Abstract
Objective:
The immune makers including CD4+CD25+ T cells, natural killer cells, and T cells subgroup were retrospectively analyzed to find the relationship between apatinib and the immune system in the patients treated with apatinib.
Method:
Forty-two patients with advanced malignant tumors orally took apatinib as treatment and 16 patients with the same situation did not take apatinib as a control group. These patients were all included in the study, and they orally received apatinib 500 mg daily as monotherapy or combination. The treatment was continued until disease progression or intolerable toxicity. CD4+CD25+ T cells, natural killer cells, and T cells subgroup were detected before and 1 month after therapy for all the patients. The relationship between the changing number of immune cells and progression-free survival was analyzed in this study.
Result:
For the apatinib group, the rate of CD4+CD25+ T cells significantly increased (
Conclusion:
The rate of CD4+CD25+ T cells is very important in patients with apatinib treatment. The changing number of CD4+CD25+ T cells may be a good indicator for apatinib prognosis. Natural killer cells and T cells subgroup did not change much after apatinib, and they were not independent prognostic factors for progression-free survival.
Background
Apatinib is a kind of small-molecule inhibitor of vascular endothelial growth factor receptor 2 (VEGFR-2), the evidences coming from clinical study had demonstrated its encouraging anticancer activity in many malignant tumors, such as gastric cancer, 1 colorectal cancer, 2 esophagus cancer, 3 ovarian cancer, 4 and sarcoma. 5 In clinic, we found the effect of apatinib varied widely in the different patients. Some patients obtained satisfied effect, and the others did not get any benefit from the drug in the same pathological type tumour. According to the mechanism of the drug, the difference of the clinical effects may be caused by the different expression of VEGFR-2. However, some unknown indicators may influent this different clinical efficacy. Apatinib have an extensive antitumor activity, and the oncologists did not need to check any markers to use it, so it is widely used in a variety of tumors now. So, this status quo cause the drug abuse to some extent, and some patients did not get benefit from the drug. As a result, for the oncologist there is an urgent need to identify some markers, which can help screening patients and predicting the prognosis.
Traditionally, some chemotherapy drugs can cause temporarily impaired blood cell production in the marrow and depressed immune system functions. However, in recent years, many studies have found some chemotherapy drugs can enhance the immunogenicity of the patients with tumor. 6 Up to now, the influence of apatinib in the immune system has not reported. So, we create the hypothesis that there is some relationship between apatinib and the immune system, and we may try to find some immunity indicators which can be used to select the right patient. And then we may find some immune prognostic indicators for the use of apatinib.
In this study, we retrospectively analyzed the relationship between apatinib and the immune system. We attempt to analyze the immune markers, which included CD4+CD25+ T cells, natural killer (NK) cells, and T cells subgroup, in patients who take apatinib as treatment. And we try to find some immunity indicators which may be used for selecting the right patient or to find some immune prognostic indicators for the use of apatinib.
