Abstract
Purpose:
Advanced radiotherapeutic techniques and apparatus have been developed and widely applied in stereotactic body radiation therapy for early-stage non-small cell lung cancer, but their clinical benefits have not necessarily been confirmed. This study was performed to review our 10-year experience with therapy for the disease and to evaluate whether the advanced radiotherapeutic system implemented in our hospital 5 years after we began the therapy improved the clinical outcomes of patients.
Materials and Methods:
Patients who underwent the therapy at our hospital between April 2008 and March 2018 were retrospectively reviewed. They were divided into 2 groups treated with the conventional system or the advanced system, and the characteristics and clinical outcomes were compared between the groups. The same analyses were also performed in propensity-matched patients from the 2 groups.
Results:
Among the 73 patients eligible for this study, 42 were treated with the conventional system and 31 with the advanced system. All were treated as planned, and severe adverse events were rare. The local progression-free survival rate in the advanced system group was significantly higher than in the conventional system group (P = 0.025). In the propensity-matched patients, both the local progression-free survival rate and the overall survival rate were significantly higher compared in the advanced system group than the conventional system group (P = 0.089 and 0.080, respectively).
Conclusion:
The advanced system improved the outcomes of patients with the disease, suggesting that technological development has had a strong impact on clinical outcomes.
Introduction
Although the standard therapy for early-stage non-small cell lung cancer (NSCLC) is lobectomy, many patients who are medically inoperable or who refuse the operation receive stereotactic body radiation therapy (SBRT). 1 SBRT for early-stage NSCLC is recognized as an alternative to curative surgery for the disease. 2 This therapy was initially developed in the 1990s and has progressed over the last 2 decades. 3 Various techniques and equipment have been developed and incorporated into the practice of SBRT for early-stage NSCLC. 4,5 However, it has not been clarified how these technological developments have improved clinical outcomes of patients receiving the therapy. Our hospital initiated SBRT for early-stage NSCLC in 2008. Five years later, we adopted an advanced treatment system. All SBRT at our hospital has since been performed with the advanced system. This study was performed to review our 10-year experience of SBRT for early-stage NSCLC and evaluate whether the advanced system improved the outcome of the patients.
Materials and Methods
Patients
Patients with early-stage NSCLC who underwent stereotactic body radiotherapy at our hospital between April 2008 and March 2018 were retrospectively reviewed. This retrospective study was conducted in accordance with the Declaration of Helsinki. Our Institutional Review Board approved the study (approval number: 4070) and waived the need for informed consent. Patients with metastatic lung cancer were excluded from the study. A few patients who received more than one course of SBRT were also excluded to simplify the study.
Treatment
We initiated SBRT for early-stage NSCLC with a conventional system in April 2008. In this system, respiratory motion was assessed by fluoroscopy or inhale/exhale breath-hold computed tomography (CT) images. Patients who had large motion were trained to practice shallow breathing during each treatment session. The radiotherapy planning system (RTPS) was Eclipse (Varian Medical Systems, Palo Alto, CA, USA), and the dose-calculation algorithm in the RTPS was pencil-beam convolution (PBC), which is equivalent to the Clarkson method, with heterogeneity correction using the Batho power law. The linear accelerator did not have any online image guidance functions, so we acquired CT images before and after each session with a CT scanner next to the radiotherapy room to determine whether the set-up marks on the patient were accurate, and corrected the positions if necessary (offline image guidance). Five years after initiating the therapy, we updated the conventional system and began advanced SBRT for early-stage NSCLC. In the advanced system, respiratory motion was thoroughly assessed on 4-dimensional CT images. The voluntary inspiration breath-holding technique with Abches (APEX Medical, Tokyo, Japan) and RPM (Varian Medical Systems, Palo Alto, CA, USA) was also incorporated into the advanced system and was routinely applied to tumors that moved more than 1 cm to reduce internal margin (IM). RTPS was updated, and the dose-calculation algorithm was replaced by the anisotropic analytical algorithm (AAA), which is a more accurate algorithm than PBC.
The linear accelerator was also replaced in the update. The new one was equipped with a few online image-guidance functions. In each session, we acquired cone-beam CT images and corrected the patients’ positions accordingly (online image guidance).
