Abstract
The annual incidence of papillary thyroid carcinoma has increased dramatically. T cell factor 4 (TCF4) is an important component of Wnt signaling pathway.However, the role of TCF4 in PTC remains unknown. In this study, TCF4 was observed to overexpress in PTC patients and cells by qRT-PCR assay. The colony formation assay, Edu staining and transwell assay indicated thatoverexpression of TCF4 promoted cell proliferation and invasion of TCP-1 cells, whereas knockdown of TCF4 inhibited cell proliferation and invasion of IHH-4 cells. To investigate the mechanism of TCF4 in PTC cells, the luciferase assay demonstrated that TCF4 could modulate HCP5 expression. Besides, GLuc-ON promoter reporter assayproved that TCF4 could bind to HCP5 promoter. Further, knockdown of HCP5 could significantly up-regulated miR-15a, miR-216a-5p, miR-22-3p, miR-139-5p, miR-203, miR-27a-3p and miR-320, and down-regulated miR-186-5p in IHH-4 cells, which might be potential downstream of TFC4/HCP5 axis. In conclusion, up-regulation TCF4 can promote HCP5 expression via binding to HCP5 promoter. It may be the first time to prove that TCF4 regulates HCP5 in PTC, which provides a novel sight for treatment of PTC.
Introduction
Thyroid cancer is the most common endocrine system cancer, which can be divided into 4 types, papillary thyroid carcinoma (PTC), follicular thyroid cancer (FTC), medullary thyroid cancer (MTC), and anaplastic thyroid cancer (ATC). 1-2 PTC, accounting for ∼80% of all thyroid cancers, the annual incidence has increased dramatically. 2 Besides, the poor prognosis of PTC is closely associated with clinical characteristics, such as tumor size and lymph node metastasis. 3-4 PTC tumors often have a strong ability to metastasize, and can spread to locoreginal lymph nodes, leading to death. Therefore, it is necessary to find effective diagnostic markers and therapeutic targets for PTC.
T cell factor 4 (TCF-4) is a vital member of TCF-4/LEF gene family in thyroid. 5 TCF4plays an important role in downstream of Wnt signaling pathway, which mainly regulates the transcription of target genes. 6 -8 Recent studies have reported that TCF4 expression is significantly enhanced in lung cancer, kidney cancer, colon cancer, gastric cancer, liver cancer and other tumors, and the stimulation of downstream target gene expression by abnormally elevated TCF4 is a common early event during the occurrence of tumors. 9 -12 More importantly, the expression of TCF4 has been observed up-regulated in PTC tissues, while the role and underlying mechanism of TCF4 in PTC needs to be elucidated. 5
Accumulating evidence has shown that long non-coding RNAs (lncRNAs) are crucial regulators in many cancers, especially PTC. 13 LncRNA H19 and HOTAIR are overexpressed in thyroid cancer cells and promote proliferation, migration and invasion. 14-15 HCP5 is a novel lncRNA and its investigations about cancers are limited. A recent study has reported that a single-nucleotide polymorphism (SNP; rs2244546) in HCP5 is considered to be a susceptibility locus for hepatocellular carcinoma. 16 HCP5 is upregulated in FTC tissues and promotes tumor progression. 17 LncRNA HCP5 is regarded as a new clinical thyroid disease-related susceptibility gene, and it promotes follicular thyroid cancer progression via sponging miRNAs. 18-19 However, the relationship between TCF4 and HCP5 remains unknown.
Firstly, the lncRNA HCP5 was selected which could respond to TCF4 expression. Predicted by bioinformatics analysis, TCF4 could bind to HCP5 promoter. In this study, we demonstrated that the expression of TCF4 was increased in PTC tissues and cells, accompanied by increase HCP5 expression. Thus, the potential relationship between HCP5 and TCF4 can be illustrated, which may provide a new sight for PTC treated strategies.