Methods
Patients
Between February 2017 and March 2018, 69 patients who took apatinib in Anhui Provincial Cancer Hospital (The First Affiliated Hospital of University of Science and Technology of China) were included in the study. Progression-free survival (PFS) time could be followed up of the patients. All the patients were diagnosed with advanced malignant tumor, and previously took more than one regime of treatment at least. Consents were obtained from all patients before the therapy of apatinib. In our department, the blood test of immune cells is a conventional checking for all patients. We got the rate of serum CD4+CD25+ T cells, NK cells, and T cells subgroup data from our hospital information system (HIS) system. The patients were treated with apatinib alone or combination. CD4+CD25+ T cells, NK cells, and T cells subgroup were detected in 3 days before receiving the apatinib treatment, and 1 month after treatment for all the patients. Forty-two of 69 patients who finished the immune cells detection before therapy and 1 month after therapy were included in the study. Of the 42 patients, 12 patients were gastric cancer. The other 30 patients were consisted of 9 patients with esophageal squamous cell carcinoma, 13 patients with colorectal cancer, 3 patients with ovarian cancer, 3 patients with sarcoma, 1 patient with malignant melanoma, and 1 patient with invasive thymoma. The median age was 57 years, the range from 24 to 76 years. The ratio of males and females in the study was 1.5:1 (25 males and 17 females). Seventeen patients had received regimens of apatinib in combination with one chemotherapy drug, the other 25 patients took apatinib as monotherapy. Fourteen patients had taken one regime of chemotherapy treatment previously and the other 28 patients had taken multiline chemotherapies before apatinib. Thirty-four of 42 patients had more than 1 organ metastasis. The primary analysis data consisted of age, gender, the rate of serum CD4+CD25+ T cells, the rate of serum NK cells and the rate of serum T cells subgroup, diagnosis, the therapy line of apatinib, chemotherapy regimens, and the number of metastatic organ. We also choose 16 patients who did not take apatinib as a control group (nonapatinib group). All of the 16 patients were diagnosed with advanced malignant tumor and did not take any therapy before the first time immune cells checking.
Immune Cell Detection Method
The rate of CD4+CD25+ T cells, NK cells, and T cells subgroup were all measured by flow cytometry. The following antibodies were used in our study: anti-CD4 antibody (Abcam, Cambridge, UK), anti-CD25 antibody (Abcam, Cambridge, UK), anti-CD127 antibody (Abcam, Cambridge, UK), anti-CD3 antibody (Abcam, Cambridge, UK), anti-CD16 antibody (Abcam, Cambridge, UK), anti-CD56 antibody (Abcam, Cambridge, UK), anti-CD8 antibody (Abcam, Cambridge, UK), and anti-CD45 antibody (Abcam, Cambridge, UK). Flow cytometry analysis was performed on a FACScan (BD Biosciences, Mountain View, California) using CellQuest software.
Statistical Analysis
Statistical analysis was conducted with SPSS version 16.0 (SPSS Inc, Chicago, Illinois). The difference of immune cells was analyzed by
Results
In this study, 42 patients had finished the blood test of CD4+CD25+ T cells, NK cells, and T cells subgroup before and 1 month after apatinib therapy. The other 16 patients had also finished the above examination before and 1 month after chemotherapy (nonapatinib group). The results of immune cells were recorded in percentage form. For the apatinib group, the median age was 57 years, PFS was 3.25 months. There are 32 patients still alive at the end of the follow-up period (April 27, 2018), so the overall survival was not analyzed in this article. The median value of CD4+CD25+ T cells, NK cells, and T cells subgroup before the treatment was 12.06%, 16.75%, and 74.45%, respectively. The relationship between immune cells and patients’ characteristics is shown in Table 1. The number of patients having gastric cancer with elevated rate of T cells subgroup was larger than that of the other patients with malignant tumor (χ2 = 4.200,
Characteristics of 42 Patients and Relationship Between Immune Cells.

Abbreviations: GC, gastric cancer; NK, natural killer
Kaplan-Meier Analysis of the Relationship Between PFS and Clinical Characteristics (Apatinib Group).

Abbreviations: CI, confidence interval; mPFS, median progression-free survival.
Influence of Serum CD4+CD25+ T Cells, NK Cells, and T Cells Subgroup in PFS (Apatinib Group).
