Abstract
Delta-like ligand 4 is a notch ligand that is predominantly expressed in the endothelial tip cells and plays essential roles in the regulation of angiogenesis. In this study, we explored the therapeutic effects of delta-like ligand 4 gene vaccine overexpression on the syngeneic model mouse model of liver cancer and the underlying mechanisms. Mouse hepatocellular carcinoma cell line H22-H8D8 was used to generate subcutaneous syngeneic model liver cancer in Kunming mice, and the effects of recombinant plasmid pVAX1 containing delta-like ligand 4 vaccine on tumor growth was examined. Compared to controls, delta-like ligand 4 vaccination reduced syngeneic model tumor size by 70.31% (from 17.11 ± 9.30 cm3 to 5.08 ± 2.75 cm3, P = .035) and tumor weight by 34.19% (from 6.26 ± 3.01 g to 4.12 ± 2.52 g, P = .102), while the mouse survival was significantly increased (from 27.7 ± 6.0 days to 33.1 ± 6.1 days, P = .047). High level of delta-like ligand 4 antibody, together with a significantly increased number of CD4+ and decreased CD8+ cells were identified in the mouse peripheral blood serum samples after delta-like ligand 4 immunization. In addition, elevated serum levels of interleukin 2, interleukin 4, and interferon γ were detected in the delta-like ligand 4–vaccinated mice when compared to the controls. Further studies have revealed increased CD31 and decreased Ki67 expression in the syngeneic model tumor tissues of vaccinated mice. Taken together, our studies suggest that delta-like ligand 4 gene vaccine can inhibit the growth of hepatocellular carcinoma in mice through inhibiting tumor angiogenesis and boosting antitumor immune responses. Hence, delta-like ligand 4 gene vaccination may be a promising strategy for the treatment of transplanted liver cancer.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common and the second leading cause of cancer-related death worldwide. 1 Hepatocellular carcinoma is particularly common in Asian countries including China. 2 Most of the patients with HCC are diagnosed at advanced stage due to a lack of specific diagnostic marker, and patients with advanced HCC generally have very poor prognosis, as the tumors are generally resistant to the existing treatments and are prone to local and distant metastasis. Curative treatments such as surgery and liver transplantation are only possible for less than 20% of patients with early diagnosed HCC, 3 with the reported 5-year post-surgery recurrence rate of 75%. 3 -6 Clearly, more effective treatments are urgently needed.
Immunotherapy for cancers has been regarded as one of the most important and promising advances in cancer therapy. 7 Ideally, cancer treatments should specifically eradicate tumor cells without damaging normal cells. 8 In this aspect, immunotherapy differs from the traditional cancer therapies such as surgery, radiation, and chemotherapy in that it can be made highly tumor-specific. 9 The goal of immunotherapy is to recover or boost the host immune response to tumor-associated antigens or any specific proteins expressed by tumor cells through regulating the body’s immune system. 10
Antitumor gene vaccines have become a promising tool for anticancer immunotherapy because they can exert a specific antitumor effect through increasing the tumor antigen expression on the host cells, which then reenforces the innate immune function. Antitumor gene vaccines differ from traditional vaccines in that the former can induce both humoral and cellular immune response, whereas the traditional vaccines only induce antigen-specific humoral immune response. 11 Gene vaccine may open a new avenue for cancer immunotherapy. 12,13
Functional angiogenesis is essential for tumor development, growth, invasion, and metastasis. 14,15 As such, antiangiogenesis has been demonstrated a promising approach for the treatment of solid tumors, with blocking vascular endothelial growth factor (VEGF) receptor signaling pathway being a classical example. 16 However, despite a short improvement in the overall survival rate in patients receiving the combinatorial therapy of VEGF monoclonal antibody and traditional chemotherapy, most patients developed resistance to anti-VEGF treatments after a period of drug exposure. 17 Clearly, drug resistance is a critical hurdle for successful cancer treatment, and more effective approaches need to be developed.
