Abstract
Prostate cancer is the second leading cause of cancer death in men. Its current treatment includes various physical and chemical approaches for the localized and advanced prostate cancer [e.g. metastatic castrate resistant prostate cancer (mCRPC)]. Although many new drugs are now available for prostate cancer, none is suitable for local treatment that can reduce adverse effects often associated with the current physical treatment. Of the drugs approved by FDA for mCRPC, the best mean improvement in overall survival is only about 4.8 months. Therefore, there is a need for improved treatment approaches for prostate cancer, especially drug-resistant cancer.
Ultrasound therapy represents a useful new physical approach for the drug-resistant cancer treatment by facilitating the entry of the related chemotherapy drug into the target cancer cells. There are two versions of ultrasound: High Intensity Focused Ultrasound (HIFU) and Low Intensity Pulsed Ultrasound (LIPUS). HIFU has been a promising treatment option for prostate cancer due to its noninvasiveness and various biological effects on cancer tissue. It has been approved for the treatment of cancer and in recent years there have been numerous findings suggesting HIFU can reduce cancer cell viability and possibly reverse the spread of cancerous tumors. LIPUS is currently being studied as an alternative treatment option for prostate cancer. Preliminary studies have found LIPUS to reduce cancer cell viability without the side effects seen in HIFU. Reversible cell membrane damage caused by LIPUS could allow increased uptake of anticancer drugs, enhancing cytotoxicity and death of cancer cells. In this way, a low dose of anticancer drug is more effective toward cancer cells while there is less damage to normal cells. The combination of LIPUS with certain chemotherapeutic agents can be an exciting physical-chemical combination therapy for prostate cancer. This review will focus on this topic as well as the clinical use of HIFU to provide an understanding of their current use and future potential role for prostate cancer therapy.
Introduction
Since the discovery of prostate cancer by Dr. J. Adams in 1853 as “a very rare disease”, prostate cancer is now the most common diagnosed cancer in men, with over 174,000 new cases in 2019 and is the second leading cause of cancer death of men in the United States. 1 -3 Currently, there is a wide variety of treatment options for prostate cancer depending on its severity. For low or intermediate risk prostate cancer, patients can be treated with options such as active surveillance, minimally invasive ablative therapies, radiation therapy, or prostatectomy. 4 For localized cancer the current recommended options are radiation therapy, brachytherapy or prostatectomy. 1,2 As the disease progresses with metastases, chemotherapy is always a final option if it can be tolerated by the patient. However, recurrences from chemotherapy are common. Resistance to chemotherapy is the reason for 90% of drug failures in patients with metastatic cancer. 5 Of the 6 drugs approved by FDA for the treatment of metastatic resistant prostate cancer (mCRPC), the best mean improvement in overall survival is only 4.8 months, most likely due to resistance development. 6 In addition, chemotherapy often induces various severe side effects. Therefore, there is a need for new therapeutic approaches that can not only enhance efficacy but also reduce side effects.
Over the past decades, many new therapeutic approaches have been developed. These include chemical entities (currently there are 34 drugs approved by FDA for prostate cancer), physical entities (radiation, ultrasound) and their combinations. While there are many examples of chemical-chemical combinations 1,2 physical-chemical combinations are less common. One such example is radiation in combination with androgen-ablative therapy. This combination is well known for improving time of relapse and survival. 1 As of to date, there is no established ultrasound-drug combination to reach clinical trial stage, although the benefit of such combination can be considerable.
The present article will review the use of ultrasound for the treatment of prostate cancer, especially pertaining to its combination with anticancer drugs for potential treatment of prostate cancer. Since ultrasound can induce diverse effects to human cells (from harmless to lethal effect), it can offer a unique opportunity to optimize therapy when used in combination with chemotherapeutic agents.
