Abstract
Objective:
Systematic biopsy plays a vital role in diagnosing prostate cancer, but it can lead to misdiagnoses or undertreatment. Advances in magnetic resonance imaging (MRI) and its guided targeting technology provide the possibility of improving the use of biopsies. This study aimed to evaluate the performance of MRI screening and MRI/ultrasound (MRI/US) fusion-guided transperineal biopsy in the detection of prostate cancer.
Methods:
We performed a retrospective study on patients with suspected prostate cancer in the Kunshan Hospital Affiliated with Jiangsu University from January, 2017, to December, 2019. All of the patients underwent MRI examinations, followed by a systematic biopsy (either alone or in combination with MRI/US fusion-guided targeted biopsy, based on MRI-visible lesions). We evaluated the diagnostic accuracy of MRI screening and compared biopsy methods by considering sensitivity, specificity, and area under the curve (AUC) values.
Results:
A total of 157 patients were enrolled, including 112 patients with MRI-visible lesions and 45 patients without MRI-visible lesions. The cancer detection rate (CDR) was higher in patients with MRI-visible lesions (P < 0.001); however, the serum prostate-specific antigen (PSA) indicators were similar (P > 0.05). The AUC of MRI was 0.63, which was superior to the AUC values of ultrasound (AUC = 0.55, P = 0.031) and digital rectal examination (AUC = 0.52, P = 0.041) for screening prostate cancer. Both overall CDR and clinically significant prostate cancer detection rates were improved if we combined systematic biopsy and MRI/US fusion-guided targeted biopsy procedures.
Conclusion:
Overall, prior MRI screening may serve as a classifier for avoiding the overuse of biopsies. A combination of systematic and MRI/US fusion-guided targeted biopsy procedures offers an optimal regimen for detecting prostate cancer.
Keywords
Introduction
Prostate cancer remains the second most common cancer and ranks as the fifth leading worldwide cause of cancer-related deaths among males, with approximately 1.3 million new cases and 360,000 deaths occurring every year. 1 To date, multiple risk factors have been discovered, including age, obesity, ethnic predispositions, and genetic predispositions, 2,3 but the etiology remains unclear. Although approximately 80% of patients are diagnosed with localized prostate cancer, over 30% of them will undergo postoperative recurrence in the following 5-10 years. 4 When compared with the >98% 5-year survival rate in the United States, the 5-year survival rate of prostate cancer in China is less than 60%, which is mainly due to delayed diagnoses. 5 Currently, the serum prostate-specific antigen (PSA) test and digital rectal examination (DRE) are the main methods for screening prostate cancer. 6 Specifically, males with elevated PSA or abnormal DRE results would undergo transrectal or transperineal ultrasound-guided biopsy procedures. However, there is a great controversy considering overdiagnoses and overtreatment, due to the low specificities of these procedures. 7 Therefore, it is necessary to optimize the screening and diagnosis processes for prostate cancer.
