Abstract
Objective:
Our objective is to assess whether the outcome of intrahepatic unresectable or ablative hepatocellular carcinoma (HCC) could be improved by supplemental transarterial chemoembolization (TACE) following initial treatment of TACE with 3-dimensional conformal radiotherapy (3DCRT), compared to TACE followed by 3DCRT alone.
Methods:
We retrospectively analyzed intrahepatic unresectable or ablative HCC patients who underwent TACE, followed by 3DCRT with or without additional TACE, from June 2010 to December 2016 at our institution. Survival was assessed using the Kaplan-Meier method and compared with the log-rank test. Cox regression analyses were used to identify factors that influenced prognosis. Toxicity profiles were evaluated using CTCAE 4.0.
Results:
27 patients received TACE with 3DCRT (TR group) and 11 received additional TACE following TACE and 3DCRT (sandwich group), respectively. The median intrahepatic progression-free survival (IPFS), progression-free survival (PFS), and overall survival (OS) in the TR group and sandwich group were 5.4 months vs. 17 months (P = 0.018), 5.4 months vs. 17 months (P = 0.008), and 13.5 months vs. 29.2 months (P = 0.011), respectively. Multivariate Cox regression demonstrated that TACE followed by radiotherapy alone had a shorter IPFS (HR: 2.516, 95% CI (1.136-5.570), P = 0.023) and PFS (HR: 2.637, 95% CI (1.182-5.880), P = 0.018) compared with the sandwich treatment. Hepatitis B virus reactivation occurred in 1 patient in the sandwich group. Myleosuppresion was considered a grade 3/4 adverse event.
Conclusion:
Unresectable or ablative HCC patients possibly benefit from the combination of TACE and 3DCRT followed by additional TACE therapy, compared with TACE followed by 3DCRT alone.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors with high morbidity and mortality in China, as well as worldwide. 1,2 Curative treatment options include surgical resection, liver transplantation, and ablation therapy, which includes radiofrequency ablation (RFA), microwave ablation, or percutaneous ethanol injection. 3 Unfortunately, not all patients are suitable for these treatments due to underlying liver function, a shortage of donor livers, tumor burden, or the location of the tumor (such as lesions adjacent to the gastrointestinal tract or major blood vessels). 4 Therefore, other local or systemic treatments are necessary for these patients.
Transarterial chemoembolization (TACE) has been widely used as a palliative treatment option for HCC unsuitable for curative treatment options. 5 Compared with the best supportive care, TACE imparts survival benefits for patients with unresectable HCC. 6 However, patients rarely exhibit complete responses to TACE alone due to larger lesions in and around the tumor that are nourished by portal veins (rather than the hepatic artery), generally resulting in incomplete embolization. 7,8 Furthermore, it is not recommended to undergo multiple repetitive sessions of TACE. Not only does this result in less therapeutic efficacy, but it also contributes to the deterioration of liver function and increases TACE-related adverse effects. 9 Therefore, additional treatment regimens following TACE therapy are often required.
Previously, radiotherapy was not a recommendation for HCC treatment due to poor tolerance to radiation and a high risk of radiation-induced liver damage. 5 However, there have been numerous advances in radiation technology over the past few decades, with current treatments including 3-dimensional conformal radiation therapy (3DCRT), intensity-modulated radiotherapy (IMRT), and stereotactic body radiation therapy (SBRT). These improvements have expanded the indications of radiation therapy for HCC. Radiotherapy alone or with the combination of other local or systemic treatments are both options for HCC patients with adequate liver function. 10 Regarding combination therapy, radiotherapy combined with TACE has become a common and effective treatment. However, with this combination, the 3-year overall survival rate is 29% and the median survival time is 18 months, 11 respectively, which is far away from our expectations. Reasons for the poor outcomes with the combination of TACE and radiotherapy are predominantly due to intrahepatic or local recurrence, or even distant metastases. Therefore, additional treatment following completion of the combined treatments of TACE and radiotherapy is necessary to improve patient outcomes.
Hence, in the present study, we aimed to assess whether additional TACE following the completion of TACE and 3DCRT combination therapy could improve outcomes. We also aimed to evaluate its safety.