On the other hand, there were a few specific points in the advanced system that were consistent with the conventional system. Patient immobilization was performed with a BodyFIX system (Elekta Medical Intelligence, Schwabmuenchen, Germany), in both and the conventional and advanced systems. The new linear accelerator was manufactured by the same company (Varian Medical Systems, Palo Alto, CA, USA) as the original unit, and the multi-leaf collimator in the new linear accelerator was the same model as the original unit. The accelerators were beam-matched, and 4 MV photons were consistently employed in SBRT for early-stage NSCLC in both systems. With regard to contouring, we applied the same policy in each case in both systems. Gross tumor volume (GTV) was contoured on CT images with thickness of 2.5 mm with a pulmonary window setting. Clinical target volume (CTV) was set to be identical to GTV. Internal target volume (ITV) was generated from the GTV by putting adequate IM according to the results of respiratory motion assessment. A margin of 5 mm was mechanically placed on the ITV to generate planning target volume (PTV).
Patients were consistently prescribed a dose of 48 Gy given in 4 fractions, except for a few cases mainly with “central tumors” 6 who received 65 Gy in 10 fractions. We felt that 65 Gy in 10 fractions was equivalent to 48 Gy in 4 fractions in terms of the biologically equivalent doses calculated with an α/β ratio of 10 (BED10). We initially prescribed these radiation doses to isocenter (the isocenter prescribing method), but changed the method 3 years after the update and began prescribing a dose covering 95% (D95%) of the PTV (the D95% prescribing method).
Analysis
Statistical comparisons between the whole conventional and advanced system groups were performed using Fisher’s exact test and the Mann–Whitney U test. Survival analyses of the entire cohort were then performed. Either local failure or death due to any cause was counted as an event in local progression-free survival (LPFS) analysis. Any failure and deaths due to any cause were counted as events in progression-free survival (PFS) analysis. Death due to any cause was counted as an event in overall survival (OS) analysis. These survival rates of each of the groups were calculated from the start of treatment, and the survival curves were calculated by the Kaplan–Meier method. Statistical comparisons between the curves of the conventional and advanced system groups were performed by the log-rank test. We also performed propensity-matched analysis to reduce biases between the groups. A propensity score difference of 0.20 was used as a maximum caliper width for matching the groups. All matching was performed in a 1:1 ratio. The same survival analyses described above were performed on the matched patients. All statistical analyses were performed with the statistical software JMP version 14.2.0 (SAS Institute Inc., Cary, NC, USA). In all analyses, P < 0.05 was taken to indicate statistical significance. Toxicity was assessed using the Common Terminology Criteria for Adverse Events version 4.0 (National Cancer Institute, Rockville, MD).
Results
Seventy-three patients with early-stage NSCLC who received SBRT at our hospital between April 2008 and March 2018 were identified as appropriate for this study. Forty-two were treated with the conventional system and 31 were treated with the advanced system. Table 1 shows the characteristics of both groups. Performance status was evaluated with the Eastern Cooperative Oncology Group scale, 7 and geriatric assessment was performed with Charleston Comorbidity Index (CCI). 8 Medical inoperability was determined by cardiothoracic surgeons and/or pulmonologists. The medical operability of patients who refused surgery was not necessarily assessed. There were significantly more patients with CCI ≥ 4 in the advanced system group than in the conventional system group. The mean follow-up period in the advanced system group was significantly shorter than that in the conventional system group.
Patient Characteristics.

Abbreviations: M, median; PS, performance status; CCI, Charleston Comorbidity Index; SCC, squamous cell carcinoma; SBRT, stereotactic body radiation therapy.
Table 2 shows the characteristics of the treatment for both groups. Although there was no difference in tumor size between the groups, as shown in Table 1, PTV was significantly smaller in the advanced system group than the conventional system group. There were more patients with suppression of respiratory motion in the advanced system group than the conventional system group. The percentage of irradiated lung volume exceeding 20 Gy (V20) was significantly lower in the advanced system group than the conventional system group.
Treatment Characteristics.

Abbreviations: PTV, planning target volume; M, median; fx, fraction; PBC, pencil-beam convolution; AAA, the anisotropic analytical algorithm; D95%, a dose that covers 95% of the PTV; V20, the percentage of irradiated lung volume exceeding 20 Gy.
All patients underwent their treatment as planned. No acute adverse events of grade > 2 were seen. Grade 3 and 4 pneumonitis as late adverse events were seen in 2 and 1 patient in the conventional system group, and 2 and no patients in the advanced system group, respectively. No other grade ≥ 3 late adverse events were seen in either group.
Figure 1 shows the LPFS, PFS, and OS curves of the conventional and advanced system groups from the entire cohort. Three-year LPFS, PFS, and OS rates in the conventional and advanced system groups were 56.0% (95% confidence interval [CI]: 39.7-71.1) and 88.2% (95% CI: 68.9-96.2), 53.3% (95% CI: 37.2-68.8) and 85.0% (95% CI: 66.0-94.3), and 64.2% (95% CI: 47.5-78.0) and 88.2% (95% CI: 68.9-96.2), respectively. The LPFS rate was significantly higher in the advanced system group than the conventional system group (P = 0.025). However, the differences in PFS and OS between the 2 groups were not statistically significant (P = 0.089 and 0.080, respectively).