Method and Materials
Patients Samples
Fifty-two pairs of PTC tissues and paired normal tissues were obtained from XXX Hospital. This study was based on the principle of the World Medical Association Declaration of Helsinki and approved by the Ethics Committee ofXXX Hospital. All the patients have written informed consent. The patients’ characteristics were recorded in Table 1. All the patients were not treated with radiotherapy or chemotherapy before surgery and were immediately stored in liquid nitrogen after surgery.
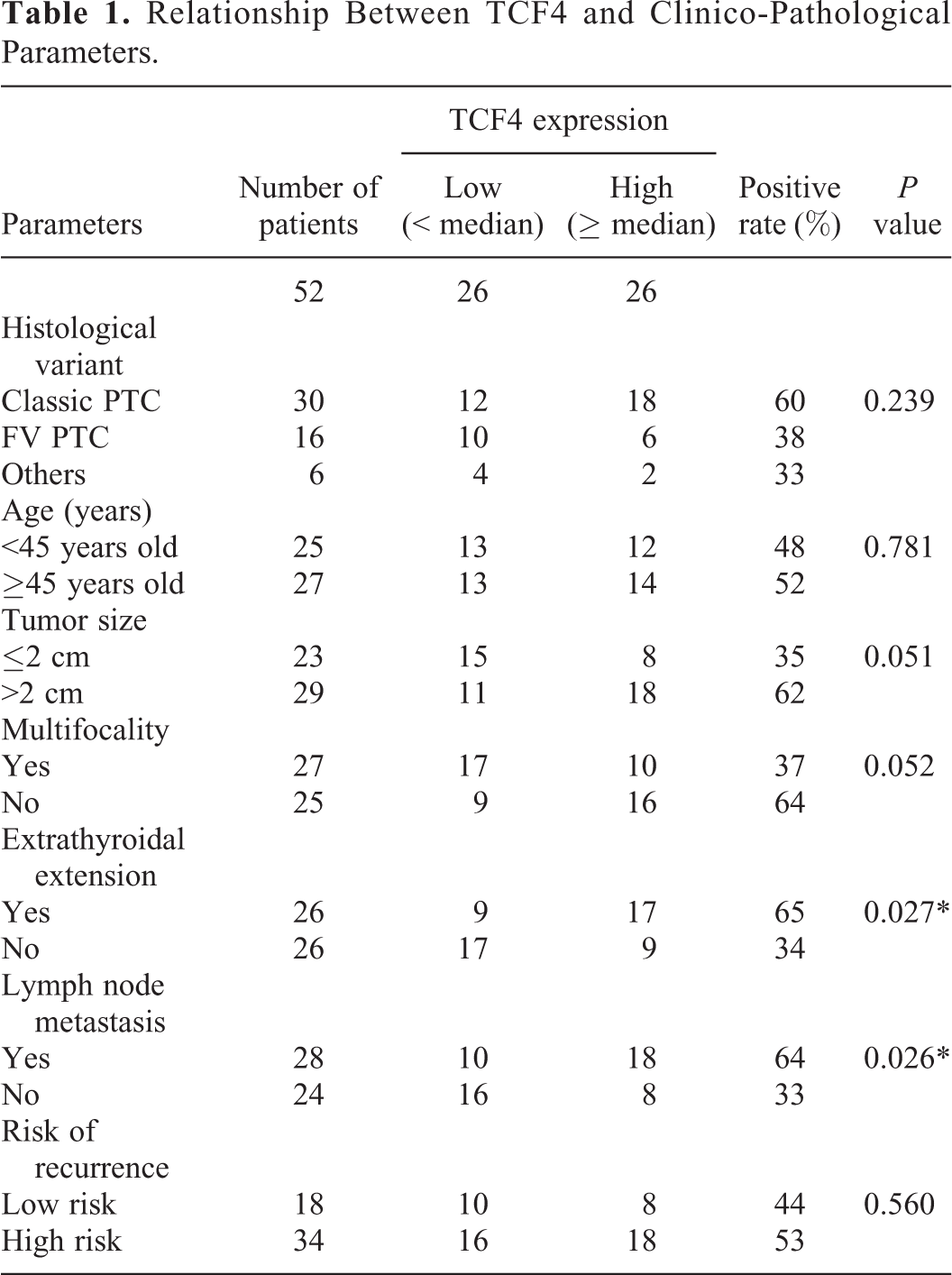
Relationship Between TCF4 and Clinico-Pathological Parameters.
