Abstract
Objective:
The purpose of this study was to evaluate the changes in pulmonary function after wedge resection, segmentectomy, and lobectomy.
Methods:
The patients were divided into 4 groups: lobectomy group, segmentectomy group, wedge resection group, and mediastinal surgery group. The pulmonary function was evaluated at various time points: 3 days before surgery, 1 month, 6 months, 12 months, and then 24 months after surgery.
Results:
The analysis results of forced expiratory volume in one second difference and breath-holding tests difference were consistent: Lobectomy group was higher than segmentectomy group, wedge resection group, and mediastinal surgery group at 4 postoperative time points (P < .05); segmentectomy group was higher than the wedge resection group and mediastinal surgery group (P < .05), and there was no statistical difference between the wedge resection group and the mediastinal surgery group (P > .05). Analysis results of tidal volume difference: Lobectomy group was higher than segmentectomy group, wedge resection group, and mediastinal surgery group at 4 postoperative time points (P < .05); segmentectomy group was higher than wedge resection group and mediastinal surgery group at 1 month after surgery (P < .05). Analysis results of the diffusion capacity of the lung for carbon monoxide difference: lobectomy group and segmentectomy group were higher than wedge resection group and mediastinal surgery group at 4 postoperative time points (P < .05). Analysis results of the stair-climbing test difference: lobectomy group and segmentectomy group were higher than the wedge resection group and mediastinal surgery group at 4 postoperative time points (P < .05).
Conclusion:
Segmentectomy was superior to lobectomy in the protection of pulmonary function, while wedge resection was superior to segmentectomy. The recovery process of the pulmonary function was the fastest during the first 6 months after surgery.
Introduction
Lung cancer is currently the most common malignancy with the highest morbidity and mortality. 1 With the popularization of low-dose spiral computed tomography (CT) screening, many patients with pulmonary nodules requiring surgery have been screened out. 2 Due to advances in surgical instruments and techniques, these pulmonary nodules can be removed with minimal invasive surgery. 3,4 Over the last few years, the 3 surgical methods for pulmonary nodules resection under uniportal video-assisted thoracoscopic surgery (VATS), namely, wedge resection (W), segmentectomy (S), and lobectomy (L), have been commonly used. 5 Although segmentectomy can preserve more lung parenchyma and improve postoperative pulmonary function theoretically, it remains unclear whether this specific procedure can provide patients with tangible pulmonary function protection compared to lobectomy. Kobayashi et al 6 reported that lobectomy decreased more pulmonary function than a segmentectomy or partial resection, while Deng et al 7 believed that there was no significantly different effect on the pulmonary function after segmentectomy or lobectomy. As fascinating as it may be, there have been numerous studies already conducted regarding this topic and only a few toward the changes in wedge resection and segmentectomy on pulmonary function under uniportal VATS. Therefore, our main focus will be to evaluate the changes in wedge resection, segmentectomy, and lobectomy under uniportal VATS on lung function.
Materials and Methods
Patients and Data Collection
We retrospectively reviewed the medical records of patients who underwent pulmonary nodules resection under uniportal VATS from July 2016 to December 2017.
To make the research results more reasonable, we have formulated inclusion and exclusion criteria. Inclusion criteria: (1) The surgical method was uniportal VATS and (2) the patient’s diagnosis before surgery was pulmonary nodules or mediastinal tumor. Exclusion criteria: (1) patients with myasthenia gravis; (2) patients who need adjuvant therapy such as chemotherapy after surgery; (3) patients with the previous history of lung surgery; and (4) patients with incomplete follow-up data. A total of 312 patients met the inclusion criteria. According to the exclusion criteria, 199 cases were excluded, and finally 113 cases were included in the group. The details are shown in Figure 1. None of the enrolled patients experienced recurrence or metastasis during the 2-year follow-up. There were 28 cases in group W, including 9 cases of benign lesions, 5 cases of atypical adenomatous hyperplasia (AAH), 12 cases of adenocarcinoma in situ (AIS), and 2 cases of stage IA non-small cell lung cancer (NSCLC). There were 33 patients in group S, including 2 benign lesions, 7 cases of AIS, and 24 cases of stage IA NSCLC. A total of 42 patients in group L included 2 benign lesions, 19 cases of stage IA NSCLC, and 21 cases of stage IB NSCLC. There were 10 cases in mediastinal surgery group (C).