Delta-like ligand 4 (DLL4), an important ligand for notch signaling, is a critical component of neovascularization both under the physiological and the pathological conditions. 18,19 Delta-like ligand 4 is localized downstream of the VEGF signaling and plays a tumor-suppressive role in tumor cells. 20,21 Recent studies have identified that DLL4 is a potential drug target, 22 -24 as DLL4 blockade (eg, by anti-DLL4 antibodies) inhibited tumor growth despite it increased blood vessel density. It is well known that structural defects and dysfunction of neovascularization impair the ability of tumor tissues to grow. 22,23,25 -27 Since DLL4 is predominantly expressed in endothelial tip cells during angiogenic sprouting, it constitutes a unique target for cancer immunotherapy. Thus, targeting DLL4 may be a useful approach for the treatment of anti-VEGF therapy-resistant HCC. So far, treatment of liver cancer with gene vaccine has been rarely reported.
In this study, we aimed to investigate the therapeutic efficacy of DLL4 gene vaccine using syngeneic model liver cancer in mice as a model. We demonstrated that DLL4 gene vaccine inhibited syngeneic model tumor growth via disrupting functional tumor angiogenesis and inducing both humoral and cellular immune response in mice.
Materials and Methods
Reagents and DNA Constructs
The pVAX1-DLL4 gene vaccine plasmids 28 were kindly provided by Dr Qiqi He (Key Laboratory of Urinary Diseases of Gansu Province; Department of Urological Surgery, Lanzhou University Second Hospital, Lanzhou, China). Transfection reagent polyester amines (PEAs) 29 was a kind gift from Dr Tingting Liu (Key Laboratory of Heavy Ion Radiation Biology and Medicine, Institute of Modem Physics, Chinese Academy of Sciences, Lanzhou, China). FITC-conjugated DLL4 antibody (Cat No: 131093) was purchased from Zeye Biological Co, Ltd. Rabbit polyclonal antibody against human DLL4 (Cat No: ab7280) was purchased from Abcam. FITC-conjugated rat anti-mouse CD4 and PE-labeled rat anti-mouse CD8 were purchased from Biolegend. Mouse polyclonal antibodies against CD31 and Ki67 were purchased from Zhongshan Golden Bridge Biological Technology Co. Ltd. Antibody against β-actin was purchased from Santa Cruz Biotechnology. All primary antibodies were diluted at 1:200 in phosphate-buffered saline (PBS). Secondary antibodies including horseradish peroxidase–conjugated goat anti-rat immunoglobulin G (IgG) and anti-rabbit IgG were purchased from Zhongshan Golden Bridge Biological Technology Co Ltd. TdT-mediated dUTP Nick-End Labelling (TUNEL) Assay Kit (Cat No: G3250; Promega) was purchased from Gansu Baoxin Biological Technology Co Ltd. Amersham ECL Western Blotting Detection Kit was purchased from GEHealthcare Life Science.
Cell culture, transfection, and Western blot
Mouse HCC cell line H22-H8D8 (H22) was purchased from Focusbio and were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum, 1% glutamine, 100 U/mL penicillin, and 100 µg /mL streptomycin. Cells were cultured in fresh culture medium for 12 hours before transfection. To optimize the transfection conditions, PEAs (0.15 μg/µL) were mixed with pVAX1-DLL4 DNA at various volume ratios (1:0.5, 1:1, 1:2, and 1:4) and incubated at room temperature for 15 minutes to generate the DNA-lipid complexes. H22 cells were then transfected by adding these complexes drop wise into the culture medium. After 48 hours, the transfected cells were lysed with modified radioimmune precipitation assay buffer (Cell Signaling Technology), total proteins were extracted, and the concentration was measured by BCA protein assay kit (Beyotime).
For Western blot analysis, 25 µg of total cell proteins from each treatment condition were subjected to sodium salt polyacrylamide gel electrophoresis. The proteins were transferred to polyvinylidene fluoride membrane (Millipore) at 120 v for 1.5 hours and probed with primary antibodies against rabbit-anti human DLL4 (1:500) or β-actin for 1.5 hours at room temperature. After 3 washes in Tris-buffered saline with 0.05% Tween, the membranes were incubated with appropriate secondary antibodies (peroxidase-conjugated goat anti-rabbit immunoglobulin or goat anti-mouse immunoglobulin, both from Santa Cruz Biotechnology). The signals were detected by Amersham ECL Western Blotting Detection Kit.
