Abstract
Background
As the number of cancer survivors increases, the prevalence of the second type of primary cancer will increase. In clinical trials, patients with a history of malignant tumors in the past are usually excluded. It is unknown whether previous cancers affect survival outcomes. The purpose of this study was to investigate the impact of previous malignant tumors on the long-term prognosis of patients with gallbladder cancer.
Methods
By using the Surveillance, Epidemiology and END Results (SEER) database, we collect patient data and obtain patients who had gallbladder cancer diagnosed in 2004–2015 and had an adaption and contrast 1:1 with cases. We applied the Kaplan–Meier analysis and Cox regression models to assess the influence of prior malignancy on gallbladder cancer survival outcomes.
Results
Among 8338 patients who had mainly gallbladder cancer, 525 (6.3%) suffered prior cancer. Prostate cancer (22.29%), Breast cancer (21.14%), and Genitourinary (14.67%) are the most common types. Before propensity score matching (PSM), two groups of different Kaplan–Meier curves were obtained by classifying previous cancer history, and by comparison, the all-cause difference in the group with previous cancer history was not salience (P = 0.31), but there is a protective effect on the Cancer-specific fatality rate (p < 0.001). Similar results were obtained after propensity score matching (PSM). Among the multivariate Cox analysis, previous malignancy had no obvious relation, including all causes (HR = 0.98, 95% CI: 0.86–1.12, p = 0.70) but a better gallbladder cancer-specific survival (HR =0.64, 95% CI: 0.55–0.75, P < 0.001).
Conclusion
Prior cancer may not be an obvious factor impacting the survival of cancers of all-cause including the gallbladder. In clinical trials of gallbladder cancer, exclusion criteria based on cancer history should be assessed.
Introduction
Being a frequent malignancy in the biliary system, gallbladder cancer (GBC) accounts for about 2/3 of all tumors of the biliary system, and its incidence has increased in recent years.1,2 Unfortunately, GBC is associated with poor prognosis, with a 5-year survival rate of less than 2%. 3 However, significant advancements have been made in the treatment and prognosis of GBC, particularly due to the understanding of its unique tumor biology and the emergence of molecularly targeted therapies. One promising avenue for the treatment of metastatic solid tumors, including GBC, lies in targeting genomic changes. 4 The latest progress in this area is the development of the PD-L1 inhibitor durvalumab, which opens a therapeutic prospect for the suppression of immune checkpoints in GBC.5,6 In the United States, with the improvement of clinical diagnosis and treatment, there has been a rapid increase in the number of cancer survivors (CS), which has quadrupled recently. 7 However, it is crucial to remain vigilant about the risk of developing a second type of cancer. Despite this, there is currently limited understanding regarding the clinical features and prognostic variables of GBC as secondary cancer or other forms of premalignancy.
The use of clinical trials to enhance the survival of patients with GBC is becoming increasingly common. However, patients with a history of malignancy are typically excluded from these trials.8–10 The rationale behind such exclusion criteria lacks verification and support from evidence-based guidelines. 11 It is worth noting that less than 5% of cancer patients are deemed eligible for participation in clinical trials.12,13 Overly stringent exclusion criteria can significantly limit the generalizability and accuracy of clinical trial results. To shed light on the impact of previous cancer on overall survival, a comprehensive pan-cancer investigation was conducted. The findings revealed that the influence of a prior cancer on survival outcomes varied depending on the specific type of cancer previously diagnosed. 14 Building upon this research, some researchers have reported the effect of a previous cancer history in various contexts. For example, a retrospective survey conducted by Laccetti et al. illustrated that the presence of cancer history does not negatively affect the clinical prognosis of patients with advanced-stage lung cancer, regardless of the previous cancer type or timeframe.15–17 Another study also showed that a history of cancer has no significant impact on the prognosis of patients with intrahepatic cholangiocarcinoma. 18 However, in the case of laryngeal and gastric cancers, previous cancer history has been found to adversely affect prognosis.19,20 Despite these findings, the influence of a prior cancer diagnosis on the prognosis of patients with gallbladder cancer remains understudied and requires further investigation.