The study flowchart.
All the patients who met the inclusion criteria had to go through the breath-holding tests (BHT) and stair-climbing test (SCT) tests. Each patient has to complete 3 sets of BHT, 5 minutes apart and the average was then calculated. The SCT test is to climb 80 steps and calculate the time. Pulmonary function tests (PFTs), SCT, and BHT were performed 3 days before surgery, 1 month after surgery, 6 months after surgery, 12 months after surgery, and 24 months after surgery. The corresponding difference was then acquired by subtracting the postoperative data from the preoperative data at each time point. Finally, the difference was statistically analyzed. Patient characteristics are shown in Table 1. There were no significant differences in age, gender composition, smoking index, forced expiratory volume in one second (FEV1), tidal volume (VT), diffusion capacity of the lung for carbon monoxide (DLCO), SCT, and BHT among the 4 groups.
Demographic Characteristics and Preoperative Pulmonary Function.

Abbreviations: S, segmentectomy group; W, wedge resection group; C, mediastinal surgery group; L, lobectomy group; SCT, stair-climbing test; BHT, breath-holding tests; VT, tidal volume; FEV1, forced expiratory volume in one second; DLCO, diffusion capacity of the lung for carbon monoxide.
Surgical Procedure
Wedge resection was performed when the pulmonary nodules located in peripheral and the frozen section analysis of the lesion is benign lesions, AAH, AIS, or minimally invasive adenocarcinoma (MIA). Segmentectomy is performed when the pulmonary nodules are <2.0 cm in diameter and located near the segmentary bronchial structures. Lobectomy was performed for pulmonary nodules located in lobular bronchial structures. During wedge resection, when the intraoperative frozen section diagnosis is invasive lung cancer, then lobectomy or segmentectomy is required. And lymph node dissection is performed when the intraoperative frozen section diagnosis is invasive lung cancer.
Statistical Analyses
The continuous data are expressed as mean ± standard deviation (x ± s) and were analyzed by analysis of variance (ANOVA). Furthermore, when the statistical results are statistically significant, 1-way ANOVA was used to compare the 2 groups. Differences between groups with categorical variables were assessed by the chi-square test. Statistical significance was accepted as a P value of < .05 throughout the study. All data entry and analyses were performed with SPSS17.0 statistical software (SPSS Inc.).
Results
Postoperative Changes in Pulmonary Function
The FEV1, VT, DLCO, SCT, and BHT differences were analyzed at 4 different time points: 1 month, 6 months, 12 months, and 24 months after the operation. Table 2 suggested that FEV1 difference and BHT difference analysis results were consistent: group L was higher than the group S, W, and C at 4 postoperative time points (P < .05); group S was higher than group W and C (P < .05), and there was no statistical difference between group W and group C (P > .05).
Analysis Results of Postoperative FEV1 Difference and BHT Difference.