Materials and Methods
Patients
From June 2010 through December 2016, patients who received the combination treatment of TACE and 3DCRT followed by TACE (sandwich group), as well as those who did not follow up with TACE after the initial combination treatment (TR group), were retrospectively analyzed. The criteria for eligibility for these treatments included: (1) intrahepatic disease either unresectable or ablative due to near macrovascular or porta hepatis, or patients unwilling to receive operation; (2) Barcelona clinical liver cancer (BCLC) stage B or C; (3) a single nodule greater than 5 cm or 2 to 3 nodules with a sum of diameters ≤10 cm; (4) Child-Pugh score ≤8; and (5) partial TACE before radiotherapy. The efficacy was assessed in accordance with the Modified Response Evaluation Criteria in Solid Tumors, version 1.0 (mRECIST 1.0). The exclusion criteria in the present study included extrahepatic metastasis, with previous radiotherapy in the upper abdomen, or other malignancies within the past 5 years. This study was approved by our institute. The written informed consent was waived due to the nature of the retrospective study.
Treatments
Details of TACE procedure are as follows. An angiographic catheter using Seldinger technique was inserted into the hepatic artery from the femoral artery to assess tumor-feeding arteries and portal vein tumor thrombosis. Afterward, the feeding artery was selectively catheterized and the chemotherapeutic lipiodol and doxorubicin mixture were administered as an oil emulsion, followed by embolic materials., either before or after 3DCRT.
For the CT simulation, metal markers were used in the bilateral hypochondriac regions, and patients were immobilized in the supine position using vacuum casts with their arms placed above their heads. An active breathing coordinator or abdominal compression was used to reduce potential bias caused by respiratory movements. Upper abdomen axial dynamic contrast-enhanced computed tomography (CT) images with a slice thickness of 3 mm were obtained. The resolution of the CT image was 512 mm × 512 mm, and the voxel size was 0.69 mm × 0.69 mm × 0.5 mm. The image sets were transferred to a Varian treatment planning system for 3DCRT planning. The gross tumor volume (GTV) was defined as the all visible tumors in dynamic contrast-enhanced CT images and portal vein tumor thrombus (PVTT). Clinical target volume (CTV) was created with a 5 mm outer margin from the GTV. The planning target volume (PTV) was generated from the CTV with a 5-10 mm external margin, taking into account respiratory liver motion and patient set-up errors. Organs at risk, such as the total liver, non-target normal liver (NTNL), stomach, kidneys, and spinal cord were also delineated for evaluation of the irradiation dose. The 3DCRT planning was designed using photon beams of 6 or 10 MV in the Eclipse V13.1 treatment planning system with a prescription dose of 50-60 Gy with a conventional fraction (1.8-2.0 Gy/fraction), and at least 95% of the PTV receiving the prescribed dose. 120 KV fluoroscopy or cone-beam computed tomography were used to assess the treatment set-up and field verification with the intrahepatic lipiodol as the fiducial markers. Patients with an abnormal serum AFP concentration (>25ug/L), stable disease, or partial disease response 3-6 weeks after 3DCRT (according to mRECIST 1.0) received additional TACE. Treatment options after failure with these treatments were determined by multi-disciplinary teams at our institute.
Evaluations
After treatment completion, patients were to follow-up every 3 to 6 months for the first 2 years, and then every 6 to 12 months until death. Follow-up appointments included routine blood examinations, blood biochemistry, AFP, HBV-DNA, and liver dynamic contrast-enhanced CT or MRI scans. The efficacy was estimated using mRECIST v 1.0 according to liver dynamic contrast-enhanced CT or MRI scans. Toxicities were evaluated using the Common Terminology Criteria of Adverse Events (CTCAE), version 4.0. Both the efficacy and AFP response were assessed every 3 months after finishing the treatments. Classic radiation-induced liver disease (RILD) was diagnosed if patient had anicteric ascites or least a 2-fold increase in serum alkaline phosphatase levels compared to pretreatment or baseline values in the absence of tumor progression after hepatic irradiation.
