Abstract
Objective:
This study aimed to investigate the correlation of L-asparaginase (L-asp) activity, anti-L-asp antibody, asparagine and glutamine levels with the risks of adverse events (AEs), especially anaphylaxis, in pediatric acute lymphoblastic leukemia (ALL) patients who underwent polyethylene glycol-conjugated L-asp (PEG-asp)-contained treatment.
Methods:
Plasma samples were collected from 91 pediatric ALL patients who underwent PEG-asp-contained treatment on the 7th day after drug administration. Plasma L-asp activity, anti-L-asp antibody level, asparagine level and glutamine level were detected. Meanwhile, AEs related to PEG-asp administration were recorded.
Results:
AEs occurred in 13 (14.3%) patients, among which 7 (7.7%) patients had anaphylaxis, while another 6 patients had non-anaphylaxis AEs (including 4 (4.4%) patients who had acute pancreatitis, 1 (1.1%) patient who had abdominal pain and diarrhea, as well as 1 (1.1%) patient who had nausea and vomiting). L-asp activity was decreased, while asparagine and glutamine levels were increased in patients with AEs compared to patients without AEs, and ROC curves showed that they were correlated with higher AEs risk. Notably, further analyses revealed that L-asp activity, anti-L-asp antibody, asparagine and glutamine levels were highly correlated with anaphylaxis risk, but they were not associated with the risk of non-anaphylactic AEs.
Conclusion:
The measurement of L-asp activity, anti-L-asp antibody level, asparagine level and glutamine level might assist the prevention of anaphylaxis-related AEs in pediatric ALL patients who underwent PEG-asp-contained treatment.
Keywords
Introduction
Pediatric acute lymphoblastic leukemia (ALL), one of the most common pediatric malignancies, accounts for approximately 75%-80% of pediatric acute leukemia. 1 Meanwhile, ALL is also the top cause of death from cancer in patients less than 20 years of age. 1 In the past 50 years, the induction of chemotherapy protocol for pediatric ALL has greatly improved the survival of pediatric ALL patients, among which L- asparaginase (L-asp) is a key component. 2,3
Currently, polyethylene glycol conjugated L-asp (PEG-asp) is vastly administrated in pediatric ALL patients; it possesses longer half-life and is less likely to induce hypersensitivity reactions compared to native L-asp derived from Escherichia coli or Erwinia. 4 By specifically catalyzing the hydrolysis of the amide bond of asparagine (Asn), PEG-asp is able to deplete Asn level in the blood, and the latter one is critical for the protein synthesis of several malignancies including ALL; thus, PEG-asp exerts therapeutic property in pediatric ALL patients. 5,6 Besides, PEG-asp is also able to hydrolyze glutamine (Gln), but with relatively low activity and binding affinity; therefore, PEG-asp would hydrolyze Gln after the depletion of Asn, which is closely related PEG-asp treatment-related AEs in pediatric ALL patients including pancreatitis, damage in liver function, etc. 7 Meanwhile, the administration of PEG-asp might induce the production of anti-L-asp antibody, which could reduce the treatment efficacy of PEG-asp, causes hypersensitivity response that leads to anaphylaxis, and is correlated with worse prognosis in high risk pediatric ALL patients. 8 In our preliminary study (not reported), we found L-asp activity was reduced, while anti-L-asp antibody, Asn and Gln were elevated in ALL patients who occurred AEs. Based on the above-mentioned information, we hypothesized that PEG-asp activity, the levels of its antibody and substrates in pediatric ALL patients might be indicators of AEs (especially anaphylaxis) during the administration of PEG-asp. However, relevant information is rare.
In this study, we aimed to investigate the correlation of PEG-asp activity, anti-L-asp antibody level, Asn level and Gln level with AEs (especially anaphylaxis) in pediatric ALL patients who underwent PEG-asp-contained treatment, thus providing evidence for potentially predicting anaphylaxis-related AEs in these patients to improve their individualized treatment.
