Abstract
Introduction
This study primarily aims to investigate the suitability of Halcyon in the context of cervical-thoracic esophageal cancer by exploring the dosimetric quality and delivery efficiency of Halcyon plans with different arc configurations. Additionally, it compares these findings with the dosimetric indices and delivery efficiency of TrueBeam and TomoTherapy accelerators, focusing on their capability to optimize protection for organs at risk (OARs) while maintaining efficient treatment delivery strategies.
Methods
This retrospective study involved 26 patients diagnosed with cervical-thoracic esophageal cancer, and new radiotherapy plans were created using Halcyon, TrueBeam, and TomoTherapy. Dose volume histogram (DVH) metrics and delivery efficiency for plans involving different arc numbers on Halcyon (2, 3, and 4 arcs) were compared with those from TrueBeam and TomoTherapy. T-tests were employed to evaluate differences in organ protection among the accelerators.
Results
The Halcyon plans, especially those with 4 arcs, provided superior protection for organs at risk, including the heart, lungs, and spinal cord, while maintaining excellent delivery efficiency. Compared to TrueBeam 2arc plans and TomoTherapy helical plans, Halcyon plans with 3 arcs also showed slight advantages. Although TomoTherapy offered better uniformity in dose distribution, it did not demonstrate a clear advantage over the other accelerators in terms of OAR protection or treatment efficiency. Furthermore, despite the lack of clear advantages in TrueBeam 2arc plans with flattening filter (FF), TrueBeam with flattening filter free (FFF) plans may hold potential in the treatment.
Conclusion
Halcyon, particularly with 4 arcs, offers an optimal balance between reducing toxicity to organs at risk and maintaining treatment efficiency, making it a preferred choice in cervical thoracic esophageal cancer radiotherapy. The findings highlight the need for careful selection of radiotherapy accelerators based on specific clinical goals, with Halcyon showing potential advantages in scenarios where both treatment efficiency and OAR protection are paramount.
Introduction
Radiotherapy plays a pivotal role in managing esophageal cancer, 1 significantly reducing the risk of recurrence after surgery in early stages 2 and alleviating symptoms like dysphagia and pain in advanced cases. 3 Moreover, the integration of chemotherapy and radiotherapy has been shown to substantially improve treatment outcomes. 4 , 5 However, constrained by organ toxicity and target volume, many esophageal cancer patients cannot undergo curative doses of radiation therapy, leading to localized treatment failures in combined chemoradiotherapy and potential recurrence of the primary tumor or local lymph nodes. 6 , 7 Advances in precise radiotherapy techniques, especially in accelerator development, typically allow for the delivery of sufficient treatment doses while better protecting organs at risk (OARs), and thus enhancing treatment outcomes. 8
The denser and taller multileaf collimators (MLCs) can effectively reduce leakage, thus better protecting OARs. 9 Varian Medical Systems’ premier C-arm accelerator, TrueBeam (TB), is equipped with a HD120 MLC featuring leaves with widths of 2.5 mm and 5 mm at the isocenter, as well as jaw tracking technology, both of which ensure excellent organ protection ability. 10 Varian's Halcyon (HL) O-ring accelerator features a dual-layer SX2 MLC with an effective width of 5 mm leaves at the isocenter and a 234 mm radius of curvature, which reduces the dosimetric leaf gap (DLG) compared to HD120's radius of 160 mm. Additionally, the dual-layer MLC in HL, with 77 mm height (greater than HD120's 67 mm) and 5 cm/s moving speed, substantially lowers leaf transmission and enhances treatment efficiency. 11 , 12 TomoTherapy (TOMO), another O-ring linear accelerator like HL, is among the advanced equipment developed in the field of radiotherapy in recent years. TOMO offers two delivery modes 13 : TOMO-Direct, which delivers treatment at discrete angles with a fixed gantry, and the more commonly used TOMO-Helical, which synchronizes gantry and treatment couch movement for a more uniform dose distribution in the Planning Target Volume (PTV), enhancing treatment efficacy. 14 , 15 its 10 cm high MLC reduces leakage, and the unique binary MLC dynamic modulation at 250 cm/s enables more subfield intensity modulation than conventional accelerators, ideal for complex plans. 16 , 17