Statistical Analysis
The continuous variable was represented as the mean ± standard deviation and compared with the Student’s t-test or Mann-Whitney U test, or it was transformed into a categorical variable. Fisher’s exact test was used to compare the categorical variable between groups. The intrahepatic progression-free survival (IPFS) was defined as the time from radiotherapy completion to the first time of intrahepatic recurrence. PFS was defined as the time from radiotherapy completion to the first time of any recurrence. OS was defined as the time from radiotherapy completion to the time of death. Survival was calculated using the Kaplan-Meier method and differences between groups were identified using the log-rank test. The survival curves were contoured using GraphPad Prism version 7.0. Factors that influenced prognosis were identified by Cox regression analyses using the method of forward: conditional. All statistical analyses were performed using SPSS, version 24.0 (IBM SPSS, Inc, Chicago, IL). A p-value <0.05 was considered statistically significant.
Results
Baseline Characteristics of Patients
A total of 38 patients with unresectable HCC who underwent the combination treatment of TACE and 3DCRT were analyzed. Of them, 27 did not receive any anti-tumor treatment after the completion of 3DCRT (TR group). The remaining 11 patients received a cycle of TACE within 3 to 6 weeks after finishing 3DCRT (sandwich group). All patients received at least 1 cycle of TACE treatment before 3DCRT. Of the patients, 92.1% (35/38) were male and 73.7% (28/38) had an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0. The mean age of the patients was 58.2 years old. Most of the patients (86.8%, 33/38) were Hepatitis B virus-positive. Patients with cirrhosis accounted for 65.8% (25/38) and most of the patients (86.8%, 33/38) had a Child-Pugh score of 5. Approximately 65.8% (25/38) of the patients had 1 to 2 lesions, and half of them had a vascular invasion. T1-T2 stage (American Joint Committee on Cancer TNM system, Version 7) applied to up to 55.2% (21/38) of the patients, while BCLC stage (Barcelona clinical stage) applied to up to 63.2% (24/38). The serum AFP concentration was above the upper limit of normal (≥25ng/mL) in all patients, and 57.9% (22/38) had a serum AFP concentration of less than 200 ng/ml. There were no significant differences in the baseline characteristics among groups. These details are summarized in Table 1.
The Baseline Characteristics of Patients.

TACE Transarterial chemoembolization; 3DCRT 3-dimensional conformal radiotherapy; AFP Alpha-fetoprotein; BCLC Barcelona Clinic Liver Cancer.
Response
Regarding serum AFP concentrations, 18 patients had a concentration of less than 25 ng/ml in the TR group, whereas the serum AFP concentration was no more than 25 ng/mL in all patients of the sandwich group (P = 0.038). The responses between groups are summarized in Table 2. The objective response rates in the TR group and the sandwich group were 66.7% and 90.9%, respectively (P = 0.225).
The Responses Between Groups According to mRECIST 1.0.

TACE Transarterial chemoembolization; 3DCRT 3-dimensional conformal radiotherapy.
Survival
All of the patients had intrahepatic or extrahepatic recurrence at the end of the last follow-up at September 2018, 2 of the patients were still alive. The Kaplan-Meier survival curves are shown in Figure 1. The median IPFS was 5.4 months (95% CI: 4.5 to 6.3) in TR group, whereas the median IPFS was 17 months (95% CI: 8.6 to 25.4) in the sandwich (Figure 1A, P = 0.018). The median PFS for patients treated with TACE followed by radiotherapy was 5.4 months (95% CI: 5.2 to 5.6) and for patients received sandwich treatment was 17 months (95% CI: 8.6 to 25.4) (Figure 1B, P = 0.008). The median OS in TR group and sandwich group was 13.5 months (95% CI: 7.1 to 19.9) and 29.2 months (95% CI: 17.5 to 47.8), respectively(Figure 1C, P = 0.011). The addition of TACE to TACE followed by 3DCRT for unresectable or ablative HCC significantly improved outcomes, compared to patients who did not receive additional anti-tumor treatment.

Kaplan-Meier Curves: (A) intrahepatic progression-free survival (IPFS), (B) progression-free survival (PFS), (C) overall survival (OS).
