied in combination to help screen out patients with poorer prognosis early. It is hoped that the results of this study will provide new ideas for early clinical prediction of PTC recurrence in order to improve the prognosis of thyroid cancer and reduce recurrence.
Footnotes
Authors’ Note
This retrospective study was approved by Ethics Committee of clinical scientific research of the First Affiliated Hospital of Chongqing Medical University, and the requirement for written consent was waived. All authors have confirmed its accuracy. The Ethics Committee of the First Affiliated Hospital of Chongqing Medical University has reviewed the study protocol and informed consent form submitted for this project, which are in accordance with the requirements of the Declaration of Helsinki, and the study design is scientifically based and does not pose unnecessary risks to the subjects, and the safety and privacy of the subjects are protected to the maximum extent. (IRB NO. 2021-065).
Acknowledgments
Zhou Chunyan: conception and design, writing, review of the manuscript, and was a major contributor in writing the manuscript. Duan Dong: writing and revision of the manuscript. Liu Shuang: analysis and interpretation of data and took part in drafting the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Natural Science Foundation Project Committee of Chongqing Science and Technology Bureau. Grant (cstc2019jcyj-msxmX0327).