General Properties of Ultrasound
Ultrasound waves refer to wave frequencies beyond the range the human ear can hear (>20 kHz). 7 The ultrasound vibrations can cause a transfer of energy along a directed path and can propagate through the body, oscillating at varying pressures depending on the delivery of the ultrasound. 8 There are usually three characteristics about ultrasound waves: frequency, wavelength, and velocity. 9 Frequency is the number of times a particle experiencing a complete compression and rarefaction cycle in one second. Therapeutic ultrasound usually uses frequency ranges between 1 and 3 MHz and medical imaging applications use 1-20 MHz frequency ranges (Figure 1A). Ultra-high frequency ultrasonic transducers can also be used for research applications such as cellular stimulation and manipulation. 10 Wavelength is the distance between two equivalent points on the waveform in a particular medium (Figure 1B). Depending on applications, appropriate pulse parameters are required. PRT (PRF) represents pulse repetition time. The duty factor is the fraction of time that an ultrasound pulse is actually being produced. The amplitude (Vp-p) of a sound wave is the maximum distance moved by a point on a wave measured from its equilibrium position and determines its intensity. Its intensity is an important parameter delivered from the ultrasound machine which is defined as the concentration of energy within the beam with units of W/cm2. The velocity of ultrasound is approximately 1500 m/sec in water and it can travel more rapidly in a denser medium.

Sound waves and ultrasound parameters. (A) In ultrasound (US) field, different frequencies are used in water or different biological targets for different purposes. (B) Depending on applications, appropriate US pulse parameters are required. (C) In focused ultrasound (FUS), the stimulation parameters are easily adjusted by using a function generator and the focuses by an oscilloscope.
Focused ultrasound (FUS) produces a focused beam of acoustic energy that precisely and accurately reaches large targets in the body without damaging surrounding normal cells. 11 The FUS stimulation parameters are easily adjusted by using a function generator (Figure 1C). The focal distance from the transducer to the surface of culture dish is aligned using a pulser-receiver (oscilloscope). FUS technology is approved for prostate cancer (see below) but also has been proposed for use in other cancer therapy. 12,13
In this respect, there are two general types of ultrasound intensities used for therapeutic purposes: High Intensity Focused Ultrasound (HIFU) and Low Intensity Pulsed Ultrasound (LIPUS). High-intensity (>5 W/cm2) continuous FUS generates a systemic immune stimulatory effect resulting in tumor ablation. 14 Pulsed FUS (i.e., non-continuous stimulus to minimize heat generation) 15,16 may induce a more refined cellular/molecular immune response 17 by initiating inflammatory responses which boost cancer immunotherapy. 18 HIFU can lead to inertial cavitation of gas bubbles leading to cell death. 19 This inertial cavitation can in turn lead to increased temperatures within cells. 20 On the other hand, LIPUS causes stable cavitation without increasing temperature.
HIFU
HIFU was first approved by FDA in 2015. According to the recent NCCN Guidelines, ultrasound is recommended as a local therapeutic option for prostate cancer if radiation therapy fails.2 HIFU is usually defined as delivering ultrasound waves with an intensity of greater than 5 W/cm2. 21 HIFU can generates tumor ablation through a systemic immune stimulatory effect. 14 The main mechanisms of HIFU ablation involve mechanical and thermal effects. 22 The thermal effect of HIFU is heat generation due to absorption of the acoustic energy with a rapid elevation of temperature in the local tissue. The local tissue temperature could be elevated to higher than 60°C by the thermal effect, causing tumor cell destruction via coagulation necrosis. The thermal effect is the major source from thermal ablation therapy using HIFU. In fact, HIFU with intensities in the range of 100 – 10,000 W/cm2 has been utilized clinically to destroy tumor cells. 21 A spherical shaped transducer is thus used to focus the heat of the ultrasound on specific targeted cells. Many oncology centers use ultrasound with transducers between 0.8 and 3.5 MHz to not only treat prostate tumors, but also liver, kidney, or breast cancers because ultrasound therapy with frequencies between 0.8-3.5 MHz is more effective than standard diagnostic US for cancer treatment. 23
Mechanical effects induced by HIFU are associated with acoustic pulses only at high intensities, including cavitation. 22 Cavitation is defined as the physical forces of the sound waves on microenvironmental gases within fluid. As the sound waves propagate through the medium, the characteristic compression and rarefaction causes microscopic gas bubbles in the tissue fluid to contract and expand. It is generally thought that the rapid changes in pressure, both in and around the cell, may cause damage to the cell. There are two forms of cavitation: stable and inertial cavitation. Stable cavitation is the stable oscillation of the size of the bubble when exposed to a low-pressure acoustic field, and may play a role in US-enhanced drug and gene delivery. In contrast, inertial cavitation is violent oscillations of the bubble and rapid growth of the bubble during the rarefaction phase when they reach their size of resonance, eventually leading to the violent collapse and destruction of the bubble. The violent collapse will produce shock waves that produce free radicals and a cascade of molecular events and, in turn, damage the cancer cells. The mechanical effects can be explained by a concept of inertial cavitation. The heat generated by HIFU results in compression and rarefaction which then leads to formation of bubbles within the tissues. As the tissues are constantly heated and pressured by the HIFU, the bubbles are oscillating and expanding until reaching a size that would collapse asymmetrically, leading to tissue damage and cell death. However, it is less predictable and not as significant as the thermal effect, yet it can also play a role in damaging the targeted tissues.