Factors Affecting Prognosis
The multivariate Cox analyses for the predictors of PFS, IPFS, and OS are summarized in Table 3. The results demonstrated that ECOG PS 1-2 (HR: 3.063, 95% CI (1.358-6.911), P = 0.007), 3 tumors (HR: 2.154, 95% CI (1.059-4.380), P = 0.034), and TACE followed by radiotherapy (HR: 2.516, 95% CI (1.136-5.570), P = 0.023) had poor IPFS. Male gender (HR: 4.380, 95% CI (1.151-16.666), P = 0.030), ECOG PS 1-2 (HR: 3.153, 95% CI (1.378-7.218), P = 0.007), 3 tumors (HR: 2.691, 95% CI (1.309-5.529), P = 0.007), and TACE followed by radiotherapy (HR: 2.637, 95% CI (1.182-5.880), P = 0.018) were poor prognostic factors for PFS. The presence of 3 tumors (HR: 2.533, 95 CI% (1.203-5.331), P = 0.014) was the only poor prognostic factor for OS. TACE followed by radiotherapy (HR: 2.232, 95% CI (0.991-5.027), P = 0.053) seemed to have no effect on OS.
Multivariate Analyses of Independent Prognostic Factors Affecting IPFS, PFS and OS.
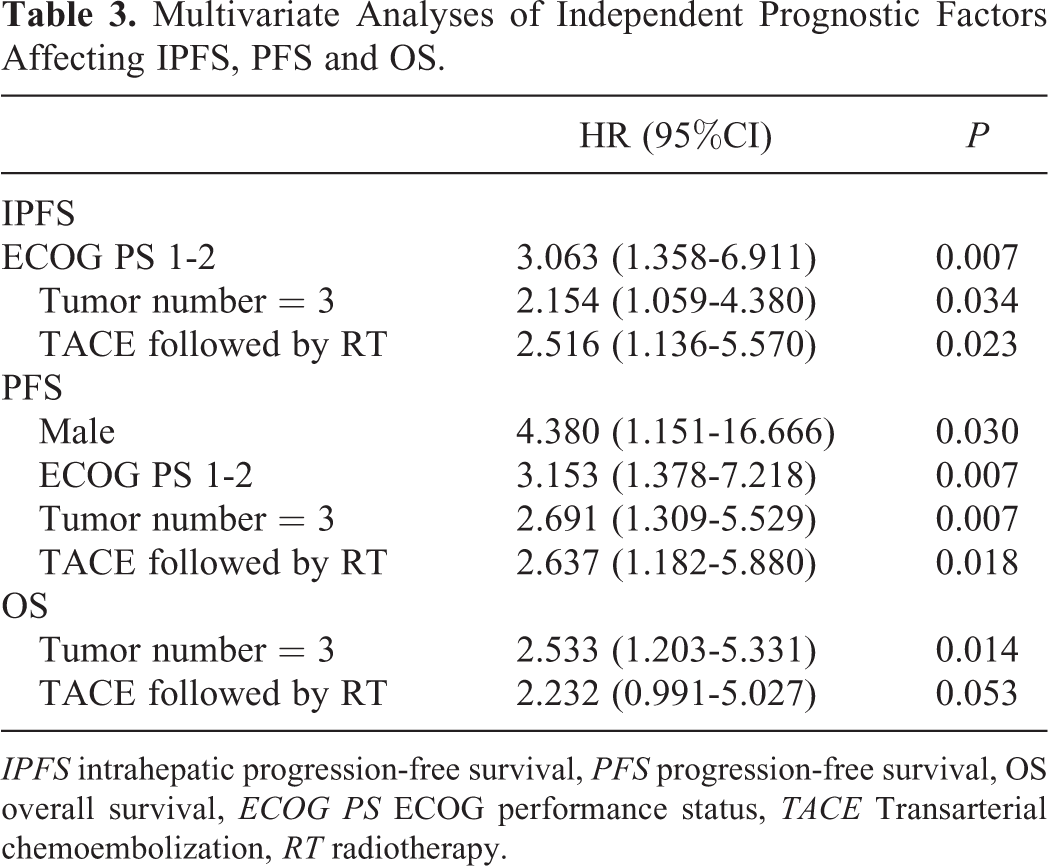
IPFS intrahepatic progression-free survival, PFS progression-free survival, OS overall survival, ECOG PS ECOG performance status, TACE Transarterial chemoembolization, RT radiotherapy.