Comparisons between HL and TB have been widely conducted across intensity-modulated radiation therapy (IMRT) and volumetric modulated arc therapy (VMAT), covering rectal cancer, craniospinal irradiation, and various stereotactic body radiation therapy (SBRT) sites, with studies examining differences in dose distribution, modulation complexity, gamma pass rate (GPR), and delivery efficiency.18–23 While increasing the number of beams improves modulation, it also lengthens treatment time, necessitating further research to determine the optimal number of beams for HL based on tumor characteristics. Yokoyama et al compared 1 to 4 arcs and found that more arcs offer better hippocampus protection. 24 Seok et al noted that increasing arcs in breast plans with HL significantly reduces the initially higher skin dose compared to TB. 25 Comparing the performance of HL and TOMO has become a focal point of research in radiation therapy due to their shared O-ring design. Panda et al compared HL and TOMO for cervical cancer 26 while Ju et al compared HL-VMAT and TOMO-Helical for nasopharyngeal carcinoma, finding similar dosimetric profiles. HL-4arc plans outperformed HL-2arc plans in dosimetry. 27 Vaidya et al further evaluated the advantages and limitations of TB, HL, and TOMO in whole breast treatment. 28
Despite extensive studies on the three accelerators for treating tumors in various locations, research specially addressing esophageal cancer remains rare. Gu et al compared TOMO-Helical and IMRT for esophageal cancer, finding TOMO-Helical offered better target coverage, dose uniformity, and reduced OAR exposure. 29 As treatment shifts from IMRT to VMAT, 30 further evaluation is needed, with Kataria et al favoring VMAT over IMRT for midterm esophageal cancer. 31 Other studies also compared Elekta accelerators with TOMO for this cancer type. However, research into the treatment of esophageal cancer using HL remains rare. This deficiency prompted our study, which conducted a comprehensive review and comparison of simulated treatment plans for 26 esophageal cancer cases using HL-VMAT, TB-VMAT, and TOMO-Helical technologies to assess planning quality and delivery efficiency. This investigation seeks to enhance the understanding of treatment planning for esophageal cancer using HL-VMAT, thereby contributing to more informed decisions in treatment strategy development.
Methods
Patient Selection
This retrospective study, conducted from August 20, 2023, to August 10, 2024, randomly selected 26 patients with cervicothoracic esophageal cancer who underwent radiotherapy, including 20 males and 6 females, with a median age of 69 years (range 53-87 years). The reporting of this study conforms to the STROBE statement. 32 The sample size was based on the availability of eligible cases rather than a formal calculation. Previous studies in this area, which we reviewed, included sample sizes ranging from 16 to 20 patients, 18 , 22 so the sample size of 26 patients is within a reasonable range. However, it may still limit the generalizability of the findings. Written informed consent was obtained from all patients for the use of their treatment information for scientific research purposes. All patient details have been de-identified to ensure that their identities cannot be ascertained. For CT imaging, these patients were immobilized at the supine position with thermoplastic masks using a Brilliance Big Bore CT scanner (Philips, Eindhoven, The Netherlands) with a slice thickness of 0.5 cm. The PTV and OARs were delineated by experienced oncologists, adhering to the guidelines established by the ICRU Report 83. 33 For optimization and evaluation purposes, a new PTV was created by clipping the original one from the body contour by 3 mm. Detailed PTV and OARs characteristics are shown in Table 1. The prescription dose was 61.2Gy in 34 fractions (180cGy/fraction), aiming for 100% of the PTV to be covered by at least 95% of the prescription dose (V100% > 95%) and the maximum dose not exceeding 110% of the prescribed dose. The OARs primarily considered for optimization and evaluation were lungs excluding the PTV (lungs-PTV), heart, and spinal cord.
PTV and OARs Characteristics of Selected Patients with Cervical Thoracic Esophageal Cance.

Treatment Planning
Eclipse (Version 15.6, Varian Medical Systems, Palo Alto, USA) was used to develop VMAT plans for TB and HL, while Precision™ (Version 1.1.1.1, Accuray®, Sunnyvale, USA) was utilized for TOMO-Helical plans. The treatment isocenter was set to the mass center of the PTV. When optimizing plans, we prioritized PTV coverage while trying to minimize the dose to OARs as much as possible. After optimization, HL and TB plans could be further normalized, while TOMO plans remained unchanged. Additionally, we have developed auxiliary structures (blocks) for all plans including (HL, TB and TOMO), to limit the angle of arcs, thereby reducing the dose to the lungs. To reduce potential bias, each patient's treatment plan was independently created by two physicists, and the final plan was selected by two senior physicists.