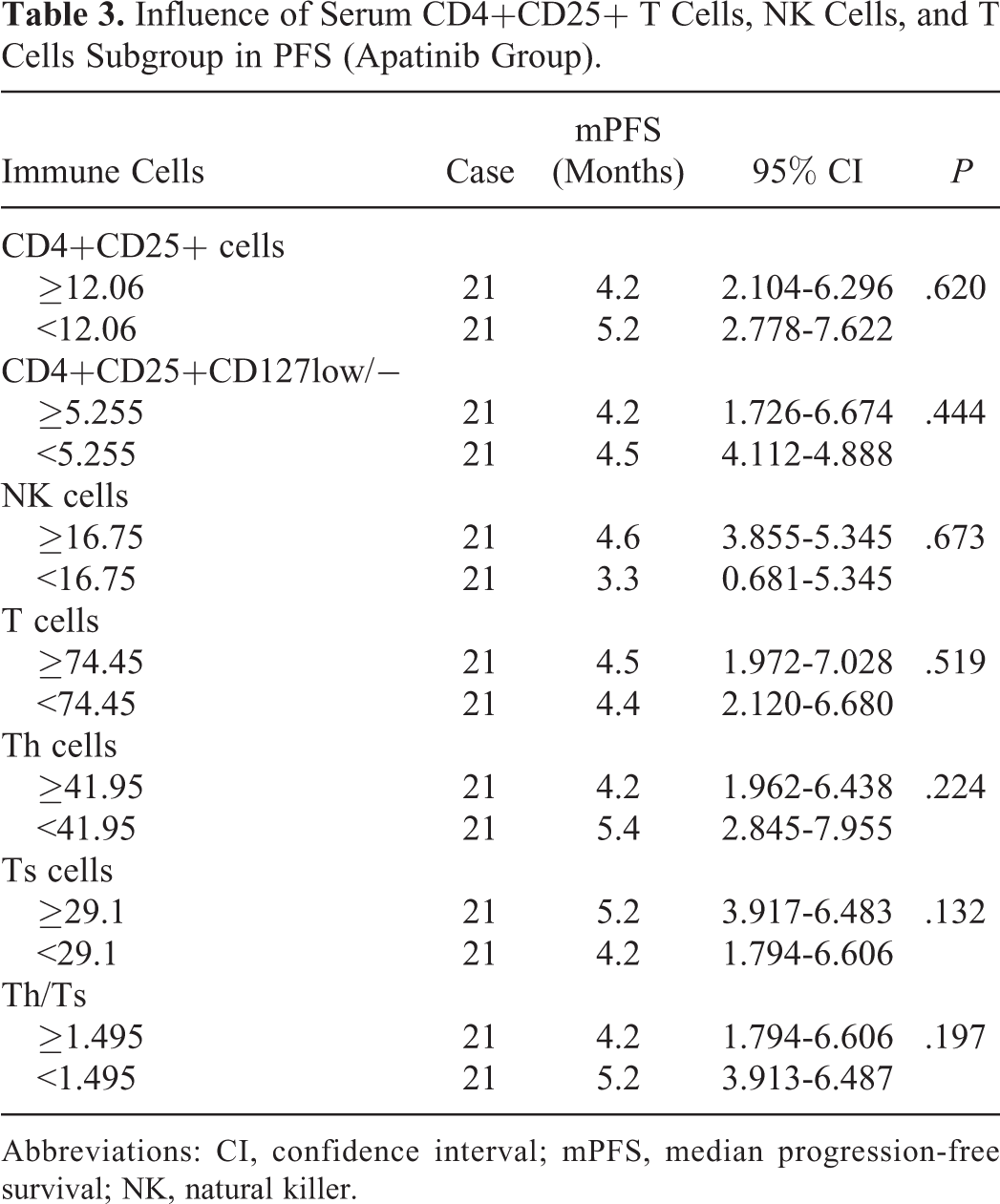
Abbreviations: CI, confidence interval; mPFS, median progression-free survival; NK, natural killer.
The Characteristics of 16 Patients and the Relationship Between Immune Cells(Nonapatinib Group).

Abbreviations: GC, gastric cancer; NK, natural killer.
After 1 month therapy of apatinib, the rate of CD4+CD25+ T cells elevated significantly (T = −2.042,
The Difference of Immune Cells Before and After Therapy (Apatinib Group).

Abbreviation: NK, natural killer.
The Influence of Serum CD4+CD25+ T Cells, NK Cells, and T Cells Subgroup Before and After Therapy in PFS (Apatinib Group).