Standard systematic 6-core transrectal ultrasound biopsy (TR) is commonly used for preoperative diagnoses 8 ; however, over 30% of patients are misdiagnosed with prostate cancer. 9 An increasing number of studies have been conducted to improve its accuracy by sampling 12, 20, or >20 cores; however, this has resulted in more cases lacking clinical significance and increased risks of bleeding, infection, and other complications. 10,11 The 6-core systematic biopsy is still recommended for patients with PSA >20 ng/ml. 12 Recently, transperineal systematic prostate biopsy (TP) has attracted much more attention. TP shows a significantly higher cancer detection rate (CDR) among patients with persistently elevated PSA levels, as well as abnormal DRE and previous negative TR results 13,14 ; in addition, it also results in fewer complications. TP is faster and the patient doesn’t need to be hospitalized. TP-targeted biopsy with electromagnetic tracking was found to be comparable to the TR fusion-guided approach in the detection of prostate cancer, whereas Wajswol et al found that TP demonstrated more accuracy for detecting lesions in the anterior prostate, compared to transrectal biopsy. 15,16
Imaging examinations help to improve the accuracy of diagnosis and to reduce unnecessary biopsy procedures. The procedure mainly relies on the use of ultrasound (US), but it has been criticized for its low sensitivity. Magnetic resonance imaging (MRI) can provide descriptions of tissue anatomy and tissue characteristics; thus, it is regarded as a prospective technique to improve the diagnostic ability of prostate cancer. 17 Recently, the US/MRI fusion technique has been introduced into clinical practice to improve the diagnostic accuracy of targeted biopsy. This technique may reduce unnecessary biopsies among patients with a clinical suspicion of prostate cancer when performing the 4 K score test in conjunction with MRI. 18 Previous studies have suggested that the combination of prostate-specific antigen density and MRI findings allows for the avoidance of unnecessary prostate biopsy. 19 Although the accuracy rates of MRI use in staging prostate cancer were comparable in African American and Caucasian American patients, 20 it should be noted that the assessment of inter-reader variability, in combination with the prediction of clinically significant prostate cancer and the cancer detection rate, is a pivotal factor for performing MRI-based screening. However, it has not been systematically evaluated and widely applied for prostate cancer diagnosis in China, especially for hospitals in different economic locations.
In the current study, we retrospectively collected clinical data in a Chinese population and compared the diagnostic accuracy of MRI imaging screening and MRI/US fusion-guided transperineal targeted biopsy in prostate cancer.
Materials and Methods
Ethics Statement
This study was approved by the Ethics Committee of the First People’s Hospital of Kunshan (IEC-C-012-A03-V2.0). Informed consent was obtained from all of the participants. This study was conducted in accordance with the Declaration of Helsinki.
Study Population
We enrolled patients who were suspected of having prostate cancer in the Kunshan Hospital Affiliated with Jiangsu University from January, 2017, to December, 2019. They received an MRI test and underwent transperineal prostate biopsy procedures. The inclusion criteria for biopsy referred to the “2011 edition of Chinese Guidelines for the Diagnosis and Treatment of Urological Diseases.” Patients meeting the following criteria underwent 6+X systematic biopsy and transrectal ultrasonography (TRUS)-guided biopsy for suspicious nodules: 1) DRE revealing the presence of nodules; 2) hypoechoic or isoechoic nodules of the prostate being detected with ultrasound examination; 3) PSA of >10 ng/ml; and 4) PSA of 4-10 ng/ml, along with an abnormal ratio of free/total PSA or abnormal PSA density (PSAD). For patients with PSA levels of 4-10 ng/ml and a normal ratio of free/total PSA and PSAD, we measured the prostate volume via ultrasound and compared the difference between the actual value and the predicted value of PSA. The predicted PSA = prostatic volume*0.12 (prostatic volume = L*W*H*0.523). For patients with elevated PSA, we first performed a systematic biopsy, followed by the use of MRI/US fusion-guided targeted biopsy. For elderly patients who could not tolerate systemic biopsy, only suspicious lesions detected via TRUS or MRI were punctured.
MRI Examination Protocol
For patients with PSA levels greater than 10 ng/ml, MRI examination is recommended, regardless of whether lesions are indicated by digital rectal examination and rectal ultrasound examination. For cases in which there are PSA levels of 4-10 ng/ml and a ratio of free/total PSA less than 0.16, MRI examination is similarly recommended. Before the biopsy, patients received an MRI examination using a 1.5-T or 3.0-T scanner with a pelvic phased-array coil, T2-weighted, dynamic contrast-enhanced, diffusion-weighted imaging, and MR spectroscopy. 21,22 Two experienced radiologists carefully read the MRI and interpreted the results. Moreover, MRI/US cognitive fusion was applied to determine the targeted sites before introducing fusion navigation software.