Based on our optimization studies, we have determined that 4 g per reaction was the most optimal dose for the pVAX1-DLL4 gene vaccine plasmids that can give most efficient transfection. Hence, in the subsequent studies, 4 g of plasmids were used.
Syngeneic model, tumor model, and DNA vaccination
Male specific pathogen free Kunming (KM) mice (4 weeks, average weight 14.8 ± 2.7 g) were purchased from the Experimental Animal Center of Gansu Provincial College of Traditional Chinese Medicine. Each mouse was inoculated with H22 cells (2×106) in the left hind leg. Tumors were measured with a caliper weekly, and the tumor volume was calculated using the formula: L × W × π/6, 30 where L is the largest diameter (mm) and W is the smallest diameter (mm). When the tumor mass reached approximately 5 × 5 mm, mice were randomized into 2 groups and were intramuscularly injected into the quadriceps with 50 µg of DLL4 gene vaccine (n = 14) or the same amount of pVAX1 empty plasmids (n = 14). Mice were immunized once every 2 weeks for a total of 3 doses. Twenty-four hours prior to each dose of vaccine, each mouse was injected with 200 µL of 0.25% bupivacaine at the same site of vaccine injection. 31 Four weeks after the last immunization, mice were killed. All animal studies were approved by Lanzhou University Institutional Animal Care and Use Committee.
Detection of DLL4 antibody
One week after the last immunization, serum samples were collected for the detection of DLL4-specific antibody by using DLL4 ELISA kit (JL19084, Shanghai Jianglai Biotechnology, Co, Ltd). Briefly, 96-well enzyme-linked immunosorbent assay (ELISA) plates were prepared by coating with human DLL4 antibody. Absorbance was measured at 450 nm with an ELISA reader, per the instructions from the supplier.
Assessment of Interleukin-2, Interleukin-4, and interferon γ by ELISA
Peripheral blood samples were collected from mice via eye ball and diluted 100 times with PBS. Interleukin-2 (IL-2), Interleukin-4 (IL-4), and interferon γ (IFN-γ) titers were evaluated by respective ELISA kits (ZSGB Bio). Normal mouse serum samples were used as negative controls. Absorbance was read at 450 nm. All samples and controls were measured in duplicates.
Flow cytometry analysis
Anticoagulant blood sample from each mouse (100 μL) was incubated with 2 µL of FITC-labeled rat anti-mouse CD4 (0.5 mg/mL) or 5 µL of PE-labeled rat anti-mouse CD8 (0.2 mg/mL; BioLegend) for 30 minutes, followed by incubating with 2 mL of ammonium–chloride–potassium lysis buffer for 15 minutes in the dark and centrifuged for 5 minutes at 1500 rpm. After 2 washes with 2 mL of PBS, the resultant cell pellet was resuspended in 500 µL of PBS and analyzed by Calibur Flow Cytometry.
Immunohistochemical staining
The expression of CD31 and Ki67 in the syngeneic model tumor tissues was stained with mouse polyclonal antibodies against CD31 and Ki67. Mouse IgG (Abcam) was used as a control. Slides were developed with diaminobenzidine tetrahydrochloride and counterstained with hematoxylin. The staining results were semi-quantitatively scored from 3 randomly chosen fields as previously reported. 32
TdT-mediated dUTP nick-end labelling assay
The TUNEL assay was used to detect apoptosis in paraffin-embedded syngeneic model tumor tissues. The TUNEL procedure was performed according to the manufacturer’s instruction. Briefly, the sections were treated with 1 mg/mL of DNase I at room temperature for 10 minutes as a positive control. Paraffin-embedded blocks were cut into sections of 5-µm thickness, incubated with 100 µL of protease K for 10 minutes, followed by incubation with 50 µL of rTdT buffer for 60 minutes. The sections were stained with NaCl and sodium citrate solution for 15 minutes and counterstained with pyridine iodide solution for 15 minutes in the dark. The slides were observed under a fluorescence microscopy (ZEISS LSM 510 META) at 520 ± 20 nm excitation and 620 nm emission. Each slide was scored in 10 random fields.