Cell Culture and Transfection
The human Nthy-ori 3-1, IHH-4 and TPC-1 cells were obtained from the American Type Culture Collection (ATCC;USA) and supplemented with Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS; Gibco, USA).HTori-3 cells were purchased from ATCC and cultured in F-12 K medium (ATCC, USA) containing 10% FBS. SNU790 cells (Cobioer, China) were cultured in RPMI-1640 (Gibco, USA) medium supplemented with 10%FBS. Cells were incubated at 37°C in a humidified atmosphere containing 5% CO2.
The shTCF4#1(5’-GAACGGAGGATGGCCAATAAT-3’) or shTCF4#2 (5’-GGTCAAGATCTAGCAATAACG-3’) was inserted into pSUPER plasmid to knockdown TCF4. The full length of TCF4 was inserted into pcDNA3.1 plasmid to overexpress TCF4. The recombinant plasmid shTCF4#1 or shTCF4#2 was transfected into IHH-4 cells and the pcDNA3.1-TCF4 was transfected into TPC-1 cells.
The shNC, shHCP5#1 and shHCP5#2were purchased from GenePharma (Shanghai, China) and transfected into IHH-4 cells by Lipofectamine® 2000 (Invitrogen, USA).
Immunohistochemistry (IHC) Assay
Human PTC tissuesand paired normal tissues were embedded in paraffin blocks, cut into 4-µm serial sections and incubated with TCF4 antibody (Abcam, UK). Immune complexes were visualized using HRP Rabbit Anti-Goat (IgG)(Abcam, UK) and DAB substrate kit (Abcam, UK). Hematoxylin slightly reversed staining of the nucleus and sections were mounted.
Colony Formation Assay
After transfection, 1 × 103 cells were seeded in six-well plates. Then the cells were mixed and cultured for 10 days. Clusters more than 40 cells were regarded as a single colony.
Ethynyldeoxyuridine (Edu) Analysis
The cells were cultured in a 96-well plate at a final density of 1 × 104 cells/well. After 48 h of transfection, 20 μM Edu-labeled medium (NanJingKeyGenBiotech, China) were added into 96-well plates and incubated at 37°C and 5% CO2 for 2 h. After treated with 4% paraformaldehyde and 0.5% Triton X-100, the cells were dyed with anti-EDU solution. The cell nucleus were stained with DAPI. The EDU positive cells were calculated by fluorescence microscopy.
Transwell Assays
After transfection, the cells were cultured in the upper chamber of a Transwell membrane filter (Corning, USA) coated with Matrigel (Corning) for invasion test.A medium containing 10% FBS was added to the lower compartment of the compartment. After incubation for 24 hours, the cells were stained with methanol and 0.1% crystal violet and counted with an Olympus microscope (Tokyo, Japan).
GLuc-ON Promoter Reporter Assay
Cells were co-transfected with the plasmids of the GLuc-HCP5 promoter (Promoter-FL, Promoter#1-4) and Flag-TCF4. Based on manufacturer’s protocols, the relative GLuc activitywas performed using a Secrete-Pair Dual Luminescence Assay Kit (GeneCopoeia, USA).