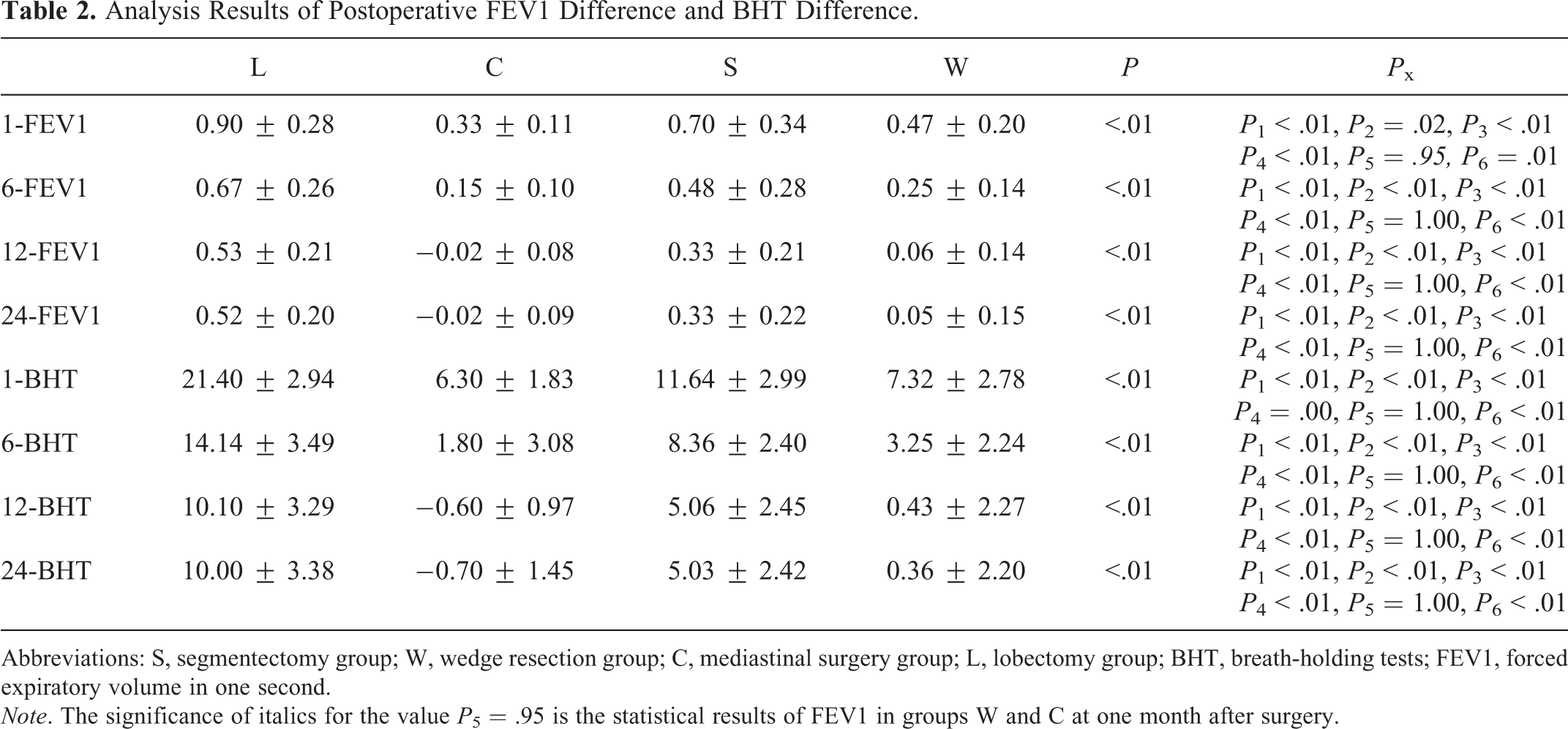
Abbreviations: S, segmentectomy group; W, wedge resection group; C, mediastinal surgery group; L, lobectomy group; BHT, breath-holding tests; FEV1, forced expiratory volume in one second.
Note. The significance of italics for the value P 5 = .95 is the statistical results of FEV1 in groups W and C at one month after surgery.
Table 3 shows the analysis results of VT differences among the 4 groups: group L was higher than group S, W, and C at 4 postoperative time points (P < .05). Group S was higher than group W and C at 1 month after surgery (P < .05), and there was no statistical difference between group S compared with group W and C (P > .05) at 6, 12, and 24 months after surgery (P > .05). There was no statistical difference between group W and group C at 4 postoperative time points (P > .05).
Analysis Results of Postoperative VT Differences.