For HL version 3.0, all treatment fields were set to the default 6MV-FFF mode at 800 MU/min dose rate. The HL plans were designed with varying number of arcs, ranging from 2 to 4, to investigate the impact of arc quantity on treatment efficacy. The 2-arc plans utilized two full arcs, one clockwise from 181 to 179 degrees with a collimator angle of 5 degrees, and the other counterclockwise with a collimator angle of 355 degrees. The 3-arc and 4-arc plans extend this by adding 1 and 2 arcs, respectively, each with a collimator angle of 90 degrees. These additional arcs, which leveraged the “limit leaves” feature in Eclipse to confine the arcs to specific parts in the superior-inferior direction, were primarily used to irradiate specific segment of the thoracic esophagus cancer thereby improving the dose distribution in the region.
For TB, all treatment fields were set to 6MV-FF mode at 600 MU/min. The TB employed an HD 120 MLC and jaw tracking technology. The TB plans consisted of 2 full arcs with settings identical to the gantry start, gantry end, and collimator angle of the HL plans. Both HL and TB employed the same optimization engine (Photon Optimizer) and dose calculation algorithm (Analytical Anisotropic Algorithm, AAA), with a 2 mm dose calculation grid size (CGS).
TOMO-Helical plans utilized a 6MV FFF beam alongside dynamic jaw mode. The jaw width was configured to 2.5 cm, with a modulation factor of 2.0 and a pitch of 0.287. Optimization dose resolution was set to medium, featuring a dose grid spacing of 2.37 mm in the x–y plane and 2.5 mm in the z-axis, and the optimization terminates after 100 iterations. The final dose calculation employed a higher resolution (1.37 mm in the x–y plane and 5 mm in the z-axis).
Plan Evaluation
The plan quality was assessed by using Conformity Index (CI), Homogeneity Index (HI), and Gradient Index (GI) for the PTV, along with various DVH parameters for critical OARs, primarily the spinal cord, heart and lungs-PTV. Both the RTOG Conformal Index (
Statistical Analysis
All analysis was performed using the SciPy 1.11.1 package in Python 3.10.12. Specifically, paired t-tests were conducted with the scipy.stats.ttest_rel function to compare dosimetric indices and delivery efficiency indices between different accelerator plans. Statistical significance was set at a p-value of less than 0.05.
Results
Table 2 presents the means and standard deviations of dosimetric indices for all plans, pairwise t-tests on these indices, detailed in Table 3, revealed no significant differences in PTV dose indices across different numbers of arcs in HL plans (as shown in Figure 1) while OAR sparing improved with additional arcs. Regarding lung dosimetric indices, including the mean dose, V20Gy, and V5Gy, significant differences were observed when comparing HL-2arc plans, HL-3arc and HL-4arc plans. For mean dose, V30Gy, and V40Gy to the heart, significant differences were observed between HL-2arc plans and both HL-3arc and HL-4arc plans, while no significant differences were noted between HL-3arc and HL-4arc plans. Significant differences were observed in the maximum dose of the spinal cord across all arc configurations, with the maximum dose decreasing as the number of arcs increased. It can be concluded that the HL plans with 2, 3, and 4 arcs showed no significant differences in PTV evaluation indices, affirming consistent target coverage. However, the HL-4arc plans exhibited a clear advantage over the HL-2arc plans in reducing toxicity across three critical OARs mentioned before. While the benefits of increasing from 3 to 4 arcs in the HL plans were smaller compared to the transition from 2 to 3 arcs, the reductions in lung dosimetric indices and maximum spinal cord dose were still substantial. The analysis, supported by data from Table 2, Table 3 and Figure 2, demonstrated that TB and HL plans did not differ significantly in PTV dose evaluation metrics, indicating similar dose distributions for the PTV. Notably, the HL data presented in Figure 2 was generated using the HL-4arc plans only. Both plans exhibited better conformity and 50% dose attenuation compared to TOMO plans, but TOMO demonstrated superior dose uniformity. In terms of OARs, HL showed significant advantages in the mean dose, V20Gy, and V5Gy to the lungs-PTV over both TB and TOMO, effectively reduced lung toxicity. Additionally, TOMO slightly outperformed TB in the V20Gy. Regarding the heart, HL demonstrated benefits in the mean dose compared to other accelerators and especially being superior to TOMO in V30Gy while no differences were observed between TOMO and TB. HL recorded lower spinal cord maximum doses than TOMO without statistical significance; however, it significantly reduced toxicity compared to TB.