Western Blot
Total proteins from cells were extracted using lysis buffer (Pierce, USA) and the protein concentration was detected by a BCA protein assay kit (Shzysw Biomart, China). Equal protein was separated with SDS-PAGE, transferred to PVDF membranes (Millipore, USA). Then, the membranes were blocked with 5% skim milk and incubated with TCF4 antibody (dilution 1:500; Abcam, UK) or β-actin (dilution 1:1000; Abcam, UK). After incubation with Goat Anti-Mouse IgG H&L (HRP) or Goat Anti-Rabbit IgG H&L (HRP) (dilution 1:5000; Abcam, UK) secondary antibody for 2 h, the membranes were detected using an EfficientChemiluminescence (ECL) kit (Proandy, China).
qRT-PCR Assay
Total RNA was extracted from cells using RNA simple kit (Tiangen, China). The cDNA was generated by Tianscipt RT kit (Tiangen, China). The qRT-PCR was carried out with the ABI 7000 (USA) using FastQuant RT Super Mix (Tiangen, China). Each sample was performed in triplicate. Gene regulation was determined by the 2−ΔΔCt method and normalized or compared to U6 or β-actin levels. The primers were used as follows: TCF4-F: 5’-GACCTTTGCTCTCATTTCC-3’, TCF4-R: 5’-CCACATCATACGCTACACAC-3’; β-actin-F: 5’-AAAATGGCAGTGCGTTTAG-3’, β-actin-R: 5’-TTTGAAGGCAGTCTGTCGTA-3’; HCP5-F: 5’-CCGCTGGTCTCTGGACACATACT-3’, HCP5-R: 5’-CTCACCTGTCGTGGGATTTTGC-3’; U6-F: 5’-AAAGCAAATCATCGGACGACC-3’, U6-R: 5’-GTACAACACATTGTTTCCTCGGA-3’; miR-15a-F: 5’-GGGTAGCAGCACATAATGGTTTG-3’, miR-15a-R: 5’-GCGAGCACAGAATTAATACGACTC-3’; miR-216a-5p-F: 5’-TGTCGCAAATCTCTGCAGG-3’, miR-216a-5p-R: 5’-CAGAGCAGGGTCCGAGGTA-3’; miR-186-5p-F: 5’-AAGAATTCTCCTTTTGGGCT-3’, miR-186-5p-R: 5’-GTGCGTGTCGTGGAGTCG-3’; miR-22-3p-F: 5’-AAGCTGCCAGTTGAAGAACTGTA-3’, miR-22-3p-R: 5’-GCTGTCAACGATACGCTACGTAAC-3’; miR-139-5p-F: 5’-TCTACAGTGCACGTGTC-3’, miR-139-5p-R:5’-GAATACCTCGGACCCTGC-3’; miR-219-5p-F: 5’-GGTGATTGTCCAAACGG-3’, miR-219-5p-R: 5’-CAGTGCGTGTCGTGGA-3’; miR-203-F: 5’-ACACTCCAGCTGGGGTGAAATGTTTA-3’, miR-203-R: 5’-TGGTGTCGTGGAGTCG-3’; miR-140-5p-F: 5-’TGCGGCAGTGGTTTTACCCTATG-3’, miR-140-5p-R: 5’-CCAGTGCAGGGTCCGAGGT-3’; miR-27a-3p-F: 5’-CGCGTTCACAGTGGCTAAGT-3’, miR-27a-3p-R: 5’-GTGCAGGGTCCGAGGTATTC-3’; miR-320-F: 5’-TGCGGAAAAGCTGGGTTGAGAG-3’, miR-320-R: 5’-CCAGTGCAGGGTCCGAGGT-3’.
Statistical Analysis
The data in each group were presented with mean and standard deviation (SD) of 3 replicates. Student’s t-test and One-way analysis of variance (ANOVA) were used to compare values. The p values less than 0.05 indicated statistical significance. All calculations were performed with GraphPad Prism 7.0 (GraphPad, USA).