Materials and Methods
Patients
Between Dec 2019 and Feb 2020, 91 pediatric ALL patients admitted to our hospital were enrolled in this study. The inclusion criteria were as follows: (1) diagnosed as ALL according to the guidelines for the diagnosis and treatment of childhood acute lymphoblastic leukemia 9 ; (2) age within 1 month to 18 years; (3) about to receive PEG-asp contained Chinese Children’s Cancer Group (CCCG)-ALL-2015 therapy regimen. The exclusion criteria were: (1) concomitant with immunodeficiency disease; (2) complicated with other malignancies. This study was approved by the Ethics Committee of our hospital with an ethical approval number of SCMCIRB-K2017065. All guardians of pediatric patients signed the informed consents before enrollment.
Clinical Data Collection
After enrollment, patients’ clinical characteristics were documented, which included age, gender, allergic history, disease status, immunophenotype as well as risk stratification which was evaluated in accordance with the guideline. 9
Treatment
All patients received the CCCG-ALL-2015 regimen, and the detailed process of drug management was described in the CCCG-ALL-2015 study. 10 In the CCCG-ALL-2015 regimen for induction remission therapy, PEG-asp injection (Jiangsu Hengrui pharmaceutical Co. LTD, Lianyungang, Jiangsu, China; National Medicine Approval Number: H20090015) was administered at the dose of 2000 U/m2 by intramuscular injection on day 6 and day 26.
Blood Sample Collection
Peripheral venous blood of 1 mL was collected on the 7th day after the use of PEG-asp, 11 and heparin was used for anticoagulation (100 U/mL). The plasma was separated by centrifugation at 850g within 20 minutes and stored in a refrigerator at -70 °C for the measurement of L-asp activity, Anti-L-asp antibody, Asn level, and Gln level.
Plasma L-asp Activity Measurement and Drug Adjustment
The L-asp activity in the plasma was measured using the method described in a previous study. 12 Briefly, in each test tube, 40 µL plasma or standard solution (100, 200, 500, 1000, 2000, 4000, 6000 IU/L) or blank control solution, 110 µL 0.05 mol/L Tris-HCl (pH 8.5) buffer and 50 µL 0.04 mol/L Asn solution were added, respectively, and were incubated in 37°C water for 30 min. The reaction was terminated by adding 50 µL 15% trichloroacetic acid. After centrifugation, 200 µL supernatant was collected and diluted with 650 µL double-distilled water followed by adding 100 µL Nessler’s reagent and standing for 15 minutes at room temperature. After that, the optical density (OD) value at 405 nm of the solution was measured by a microplate reader (Infinite F50, TECAN, Männedorf, Switzerland), and a standard curve was obtained. Finally, the OD value of the sample was substituted into the standard curve to calculate the corresponding drug activity. For patients with plasma drug activity <100 IU/L, it was suggested to switch from PEG-asp to Erwinia L-asp (Erw-asp injection). As for patients with the occurrence of AEs, drug discontinuance or switching to Escherichia coli L-asp (E. coli-asp) or Erw-asp injection was conducted according to the specific status.
Plasma Anti-L-asp Antibody, Asn, and Gln Measurement
The anti-L-asp antibody in plasma was determined by enzyme-linked immunosorbent assay (ELISA) with the use of ELISA kit (Shanghai Enzymatic Biotechnology Co., LTD., Shanghai, China) according to the manufacturer’s manual. The Asn and Gln levels in plasma were measured by AB-QTRAP 4000 (Applied Biosystems Inc, Massachusetts, Waltham, USA) and Agilent C18 chromatographic column (Agilent, California, Santa Clara, USA) using liquid chromatography-tandem mass spectrometry (LC-MS/MS) method. Briefly, 50 µL plasma sample was mixed with 150 µL methanol solution containing cycloserine (500 ng/mL). After vortex mixing, it was centrifuged for 10 min at high speed (10000 rpm). Next, 100 µL supernatant was collected, then 100 µL triple distilled water was added. After vortex mixing, 5 µL sample was injected for detection.
AEs Recording
Referring to the adverse reactions in the pharmaceutical instructions of PEG-asp, any adverse reactions associated with the use of PEG-asp during treatment were recorded in detail, including anaphylaxis, pancreatitis, thrombosis, gastrointestinal reactions, etc.