Prostate Biopsy Procedure
Patients underwent transperineal prostate biopsy by 2 experienced urologists in an extended lithotomy position and received a periprostatic block with 2% lidocaine. MRI/US fusion-guided targeted biopsy was first performed, followed by systematic biopsy. Generally, we performed 2 cores per MRI lesion. For the systematic biopsy, the specific cores, which were simultaneously adjusted to the volume of the prostate, were obtained from the base, middle, and apex portions of the prostate under transrectal ultrasound guidance and were individually processed. Targeted biopsy was performed on suspicious lesions that were indicated by MRI/US fusion imaging in patients with MRI-visible lesions (Figure 1). Biopsy specimens were maintained in a 10% formaldehyde solution for histopathological examination.

Visualization of the performance of MRI/US fusion targeted biopsy. Patients with MRI-visible lesions underwent targeted biopsy guided by MRI/US fusion imaging. MRI/US indicates magnetic resonance imaging/ultrasound.
Histopathology
Biopsy specimens were reviewed by at least 2 experienced uropathologists who were blinded to the biopsy procedures. The Gleason score of suspicious lesions was evaluated according to the International Society of Urological Pathology (ISUP) 2005 guidelines. Specifically, lesions were defined as indicating clinically significant prostate cancer (csPCa) if the Gleason score ≥3 + 4 and indicating low-risk prostate cancer if the Gleason score ≤3 + 3.
Statistical Analysis
We applied chi-square or Fisher exact tests to compare the CDR results between the different methods. The nonparametric rank-sum test was used to compare the continuous data between the groups. The receiver operating characteristic (ROC) curve was plotted, and the sensitivity, specificity, and area under the curve (AUC) values were obtained to assess the diagnostic values of the different methods. Statistical analyzes were performed with Stata 15.0 (College Station, TX, USA). A P-value less than 0.05 was considered to be statistically significant.