Results
TCF4 Is Overexpressed in PTC Patients and Cells
To explore the role of TCF4 in PTC, a qRT-PCR analysis was applied to evaluate the expression of TCF4 in PTC patients and cells lines. As expected, TCF4 was upregulated in PTC tissues (n = 52) compared to adjacent normal tissues (n = 52) (Figure 1A). Consistent with this result, the level of TCF4 was higher in PTC cell lines (IHH-4, TPC-1 and CGTH-W3 cells) compared to Nthy-ori 3-1 andHTori-3 cells (Figure 1B). In accordance with the results above, IHC assay confirmed that TCF4 expression was enhanced in PTC tissues, compared to adjacent normal tissues (Figure 1C). Moreover, the median value of TCF4 expression was served as the cutoff value, all the PTC patients were divided into 2 groups (TCF4 high expression and TCF4 low expression). The relationship between TCF4 expression and clinical features was analyzed, indicating that higher expression of TCF4 was significantlyassociated with extrathyroidal extension (p = 0.027) and lymph metastasis (p = 0.026) (Table 1). Thus, these findings concluded that TCF4 was overexpressed in PTC patients and cells.
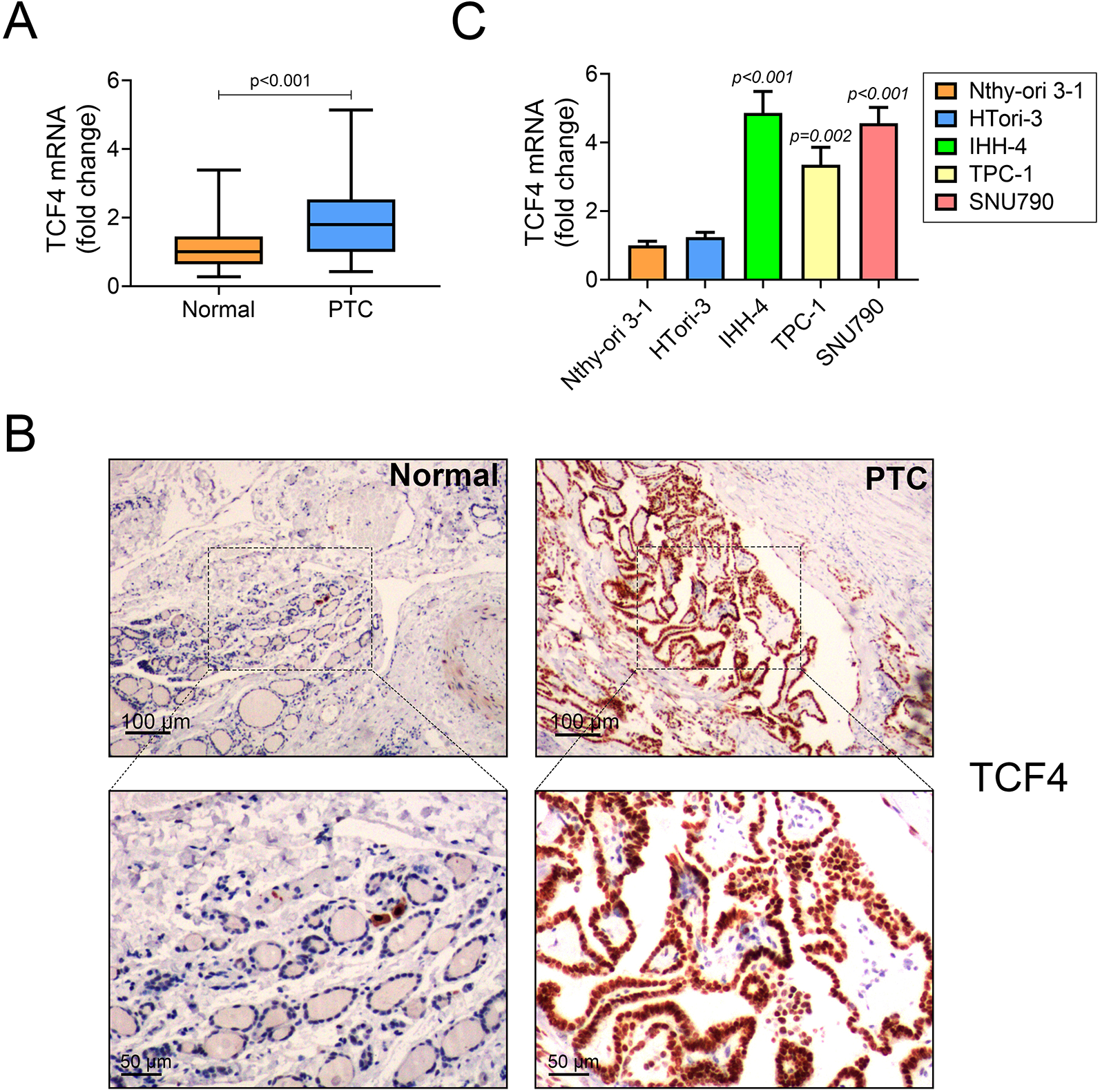
TCF4 is overexpressed in PTC patients and cells.
