Abstract
We do not know the clinical and prognostic factors that influence the survival of patients with gastric signet ring cell carcinoma (SRC). Therefore, a retrospective review was undertaken of 219 patients with SRC who had undergone gastrectomy between January 2009 and December 2012 in our hospital. Patient age, sex, TNM stage, vessel carcinoma embolus, perineural invasion, tumor site and operation type, postoperative chemotherapy, and five-year overall survival were recorded and evaluated. In our study, 93 cases (42.5%) were signet ring cell carcinoma only, and 126 cases (57.5%) were signet ring cell carcinoma coexisting with other components (such as adenocarcinoma or mucus adenocarcinoma). Eighty-three patients were female, 136 were male, 46 occurred at the gastroesophageal junction (21.0%), 63 at the fundus/body (28.8%), 80 were antrum/pylorus (36.5%), and 30 were whole stomach (13.7%). The prognosis of gastric antrum/ pylorus cancer was the best (P < 0.05). There were 133 patients (60.7%) with stage III, and the single factor analysis showed that the earlier the stage, the better the prognosis. The overall five-year survival rate was 30.1% in all patients. One-hundred and 41 patients (64.4%) received D2 radical surgery, 64 (29.2%) received D1 radical operation, and 14 (6.4%) received palliative resection, and the patients who received D2 had the best overall survival (P < 0.05). The survival time of the paclitaxel-based regimen in postoperative adjuvant chemotherapy tended to be prolonged. There was no statistical difference in overall survival between the percentage of signet-ring cells and sex. In summary, age, tumor stage, and surgical resection combined with D2 lymphadenectomy were independent prognostic factors for SRC. Adjuvant chemotherapy with a paclitaxel-based regimen may improve the survival of patients with SRC.
Keywords
Introduction
Gastric cancer (GC) is still one of the main causes of cancer-related deaths worldwide. 1 In China, the incidence of GC is second only to lung cancer, and the mortality rate is the third highest of all cancers. 2 Histologically, GC exhibits obvious heterogeneity and usually coexists with several histological components. 3 Gastric signet ring cell carcinomas (SRCs) are histological diagnoses based on microscopic characteristics described by the World Health Organization. SRCs are described as being an isolated micro-community of malignant cells with intracytoplasmic mucins that account for more than 50% of the tumors. 4 Currently, the impact of SRC on the prognosis of patients with GC is controversial. 5,6 Some studies 7 -9 have reported that patients with SRC have a poorer prognoses, while others 10,11 reported that patients with SRC have better five-year survival rates than patients with other types of GCs. Therefore, we retrospectively collected the clinical characteristics and overall survival of patients with gastric SRC and evaluated the independent prognostic value of SRC in relation to those of other variables. We investigated the prognostic value of SRC and whether it was influenced by select clinicopathological factors.
Materials and Methods
Patients
From January 2009 to December 2012, a total of 219 patients diagnosed with gastric SRC underwent curative resection for GC in our hospital. The overall survival (OS) was defined from the date of operation to the date of death or the latest follow-up (December 31, 2017). The records of age, sex, tumor location, operation mode, tumor invasion depth, tumor stage (TNM stage), vessel carcinoma embolus, and perineural invasion were collected. According to the anatomic location of the tumor center, the tumor site can be divided into the gastroesophageal junction, fundus, antrum/pylorus, large part of the stomach, or whole stomach. The seventh edition of the American Joint Committee on Cancer (AJCC) 2010 was used to define the tumor stage. 12 D1 lymphadenectomy refers to the removal of a tumor that involves the proximal, distal, or total stomach, and at the same time, cleaning of the greater and lesser omentum lymph nodes. A D2 lymphadenectomy is based on a D1 lymphadenectomy, with not only the omental sac and the anterior lobe of the transverse colon mesentery being removed, but also the corresponding periarterial lymph nodes. 9
Statistical Analysis
The demographic and clinical characteristics were compared among groups by independent t-test and χ2 tests. The Kaplan Meier (KM) method was used to generate survival curves, and then, the log-rank test was performed. Long-term survival was assessed using the five-year OS rate. The Cox regression hazard model was used for univariate and multivariate analyses. A P-value of P < 0.05 was considered statistically significant for all analyses. All statistical analyses were performed using SPSS version 21.0.
Results
Patient Demographics
A total of 219 patients diagnosed with gastric SRC were included. There were 136 men (62.1%) and 83 women (37.9%), with a median age of 62 years (22-81). We identified 93 cases (42.5%) of complete SRC only and 126 (57.5%) of SRC coexisting with other components (such as adenocarcinoma or mucus adenocarcinoma) (Figures 1, 2). According to the TNM staging standard of the American Joint Committee on Cancer (AJCC), 30 patients (13.7%) were stage I, 34 (15.5%) were stage II, 133 (60.7%) were stage III, and 22 (10.1%) were stage IV. In addition, 89 cases (40.6%) had vessel carcinoma embolus and 90 (41.1%) presentedperineural invasion. According to the tumor location, 46 (21.0%)mcarcinoma cases were located at the gastroesophageal junction (GEJ), 63(28.8%) at the fundus/body, 80 (36.5%) at the antrum/pylorus, and 30 (13.7%) in the whole stomach. The single factor analysis showed that the five-year survival time of patients was related to age, tumor stage, presence of vessel carcinoma embolus, perineural invasion, and tumor location (P < 0.05), but not to sex nor the percentage of signet-ring cells (P > 0.05) (Table 1).