Toxicities
After treatment completion, 2 patients had upper gastrointestinal hemorrhages within 3 months in the TR group. However, the hemorrhages were controlled through medical procedures, including proton pump inhibitors and pituitrin. Hepatitis B virus reactivation was observed in 4 of the patients in the TR group and one of the patients in the sandwich group. Other details concerning adverse events are summarized in Table 4. The presentation of grade 3/4 toxicity was due to myelosuppression, including thrombocytopenia (11.1% vs 0% in the TR group and sandwich group, respectively), anemia (7.4% vs 0%), neutropenia (3.7% vs 0%), and leukopenia (3.7% vs 9.1%). No RILD or treatment-related deaths were observed between groups.
Adverse Events During and/or After Treatment.
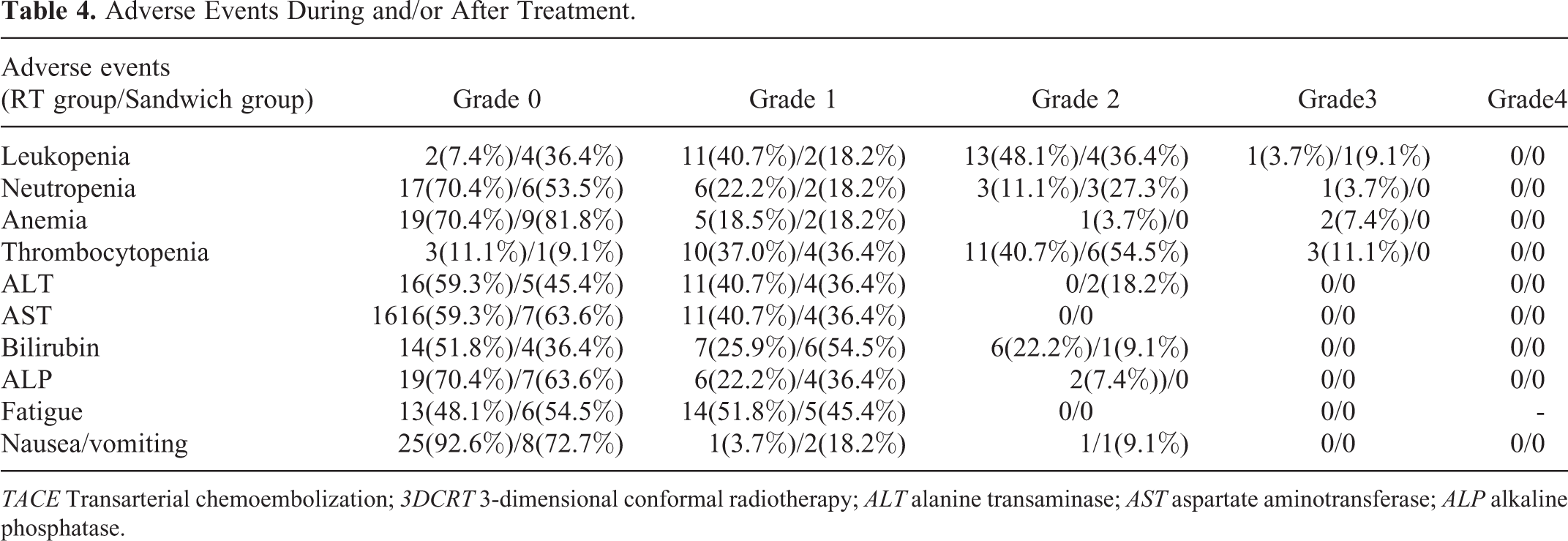
TACE Transarterial chemoembolization; 3DCRT 3-dimensional conformal radiotherapy; ALT alanine transaminase; AST aspartate aminotransferase; ALP alkaline phosphatase.
Discussion
The present study demonstrates that unresectable or ablative HCC patients may benefit from additional TACE therapy following the completion of combined TACE and 3DCRT therapy. The tolerable toxicity profiles were acceptable during and/or after the treatment.