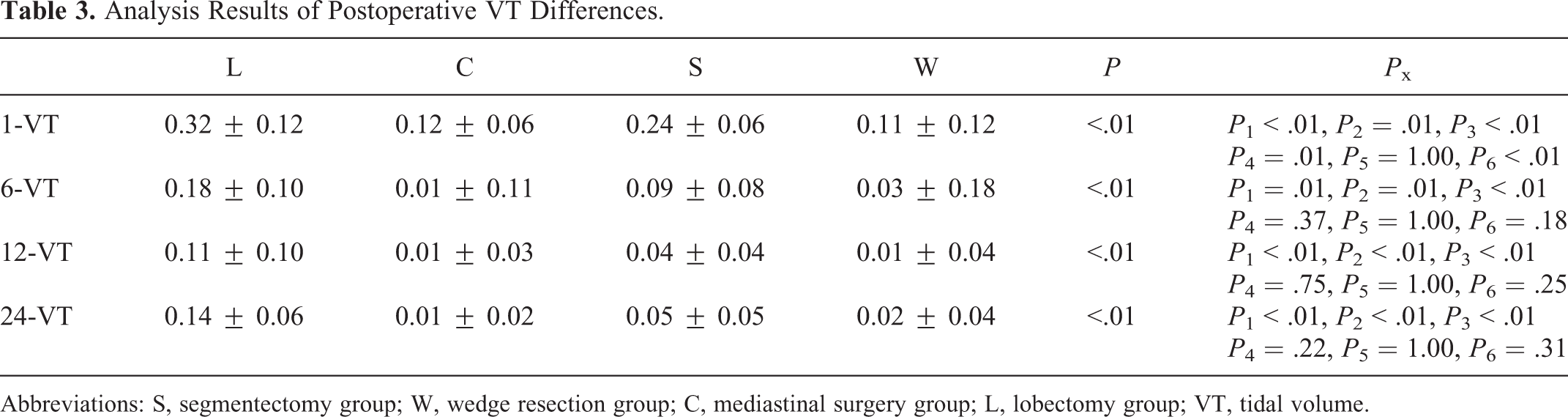
Abbreviations: S, segmentectomy group; W, wedge resection group; C, mediastinal surgery group; L, lobectomy group; VT, tidal volume.
Table 4 shows the analysis results of DLCO difference among the 4 groups: group L and group S were higher than group W and group C at 4 postoperative time points (P < .05).There was no statistical difference at 4 postoperative time points between group L and group S (P > .05) and no statistical difference at 4 postoperative time points between group W and group C (P > .05).
Analysis Results of Postoperative DLCO Difference.

Abbreviations: S, segmentectomy group; W, wedge resection group; C, mediastinal surgery group; L, lobectomy group; DLCO, diffusion capacity of the lung for carbon monoxide.
Table 5 shows the analysis results of SCT difference among the 4 groups: group L and group S were higher than group W and group C at 4 postoperative time points (P < .05). Group L was higher than group S at 1 month and 6 months after the surgery (P > .05), and there was no statistical difference at 12 months and 24 months after the surgery (P > .05). There was no statistical difference at 4 postoperative time points between group W and group C (P > .05).
Analysis Results of Postoperative SCT Difference.

Abbreviations: S, segmentectomy group; W, wedge resection group; C, mediastinal surgery group; L, lobectomy group; SCT, stair-climbing test.
1-FEV1, 6-FEV1, 12-FEV1, 24-FEV1, 1-BHT, 6-BHT, 12-BHT, and 24-BHT correspond to the difference between FEV1 and BHT at 1 month, 6 months, 12 months, and 24 months after surgery. P is the total component difference statistical result, P 1 is the comparison result between group L and C, P 2 is the comparison result between group L and S, P 3 is the comparison result between group L and W, P 4 is the comparison result between group S and C, P 5 is the comparison result of group W and C, and P 6 is the comparison result of group S and W (Tables 3 -5 are also the same).