Statistical Analysis
All data were analyzed using SPSS 13.0 software (SPSS). Significant difference was determined by unpaired, 2-tailed Student t test or independent sample t test. A P < .05 was considered statistically significant.
Results
DLL4 Gene Vaccination Inhibits Syngeneic Model Tumor Growth and Prolongs Animal Survival
To investigate whether pVAX1-DLL4 gene vaccine exerts antitumor effects in vivo, we established syngeneic model liver cancer model in KM mice using mouse HCC cell line H22. As described in the “Materials and Methods” section, each mouse was inoculated with 2 × 106 H22 cells that were previously transfected with the combination of pVAX1-DLL4 DNA and PEA (0.15 μg/µL) at a volume ratio of 1:1. Polyester amine is a low-molecular-weight polyester, poly-ε-caprolactone, and polyethyleneimine that has been shown to significantly enhance gene transfection. 29,33,34 With this transfection condition, we achieved a transfection efficiency of 50% to 70% as confirmed by fluorescence microscopy (Figure 1A and B) and Western blot analysis (Figure 1C)
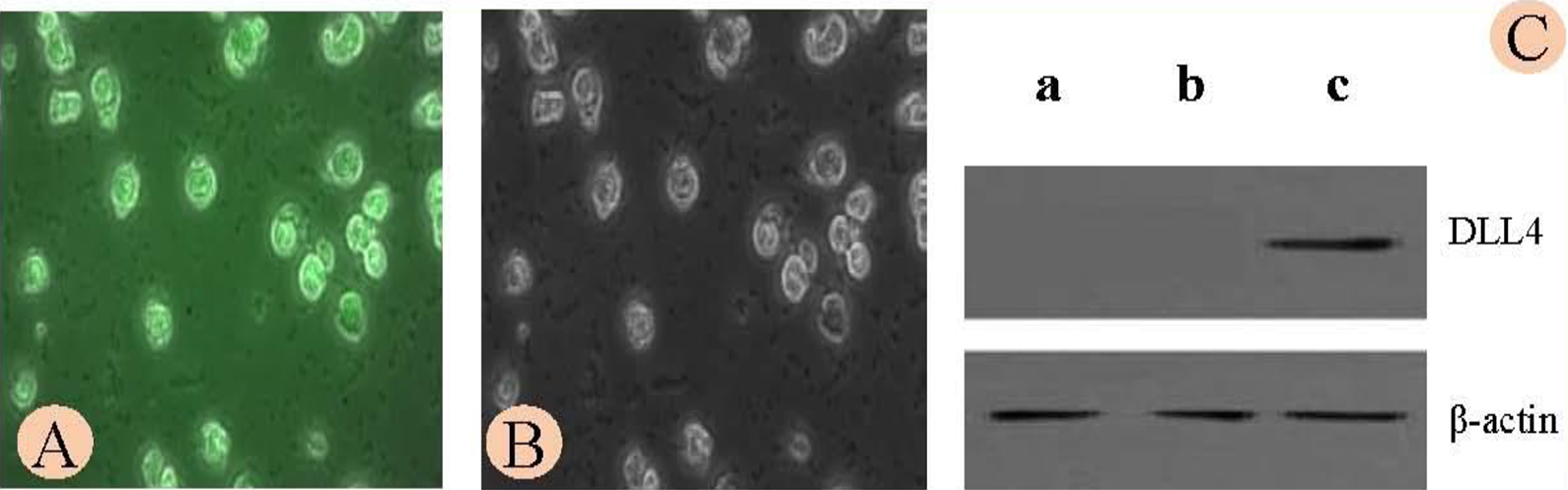
H22 cells were transiently transfected with FITC-DLL4 plasmid. A, Transfected cells were viewed under a fluorescent microscope. B, Phase-contrast image of the same field. Magnifications: ×200. C, The expression of Delta-Like Ligand 4 (DLL4) in the transfected H22 cells was confirmed by Western blot. a, Control (no transfection); b, Cells transfected with Lipo2500 alone; c, Cells transfected with DLL4 plasmids and polyester amines (PEAs) at a ratio of 1:1.