Common devices used for HIFU are Sonablate and Ablatherm. 24 Sonablate has a 4 MHz transducer with focal depth of 40 mm. Ablatherm has a 3 MHz transducer with adjustable focal length (19-26 mm). Both transducers can generate a focused ultrasound field that creates heat and cavitation which destroy the cancer tissues with a spindle-shaped elementary lesion of about 1.7 mm in diameter. The maximum depth that HIFU can penetrate prostatic tissues is about 30 mm. 25 The therapeutic benefit of HIFU using Sonablate or Ablatherm in patients with prostate cancer is well established. This includes 61.2% to 95% 5-year disease-free survival rate for Sonablate and 69% to 84% 7-8-year disease-free survival rate for Ablatherm (See Table 1). However, HIFU can induce impotence (38.8% to 55.1%) and urinary incontinence (6.1% - 12.2%). 28
HIFU for Prostate Cancer.
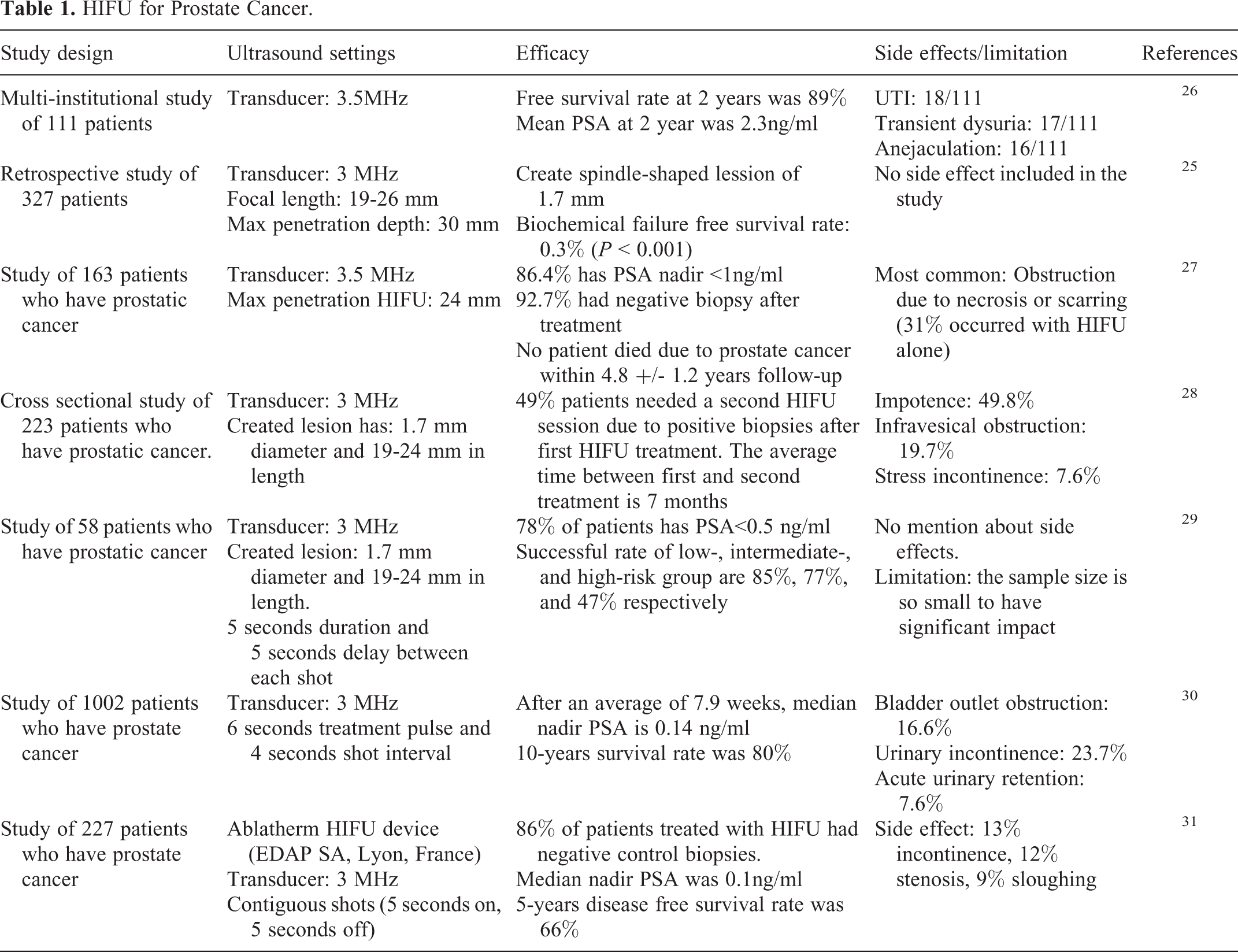
The benefit of combination of HIFU and anticancer drugs for prostate cancer is unknown. A previous study showed that the anticancer activity of anticancer drugs can be decreased by HIFU exposure when tested in human ovarian cancer cells. 32 Since HIFU, when combined with anticancer drugs is known for inducing a lethal effect rather than a therapeutic effect to a defined area, its main use is for local ablation therapy and not for metastatic cancer.
LIPUS
LIPUS is defined as ultrasound applied at an intensity less than 3W/cm2 and the energy is released at a pulsed rate. The pulsed rate, commonly referred to as duty factor or duty cycle is usually set at 1:1 (50%) or 1:4 (20%) (the amount of time the energy is released versus being off in one second). In terms of penetration, 1 MHz frequency typically reaches tissue depths of 2.3-5 cm, while a 3 MHz frequency reaches depths of 0.8 -1.6 cm. 33