Postoperative Recovery Rate of Lung Function
With the extension of recovery time after surgery, the pulmonary function indexes of all the groups recovered at different degrees, which were manifested by the decreasing difference of the observed indexes (Figure 2). The pulmonary function recovered quickly during the first 6 months after surgery and only minor changes after 1 year. The lung function of group C and group W almost returned to the preoperative level.

The trend graph of pulmonary function recovery speed.
Discussion
At present, majority of the patients with small pulmonary nodules are found by CT examination. In a case whereby the pathology of these pulmonary nodules with a diameter of less than 2 cm, is AIS or MIA, then the wedge resection could meet the treatment requirements as well as protect the lung function. Furthermore, 12 months after surgery, the pulmonary function in group W almost recovered to the preoperative level. 8 Some studies have shown that segmentectomy is as effective as lobectomy in the treatment of early-stage lung cancer, and segmentectomy was not found to be an independent predictor of recurrence or overall survival. 9,10 Hwang et al 11 indicated that although VATS segmentectomy achieves equal short-term surgical results and long-term oncological outcomes compared with VATS lobectomy, there was no significant difference in the decrease of postoperative FEV1 between segmentectomy and lobectomy. Some studies believe that segmentectomy has a protective effect on pulmonary function compared to lobectomy, especially in the early stage after surgery. 12,13 However, few studies have compared the protection of pulmonary function between wedge resection and segmentectomy. Therefore, we carried out this study to evaluate the changes in pulmonary function after wedge resection, segmentectomy, and lobectomy under uniportal VATS.
The surgical incision would cut off the muscles involved in breathing, affecting the postoperative lung function, especially the short-term recovery of postoperative lung function. The surgical methods in this study were uniportal VATS, which can reduce the impact of surgical incisions on the results of the study. In addition, we used mediastinal tumor resection as a control group, which made the results accurately show the impact of lung parenchymal loss on postoperative pulmonary function. We used FEV1 as the index of lung ventilation, VT as the index of lung volume, and DLCO as the index of diffusion function. Given that pneumonectomy changes both the lung and heart function, we innovatively included the SCT and BHT as indicators of cardiopulmonary function reserve in daily life. When we compare the indexes of group L and group S, we discovered that the loss of FEV1, VT, and BHT in group L was always higher than that of group S within 2 years after surgery (P < .05). The main reason is that FEV1, VT, and BHT are more dependent on the pulmonary parenchyma, and lobectomy causes more loss of pulmonary parenchyma than segmentectomy. The loss cannot be fully compensated by the expansion of other lung lobes. The loss of DLCO and SCT in the group L was higher than that in the group S within the first 6 months (P < .05), but the difference gradually narrowed with increasing recovery time (P > .05). The main reason is that DLCO and SCT could tolerate a certain degree of loss of pulmonary parenchyma. With the increase in postoperative recovery time, the body compensates for the decline in DLCO and SCT due to the loss of pulmonary parenchyma by other means such as increasing hemoglobin and cardiac ejection fraction. This is consistent with the results of Nagamatsu et al. 14 The comparison results of group L and group W were consistent with the comparison results of group L and group C: All the 5 postoperative pulmonary function evaluation indexes of group L were statistically different from those of group W and group C (P < .05), suggesting that wedge resection of lung had an absolute protective effect on pulmonary function compared to lobectomy, which was directly related to the retention of more lung parenchyma by wedge resection. The comparison results of group S and group W were consistent with those of group S and group C: FEV1, BHT, SCT and DLCO losses of group S were higher than those of group W and group C within 2 years after surgery (P < .05) This indicates that although segmentectomy retained more pulmonary parenchyma than lobectomy, the pulmonary parenchyma lost by segmentectomy cannot be fully compensated by the compensation mechanism. However, since more pulmonary parenchyma was preserved compared to lobectomy, the VT in group S no longer had differences compared to groups W and C after 6 months (P > .05). We assume this may be due to the expansion of the remaining pulmonary compensated for the VT, which is consistent with the results of Suzuki et al. 15 This suggested that segmentectomy had no significant loss of lung capacity compared to wedge resection in the long term after surgery. By comparing the postoperative indicators of group W and group C, we found that there was no statistical difference in pulmonary function losses between group W and group C in 2 years (P > .05). This result suggested that pulmonary wedge resection has less effect on pulmonary function. Given that mediastinal surgery does not remove the lung parenchyma, we believe that the pulmonary function that may be lost after wedge resection can be almost completely compensated by the body compensation mechanism. The results of this study are consistent with that of Gu et al. 16 According to the statistical results, whether lobectomy, segmentectomy, or wedge resection, the pulmonary function of the 4 groups recovered to different degrees after surgery. It was caused by the body’s compensatory mechanisms, which include compensatory expansion of residual lung, compensatory enhancement of cardiac function, increased hemoglobin, and so on. However, these compensatory mechanisms cannot make up for the loss of lung parenchyma indefinitely. If a lot of lung parenchyma is lost, the pulmonary function will inevitably have a certain degree of loss. Taken together, uniportal VATS wedge resection protects pulmonary function better than uniportal VATS segmentectomy than uniportal VATS lobectomy. This result is displayed intuitively in the trend graph (Figure 2).