Following cell injection, syngeneic model tumors were successfully established in 28 mice (82.35%, total mice 34) within 7 to 14 days of tumor cell injection. Two weeks after tumor cell translation, the tumor bearing mice were randomly divided into 2 groups: DLL4 gene vaccine group and control group (mice receiving pVAX1 empty plasmids). We observed that 37 days after cell inoculation, 64.7% of mice receiving DLL4 immunization survived, whereas only 16.7% of mice receiving control vaccine survived (Figure 2A). The average survival time for mice receiving DLL4 vaccines was 33.1 ± 6.1 days, which was significantly longer than the mice in control group (27.67 ± 6.02 days, P = .047).

Effects of the delta-like ligand 4 (DLL4) gene vaccine on tumor growth and mouse survival. A, Animal survival analysis. B, Tumor volume. C, Tumor weight. D, Typical pictures of excised xenograft tumors.
Upon completion of the in vivo experiments, the syngeneic model tumors were analyzed. As illustrated by the tumor volume analysis, DLL4 vaccines led to a significant (70.3%) reduction in the syngeneic model tumor volumes (5.08 ± 2.75 cm3 in DLL4 group vs 17.11 ± 9.30 cm3 in control group, P = .035; Figure 2B and D). A reduction in tumor weight by 34.2% was also observed following DLL4 vaccination (4.12 ± 2.52 g in DLL4 group vs 6.26 ± 3.01 g in control group, P = .102), although the difference was not statistically significant (Figure 2C and D). Representative pictures of syngeneic model tumors are shown in Figure 2D. These results demonstrated the therapeutic efficacy of DLL4 gene vaccine in the mouse model of HCC.
The DLL4 Gene Vaccine Elicits Both Humoral and Cellular Immune Responses in Mice
To explore the potential mechanisms of the anticancer effects of DLL4 gene vaccine, we evaluated the both humoral and cellular immune responses. High titer of DLL4-specific antibody was detected in the serum samples of the DLL4-vaccinated mice (optical density [OD] value: 1.32 ± 0.21) but not in the mice immunized with empty vector (OD value: 0.47 ± 0.11; P = .045; Figure 3A). These results indicate that the DLL4 gene vaccine stimulated antigen-specific humoral immune response in mice.

Cellular and humoral immune response in mice receiving DLL4 vaccination. A, Production of delta-like ligand 4 (DLL4). B, and (C) Increased number of CD4+ T cells and decreased number of CD8+ T cells in DLL4-vaccinated mice as determined by flow cytometry. D, Serum level of IL-2, IL-4, and IFN-γ was detected by ELISA and analyzed by Student t test. *: P < .05; **: P < .01. ELISA indicates enzyme-linked immunosorbent assay; IFN, interferon; IL, interleukin.
We then measured the number of CD4+ and CD8+ cells in the peripheral blood by flow cytometry. As shown in Figure 3B and C, a significantly higher level of CD4+ cells were detected in DLL4-vaccinated mice as opposed to the control mice (32.41 ± 4.19% vs 15.67 ± 7.02%, P = .001). Meanwhile, a significantly lower level of CD8+ cells was seen in the DLL4-vaccinated mice as compared to the control mice (12.62 ± 7.03%, 20.20 ± 6.45%, P = .043).
We further measured the serum levels of cytokines that are known to be secreted by activated T lymphocytes and are commonly used as surrogate markers of cytotoxic lymphocyte (CTL) activity. As shown in Figure 3D, significantly increased concentration of IL-2, IL-4, and IFN-γ was seen in the DLL4-vaccinated mice as compared with the control mice. These results indicate that DLL4 gene vaccine enhanced and provoked immune responses.
DLL4 Gene Vaccine Induces Defective Tumor Angiogenesis and Decreases Cell Proliferation in Syngeneic Model Tumor
Since the DLL4 is predominantly expressed in the endothelial cells and is involved in the regulation of angiogenesis, we examined the impact of DLL4 vaccination on tumor vascularity by detecting the expression of CD31, a marker for neovascularization in the syngeneic model tumor tissues. As shown in Figure 4A, DLL4 vaccination led to an increased CD31 expression in the syngeneic model tumor tissues. In addition, DLL4 vaccination also caused a marked reduction of cell proliferation as indicated by reduced expression of Ki67 (Figure 4A). However, DLL4 vaccination did not cause significant apoptosis in the syngeneic model tumor tissues as revealed by TUNEL assay (Figure 4B).