Knockdown of TCF4 Inhibits Cell Proliferation and Invasion in PTC Cells
To further verify the function of TCF4 in PTC, the shTCF4#1 and shTCF4#2 were transfected into IHH-4 cells to knockdown the expression of TCF4. Moreover, the TCF4 was inserted into Vector and transfected into TPC-1 cells to enhance the expression of TCF4 (Figure 2A).Consistent with these results, the expression of TCF4 was determined by western blot, suggesting that shTCF4#1 and shTCF4#2 reduced TCF4 protein levels in IHH-4 cells and TCF4 recombinant plasmid increased TCF4 protein levels in TPC-1 cells (Figure 2B). Then, the role of TCF4 in proliferation was detected by using a colony formation assay and an Edu assay. The colony formation assay showed that knockdown of TCF4 inhibited cell proliferation but overexpression of TCF4 promoted cell proliferation (Figure 2C). Similarly, the Edu assay confirmed that knockdown of TCF4 decreased the Edu-positive IHH-4 cells, whereas overexpression of TCF4 increased Edu-positive TPC-1 cells (Figure 2D). As shown in Figure 2E, IHH-4 cell invasion was down-regulated in knockdown of TCF4, while the opposite result was observed in overexpressing TCF4 in TPC-1 cells. Therefore, these results concluded that knockdown of TCF4 inhibited cell proliferation and invasion.

TCF4 inhibits cell proliferation and invasion in PTC cells. The mRNA levels (
TCF4 Regulates the Expression of lncRNA HCP5
To investigatethe mechanism of TCF4 in lncRNA HCP5, the expression of HCP5 were detected, indicating that knockdown of TCF4 decreased HCP5 expression in IHH-4 cells, and overexpression of TCF4 increased HCP5 expression in TPC-1 cells (Figure 3A). Moreover, as shown in Figure 3B, we constructed plasmids of the GLuc-HCP5 promoter of either full-length [FL, −2000 bp ∼ TSS (transcription start site)] or different truncations (#1 of −1500 bp ∼ TSS, #2 of −1000 bp ∼ TSS, #3 of −500 bp ∼ TSS, #4 of −2000 ∼ −500 bp) and measured with GLuc-ON Promoter Reporter assays. The high expression of TCF4induced an increased relative Gluc activity of GLuc-HCP5 promoter FL and #1 in TPC-1 cells, with no changes in #2, #3 or #4. This suggested that TCF regulated HCP5 expression via recognizing −1500 bp ∼ −1000 bp region of HCP5 promoter. These results suggest that TCF4 regulates the expression of lncRNA HCP5 in transcriptional level.