Abbreviations: CI, confidence interval; NK, natural killer; PFS, progression-free survival.

The median progression-free survival of regulatory T cells increased and decreased group (apatinib group).
Multivariate Analysis of PFS in 42 Patients (Apatinib Group).

Abbreviations: CI, confidence interval; NK, natural killer; PFS, progression-free survival.
Multivariate Analysis of PFS in 16 Patients (Nonapatinib Group).
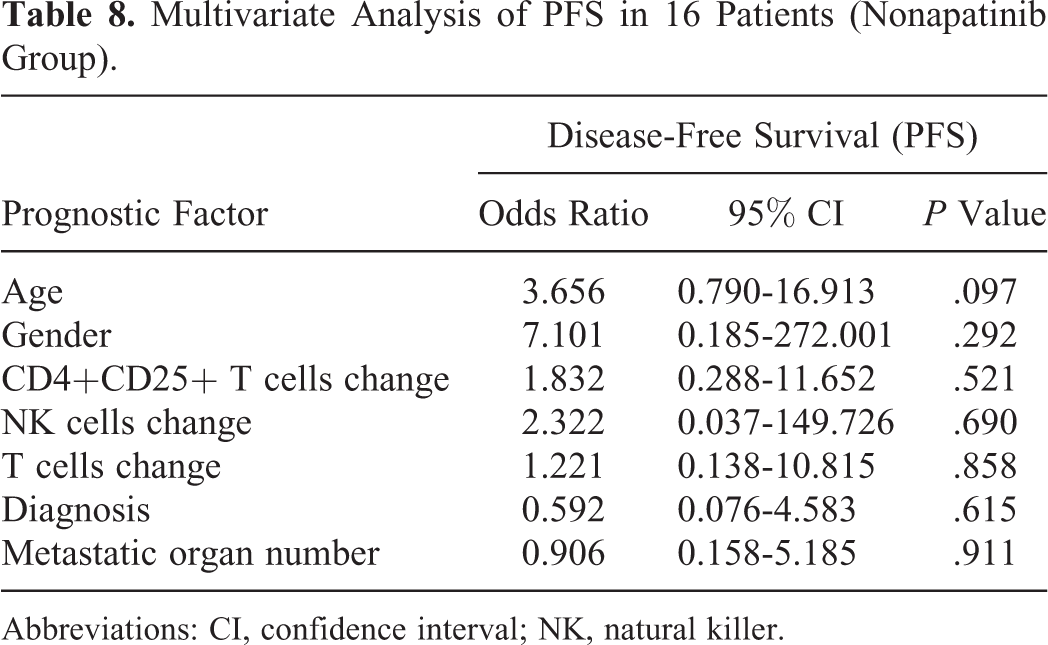
Abbreviations: CI, confidence interval; NK, natural killer.
Discussion
For many malignant tumors there were always no effective medicines available in the late stage. Apatinib is a small molecule which could inhibit VEGF-stimulated endothelial cell migration and proliferation and decrease tumor microvascular density. 7,8 It was approved and launched in People’s Republic of China in 2014 as a subsequent-line treatment for patients with advanced gastric cancer. And the clinical trials of this drug in other tumors are currently undergoing in clinic. In our clinical work, we find apatinib is a new strategy for the treatment of a variety of solid tumors, such as non-small cell lung cancer, breast cancer, and hepatocellular carcinoma, esophageal cancer, sarcoma, and so on. During the apatinib medication, we have noticed an interesting phenomenon. Some patients have obtained good results, and these patients always have a very long PFS after the apatinib treatment. However, the others have the disease progression rapidly and die very soon. As apatinib was approved in advanced gastric cancer, it was also chosen for the other types of tumors because there was no effective medicines available, as well as intolerable chemotherapy. This status quo may be related to the poor prognosis. But in our work, we also find some patients with a good eastern cooperative oncology group (ECOG) cannot get a good result from apatinib. What’s more, up to now there is no effective prognostic indicator for the use of apatinib. This phenomenon makes us think apatinib may have an influence on immune system, and the immune system may be an important factor for the therapeutic effect.