Curves of (a) local progression-free survival (LPFS); (b) progression-free survival (PFS); and (c) overall survival (OS) of the conventional system group (CSG) and the advanced system group (ASG) for the entire cohort.
Propensity-score matching was performed. Scores were calculated using a logit model with age, sex, size of the solid component, performance status, CCI, and follow-up period as factors, because these were thought to be related to survival outcomes. 8 -11 We identified 20 patients from each group who were matched for propensity score. The groups of matched patients had similar characteristics to each other in terms of not only the factors outlined above but also the reason for choosing SBRT (Table 3).
Characteristics of the Propensity Score-Matched Patients.
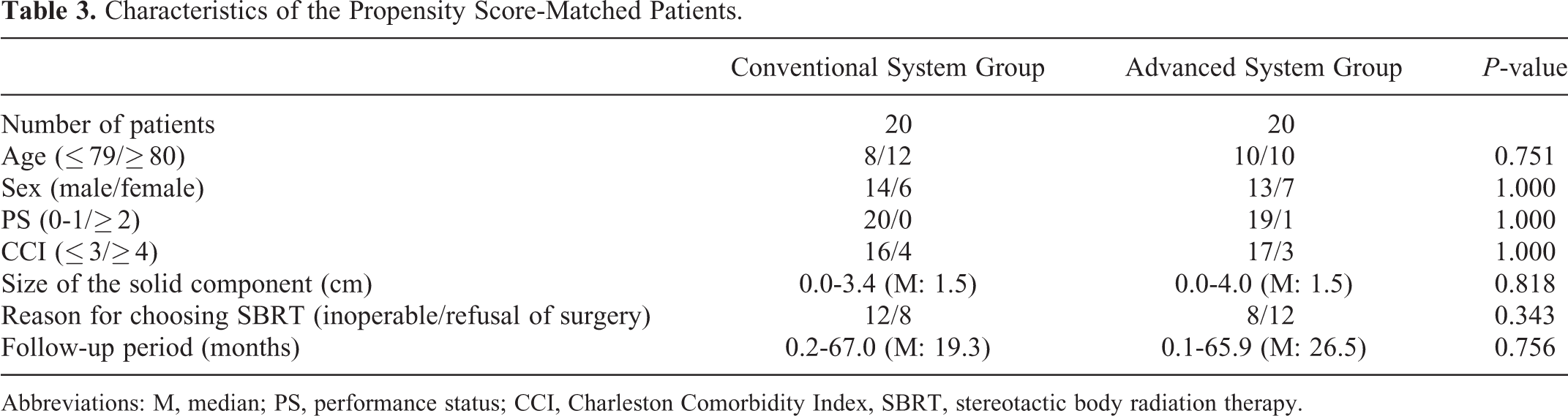
Abbreviations: M, median; PS, performance status; CCI, Charleston Comorbidity Index, SBRT, stereotactic body radiation therapy.
Figure 2 shows the LPFS, PFS, and OS curves of the conventional and advanced system groups from the matched patients. Three-year LPFS, PFS, and OS rates in the conventional and advanced system groups were 44.9% (95% CI: 22.1-70.1) and 93.8% (95% CI: 66.5-99.1), 46.3% (95% CI: 23.1-71.2) and 88.9% (95% CI: 64.8-97.2), and 51.7% (95% CI: 27.3-75.3) and 93.8% (95% CI: 66.5-99.1), respectively. The differences were statistically significant for LPFS (P = 0.009) and OS (P = 0.016), but not for PFS (P = 0.080).

Curves of (a) local progression-free survival (LPFS); (b) progression-free survival (PFS); and (c) overall survival (OS) of the conventional system group (CSG) and the advanced system group (ASG) for the propensity score-matched patients.
Discussion
Improvements in techniques for patient immobilization have facilitated the application of stereotactic radiotherapy to cancers in the trunk, i.e. SBRT. 12 In particular, it has been applied to early-stage NSCLC around the world. 13 It requires image guidance systems and motion management systems, and various types of radiotherapy technique and apparatus, including online image guidance and sophisticated motion management systems, which have seen rapid development with the widespread adoption of the therapy. 14 RTPS algorithms have also improved and achieved more accurate dose calculations. 5,15 Such technological developments are fundamentally thought to have beneficial effects on clinical outcome. However, there have been few studies to evaluate the impact of technological development on the clinical outcome of patients. 16 Randomized controlled trials are not applicable to such evaluations, and comparisons between different facilities are difficult due to the need to exclude inherent biases that hinder evaluation of the impact itself. Comparing clinical outcomes before and after updating the system in a single institution, as in the present study, is thought to be a reasonable way to evaluate the impact of technological developments on clinical outcomes.