Impact of DLL4 vaccination on angiogenesis, cell proliferation, and apoptosis in xenograft tumor tissues. A, Immunohistochemical staining of angiogenesis marker CD31 and proliferation marker Ki67 in xenograft tumor tissues. B, Apoptosis was detected in the xenograft tumor tissues by TUNEL. Green fluorescence: apoptosis detected by FITC; Red: nuclei stained by propidium iodide(PI). DDL4 indicates delta-like ligand 4; TUNEL, TdT-mediated dUTP Nick-End Labeling.
Delta-Like Ligand 4 DNA Vaccination Does Not Induce Significant Adverse Effects and Toxicity in Mice
To address whether DLL4 gene vaccine exerts any adverse effects to mice, we conducted a careful evaluation on the impact of DLL4 gene vaccine on the general health and blood biochemistry of the vaccinated mice.
Mice immunized with DLL4 gene vaccine did not have any obvious abnormalities in food intake, drinking, fur color, urine production, bowel movement, and overall activity.
Upon examining the blood biochemistry (Table 1), DLL4 immunized mice showed a significant increase in white blood cells when compared to the control mice (F = 0.821, t = 3.482, P = .006). DLL4 vaccine did not show any hepatic and hematological toxicity.
Blood Biochemistry in DLL4-Immunized Mice.

Abbreviations: HB, hemoglobin; PLT, platelets; RBC, red blood cells; WBC, white blood cells.
a P = .006.
Discussion
Gene vaccines, also known as nucleic acid vaccines, are a new class of therapeutic vaccination for cancers. Unlike the traditional attenuated vaccines and protein subunits which mainly induce humoral immune response, gene vaccines can stimulate both cellular and humoral immune responses. As previously reported, gene vaccines could elicit both prophylactic and therapeutic effects in prostatic adenocarcinoma. 12,35 -37 Our current study clearly demonstrated that DLL4 vaccines could inhibit the growth of syngeneic model liver cancer in mice.
In gene vaccination, vaccine delivery is a critical issue that may affect the efficacy of vaccination. Hence, effective delivery approaches must be used to ensure the efficient and safe delivery of vaccinating genes to the recipients. Muscle injection is the most cost effective and a safe approach when compared to other approaches such as scratches, microinjection, ultrasound, and electroporation. 38 -43 Direct intramuscular injection of naked plasmid DNA with bupivacaine has been shown to induce a strong and long-term immune response to the antigen encoded by the vaccinating gene. 31 It was reported that pVAX1-DLL4 gene vaccination via muscle injection could mobilize the antigen-presenting cells (APCs) in the host bone marrow, 44 -46 which can then activate the cytotoxic lymphocytes (CTL) via major histocompatibility complex (MHC) I pathway. However, it should be noted that the maturation and infiltration of APCs can be affected by tumors. 47,48 Based on our optimization studies in vitro, the combination of recombinant pVAX1-DLL4 plasmids and PEAs at a ratio of 1:1 gave the most efficient transfection efficiency. Hence, this condition was used in the subsequent studies and was shown to achieve a successful up-regulation of DLL4 in recipient mice.
In our study, outbred KM mice rather than the classical nude mice or inbred mice were used because the outbred KM mice show little variation in gene purity while minimum genetic heterogeneity is maintained. 49,50 In addition, KM mice can produce several biologically active peptides that are capable of stimulating the lymphoid progenitor cells into mature T lymphocytes. Hence, this type of mice is a preferred model for studying the effect DLL4 vaccine on immune response. Tumor development may cause immune dysfunction. 51 CD4+ T cells boost the antitumor immune response, whereas CD8+ T lymphocytes have the opposite effects on the antitumor immunity. 52 With the right animal model, we have observed that DLL4 vaccine provoked immune responses in mice. Due to the lack of sufficient tissues, we were unable to examine the extent of the infiltration of CD4+ and CD8+ T lymphocytes. However, we observed that vaccination with DLL4 gene vaccine increased CD4 but decreased CD8 counts in the peripheral blood, indicating DLL4 gene vaccine possesses antitumor effects.