Radiotherapy is an important treatment option in the comprehensive therapy of solid tumors, including HCC. Numerous studies have indicated that radiotherapy combined with TACE is an effective and safe treatment option for HCC. A prospective phase 2 multicenter study showed that the 2-year PFS and OS rates of 3D-CRT, following incomplete TACE in the treatment of unresectable HCC patients, were 29.0% and 61.3%, respectively. No classic radiation-induced liver disease and treatment-related deaths or hepatic failure were observed. 3 Another phase 2 trial indicated that the 2-year local control, overall survival, and progression-free survival rate of stereotactic body radiation therapy as a local salvage treatment for incomplete transarterial chemoembolization hepatocellular carcinoma were 94.6%, 68.7%, and 33.8%, respectively. However, grade 3 gastrointestinal toxicity and grade 4 gastric ulcer perforation were also observed. 12 Li and colleagues 13 reported that the combination of TACE with RT provides a significantly better survival outcome than TACE alone for unresectable HCC patients with PVTT, especially for patients with PVTT involving the right/left portal vein or the main trunk. Our study also indicated that patients who received TACE followed by 3DCRT seemed to experience worse toxicities compared with the sandwich treatment.
Recently, radiotherapy followed by TACE in the treatment of HCC was explored by several investigators. Lu et al 14 reported that 3DCRT followed by TACE may have a negative influence on liver function compared to TACE followed by 3DCRT alone. A propensity score matching study 15 indicated that compared with TACE followed by IMRT, IMRT followed by TACE yielded better survival rates and liver function in HCC patients with PVTT. Lu and colleagues 16 reported that regarding patients with HCC with PVTT, OS in patients who received 3DCRT followed by 2 to 3 cycles of TACE, as compared to those who received TACE alone, were 13 months and 9 months, respectively. Su and colleagues 17 reported that SBRT followed by TACE is superior to SBRT alone regarding OS. All of these results indicate that the addition of TACE following radiotherapy significantly prolonged OS compared to TACE alone.
However, it is unclear whether additional TACE after combination therapy could improve patient outcomes. The results of our study suggest that compared to TACE followed by 3DCRT alone, the addition of TACE after TACE followed by 3DCRT could significantly improve PFS and IPFS. The multivariable Cox regression analysis demonstrated that TACE followed by 3DCRT was a poor prognostic factor for PFS and IPFS compared with the sandwich treatment. The cell studies proved that a low dose of radiation exposure 18 or tumor hypoxia 19 could enhance the invasion and migration of HCC cells, which may be a factor of recurrence/relapse after TACE and radiotherapy. Our results showed that additional treatment with TACE can significantly reduce cancer recurrence, possibly due to severe hypoxia to the micrometastases. However, the results did not show significant improvement in OS when the additional TACE was added compared with 3DCRT followed by TACE alone. The reason for this phenomenon could be due to treatment starting after progression already occurred, or the number of patients reaching the final events at the last follow-up time.
Although esophageal gastric-fundus variceal hematemesis was observed in 2 patients in the TR group, medical professionals were able to control the hematemesis. The grade 3/4 toxicity was due to myelosuppression, which was observed in the TR group and not the sandwich group. This could be explained by preventative treatments or the limited number of patients in the sandwich group. No RILD or treatment-related deaths were observed.
Although the results indicate that the sandwich treatment seems superior to TACE followed by 3DCRT alone, several limitations exist in our study. First, although there was no significant difference in baseline characteristics of patients between groups, selection bias in our population is inevitable due to the nature of retrospective studies. Second, there was a limited sample size in both groups, especially in the sandwich group. Despite these limitations, this was the first study to report the efficacy and safety between TACE followed by 3DCRT alone and sandwich treatment for local intrahepatic hepatocellular carcinoma. A prospective randomized trial in the future with a considerable population is necessary to confirm our findings.
Conclusion
In general, our study demonstrated that unresectable or ablative HCC patients could benefit more from the combination of TACE and 3DCRT followed by additional TACE, compared with TACE followed by 3DCRT alone. Further clinical randomized trials are required to confirm these results.
Footnotes
Ethics
This study was approved by the ethics committee of Shandong Cancer Hospital and Institute (SDTHEC202001001).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Shandong Province (ZR2017MH115). All the authors listed have contributed to this study, approved the final manuscript for submission.