Statistical Analysis
SPSS 22.0 statistical software (IBM, Chicago, Illinois, USA) was used for statistical data processing. GraphPad Prism 7.02 (GraphPad Software Inc., San Diego, California, USA) was used for figure plotting. Continuous data were described as mean with standard deviation (SD) or median with interquartile range (IQR), and categorical data were presented as number and percentage (No. (%)). Comparison of continuous data between the 2 groups was determined by the Wilcoxon rank-sum test. Correlation between 2 continuous variables was determined by spearman’s rank correlation test. Receiver operating characteristic (ROC) curve analysis was used to assess the performance of variables in predicting AEs risk, anaphylaxis risk, or non-anaphylactic AEs risk. P value <0.05 was considered as statistically significant.
Results
Description of Patients’ Characteristics
The pediatric ALL patients had a mean age of 5.7 ± 3.5 years, meanwhile, 58 (63.7%) of them were male and 33 (36.3%) of them were female. Regarding disease status, 78 (85.7%) patients were newly diagnosed as ALL, and 13 (14.3%) patients were relapsed ALL. As to immunophenotype, 82 (90.1%) patients were B-ALL, while 9 (9.9%) patients were T-ALL. For risk stratification, 53 (58.2%) patients were of low risk, 34 (37.4%) patients were of intermediate risk and 4 (4.4%) patients were of high risk. The detailed characteristics of pediatric ALL patients were shown in Table 1.
Characteristics of Pediatric ALL Patients.

ALL, acute lymphoblastic leukemia; SD, standard deviation; B-ALL, B-cell acute lymphoblastic leukemia; T-ALL, T-cell acute lymphoblastic leukemia.
Description of AEs
During treatment, a total of 13 (14.3%) pediatric ALL patients had AEs. Specifically, 7 (7.7%) patients had anaphylaxis, among which 4 (4.4%) patients had rash, 1 (1.1%) patient had anaphylactic shock, 1 (1.1%) patient had shortness of breath and dyspnea and 1 (1.1%) patient had complex anaphylactic reactions (Table 2). Meanwhile, 4 (4.4%) patients had acute pancreatitis, 1 (1.1%) patient had abdominal pain and diarrhea, and 1 (1.1%) patient had nausea and vomiting (Table 2).
Adverse Events (AEs).

* Complex anaphylactic reactions including shortness of breath, cyanosis of the lip, and decreased blood oxygen saturation. AEs, adverse events.
L-asp Activity, Anti-L-asp Antibody Level, Asn Level and Gln Level in Predicting AEs
According to the occurrence of AEs in pediatric ALL patients, they were divided into AE patients (n = 13) and non-AE patients (n = 78) for further analyses. L-asp activity was reduced in AE patients compared to non-AE patients (P = 0.006) (Figure 1A), and it was correlated with increased risk of AEs (area under the curve (AUC): 0.736, 95% confidence interval (CI): 0.539-0.923) with L-asp activity value at the best cut-off point (the point on the ROC curve at which the sum of sensitivity and specificity was maximized) of 267.6 IU/L, meanwhile, the sensitivity and the specificity at the best cut-off point was 69.2% and 84.6%, respectively (Figure 1B).

Correlation of L-asp activity, anti-L-asp antibody level, Asn level and Gln level with AEs in pediatric ALL patients. (A) L-asp activity in AE patients and the non-AE patients; (B) L-asp activity in discriminating AE patients from non-AE patients; (C) Anti-L-asp antibody level in AE patients and non-AE patients; (D) Anti-L-asp antibody in discriminating AE patients from non-AE patients; (E): Asn level in AE patients and non-AE patients; (F) Asn in discriminating AE patients from non-AE patients; (G) Gln level in AE patients and non-AE patients; (H) Gln in discriminating AE patients from non-AE patients. L-asp: L-asparaginase; Asn: asparagine; Gln: glutamine; AEs: adverse events; ALL: acute lymphoblastic leukemia; AUC: area under the curve; CI: confidence interval.
In addition, anti-L-asp antibody was similar between AE patients and non-AE patients (P = 0.068) (Figure 1C); meanwhile, it had no correlation with the risk of AEs (AUC: 0.659, 95% CI: 0.465-0.852) (Figure 1D).
Besides, Asn level was increased in AE patients compared to non-AE patients (P < 0.001) (Figure 1E). Meanwhile, ROC curve showed it was correlated with higher risk of AEs (AUC: 0.728, 95% CI: 0.554-0.903) with Asn level at the best cut-off point of 566.0 ng/mL, and the sensitivity as well as specificity at the best cut-off point were 53.8% and 93.6%, respectively (Figure 1F).