TCF4 regulates the expression of lncRNA HCP5
Overexpression of HCP5 Abolished shTCF4-Induced Changes in IHH-4 Cells
To investigate the regulatory mechanism of TCF4 potential functions inIHH-4 cells, the rescue experiments are necessary. As Figure 4A shown, the expression of HCP5 was significantly decreased in IHH-4 cells after transfection with shTCF4. Compared to the shTCF4+vector group cells, overexpression of HCP5 increased HCP5 expression (Figure 4A). In Figure 4B and 4C, the colony formation and Edu assay indicated that the cell proliferation was attenuated in shTCF4+vector transfected-cells compared to the shNC+vector group cells. And this inhibition could be partially rescued by overexpression of HCP5. More importantly, the invasion of shTCF4-induced IHH-4 cells was markedly promoted by co-transfection with overexpression of HCP5 vector (Figure 4D). The results indicated that knockdown of TCF4 inhibited cell proliferation and invasion, whereas overexpression of HCP5 abolished shTCF4-induced changes in IHH-4 cells. Therefore, these findings concluded that knockdown of TCF4 inhibited cell proliferation and invasion via modulating HCP5 expression.

Overexpression of HCP5 abolished shTCF4-induced changes in IHH-4 cells. The vector or HCP5 with shTCF4 or shNC were co-transfected into IHH-4 cells
The Expression Pattern of Potential Targets of HCP5
LncRNA HCP5 participates in cancers via serving as a ceRNA and sponging miRNAs. 20 To speculate the potential target of HCP5, a series expression of potential target miRNAs was detected.As Figure 5 illustrated, knockdown of HCP5 could significantly up-regulate miR-15a, miR-216a-5p, miR-22-3p, miR-139-5p, miR-203, miR-27a-3p and miR-320, and down-regulated miR-186-5p in IHH-4 cells, with no significance change in miR-219-5p and miR-140-5p. These findings might provide novel sights for investigating the downstream of TFC4/HCP5 axis in PTC.

The expression pattern of potential targets of HCP5. After transfected with shNC, shHCP5#1 or shHCP5#2 into IHH-4 cells, the expression of miR-15a, miR-216a-5p, miR-22-3p, miR-139-5p, miR-203, miR-27a-3p, miR-320, miR-186-5p, miR-219-5p and miR-140-5p were determined by qRT-PCR assay. Every experiment repeated 3 times.
Discussion
In this study, TCF4 was observed to overexpress in PTC patients and cells. Moreover, overexpression of TCF4 promoted cell proliferation and invasion of IHH-4 and TCP-1 cells, whereas knockdown of TCF4 inhibited cell proliferation and invasion of IHH-4 and TCP-1 cells. To investigate the mechanism of TCF4 in PTC cells, we demonstrated that TCF4 could modulate HCP5 expression. Besides, TCF4 was proved to bind to −1500∼ −1000 bp region of HCP5 promoter. Further, knockdown of HCP5 could significantly up-regulated miR-15a, miR-216a-5p, miR-22-3p, miR-139-5p, miR-203, miR-27a-3p and miR-320, and down-regulated miR-186-5p in IHH-4 cells, which might be potential downstream of TFC4/HCP5 axis. It may be the first time to prove that TCF4 regulates HCP5 in PTC, which provides a novel sight for treatment of PTC.
PTC is one of the most common thyroid malignant tumors, characterized by rapid growth and metastasis, frequent recurrence and high mortality. This current study focused on the role of TCF4 in regulating the proliferation and invasion of PTC.TCF4 is a transcription factor and involved in the canonical β-catenin-mediated Wnt signaling. 21 A recent study has reported that overexpression of TCF4 significantly suppresses cell proliferation in sonic hedgehog medulloblastoma in vitro. 22 Mechanically, TCF4 knockdown causes differentially expressed and is highly involved in the cell cycle. 23-24 Besides, TCF4 isnegatively regulated by miR-137 and affects colon cancer cells migration and invasion. 25 The inhibition of TCF-4 transcription activity suppresses cell proliferation and invasion, induces cell cycle arrest and promotes apoptosis in glioma. 26 Fortunately, we investigated that knockdown of TCF4 inhibited cell proliferation and invasion but overexpression of TCF4 promoted them in PTC cells, indicating the vital role of TCF4 in PTC. It might be the first time to explore the inhibitory function of TCF4 in PTC cell proliferation and invasion.