Therefore, we utilized the Surveillance, Epidemiology, and End Results (SEER) database to investigate the impact of previous cancers on clinical outcomes in patients with GBC.
Methods
Patients and Data Sources
The patient data is obtained by SEER research plus data, 18 registries, Nov 2020 sub (2000-2018) using seer*stat software (version 8.4.0) for clinical data; Seer research data, 18 registries (excl AK), Nov 2020 sub (2000-2018), applied for gaining detailed information about a variety of cancers; Seer∗stat software (version 8.4.0) for data discovery. Due to the availability and utilization of de-identified data, the permission of our institutional review committee is not necessary.
There are qualified patients (more than 18 years of age) who diagnosed with primary gallbladder cancer from 2004 to 2015 in the database were recruited. The following patients were not the target patients of the current research: tumors in situ or benign tumors, not actively followed up, unknown months of survival and state of life, having more than one type of cancers, or prior cancer information known from autopsy or death certificates alone. Included individuals were in two groups according to have a prior malignancy or not. Dividing individuals with malignancies into subgroups by the site of previous cancer, involving prostate, lung and bronchus, colon and rectum, liver, the intrahepatic bile duct, hematologic, kidney, genitourinary, head, neck, breast, and other cancers. To avoid the influence of concurrent malignant tumors, patients who had previous cancer or gallbladder cancer diagnosed within 6 months were excluded.
Covariates and Endpoint
An analysis of 11 variables was conducted in this study, including age (<65 ≥ 65), gender (male, female), race (White; Black; Other), marital status at diagnosis (Single; Married; Unknown), grade (Grade I/II; Grade III/IV; Unknown), histological. Type (adenocarcinoma (AC), Others), tumor size (cm) (≤3.6; 3.6-6;>6; Unknown), surgery (Yes; No; Unknown), chemotherapy (None/Unknown; Yes), radiation (Yes; No; Unknown) and AJCC.6th. stage(I; II; III; IV; Unknown). The outcomes included overall survival (OS) and cancer-specific survival (CSS). The overall survival is the survival period with any reason from diagnosis of gallbladder cancer to death, and the cancer-specific survival is the period from diagnosis of gallbladder cancer to death from gallbladder cancer.
Measures
In previous studies, the previous cancer history is identified by the SEER serial code recording. In summary, each individual's tumor was numbered in the order in which each SEER was submitted; and these orders were based on the date and order of diagnosis. The interval between gallbladder cancer and previous cancer was calculated by using the SEER survival months of the indicator cancer and the recent survival months of any previous cancer. Median interval: We calculated the period from the diagnosis of a previous cancer date to the diagnosis of the target cancer date, ie, the period lapse between two cancer records.
Statistical Analysis
The Pearson chi-square test was applied for investigating categorical variables. Kaplan Mayer's analysis was used to compare overall survival (OS) and cancer-specific survival (CSS) by log-rank test before and after PSM, and Cox proportional hazards models adjusted by multivariate propensity scores were constructed to determine whether previous cancer history is an independent prognostic factor for OS. In addition, subgroup analysis was performed to examine the gallbladder cancer according to age, gender, grade, histological type, chemotherapy, marital status, surgery, tumor size.cm and AJCC stage of gallbladder cancer, as well as time, type, and staging of previous cancer in more depth to further investigate the prognostic impact of previous malignancy. Two-tailed P values < 0.05 were considered statistically significant. SPSS (version 20.0) and R software (version 4.1.2) were used for statistical analysis and image generation.
PSM Analysis
To minimize the baseline confounding factors, A caliper of 0.2 was used to perform a one-to-one closest PSM between previous cancer and non-previous cancer. The adjusted variables included age, race, marital status, histology, pathological type, tumor size, primary tumor surgery, radiotherapy, chemotherapy and AJCC.6th. stage. In addition, the adjusted p value of propensity score is also provided. The PSM program uses R packages of Matching (ver. 4.1.0).