Also, Gln level was elevated in AE patients compared to non-AE patients (P = 0.022) (Figure 1G), and it was associated with elevated risk of AEs (AUC: 0.698, 95% CI: 0.498-0.899). Gln level at the best cut-off point was 18.2 μg/mL, and the sensitivity as well as specificity at the best cut-off point were 61.5% and 89.7%, respectively (Figure 1H).
L-asp Activity, Anti-L-asp Antibody Level, Asn level and Gln Level in Predicting Anaphylaxis
Pediatric ALL patients were divided into anaphylaxis patients (n = 7) and non-anaphylaxis patients (n = 84) according to the occurrence of anaphylaxis among them. L-asp activity was reduced in anaphylaxis compared to non-anaphylaxis (P < 0.001) (Figure 2A). Meanwhile, ROC curve showed it was closely correlated with increased anaphylaxis risk (AUC: 0.927, 95% CI: 0.864-0.990) with L-asp activity at the best cut-off point of 267.6 IU/L, and the sensitivity as well as specificity at the best cut-off point were 100.0% and 83.3%, respectively (Figure 2B).
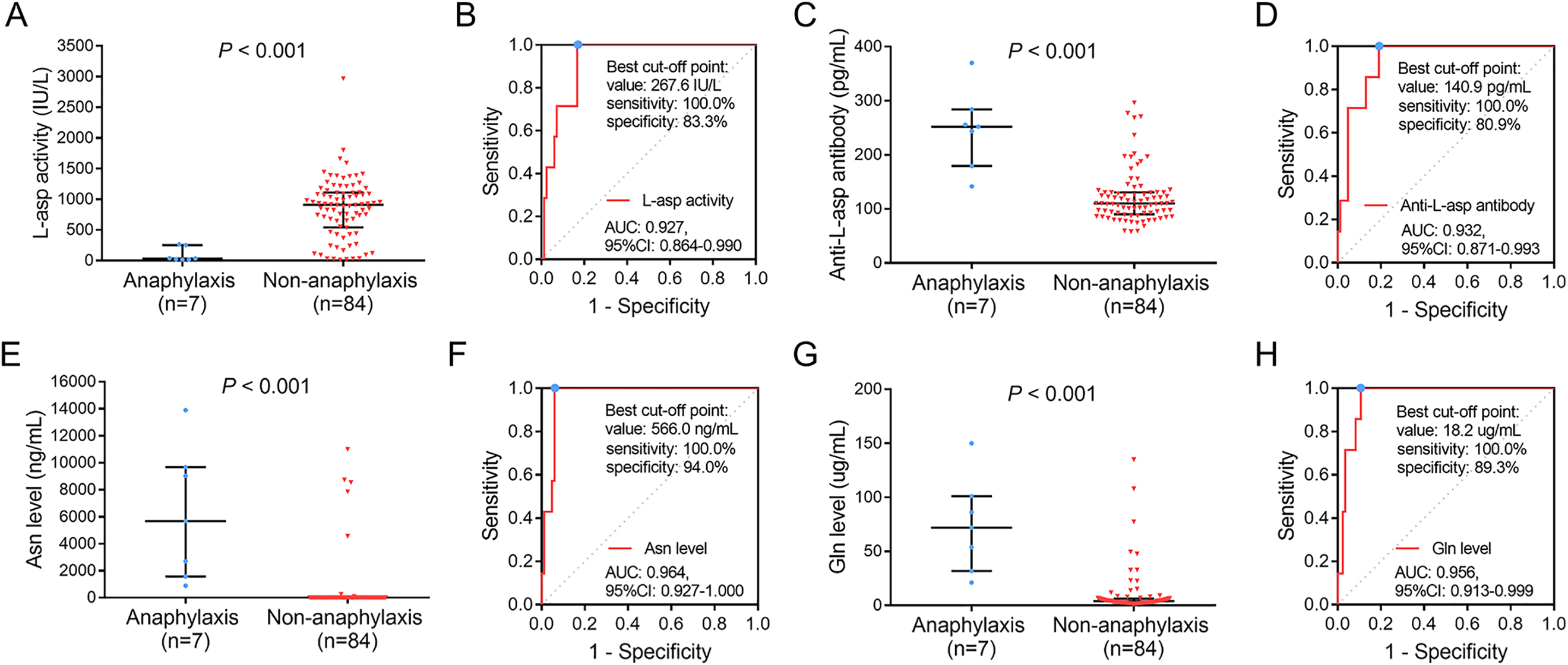
Correlation of L-asp activity, anti-L-asp antibody level, Asn level and Gln level with anaphylaxis in pediatric ALL patients. (A) L-asp activity in anaphylaxis patients and non-anaphylaxis patients; (B) L-asp activity in discriminating anaphylaxis patients from non-anaphylaxis patients; (C) Anti-L-asp antibody level in anaphylaxis patients and non-anaphylaxis patients; (D) Anti-L-asp antibody in discriminating anaphylaxis patients from non-anaphylaxis patients; (E) Asn level in anaphylaxis patients and non-anaphylaxis patients; (F) Asn in discriminating anaphylaxis patients from non-anaphylaxis patients; (G) Gln level in anaphylaxis patients and non-anaphylaxis patients; (H) Gln in discriminating anaphylaxis patients from non-anaphylaxis patients. L-asp: L-asparaginase; Asn: asparagine; Gln: glutamine; ALL: acute lymphoblastic leukemia; AUC: area under the curve; CI: confidence interval.
Meanwhile, anti-L-asp antibody level was higher in anaphylaxis patients compared to non-anaphylaxis patients (P < 0.001) (Figure 2C), and it was also highly associated with elevated anaphylaxis risk (AUC: 0.932, 95% CI: 0.871-0.993) with anti-L-asp level at the best cut-off point of 140.9 pg/mL; the sensitivity and specificity at the best cut-off point were 100.0% and 80.9%, respectively (Figure 2D).