Comparison of dosimetric indices and t-test results for the number of arcs on Halcyon.

Comparison of PTV and OAR dosimetric indices and t-test results across Halcyon (4 arcs), TrueBeam, and TomoTherapy.
Comparison of PTV and OAR Dosimetric Indices Across Halcyon, TrueBeam, and TomoTherapy Plans.

Note: Mean ± SD (range).
Statistical Significance (p-Values) of Dosimetric Differences Between Halcyon, TrueBeam, and TomoTherapy Plans Using Paired t-Test.
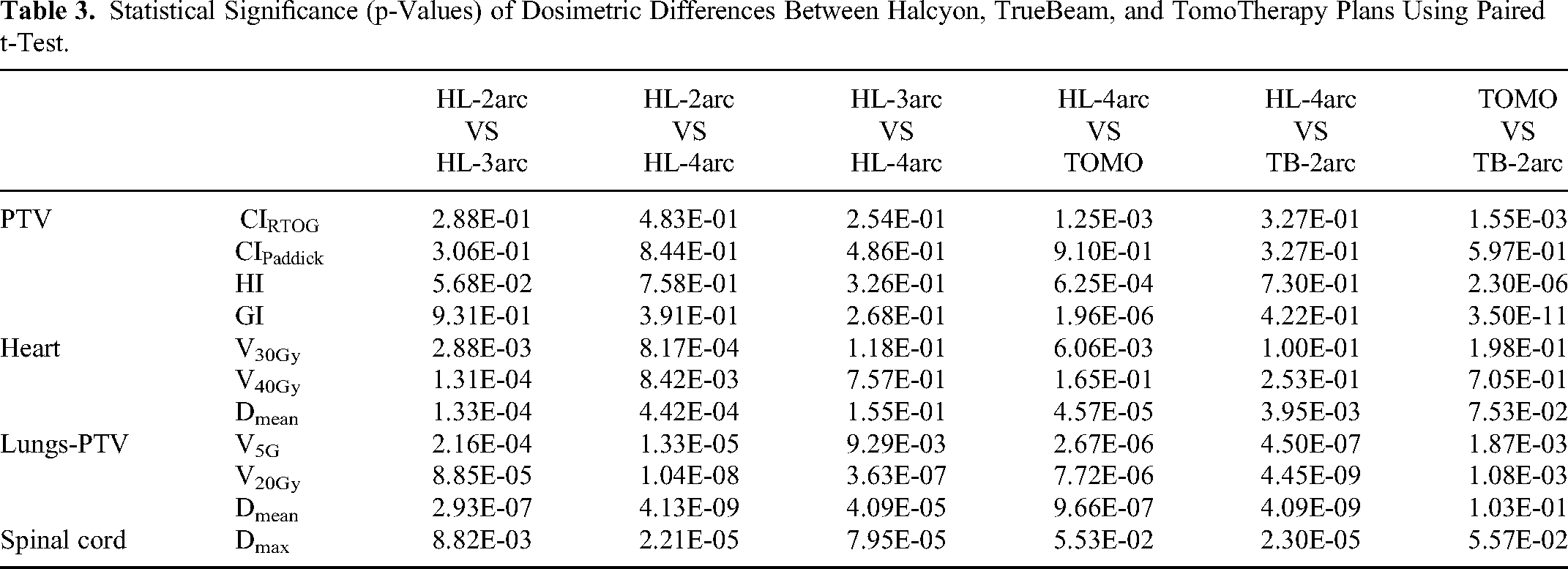
Figure 3 illustrates the dose distribution across the transverse, sagittal, and coronal planes for a representative case's plans using HL, TB, and TOMO. All accelerators achieved satisfactory target coverage, with the HL-4arc plan notably showing more confined areas covered by the 5Gy and 40Gy isodose lines compared to the TB-2arc and TOMO plans, indicating more precise dose delivery and better sparing of adjacent normal tissues.