Histological diagnosis of SRC and with other components. SRC:Signed Ring Cell carcimona; MAC:Mucinous AdenoCarcinoma; PDA:Pooriy Differentiated Adenocarcinoama; MDA: Moderately Differentiated adenocarcinoma.
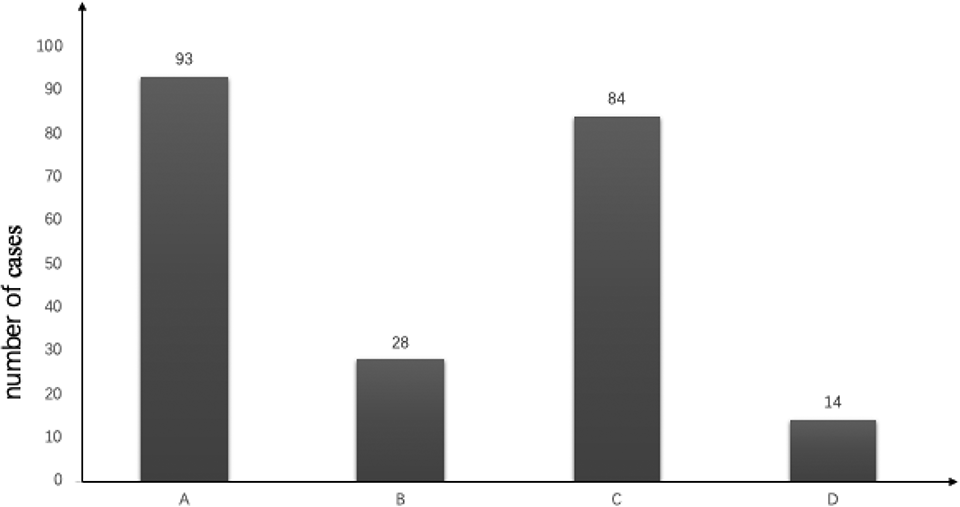
Number of SRC cases and with other components’ cases. (n = 219). A: Gastric SRC carcinoma; B: The corxistense of SRC and MAC; C: The coexistense of SRC and PDA; D: The coexistense of SRA and MDA.
Clinical Characteristics and Single Factor Survival Analysis of Patients With Gastric SRC (n = 219).

Clinical Treatments
Of the 219 patients, 141 (64.4%) underwent surgical resection combined with D2 lymphadenectomy, 64 (29.2%) had a standard D1 lymphadenectomy, and 14 (6.4%) had only palliative surgical resection. A total of 139 patients (63.5%) underwent total gastrectomy, and 139 patients (63.5%) underwent postoperative chemotherapy. Of the 139 patients, 110 patients (79.1%) had a 2-drug regimens that included oxaliplatin-based or docetaxel/paclitaxel-based regimens. The average chemotherapy period was 6 cycles (2-12 cycles). Thirty-nine patients (17.8%) received intraperitoneal perfusion chemotherapy. The single factor analysis showed that the five-year overall survival of patients was related to surgical procedures and adjuvant chemotherapy, but not to intraperitoneal perfusion. In adjuvant chemotherapy, the OS of patients who received a combination of the 3 drugs was the longest, but there was no statistical difference compared with the combination of 2 drugs (Table 2).
Clinical Treatment and Single Factor Survival Analysis of Patients With Gastric SRC.

P < 0.05.
Survival
Using the Cox proportional risk model to further analyze the prognostic factors of gastric SRC by multifactor analysis, the five-year survival rate of patients was found to be affected by age, tumor stage, operation mode, adjuvant chemotherapy, and tumor location, but had nothing to do with the patient’s sex, number of signet ring components, or intraperitoneal perfusion after operation. Notably, patients who were young, had early stage disease, and completed a standard D2 lymph node dissection had the best prognosis. (Table 3 and Figure 3)
Multivariate Analysis of Survival and Prognosis of Patients With Gastric SRC.


Five-year survival curve of patients with SRC by age (A), differentiation stage (B), and operation method (C).
Discussion
GC is the fourth most common malignancy worldwide. Despite the advances in diagnosis and treatment strategies, the prognosis of GC has not improved much over the last 2 decades, with a five-year OS rate of only 25-30%. 13 -15 The proportion SRC 3,8,16 in GC has been reported to be increasing in recent years. Except in the early stage, 17,18 SRC features have been reported to be a poor prognostic marker in GC. SRC has unique clinical characteristics, biological properties, and behavior, and it is necessary to expand research to improve the treatment of this type of GC.