Additionally, Asn level was increased in anaphylaxis patients compared to non-anaphylaxis patients (P < 0.001) (Figure 2E). Further ROC curve analysis revealed it was closely correlated with elevated anaphylaxis risk as well (AUC: 0.964, 95% CI: 0.927 -1.000) with Asn level at the best cut-off point of 566.0 ng/mL, and the sensitivity as well as specificity at the best cut-off point were 100.0% and 94.0%, respectively (Figure 2F).
Besides, Gln level was also raised in anaphylaxis patients compared to non-anaphylaxis patients (P < 0.001) (Figure 2G). Moreover, ROC curve showed it was also highly associated with increased anaphylaxis risk (AUC: 0.956, 95% CI: 0.913-0.999) with Gln level at the best cut-off point of 18.2 μg/mL, and the sensitivity as well as specificity at the best cut-off point were 100.0% and 89.3%, respectively (Figure 2H).
L-asp Activity, Anti-L-asp Antibody Level, Asn Level and Gln Level in Predicting Non-Anaphylactic AEs
Pediatric ALL patients were divided into non-anaphylactic AE patients (n = 6) and other patients (n = 85) according to the occurrence of AEs other than anaphylaxis among them. No difference was found in L-asp activity, anti-L-asp antibody level, Asn level and Gln level between non-anaphylactic AE patients and other patients (all P > 0.05) (Figure 3A, C, E, G), and ROC curves showed that no correlations were found between them and risk of non-anaphylactic AEs (Figure 3B, D, F, H).

Correlation of L-asp activity, anti-L-asp antibody level, Asn level and Gln level with non-anaphylaxis AEs in pediatric ALL patients. (A) L-asp activity in non-anaphylactic AE patients and other patients; (B) L-asp activity in discriminating non-anaphylactic AE patients from other patients; (C) Anti-L-asp antibody level in non-anaphylactic AE patients and others patients; (D) Anti-L-asp antibody in discriminating non-anaphylactic AE patients from other patients; (E) Asn level in non-anaphylactic AE patients and others patients; (F) Asn in discriminating non-anaphylactic AE patients from other patients; (G) Gln level in non-anaphylactic AE patients and others patients; (H) Gln in discriminating non-anaphylactic AE patients from other patients. L-asp: L-asparaginase; Asn: asparagine; Gln: glutamine; AE: adverse event; ALL: acute lymphoblastic leukemia; AUC: area under the curve; CI: confidence interval.