Dose distribution across the transverse, sagittal, and coronal planes for a representative case's plans using Halcyon, TrueBeam, and TomoTherapy.
Table 4 lists delivery efficiency and accuracy metrics for HL, TB and TOMO plans. The total MUs and beam-on times for HL and TB were derived from trajectory log files, while the data for TOMO, due to the unavailability of log files, were collected directly from TPS. Generally, HL plans required more MUs than TB-2arc plans: 190.53 s more for HL-2arc, 262.6 s for HL-3arc, and 330.66 s for HL-4arc. Given the rapid decrease in intensity with off-axis distance for the FFF mode used by HL plans, more MUs are naturally required to achieve uniform PTV dose distribution, compared to the FF mode used by TB plans. Although HL plans with 2 and 3 arcs required more MUs than the TB-2arc plan, their actual beam-on times were shorter, and the beam-on time for the HL-4arc plan was only 13.48 s longer than that of TB. Considering the CBCT scanning time (approximately 30– s for HL and 60 s for TB), it can be concluded that the overall treatment efficiency of HL is superior to TB. Figure 4 presents a gantry angle versus time plot from trajectory log files for a case study, highlighting HL's significantly faster gantry rotation speed. This high speed effectively compensated for the efficiency loss due to the higher MUs. However, in contrast, TOMO's treatment efficiency was significantly lower than both HL and TB, indicating a substantial difference in performance across these accelerators. Delta4 measurements revealed that under the 3 mm/3% criteria, all accelerators met the standard, with pass rates exceeding 95%. However, under the stricter 2 mm/2% criteria, HL showed more stable performance, maintaining a pass rate of over 99% while both TOMO and TB experienced a decline in performance, yet they still met the minimum threshold with pass rates above 90%.

The gantry angle over time plot of one case on Halcyon and TrueBeam.
Comparison of Delivery Parameters of Halcyon, TrueBeam and TomoTherapy Plans.

Discussion
This study analyzed DVH metrics and delivery efficiency for cervical-thoracic esophageal cancer radiotherapy plans utilizing HL, TB, and TOMO. Previous Research consistently shows that higher radiation doses to OARs increase the risk of radiation-induced damage; specific thresholds have been identified, such as heart Dmean below 15Gy reducing the incidence of Grade 3 or higher events and heart V30Gy as a predictor for pericardial effusion, while lowering heart V40Gy significantly reduces the likelihood of cardiac disease.34–38 In this context, although the heart Dmean from all three accelerators remained below 15Gy in our study, HL-4arc exhibited superior performance across all heart metrics when compared to TB and TOMO. Radiation-induced lung injury is another common concern in esophageal cancer radiotherapy, closely linked to dosimetric parameters such as V20Gy, V5Gy, and Dmean to the lung. Existing studies recommend maintaining V20Gy below 23% to 30% to significantly lower the risk of radiation pneumonitis (RP).39–42 While V20Gy is critical, the impact of Dmean is also significant; keeping Dmean under 15Gy is advised to reduce RP risk, 42 with some evidence suggesting that an Dmean exceeding 12Gy notably increases this risk. 43 While some studies link V5Gy with RP risk, evidence also shows that V5Gy levels above 71% may not lead to Grade 2 or higher RP, suggesting its impact may be less critical than V20Gy or Dmean. In our study, HL-4arc plan demonstrated superior lung protection, achieving the lowest V20Gy at 19.93%, well below the recommended threshold to minimize the risk of RP. Although all accelerators maintained the lung Dmean under the critical 15Gy limit, HL-4arc achieved even lower values than TB and TOMO. This highlights HL's enhanced efficacy in managing radiation exposure to critical lung areas. Additionally, our findings suggest that, for HL, increasing the number of arcs may enhance plan quality, with the 4arc plan showing a trend toward better overall performance. However, the differences between 4arc and 3arc plans in heart-related metrics were not statistically significant, which aligns with previous research indicating that, although more arcs may offer better OAR protection, the benefits tend to plateau beyond 3 arcs.24,27 This alignment with other studies suggests that 3 to 4 arcs may provide a reasonable balance for optimizing protection and efficiency in HL-based treatment plans.