In our study, it was found that older (>60 years; 57.1%) patients with a more advanced stage (AJCC stage III; 60.7%) had worse prognoses. It was also confirmed by Shuai Guo 19 that elderly patients and advanced tumor stage were poor prognostic risk factors for gastric SRX, and the prognosis of SRC worsened with the progression of the disease. Tumor location can also affect the survival of patients with gastric SRC. In our study, we found that the prognosis of gastric antrum cancer was the best, followed by gastric body, and tumor in most or all of the stomach was the worst (P < 0.003). This is consistent with previous reports. 20 Park 21 et al. found that the five-year survival rate of patients with proximal GCr was significantly lower than that of those with distal GC (59.6% vs. 73.7%, P < 0.001). Chen Zhixin 22 also reported similar results. This may be related to the anatomic structure of the stomach. Notably, the anatomical space between the upper part of the stomach and the lower part of the diaphragm is narrow. Approximately 30% of the posterior wall of the stomach has no serous cover; thus, a proximal GC that invaded the muscular layer can easily penetrate the whole layer of the gastric wall and invade the surrounding fatty tissue and adjacent organs. In addition, in terms of molecular biology, compared with distal gastric cancer, proximal gastric cancer has a higher TP53 gene mutation rate, and this suggests that there is a difference in the molecular mechanism of the pathogenesis of proximal GC and distal GC. 23 In some studies, TP53 mutations and high p53 protein expression have been found to negatively correlated with the prognosis of patients with GC. 24 It is necessary to further study the molecular mechanism of GC onset in different parts of the stomach.
It is widely accepted that survival rates improve with a standardized pattern of surgical resection combined with D2 lymphadenectomy. 25,26 In our study, 141 patients were treated with D2 lymphadenectomy, and the five-year survival rate of these patients was statistically different from those who received D1 lymphadenectomy. This suggests that D2 lymphadenectomy can improve the prognosis of patients with gastric SRC, reduce the risk of tumor recurrence and metastasis, and prolong survival. Chinese studies 27 reported that the median survival time of patients with advanced GC undergoing D2 lymphadenectomy was 55.0 months (95% CI: 51.1-58.9), significantly longer than the 28.5 months for patients undergoing D1 lymphadenectomy (95% CI: 21.3-34.7, P < 0.01). These observations are consistent with the general belief that D2 lymphadenectomy improves patient survival.
In this study, we found that adjuvant chemotherapy affected the OS of patients with SRC after surgery. The combination of 2 drugs, 5-FU combined with oxaliplatin or paclitaxel, was the main treatment program. Compared with chemotherapy regimens, although there was no significant difference in OS, the patients who received paclitaxel-based chemotherapy tended to have prolonged survival. Chen L et al. 28 found that patients with mixed SRCs had longer disease-free survival and OS than patients who received chemotherapy containing oxaliplatin. This indicated that adjuvant chemotherapy was beneficial and that a docetaxel-based regimen should be considered for patients with mixed SRC. In our other research, we used the “fluorouracil/leucovorin-paclitaxel-oxaliplatin (POF)” regimen as the first-line treatment against advanced gastric SRC, and the results showed a disease control rate of 89.4%and response rate of 48.5%, which indicate that the paclitaxel-based regimen was effective in advanced gastric SRC. 29 In clinical practice, we found that adjuvant chemotherapy has a poor effect on patients with SRC of the stomach, a high recurrence rate, and a short survival period. Therefore, it is urgent to find a more effective chemotherapy scheme to reduce tumor recurrence and metastasis and prolong survival time. Patients with ECOG scores of 0 or 1 should consider adopting a paclitaxel-based chemotherapy regimen that includes 5-FU, oxaliplatin/ cisplatin.
In general, the five-year OS rate of patients with SRC of the stomach is low. Early diagnosis, D2 lymphadenectomy, and adjuvant chemotherapy are helpful in improving the prognosis. We analyzed according to age, sex, stage, pathological histology, vessel carcinoma embolus, perineuronal invasion, tumor location, surgery, and chemotherapy. As a retrospective study, there are some limitations. Notably, selection bias is inevitable, and the incomplete auxiliary treatment information of some patients may have impacted prognoses. The sample size of this study was also small, and there may be bias due to sample size. The biological behavior of GC, such as carcinoembryonic antigen (CEA) expression, mismatch repair (MMR), and human epidermal growth factor receptor-2 (HER-2) expression were not included in our evaluation. Therefore, it is necessary to further expand the sample size and conduct forward-looking research to analyze the biological behavior of gastric SRC and design more effective clinical treatment strategies to improve the survival of patients.
Footnotes
Acknowledgments
Yu Jia Mi and Zeng Qing Guo conceived and designed the study; Jing Xian Zhen, Xiao Jie Wangand Yu Chen collected the data. Yu Jia Mi, Zhou Wei Zhanand Jing Lin analyzed the data. Yu Jia Mi, Ling Chen, Zhu Li Chen and Fang Yu Huang contributed to the writing of the manuscript and Yu Jia Mi edited the manuscript.
Ethics Statement
The study protocol was approved by the ethics review board of Fujian Cancer Hospital(YKT2019-019-01) .We have obtained written informed consent from all study participants .All of the procedures were performed in accordance with the Declaration of Helsinki and relevant policies in China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from Fujian Innovation project (2019-CX-4)/(2018501267) and Fujian Provincial Health Technology project (2020J011111).