Results
Demographics and Clinical Characteristics of the GBC Patients
8338 qualified patients who had gallbladder cancer were determined in the SEER database, involving 525 (6.3%) patients with prior cancer and 7813 (93.7%) patients who had no prior cancer. Gallbladder cancer most frequently occurs in white female individuals ≥ 65 years old. After adjusting the propensity score, total variables had well equilibrium in all patients who had cancer or not (p > 0.05). The Specific baseline characteristics before and after PSM are summarized in Table 1.
Patient Demographics and Pathological Characteristics with Gallbladder Cancer in the Original/Matched Data Sets.

Note: AC: adenocarcinoma; AJCC: American Joint Committee on Cancer.
The Type, Interval time, and Stage of the most recent previous malignancy are illustrated in Figure 1 using a tree diagram. Prostate cancer (22.29%), Breast cancer (21.14%), and Genitourinary (14.67%) are the most common types of patients who have a previous cancer history. The median interval was 48 months from previous cancer diagnosis to indicator cancer. The great majority of previous cancers were diagnosed at the local (41.90%) or regional (20.19%) stage.

Type distribution (A), interval time (B) and stage (C) of previous cancer for patients who have Gallbladder cancer.
Effect of Prior Cancer on All-Cause and Gallbladder Cancer-Specific Survival
Overall, the Kaplan–Meier survival curve before PSM did not show an obvious discrepancy in overall survival (P = 0.31) and cancer-specific survival (P < 0.001) in patients who had previous cancer or not (see Additional Figure S1). After adjusting the propensity score (Figure 2A), the Kaplan–Meier diagram showed that in patients who had previous cancer history, the prognosis was not poor (P = 0.25), indicating that previous cancer had no poor influence on overall survival (OS). In terms of gallbladder cancer and non-all gallbladder-specific mortality, patients with previous malignant tumors were significantly different from patients without malignant tumors which is illustrated in Figure 2B (all P < 0.001). It turns out that prior cancer diagnosis is a favorable prognostic factor for GBC, while the reason is unknown. For patients who had prior cancer history, the 1 -, 3 -, and 5-years overall survival rates were 41.9%, 21.3%, and 16.9%, respectively. For patients without previous malignancies, the 1 -, 3 -, and 5-years overall survival rates were 42.0%, 20.3%, and 16.2% respectively.

Overall (A) and gallbladder cancer-specific (B) survival (CSS) for patients whether presence of prior cancer diagnosis following tendency score matched. Two-sided p-values were investigated and contrasted according to the log-rank test.
After adjusting grade, pathology, surgery, radiotherapy, primary tumor size, and AJCC stage, we used multivariate Cox regression to show that a history of prior cancer is not an independent prognostic factor for OS. (HR = 0.98, 95% CI:0.86–1.12, P = 0.70), while a history of prior cancer is a favorable prognostic factor for CSS (HR = 0.64, 95% CI:0.55–0.75, p < 0.001) (Table 2). It shows that patients who suffered cancer are more possible to die because of non-gallbladder cancer causes than patients who had no previous cancer.
Multivariable Cox Regression Analysis of All-Cause and Gallbladder Cancer-Specific Survival in Patients with Gallbladder Cancer.

Note: AC: adenocarcinoma; AJCC: American Joint Committee on Cancer; HR: hazard ratio; CI: confidence interval; OS: overall survival; CSS: cancer specific survival.
Figure 3 A and B show the subgroup investigation results stratified by age, gender, stage, histological type, tumor size, surgery, chemotherapy as well as AJCC stage of gallbladder cancer. Besides the subgroups with age ≥ 65, tumor size.cm (3.6–6; > 6), and stage II, other groups showed no significant correlation between the history of pre-existing cancer and the overall surviving period. For the gallbladder cancer's specific survival rate, except for tumor size > 6, stage II and stage III, other subgroup analyses showed no significant correlation with specific mortality. The rest are protective factors of specific mortality.

Prior cancer's subgroup analysis influence on all-cause (A) and gallbladder cancer-specific survival (B) stratified by age, gender, stage, histological kind, chemotherapy, marital status, surgery, tumor size(cm) as well as gallbladder cancer's AJCC stage. AJCC, American Joint Committee on Cancer.