To further compare TB and HL, additional experiments were conducted on a single patient, adjusting TB's energy mode, dose rate, and number of arcs. Figure 5 demonstrates the subfield area distributions and DVH plots for each plan, while Table 5 lists additional metrics. Compared to the TB-2arc-FF-600 plan, the TB-2arc-FFF-600 plan showed a shift in subfield area distribution towards smaller areas, enhancing protection for lung, heart, and spinal cord, like that observed with the TB-2arc-FFF-800 plan. The increased number of arcs in the TB-4arc-FFF-800 plan further shifted the subfield area distribution towards smaller fields and additionally reduced heart toxicity compared to other TB-FFF plans. For TB plans with the same number of arcs, beam-on times were nearly identical, suggesting that the FFF mode can reduce doses to OARs without compromising treatment efficiency. This was because compared to FF mode, the small fields under FFF mode can effectively reduce scatter outside the field. 44 The comparisons between TB-2arc-FFF-800 and HL-2arc-FFF-800, as well as between TB-4arc-FFF-800 and HL-4arc-FFF-800, were specifically designed to assess the characteristics of each machine, revealing that TB provided superior heart protection, while HL offered better protection for the lungs-PTV and spinal cord. Furthermore, HL demonstrated significantly higher efficiency, with operation times for comparable plans just over half those of TB. In conclusion, considering both dose metrics and delivery efficiency, HL-4arc-FFF emerged as the preferred choice for radiotherapy of cervical thoracic esophageal cancer.

Subfield distribution and DVH plots for different configurations.
Dosimetric and Delivery Efficiency Metrics for TrueBeam and Halcyon Plans.

While our study provides valuable insights, certain factors may influence the extent to which these findings can be generalized. The specific conditions and practices at our center, including the prescribed dose of 61.2 Gy and patient positioning, may not fully align with those at other institutions, potentially impacting the broader applicability of our results. The higher dose we used reflects our center's clinical practice, which may differ from other institutions where lower doses, such as 45Gy, are delivered to elective areas with a boost to the gross disease. Although this higher dose may limit generalizability, it also places greater demands on the accelerators’ modulation capabilities to adequately protect OARs, highlighting their ability to manage more challenging treatment scenarios. Additionally, our study did not include patients with mid/distal thoracic esophageal cancer, which represents another limitation. We are considering a separate, future study focused on this patient group to provide a more comprehensive understanding of treatment techniques and their impact on OAR doses.
Conclusion
The HL plans, both with 3 and 4 arcs, demonstrate a favorable balance between treatment efficiency and reduction of toxicity to OARs in cervical thoracic esophageal cancer, compared to TB and TOMO accelerators. However, for enhanced protection of OARs, the TB-4arc-FFF plan can be considered as a viable alternative when treatment efficiency is not critical. Given TOMO's significantly longer treatment times and no clear dosimetric advantages, its use may be less preferable compared to other options. Additionally, the impacts of the FFF mode in TB and HL plans on other aspects, such as skin dose, require further analysis.
Supplemental Material
sj-xlsx-1-tct-10.1177_15330338241293321 - Supplemental material for Comparative Evaluation of Dosimetric Quality and Treatment Efficiency for Halcyon, TrueBeam, and TomoTherapy in Cervical-Thoracic Esophageal Cancer Radiotherapy
Supplemental material, sj-xlsx-1-tct-10.1177_15330338241293321 for Comparative Evaluation of Dosimetric Quality and Treatment Efficiency for Halcyon, TrueBeam, and TomoTherapy in Cervical-Thoracic Esophageal Cancer Radiotherapy by Shilin Chen, Jiazhou Wang, Weigang Hu and Yao Xu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
Yao Xu conceived the work and wrote the main manuscript text with Shilin Chen, who was also responsible for the acquisition of data. Jiazhou Wang and Weigang Hu assisted with the analysis and interpretation of data.
Data Availability
The data that support the findings of this study are not publicly available due to privacy and ethical restrictions. Protecting the privacy of study participants is a priority, and therefore, the data cannot be shared openly.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was approved by the ethics committee of Fudan University Shanghai Cancer Center with approval number 1612167-18. We confirm that all methods were carried out in accordance with relevant guidelines and regulations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Key Research and Development Program of China (grant numbers 2022YFC2404603, 2022YFC2404600). Key Clinical Specialty Project of Shanghai and National Natural Science Foundation of China (grant number 12375339).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