Results
Patient Characteristics and MRI Findings
A total of 288 patients who were initially treated for prostate diseases were consecutively enrolled. After excluding 131 patients who lacked detailed MRI, histopathology, and biopsy results, or who refused to participate in the study, 157 subjects were finally involved in the analysis (Figure 2). Among them, 112 (71.34%) patients had positive MRI-visible lesions and received both systematic and MRI/US fusion-guided targeted biopsy, and 45 (28.66%) patients with negative MRI-visible lesions underwent only systematic biopsy. A total of 1,387 system biopsies and 458 targeted biopsies guided by MRI/US fusion-guided targeted biopsies were performed.
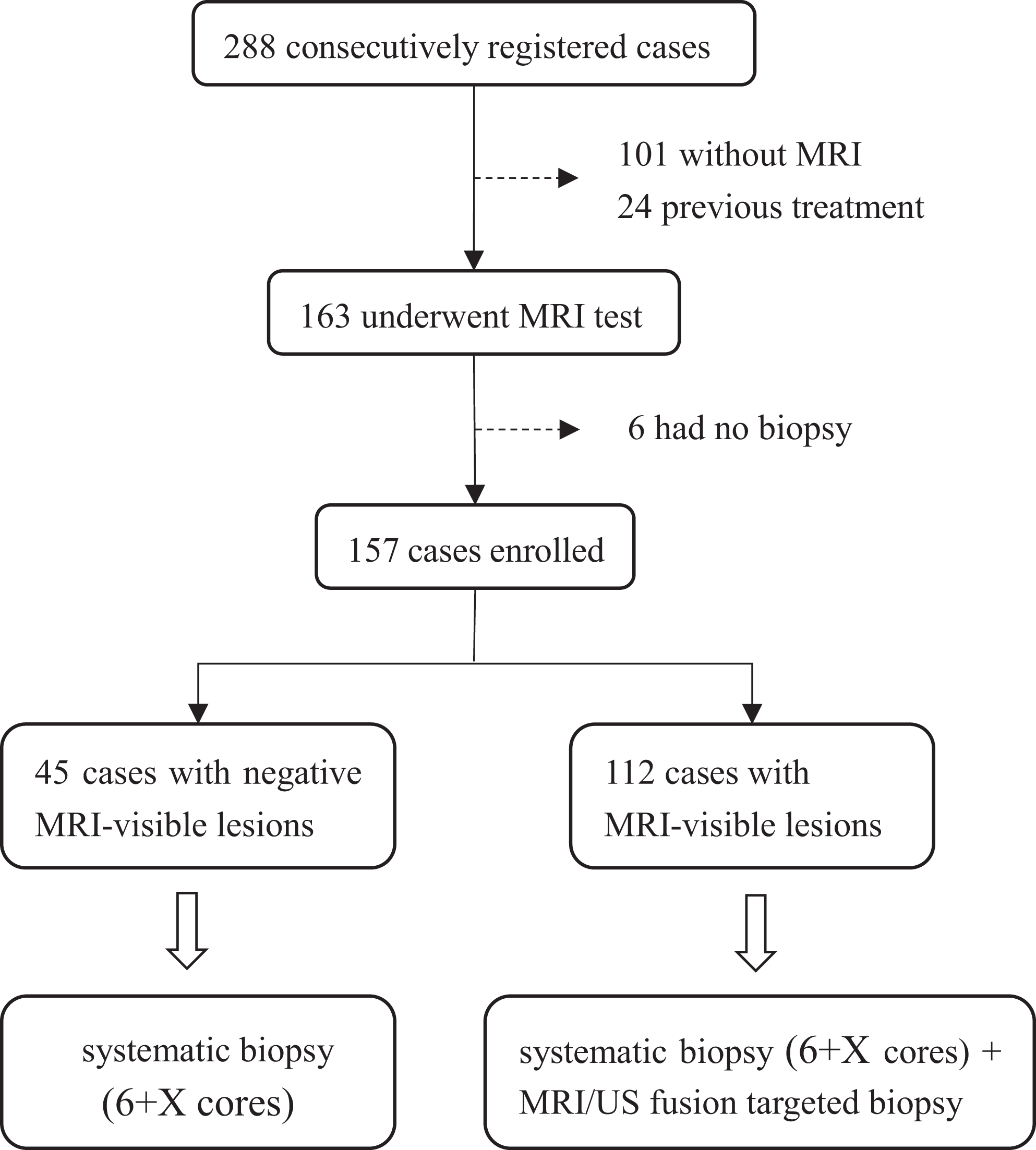
Flowchart of enrollment of study subjects. MRI indicates magnetic resonance imaging.
For patients with MRI-visible lesions, a total of 19 patients were diagnosed with a Gleason score less than 6, and 56 patients had a score greater than 6. Among them, the overall CDR was 57.14% (64/112 patients), and the clinically significant cancer detection rate (csCDR) was 43.75% (49/112 patients), which were significantly higher than those in patients without MRI-visible lesions (overall CDR: 8.89%, P = 0.0002; csCDR: 15.56%, P = 0.0009). As shown in Table 1, patients with or without MRI-visible lesions had similar levels of PSA indicators (PSA: P = 0.134; free PSA: P = 0.281; free/total PSA: P = 0.274).
Characteristics of Patients Enrolled in This Study.

Abbreviations: PSA, prostate-specific antigen; MRI, magnetic resonance imaging; DRE, digital rectal examination; US, ultrasound.