Due to the low intensity from LIPUS, the thermal effect is minimal as opposed to HIFU. Most documented effects are mechanical (or nonthermal) cellular changes. Vibrations of cellular components due to ultrasound are known to cause cavitation within cells. LIPUS can cause stable cavitation resulting from the formation of gas bubbles which will take roughly 1000 cycles to reach their maximum size. Another mechanical effect is acoustic streaming that provides a driving force capable of displacing ions and small molecules. This mechanical pressure applied by the wave produces unidirectional movement of fluid along and around cell membranes which is known to affect diffusion rates and membrane permeability. 34 Through cavitation, implosion of gas bubbles can create microjets of fluid, so possibly drugs can then enter targeted cells.35 Furthermore, non-collapsing bubbles near target cells may alter cellular membranes which can increase the influx of drugs into cells. 36
LIPUS can be further utilized to combine with microbubbles to enhance the drug effect. Microbubbles are microspheres in the range of 1-8 µm size consisting a gas core stabilized by a surrounding shell made of phospholipids or other polymers. Drugs, antibodies or other proteins can be attached to the surface of shells, encased inside the shells, or embedded in the shell membrane. 37 – 39 Injection of such microbubbles and upon exposure to ultrasound can cause cavitation (shrinking and expanding to a size that can lead to rupture) which can generate shear stress and cell membrane permeability. Such effect can allow drugs or proteins to enter the cell and improve anticancer effect.