We initiated SBRT for early-stage NSCLC with a conventional system, which was later updated to an advanced system that included several types of radiotherapeutic technique and apparatus. This study showed that the clinical outcome improved after the system was updated. This implies that the technological development had a strong impact on clinical outcomes, such as local control and overall survival, of patients with the disease. There are several possible explanations for the observed improvements.
It is obvious that motion management is an important technique in SBRT for NSCLC. 14 We adopted a sophisticated system that could precisely measure and control respiratory motion during treatment, and more patients in the advanced system group were treated under respiratory motion management than in the conventional system group. This may have reduced the geographic miss and contributed to the observed improvements in clinical outcomes. The sophisticated system enabled us to suppress respiratory motion more frequently. The voluntary inspiration breath-holding technique in the system reduces IM. As a result, PTV and V20 were significantly smaller and lower, respectively, in the advanced system group than the conventional system group, even though tumor sizes were similar. Another possible explanation for the smaller PTV and lower V20 is that we might have applied excessive IM to some of the patients in the conventional system group in compensation for uncertainty within the system. Online positional correction with image guidance was also thought to reduce the geographic miss. 17 We performed offline image guidance in the conventional system, but “online” is more accurate than “offline.”
There is some possibility that updating the algorithm used for dose calculation may have contributed to the observed improvements. The report of the American Association of Physicists in Medicine Task Group 101 did not recommend the use of pencil-beam algorithms in lung SBRT because of their inaccuracy. 15 AAA was reported to be more accurate, and it may have contributed to the observed improvement in outcomes. 18 We changed the prescribing method during the period of using the advanced system, because dose calculation using AAA is reliable even in the periphery. 15 This may have caused a virtual dose escalation. Kawahara et al. 19 reported that 48 Gy using the isocenter prescribing method was equivalent to 42 Gy using the D95% prescribing method with the superposition algorithm, which is similar to AAA. Therefore, the actual radiation dose of 48 Gy in the D95% prescribing method was 14% higher than 48 Gy in the isocenter prescribing method. Onishi et al. 20 reported that there is a dose–response relationship around BED10 = 100 Gy in SBRT early-stage NSCLC. However, it has not been fully established whether there is a further dose–response relationship. A few retrospective studies have evaluated this clinical question, and the results were not consistent. 21,22 A randomized controlled trial to answer this question is currently in progress in Japan, 23 and it is expected that the trial will solve the problem of choosing the optimal radiation dose prescription in SBRT in early-stage NSCLC.
Inappropriate use of advanced technique worsens the outcome. 5 Technological developments have to be built on an appropriate foundation to improve the outcome. We developed fundamental skills, such as patient immobilization, before initiating SBRT with the conventional system. The advanced technique and apparatus were added on this foundation. This may have been an important factor for the better outcomes with the advanced system.
This study had some limitations. First, the sample size was small because it was from a single institution. Although the results are statistically significant, we cannot completely eliminate the possibility that the advanced system only had a limited effect. Second, it is impossible to determine which technique had more impact on the outcome than others, because we adopted various advanced techniques at the same time. Third, geriatric assessments other than CCI were not performed although this cohort included a large number of elderly patients. Fourth, we could not exclude the possibility that the learning curve may have improved the outcome, although we underwent intensive training ourselves when we began SBRT for early-stage NSCLC. Fifth, neither could we exclude the possibility that advances in treatments other than radiotherapy have improved the outcome. Howlader et al. 24 reported that survival rate of NSCLC patients has substantially improved in recent years, along with the evolution of targeted therapies.
Conclusion
Our 10-year experience of SBRT for early-stage NSCLC, using the conventional treatment system in the first half and the advanced treatment system in the second half, was retrospectively reviewed. We found that the advanced system, which was equipped with several radiotherapeutic techniques and apparatus, improved the outcome of patients with this disease.
Footnotes
Abbreviations
Acknowledgments
The authors are grateful to Mrs. I. Koiwai and Mrs. Y. Ogawa for their support.
Ethics Statements
This retrospective study was conducted in accordance with the Declaration of Helsinki. Our Institutional Review Board approved the study (approval number: 4070) and waived the need for informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number JP16H05389.