Influence of Interval Time, Type, and Stage of a Malignancy History on Overall Survival
Figure 4 shows the subgroup investigation stratified by the laitance from previous cancer to gallbladder cancer diagnosis, and furthermore studying the impact of previous cancer on survival. In the 6–12 months, 1–5 years, 5–10 years as well as more than 10 years subgroups, previous cancer had no obvious influence on prognosis (p > 0.05). Multinomial covariate-adjusted Cox regression mode for subgroups analysis was stratified by latency according to Table 3, and they confirmed that a history of prior cancer would not be a separate risk element for general survivorship in the subgroup. Meanwhile, through subgroup analysis stratified by various previous cancer types which are illustrated in Table 4, just previous Hematologic(HR 0.74, 95% CI 0.56–0.99, and p = 0.04) had poorer overall survival but better gallbladder cancer-specific survival, only prior head and neck (HR 0.69, 95% CI 0.44–1.07, and p = 0.10), Liver& Other Biliary (HR 0.57, 95% CI 0.29–1.10, as well as p = 0.10) and Others cancer (HR 0.75, 95% CI 0.47–1.19, p = 0.22) showed no significant correlation with specific mortality.
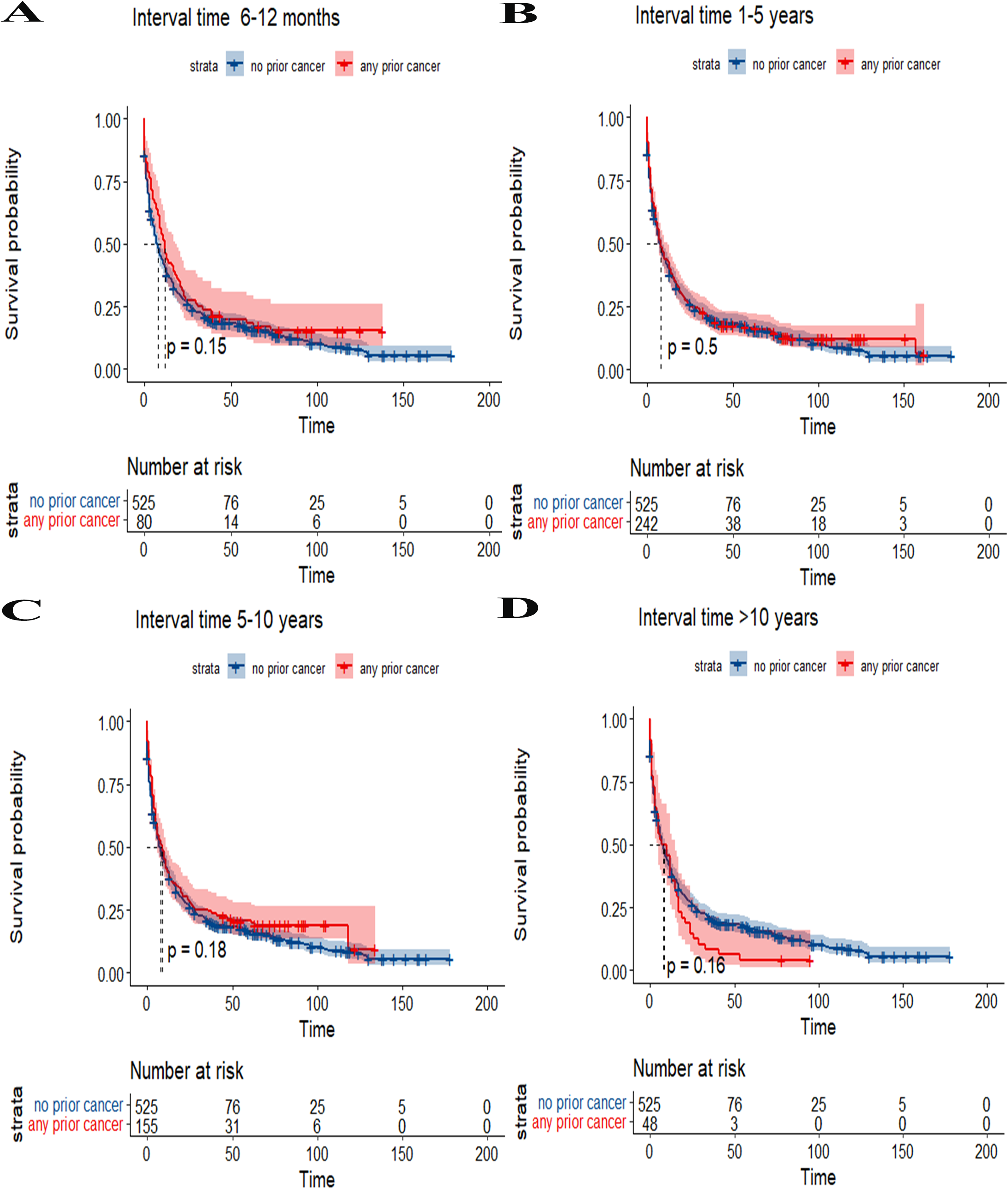
Subgroup investigation of the influence of previous cancer on overall survival stratified by the previous cancer period diagnosis in patients who had gallbladder cancer. (A) Kaplan Meier curve of the effect of previous cancer on survival, with a latency period from 6 to 12 months; (B) Kaplan Meier curve which influences prior cancer on survival had a latency from 1 to 5 years; (C) Kaplan Meier curve influences prior cancer on survival had a latency from 5 to 10 years; (D) Kaplan Meier curve influences previous cancer on the survival rate had incubation time more than 10 years.
Multivariable Cox Regression Analysis in Overall Survival in the Incubation Subgroup.

HR, hazard ratio; CI, confidence interval; AJCC, American Joint Committee on Cancer.
Multivariate Cox Regression Analysis of OS and CSS in GBC Patients Stratified Through the Original Site of Previous Cancer.
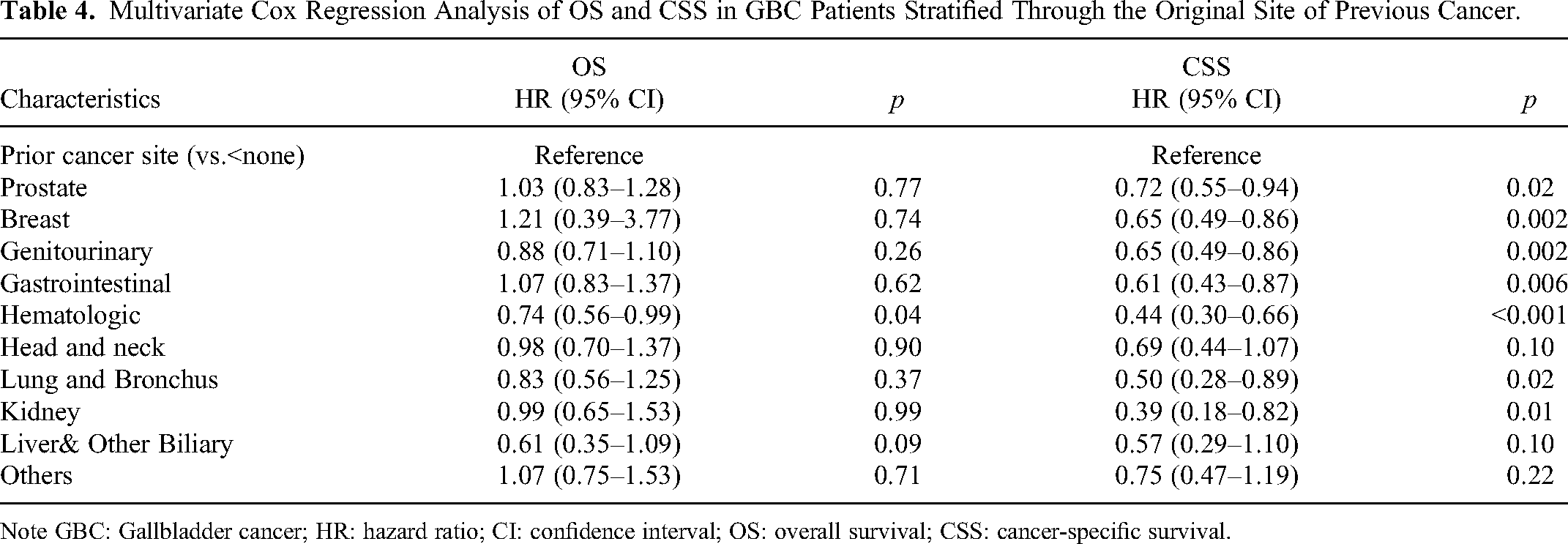
Note GBC: Gallbladder cancer; HR: hazard ratio; CI: confidence interval; OS: overall survival; CSS: cancer-specific survival.