Screening for csPCa Using MRI, US, and DRE
To estimate the diagnostic accuracies of MRI, US, and DRE for csPCa, we examined their sensitivities, specificities, and AUC values. Overall, 157 patients simultaneously underwent MRI, US, and DRE. Of these, 112 (71.79%) patients had MRI-visible lesions, 113 (72.43%) patients had positive DRE results, and 147 (94.23%) patients had abnormal indicators under US. The sensitivities of DRE, MRI, and US for the diagnosis of csPCa were 75.00% (95% CI: 61.6%-85.6%), 87.5% (95% CI: 75.9%-94.8%), and 100% (95% CI: 93.6%-100.0%), respectively. The specificities of DRE, MRI, and US for the diagnosis of csPCa were 29.7% (95% CI: 21.0%-39.6%), 37.62% (95% CI: 28.2%-47.8%), and 9.90% (95% CI: 4.9%-17.5%), respectively. The AUCs of DRE, MRI, and US for the diagnosis of csPCa were 0.52 (95% CI: 0.44-0.60), 0.63 (95% CI: 0.55-0.70), and 0.55 (95% CI: 0.49-0.63), respectively (Table 2). The overall diagnostic value of MRI was superior to that of US (P = 0.031) and DRE (P = 0.041).
Comparison of the Diagnostic Accuracy of MRI, US, and DRE for csPCa.

Abbreviations: csPCa, clinically significant prostate cancer; MRI, magnetic resonance imaging; DRE, digital rectal examination; US, ultrasound; +LR, positive likelihood ratio; −LR, negative likelihood ratio; AUC, area under the curve.
Comparison of CDR via Different Biopsy Methods
To assess the roles of different biopsy methods in the diagnosis of prostate cancer, we compared the accuracies of MRI/US fusion-guided biopsy and systematic biopsy alone against the combination of the 2 methods. Briefly, the overall CDR for MRI/US fusion-guided targeted biopsy was 50.00% (56/112), which was slightly higher than that of systematic biopsy (37.50%, 42/112) (P = 0.059). Moreover, the number of cores in MRI/US fusion-guided targeted biopsy was lower than that in systematic biopsy (271 vs. 942 cores). The addition of MRI/US fusion-guided targeted biopsy to systematic biopsy significantly improved the prostate cancer detection rate from 37.50% to 57.14% (P = 0.003). Among patients with csPCa, the csCDR varied from 39.29% in MRI/US fusion-guided targeted biopsy alone to 25.00% in systematic biopsy alone (P = 0.022) and to 47.35% in the combination of these 2 methods (P = 0.003).
The concordance detection rate of csPCa was 76.79% (86/112) between MRI/US fusion-guided targeted biopsy and systematic biopsy. Of these, 23 (20.54%) cases were diagnosed with csPCa, and 63 (56.25%) cases were diagnosed without csPCa by both methods. Furthermore, 5 patients (4.46%) had csPCa diagnosed via a systematic biopsy that was missed by MRI/US fusion-guided targeted biopsy. In contrast, 21 (18.75%) csPCa cases that were detected by MRI/US fusion-guided targeted biopsy were missed by systematic biopsy. Moreover, the addition of MRI/US fusion-guided targeted biopsy to systematic biopsy resulted in 1 more case with low-risk cancer (Figure 3).

Comparison of cancer detection rates by different biopsy methods in patients with MRI-visible lesions. csPCa indicates clinically significant prostate cancer; MRI/US, magnetic resonance imaging/ultrasound.
Discussion
The ideal diagnostic methods for prostate cancer should be noninvasive, have fewer complications and have a high differentiation ability. However, currently available tools do not adequately and completely meet the previously mentioned requirements, of which the most noteworthy are misdiagnosis and undertreatment. It is imperative to identify optimal screening strategies to identify high-risk populations and to improve the diagnostic accuracy for subsequent targeted treatment. In the present study, we explored the efficiency of MRI screening and MRI/US fusion-guided transperineal biopsy in detecting prostate cancer in a Chinese population. We observed that prior MRI screening may serve as a classifier to avoid the overuse of biopsy. The addition of MRI/US fusion-guided targeted biopsy to systematic biopsy may also significantly improve the detection rate for prostate cancer.