CD4+CD25+ T cells is a subpopulation of suppressor T cells defined based on expression of CD4 and CD25. CD4+CD25+ T cell is an important subtype of T lymphocytes. It’s a group of regulatory T cells and is differ from Th1 and Th2 cells. CD4+CD25+ T cells, 5% to 10% of peripheral CD4+T cells, resemble anergic cells
The depletion of CD4+CD25+ T cells can disrupt immunological unresponsiveness to autologous tumors, thus mediated effective immune responses to tumor cells. 17,18 The CD4+CD25+ T cells can suppress all subsets of Vα24+NKT cells, which including Vα24+CD4-CD8- cells, Vα24+CD4+ and Vα24+CD8+, in both proliferation and cytokine production, such as interferon γ, interleukin-4 (IL-4), IL-13, and IL-10. 19 So the elevated rate of CD4+CD25+ T cells suppressed the cytotoxic activity of Vα24+NKT cells and weakened the action of antitumor. Study had found CD4+CD25+ T cells was an important contributor to the development of tumors immune tolerance and they play a critical role in the induction of tolerance to tumor associated antigens and suppression of antitumor immunity. 20 CD4+CD25+ T cells consist of natural Tregs (nTregs) and peripherally induced Tregs (iTregs). 21 Natural Treg, with Foxp3 expression, are thymus derived, constitutively express CD25, and their mechanism of suppression is cell contact dependent. 22 Induced Treg, adaptive Treg, is developing in the periphery, which Foxp3 is considered unstable. Induced Treg cells variably express CD25, and they work by cells contact and cytokine dependent mechanism. 23,24 Researchers found CD4+CD25+ T cells limited beneficial responses by suppressing sterilizing immunity and compromising anti-tumor immunity. 25 Many studies showed high rate of CD4+CD25+ Treg cells was closely related with poor prognosis. 26,27 The levels of these cells in malignant tumor patients were higher than in non-neoplastic patients. 28 Up to now, the inhibitory cytokines of IL-10, IL-35, and transforming growth factor β are known as the 3 key mediators of Tregs function. 29,30
Given the above, increased levels of CD4+CD25+ T cells in the peripheral blood of patients with cancer were detected as compared to normal healthy control. And the previous research suggested the higher rate of CD4+CD25+ T cells should be connected with poor outcome. But in our study, we found the patients with CD4+CD25+ T cells increased after apatinib treatment had a better outcome. This means the elevated Treg cells were closely associated with longer PFS. But the rate of NK cells and T cells subgroup did not change much after apatinib, and they were not an independent prognostic factor for PFS. The result is contrary to conventional wisdom. This is an interesting phenomenon, and the mechanisms are worthy of further investigation.
Apatinib is a small molecule which could inhibit VEGF-stimulated endothelial cell migration. Vascular endothelial growth factor receptor 1 has been implicated in macrophage chemotaxis and tumor cell survival and invasion. Vascular endothelial growth factor receptor 2 is the primary angiogenic receptor, and apatinib is working on VEGFR2, hence inhibiting the formation of new blood vessels in the tumor tissues and the growth of tumor cells. Marek
Conclusion
CD4+CD25+ T cells are very important for patients with apatinib treatment. The changing number of CD4+CD25+ T cells may be a good indicator for apatinib, even for anti-VEGF(R) drug prognosis.
Footnotes
Authors’ Note
Our study was approved by the Ethics Committee of Anhui Provincial Hospital (The First Affiliated Hospital of University of Science and Technology of China West District; approval no. 2018-17). All patients provided written informed consents prior to enrollment in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical Scientific Research Foundation of Anhui Province, China (1808085MH234) (1408085MH179).