With the increase in postoperative recovery time, it could be found that all the observation indicators showed different degrees of recovery with the extension of time. In addition, it can be seen from the trend graph that the recovery of pulmonary function in the 4 groups recovered fastest in the first 6 months after the surgery, slowed down gradually after 6 months, and then remained stable after 12 months. We infer that the main reason for this phenomenon is that the damaged muscles involved in breathing recover quickly, and the postoperative pain disappears quickly. Compensation mechanisms such as the expansion of the remaining lungs, increase in hemoglobin, and increase in cardiac ejection fraction slowly worked and reached their peak after one year. This result is consistent with that of Yoshimoto. 17
In summary, our results suggest that uniportal VATS wedge resection has a better protective effect on pulmonary function than uniportal VATS segmentectomy, while uniportal VATS segmentectomy has a better protective effect on pulmonary function than uniportal VATS lobectomy. Therefore, for the treatment of pulmonary nodules treatment, we suggest that the surgical method with a small resection range should be selected while ensuring the treatment effect. In addition, regardless of the method used for the operation, the patient’s pulmonary function recovered rapidly within 6 months after the operation, gradually slowed down after 6 months, and remained stable after 12 months. This is especially important for patients who may need another lung surgery. Therefore, for patients who need elective surgery, the safest period for secondary lung surgery should be chosen 6 months after the previous lung surgery. However, this study still has deficiencies. For example, the lack of hemoglobin data and cardiac ejection fraction data at the observation time point cannot verify the dynamic changes of hemoglobin and cardiac ejection fraction. In addition, the lack of sample size and follow-up time made this study defective. We will further improve these problems in future research.
Footnotes
Authors’ Note
There are no ethical problems or undeclared conflicts of interest in the submission of this manuscript, and the manuscript is approved by all authors for publication. I would like to declare on behalf of my co-authors that the manuscript has not previously been published in print or electronic form and is not under consideration by any other publication, in whole or in part. All authors have contributed significantly to the content of the article. All authors have read and approved the submission of the manuscript to Technology in Cancer Research & Treatment.
All participants in this study signed informed consent. Medical Ethics Committee of Wuxi Third People’s Hospital approved the study procedures (Approval number: IEC2016060101).
Acknowledgments
I’d like to express my sincere thanks to all those who have lent me hands in the course of my writing this paper. First of all, I would like to show my deepest gratitude to my supervisor, Dr Ma Haitao, a respectable, responsible, and resourceful scholar who has provided me with valuable guidance in every stage of the writing of this paper. Second, I did like to express my gratitude to my colleagues who offered me information on time. Last but not the least, I thank the patients for their cooperation in completing this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Youth Project of Wuxi Municipal Health and Family Planning Commission (Q201803) and Youth Project of Jiangnan University Public Health Research Center (JUPH201826).