PSA, US, and DRE have been routinely applied to screen for prostate cancer, but the accuracy is not satisfactory. 23,24 Advances in MRI techniques provide novel insights into cancer screening, especially for those with clinical significance. 25 Ouzzane et al reported that targeted biopsies on MRI-detected lesions can improve the detection rate, volume, and grade of anterior prostate cancer, compared with 12-core systematic biopsy. 26 Consistent with other studies, 27 -29 we observed that patients with MRI-visible lesions are more likely to have prostate cancer, including csPCa. Additionally, we observed that the AUC value of MRI screening for csPCa was superior to either US alone or DRE alone. Some studies have reported that PSA density is different in patients with and without MRI-visible lesions. 29 According to Perez et al, the qualitative improved prostate cancer diagnosis—combination of magnetic resonance imaging and biomarkers (IMPROD) rapid biparametric MRI (bpMRI) presented a higher accuracy for predicting clinically significant cancer in males with an elevated PSA. 30 In our study, when considering the 15.56% csCDR rate in patients with negative MRI-visible lesions, it is of significance to construct a predictive model for identifying prostate cancer via MRI in combination with PSAD or other indicators.
Prostate cancer is a multifocal disease that requires multiple site sampling for pathological examination. Pokorny et al have reported that MRI-guided targeted biopsy can significantly increase the detection rate of high-risk prostate cancer. 25 Additionally, Stabile et al found that MRI-targeted biopsy is more efficient in detecting csPCa in males with a positive MRI result. A significant minority of men will have csPCa detected by systematic biopsy in cases with negative MRI results. 31 A previous meta-analysis indicated that MRI-guided targeted biopsy improved the CDR among males with a clinical suspicion of prostate cancer along with previous negative biopsy results. 32 Optimistically, MRI may offer superior anatomic details and have the ability to access cellular density based on water diffusion and blood flow (based on contrast enhancement). Currently, the most common protocols include biparametric prostate MRI (bpMRI) and multiparametric MRI (mpMRI). Of which, bpMRI are most promising since they could make acquisition time much shorter and cost-effective. 33 MRI-guided targeted biopsy may increase the diagnostic ability of clinically significant cancers by identifying specific lesions that are not visible on conventional ultrasound. 34 Although an increasing number of studies have shown that MRI-guided targeted biopsy is better than systematic biopsy in diagnosing prostate cancer, less evidence has indicated as to whether it can completely replace the latter technique. Jiang et al. reported that MRI-guided biopsy was not significantly superior to systematic biopsy, but that the combination of these 2 methods can improve the detection rate of prostate cancer. 35 It should be noted that the controversy concerning whether MRI-guided targeted biopsy is better than systematic biopsy may be primarily due to the difference in the fusion methods or a lack of a whole-mount histopathological reference. 15,29,36 In our study, we applied targeting software (rather than cognitive MRI/US), which significantly improved the positioning accuracy. Furthermore, our data indicated that the combination of systematic and MRI-guided targeted biopsy may substantially improve the CDR of prostate cancer, which was consistent with previous studies. 37
Several limitations should be noted. First, the current study was designed as a retrospective study in 1 hospital, which limited its generalizability. Prospective and multicenter designs can provide more convincing evidence. Second, we did not perform an economic evaluation of the different biopsy methods. Cost-effectiveness is an important issue that will have to be evaluated in the future.
Conclusions
Our findings suggested that prior MRI screening may serve as a classifier to avoid the overuse of biopsy. A combination of systematic and MRI/US fusion-guided targeted biopsy offers an optimal regimen for detecting prostate cancer.
Footnotes
Authors’ Note
Hongqing Yin, Jun Shao, and Huan Song contributed equally to this work. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work. All data generated or analyzed during this study are included in this published article. Informed consent was obtained from all participants. This study was approved by the Ethics Committee of the First People’s Hospital of Kunshan (IEC-C-012-A03-V2.0). All procedures involving human participants were in accordance with the Ethical standards. This study was conducted in accordance with the Declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Kunshan Social Development Science and Technology Project (KS1721), National Natural Science Foundation of China (81673249), and National Key R&D Program of China (2017YFC0907000). The funding agencies had no role in the study design, data collection, analysis, decision to publish, or preparation of the manuscript.