LIPUS has been investigated as a possible prostate cancer treatment option either alone or in combination with microbubbles [See Table 2]. Hou et al utilized LIPUS with microbubbles in order to induce microvessel damage in tumors leading to cell necrosis or apoptosis. 40 Their results indicated that with LIPUS treatment four times per day, cell proliferation was inhibited, and apoptosis was promoted. However, when treated only once per day, they observed opposite effects indicating further research is needed to clearly define the ultrasound effects. Yang et al reported the effect of LIPUS plus microbubbles on microvessel disruption in prostate cancer. 41,42 They concluded that the optimal ultrasound parameters are 20 kHz frequency, 1 W/cm2 intensity, 40% duty cycle and 3-minute duration to induce tumor ablation in mice implanted with PC3 cancer cells.
LIPUS Alone or in Combination With Drugs on Prostate Cancer.

Abbreviations: USMB, ultrasound with microbubble; ER, endoplasmic reticulum; RFA, radiofrequency ablation.
The use of LIPUS in combination with anticancer drugs has also been explored (Figure 2A, B). Anti-cancer drugs can inhibit various pathways known to regulate cancer proliferation and survival. Several classes of drugs/treatments have been developed to interfere with oncogenes/oncoproteins, androgen receptor signaling, bone metastasis, and immune response known to be involved in the progression of prostate cancer. 47,48 There are now 36 drugs by FDA as of January 14, 2021. 49 (Due to this extensive list of drugs, we like to refer those interested in therapeutic use of drugs for prostate cancer to the website and individual product information.) As is often the case with chemotherapy, drug resistance can occur with continued therapy, 50 so improved methods to treat drug-resistant cancers are needed. The use of LIPUS in combination with anticancer drugs may offer a new treatment for drug resistant cancer (Table 2) (Figure 2A and B).
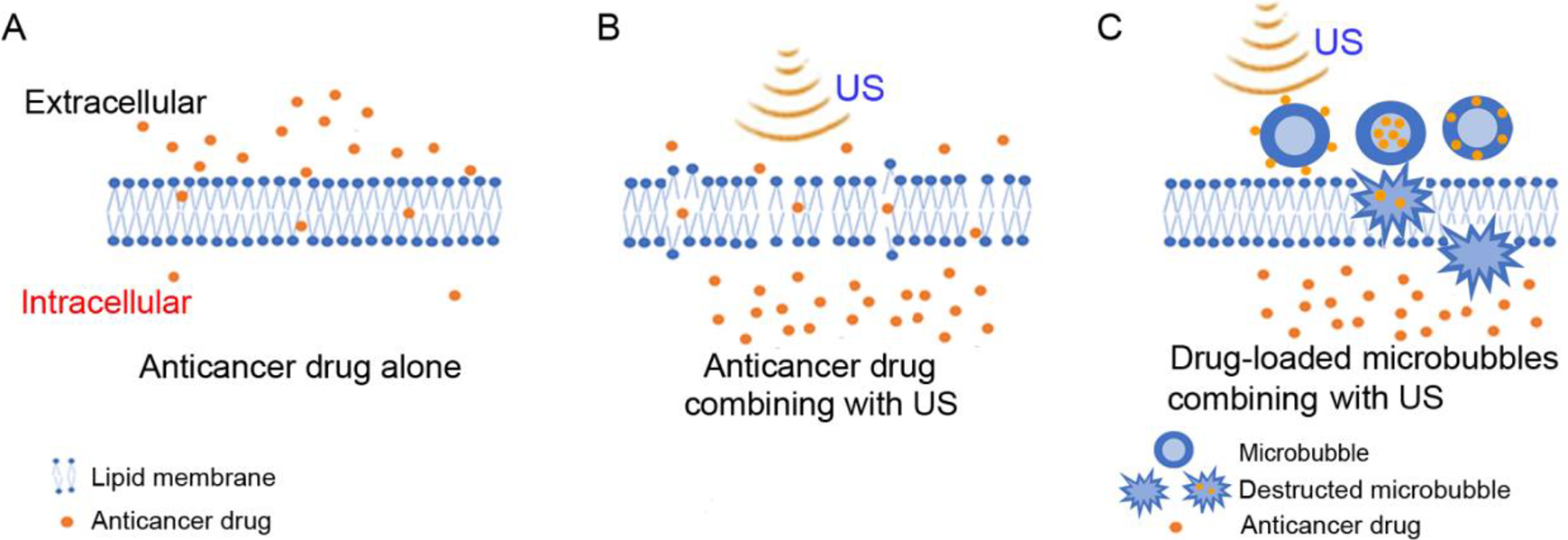
Effect of membrane poration with ultrasound (US) on enhancement of anticancer drug delivery to cells. (A) The ability of the drug alone to cross the lipid membrane. (B) The proposed enhanced ability of the anticancer drug to cross the lipid membrane in combination with the use of US. (C) The anticancer drug-loaded microbubbles combining with US in the treatment of tumors. Anticancer drugs are attached to the surface of the microbubbles, encased inside the microbubbles, or embedded in the microbubble membrane. 37 -39 After ultrasound irradiation, microbubbles are destructed in tumor cells and drugs carried by microbubbles are released into tumor cells.