As an important component Wnt signaling, it participates in Wnt/β-catenin signaling; as a transcript factor, it can bind to promoters to enhance or reduce the target molecules expression. TCF4 has been observed to directly bind to 1.5 kb of the promoter region of FGF21 in stomach cancer cells. 27 TCF4/β-catenin complex can bind to 1.5 kb of the Smo and Gli1 promoters in human fibroblasts. 28 Despite of the regulatory function of target proteins expression, TCF4 can regulate lncRNAs expression. TCF-4 can modulate lncRNA-XIST and promote M2 polarization of macrophages, thus induce lung cancer. 29 Besides, TCF-4 is observed to bind to the LINC01278 promoter and participates in hepatocellular carcinoma (HCC) metastasis. 30 In this study, TCF-4 could bind to HCP5 promoterregion and regulated HCP5 expression in IHH-4 cells. It might provide a new sight for investigating the function of TCF4 in PTC. In addition, Huang et al. have proved that β-catenin/TCF-4-LINC01278-miR-1258-Smad2/3 axis promotes hepatocellular carcinoma metastasis. 30 We hypothesized that whether there is a similar mechanism of TCF-4-HCP5 axis in PTC.
HCP5 promotes FTC progression via miRNAs sponge. 19 More importantly, HCP5 serves as a ceRNA and promotes breast cancer and pancreatic cancer progression by sponging miR-219a-5p and miR-214-3p. 20, 31 By the way, it has been reported that knockdown of HCP5 plays a tumor suppressive role by upregulation of miR-128-3p in anaplastic thyroid cancer. 32 Furthermore, miR-128-3p simultaneous over-activates Wnt/β-catenin and TGFβ signaling, eventually, can induce metastasis in non-small cell lung cancer. 33 Importantly, antagonism of miR-128-3p potently reverses metastasis of NSLC. 33 Thus, we can suspect that TCF4 could increase the HCP5 expression and then decrease the miR-128-3p in PTC cells, which needs more experiments in the future. Also, miR-128-3p suppresses breast cancer progression via targeting LIMK1. 34 Elevation of miR-128-3p inhibits apoptosis and activates Wnt/β-catenin signaling by targeting Axin1 in Parkinson’s disease. 35 Nevertheless, overexpression of miR-128-3p suppresses proliferation, migration, invasion, and inhibit Wnt signaling by targeting NEK2 in breast cancer. 36 Considering these findings, the potential role of TCF4/HCP5/miR-128-3p axis and the hypothesis need to be proved in PTC cells in the future. Besides, we examined other potential targets of HCP5, which can be confirmed in future studies.
In conclusion, up-regulation TCF4 can promote HCP5 expression via binding to −1500∼ −1000 bp region of HCP5 promoter. Our results showed that the “TCF4/HCP5” axis promotes PTC cells proliferation and invasion, though the complete axis need to be improved in the future. Above all, TCF4 could serve as a new therapeutic target for PTC.
Footnotes
Abbreviations
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Authors’ Contributions
Rui Wang and Jidong Cai designed the study, supervised the data collection, Shangnao Xie and Chunlei Zhao analyzed the data, interpreted the data, Yi Wang, Deming Zhao and Gang Li prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Our study was approved by the Ethics Committee of Hangzhou Cancer Hospital (approval no. HZCH-2017-08). All patients provided written informed consent prior to enrollment in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Key Project of Science and Technology Planning of Health in Hangzhou (Grant No. 2018ZD02).