Correlation Analysis Among L-asp, Anti-L-asp Antibody Level, Asn Level and Gln Level
L-asp activity was negatively correlated with anti-L-asp antibody level (P < 0.001, r = -0.420), Asn level (P < 0.001, r = -0.608) and Gln level (P < 0.001, r = -0.276). Meanwhile, anti-L-asp antibody level was positively correlated with Asn level (P < 0.001, r = 0.483). Additionally, Asn level was positively correlated with Gln level (P < 0.001, r = 0.616). However, no correlation was observed between anti-L-asp antibody level and Gln level (P = 0.197, r = 0.135) (Table 3).
Correlation Among L-asp Activity, Anti-L-asp Antibody, Asn Level and Gln Level.

L-asp, L-asparaginase; Asn, asparagine; Gln, glutamine.
Drug Adjustment in Patients Occurred AEs
In patients who occurred anaphylaxis (n = 7), PEG-asp was switched to Erw-asp. Meanwhile in patients who had acute pancreatitis (n = 4), 3 of them ceased PEG-asp administration, and 1 of them switched to E. coli-asp. In patients who had abdominal pain and diarrhea (n = 1) or nausea and vomiting (n = 1), due to the symptoms were relatively mild and tolerable, no adjustment was conducted (Table 4).
Drug Adjustment After Occurrence of AEs.

AEs, adverse events; PEG-asp, polyethylene glycol-conjugated L-asparaginase; E. coli-asp, Escherichia coli L-asparaginase; Erw-asp, Erwinia L-asparaginase.
Discussion
In this study, we found that: (1) L-asp activity, anti-L-asp antibody level, Asn level and Gln level were correlated with the risk of AEs, especially anaphylaxis, but not non-anaphylactic AEs; (2) negative correlations were observed in L-asp activity with anti-L-asp antibody level, Asn level and Gln level; while positive correlations were observed between anti-L-asp antibody level and Asn level, as well as between Asn level and Gln level.
PEG-asp is an important component in the first-line treatment of pediatric ALL, which catalyzes the hydrolysis of Asn, an essential amino acid for the protein synthesis of leukemic cells due to the extremely low activity of Asn synthetase. 5 Through depleting blood concentration of Asn, PEG-asp suppresses the subsequent DNA and RNA syntheses in leukemic cells and leads leukemic cells to apoptosis, thus exerting its anti-ALL property. 5,13 At the same time, PEG-asp possesses partial activity in hydrolyzing Gln (about 3%-10% of total PEG-asp activity), which is directly correlated with the adverse events of PEG-asp administration including pancreatitis and impairment of liver function. 14 Moreover, although conjugated with PEG to reduce the immunogenicity, PEG-asp would still rise the production of anti-L-asp antibody, which further results in higher drug clearance and anaphylaxis in pediatric ALL patients. 8 Therefore, PEG-asp is a critical therapeutic agent for ALL treatment with minor drawbacks.
The AEs in pediatric ALL patients who receive PEG-asp treatment have been well-documented. For example, in a randomized, open-label, phase III clinical trial, PEG-asp related AEs occur in 28% of pediatric ALL patients who receive PEG-asp treatment, and the most common AEs are bacterial or fungal infection and anaphylaxis reactions. 15 Meanwhile, previous studies show that the occurrence of anaphylaxis reactions of any grade in pediatric ALL patients who receive PEG-asp administration varies from 1% to 23%. 15 -17 However, the safety profile of PEG-asp in pediatric ALL patients in China was quite unclear. In this study, the occurrence of PEG-asp-related AEs was observed in 13 (14.3%) pediatric ALL patients, among which 7 (7.7%) AEs were related to anaphylaxis, and the other 6 AEs included 4 (4.4%) acute pancreatitis, 1 (1.1%) abdominal pain and diarrhea, as well as 1 (1.1%) nausea and vomiting. The difference in the occurrence of AEs between previous studies (mentioned above) and this study could be explained by the difference in the definition of AEs, for instance, one previous study defines pancreatitis, hypertriglyceridemia, VTE, coagulation disorder, hyperglycemia, hypersensitivity, and elevated bilirubin and liver function as AEs, 18 while another study defines AEs according to the Common Terminology Criteria for Adverse Events, 16 which could result in different occurrence of AEs. Meanwhile, different dosages of PEG-asp might also result in the difference in AEs occurrences. 18 Of note, 1 (1.1%) patient in this study occurred anaphylactic shock after the administration of PEG-asp, and PEG-asp was switched to Erw-asp since the difference in immunogenicity between PEG-asp and Erw-asp.