Yang et al studied the ultrasound combined with microbubbles (USMB) in further combination with docetaxel. 43 They found that the mean tumor inhibition was 73.33%, 46.67% and 33.33% for ultrasound-docetaxel combination, docetaxel alone and ultrasound alone, respectively (Table 2). They believe the combination effect is due to inertial cavitation caused by USMB that generates jet streams. The jet streams in turn generate pores in vessel walls and cell membranes to enhance drug delivery (Figure 2C). Xu et al found a similar combination effect with ultrasound plus simvastatin. 44 Their results indicated that USMB when combined with simvastatin significantly increased cellular apoptosis of DU-145 cells when compared to ultrasound and drug alone. This enhanced effect is proposed to be due to cavitation caused by ultrasound microbubbles and increased membrane permeability. Other prostate cancer studies also found the combination of USMB with anticancer drugs enhances chemotherapeutic effects through increased membrane permeability. 45,46 Most of these reports related to the use of combination of USMB with anticancer drugs because microbubbles can amplify the impact of low-frequency ultrasound stimulation with the capability of long-distance penetration of cells physically coupled to microbubbles, as a result of a large difference in acoustic impedance between the surrounding media and the air inside the bubbles. 51,52 However, ultrasound-anticancer drug combination without microbubble also has been demonstrated to induce significant change in prostate cancer cell viability. 48
Discussion and Conclusion
While the use of HIFU for the treatment of localized prostate cancer is well accepted, the potential of LIPUS-anticancer drug combination can be an exciting area of research. LIPUS can penetrate a greater depth than HIFU and therefore is more versatile for application. Since LIPUS causes minimal damage to cells, it is also safer to normal tissues. In addition, if the effect of LIPUS causes reversible cell membrane damage that could allow anticancer drugs to enter cancer cells more readily, such combination with or without microbubbles may induce enhanced cytotoxicity to cancer cells. This can be especially useful if cancer cells are more sensitive to anticancer drugs than normal cells. Anticancer drugs like docetaxel and cabazitaxel can achieve a much lower IC50 for PC3 and DU145 prostate cancer cells compared to normal prostate cells, suggesting specificity of the drugs to cancer cells (our unpublished data). Future research on the application of LIPUS-anticancer drug combination can target both localized as well as certain metastatic sites. For local therapy such as the prostate, a lower dose of anticancer drug in combination with LIPUS may prove to be as effective but safer and more versatile than HIFU (Figure 2B). For metastatic prostate cancer, such combination may be also applied to common metastatic sites such as long bones, pelvis, lower spine, lymph nodes, liver, lungs or pleura in the thorax. 53 -55 Technical development of implanting a small transducer inducing LIPUS in combination with systemic anticancer drug therapy is needed. Potentially the development of intravascular catheter insertion with a small transducer without microbubbles could possibly enhance the versatility of LIPUS for metastatic prostate cancer in the future. In conclusion, LIPUS when used in combination with anticancer drug therapy could be an exciting example of a physical-chemical combination (Figure 2), resulting in potentially enhanced efficacy and reduced toxicity (in normal cells). Further research in this direction could be a worthwhile endeavor, especially for a patient with resistant prostate cancer in which there is no effective treatment.
Footnotes
Acknowledgments
The authors thank Ms. Yuna Kwon for her assistance on manuscript submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health grant GM126016 and USC BME Baum chair account (KKS).