Indeed, we have observed a significant reduction of Ki67 expression in the syngeneic model tumors. DLL4-induced immune response is also supported by the significant induction of several key pro-inflammatory cytokines, including IL-2, IL-4, and IFN-γ. Numerous studies have shown that these cytokines are also associated with antitumor effects. For example, IL-2 produced by CD4+ T cells, CD8+ T cells, and large granular lymphocytes can stimulate nature killer cells to secrete various cytokines that are important in antitumor immune response. 53 Interferon γ is an important pro-inflammatory cytokine that induces chemokine secretion, resulting in chemoattraction of other immune cells such as macrophages. Hence, increased IFN-γ production may exert antitumor effects through stimulating phagocytosis by macrophages and upregulating MHC class I molecules. 54
Angiogenesis is an important mechanism for solid tumors to grow and metastasize. As such, antiangiogenic agents have become a class of promising anticancer drugs. However, in our studies, we have observed a significant increase, rather than a decrease of the vascular density, as indicated by increased expression of CD31 in the syngeneic model tumor tissues. Based on the published data 17,25,37 and our observation of the syngeneic model tumor tissues, we propose that DLL4 vaccine may have caused “nonfunctional vascular density.” Due to the lack of sufficient tumor tissues, we were unable to examine the messenger RNA or protein level of DLL4 in the tumor tissues directly. Now available data of high-expression anti-DLL4 in blood were consistent with the published literature that engineered DLL4 gene vaccine could induce antitumor effects in mice with a production of specific DLL4 antibodies. 22,23 It was proposed that when the balance between VEGF and DLL4 in angiogenesis was broken, higher level of VEGF may promote vascular density but do not effectively transport blood because of the reduced vascular integrity caused by DLL4. 55 In our study, we observed high proliferative fraction of vascular cells as demonstrated by increased CD31 positivity but a reduced vascular perfusion caused by DLL4 antibodies.
Induction of apoptosis is an important mechanism by which anticancer agents exert their effects. In our studies, DLL4 vaccine did not cause any apoptosis in the syngeneic model tumors, suggesting that the antitumor effect of DLL4 vaccine was not through induction of tumor cell apoptosis. More studies are required to further clarify the molecular mechanisms of tumor inhibitory effects of DLL4 vaccine.
Further studies showed that the intensity and duration of the induced immune response from DLL4 gene vaccine may be related to a variety of factors, including characteristics of different tumors, surface charge density, and reactive oxygen signals. 33,34 The observed antitumor effects by the DLL4 vaccine may also be attributed at least partially to the PEAs used as a part of vaccine delivery approach, as this reagent has been reported to stimulate immune responses, enhance local lymphatic drainage, trigger Toll-Like Receptor 9 recognition, activate DC maturation, induce Th1 response, and increase cytokine release in the recipients. 33 All these effects are known to be associated with antitumor effects. More studies are required to further clarify the molecular mechanisms of tumor inhibitory effects of DLL4 vaccine.
An important finding of our study is that DLL4 vaccines did not show any obvious adverse effects, suggesting an acceptable safety profile. In contrast, previously published studies have reported significant toxic effects of DLL4 antibody-mediated inhibition of DLL4 signaling to multiple organs in experimental animals 19,56 and in a dose-dependent manner increase the ulcerating subcutaneous tumors. 57 Hence, we believe DLL4 vaccine is superior to DLL4 antibody when used for therapeutic purposes.
In conclusion, our data provide solid evidence that DLL4 gene vaccine holds promise as a novel class of therapeutic strategy for liver cancer.
Footnotes
Authors’ Note
Yi Yu and Xiang Wang contributed equally to this work. This study was approved by Second Hospital of Lanzhou university Ethical Committee (approval no. 2019-176).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by Second Hospital of Lanzhou university Ethical Committee (approval no. 2019-176).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the National Science Foundation of China (grant no., 30571860).