The monitoring of L-asp activity in pediatric ALL patients has been proposed by several studies to ensure the treatment efficacy of L-asp. 19,20 For example, one previous study illustrates that in pediatric ALL patients who receive Erw-asp treatment, nadir serum asparaginase activity >0.1 IU/mL is achieved in 83% of patients after 48 h and in 43% of patients after 72 h, implying the need for therapeutic drug monitoring. 20 Meanwhile, it is reported that pediatric ALL patients who receive an individualized dose of E. coli-asp (which maintains nadir serum asparaginase activity between 0.10 and 0,14 IU/mL) present improved prognosis compared to those who receive a standard dose of E. coli-asp. 19 However, the importance of monitoring of L-asp activity level, anti-L-asp antibody level, Asn level and Gln level after PEG-asp administration and their correlations with PEG-asp-induced AEs in pediatric ALL patients were unclear. In the present study, ROC curves showed that decreased L-asp activity, but increased anti-L-asp antibody level, Asn level and Gln level were correlated with higher risks of AEs, especially anaphylaxis, but not non-anaphylactic AEs. Possible explanations could be that: (1) higher anti-L-asp antibody level might increase the clearance of L-asp by the immune system, which resulted in elevated levels of L-asp substrates (Asn and Gln) and higher immune reaction between L-asp antigen and anti-L-asp antibody, thus enhancing the risk of AEs and anaphylaxis in pediatric ALL patients 21 ; (2) increased anti-L-asp antibody level, along with reduced L-asp activity, Asn and Gln levels might induce hypersensitivity, thus causing anaphylaxis; while they might not directly cause other AEs such as pancreatitis. Therefore, they were correlated with the risks of anaphylaxis, but not non-anaphylactic AEs in pediatric ALL patients. Furthermore, we calculated the values of L-asp activity, anti-L-asp antibody level, Asn level and Gln level at the best cut-off points on the ROC curves, which could be set as the threshold values for the risks of AEs and anaphylaxis to assist the development of the individualized dosing of PEG-asp. However, these data should be further substantiated in validation cohorts. Besides, negative correlations were observed in L-asp activity with anti-L-asp antibody level, Asn level and Gln level; while positive correlations were observed between anti-L-asp antibody level and Asn level, and between Asn level and Gln level, which could further explain the prediction of AEs and anaphylaxis occurrence by these factors in pediatric ALL patients who received PEG-asp treatment.
There were several limitations in this study and should be clarified. First of all, the sample size of this study was relatively small, and further studies with larger sample sizes might be performed to investigate more general threshold values of L-asp activity, anti-L-asp antibody level, Asn level and Gln level for predicting the AEs in PEG-asp-treated pediatric ALL patients. Secondly, we detected the L-asp activity on the 7th day after L-asp administration according to previous clinical experiences. 11 However, the sustainably monitoring L-asp activity, anti-L-asp antibody, Asn and Gln levels at multiple timepoints might possess better prediction for the risks of AEs especially anaphylaxis, which could be performed in further studies. Finally, this study lacked a validation cohort, and the results in this study should be substantiated in further studies, especially the threshold values of L-asp activity, anti-L-asp antibody, Asn and Gln levels in predicting risks of AEs and anaphylaxis.
Collectively, the measurement of L-asp activity, anti-L-asp antibody level, Asn level and Gln level could be helpful for the prevention of anaphylaxis-related AEs in pediatric ALL patients who underwent PEG-asp-contained treatment by recognizing patients with high risk of the occurrence of anaphylaxis-related AEs, which might improve their individualized treatments.
Footnotes
Abbreviations
Authors’ Note
This study was approved by the Ethics Committee of Shanghai Children’s Medical Center, Shanghai Jiao Tong University School of Medicine with an ethical approval number of SCMCIRB-K2017065. All guardians of pediatric patients signed the informed consents before enrollment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Hospital Pharmacy Research Foundation of Shanghai Jiao Tong University School of Medicine (No: JDYX2017ZD002).