Figure 5 shows overall survival stratified by types and stages of prior cancer. According to all the prior cancer types recorded, OS was displayed based on prior cancer's different types. Among patients who had previous cancer history, the survival rate with previous gastrointestinal cancer is often the worst (log-rank test p < 0.05), and the survival rate is obviously lower than that of patients who had no previous cancer. Using a log-rank way to contrast the different groups’ survival rates, contrasted to patients who had no previous cancer, the other patients’ survival rates who had prior cancer history seemed to have no significant difference. In the subgroup investigation stratified through previous cancer stages, there is no obvious distinction in the survival rate of patients who suffered previous cancer at each stage contrasted with patients who had no previous cancer history (p > 0.05).

Prior cancer's subgroup analysis influences overall survival in patients with gallbladder cancer stratified by prior cancer's type (A) as well as stage (B).
Discussion
In the past few decades, the survival rate of patients diagnosed with cancer has significantly improved, resulting in an increasing number of CS within the general population. 21 In the CS population, the long-term side effects of radiotherapy and chemotherapy, advanced early screening and diagnostic techniques, and the continuous impact of risk factors have led to a significant increase in the incidence rate of multiple malignant tumors. 22 Epidemiological studies have reported that the incidence rate of multiple primary malignant tumors falls within the range of 2%–17%.23,24 However, it is worth noting that most clinical trials exclude patients with a history of previous malignancy, resulting in limited research focus on these patients. Currently, there is no research report on the characteristics and prognosis of GBC patients with previous cancer history. Therefore, it is crucial to direct attention towards these patients.
According to data from the SEER database, our research focused on investigating the influence of prior cancer on the prognosis of patients with GBC. Among more than 8000 patients, we found that 6.3% of them had a history of previous cancer. We analyzed the clinical characteristics of these patients and made some significant observations. Contrary to expectations, we discovered that the OS or CCS rates of GBC patients with previous cancer were not poor when compared to GBC patients without a history of previous cancer. To ensure the validity of our findings, we stratified the patient population based on the type, stage, and interval time of the previous cancer, and conducted a thorough analysis. Multivariate Cox regression analysis illustrated that previous malignancies were not correlated with poor all-cause survival but, surprisingly, acted as protective factors for gallbladder cancer-specific survival. These findings were consistent across different subgroups based on the time of occurrence of the previous cancer, including subgroups with intervals ranging from 6 to 12 months, 1 to 5 years, 5 to 10 years, and more than 10 years. Based on these compelling results, we believe it is crucial to reconsider the exclusion of patients with a history of prior cancer when studying the major endpoints of OS or CSS.
This study has several interesting findings. First, as the number of CS increases, prostate and breast cancer are the most common types in the study among patients with previous malignant tumors. There are several potential reasons for this, (1) prostate cancer and breast cancer are known for their relatively high incidence rates and favorable prognoses due to their indolent characteristics in men and women, respectively, (2) previous cancer treatments can increase the risk of developing a second type of cancer. The multiple primary incidences of prostate cancer patients after radiotherapy ranged from 1.14% to 8.7%. 25 The late toxicities of radiotherapy and chemotherapy can increase the risk of the second primary tumor after breast cancer. Hormone treatments used for primary breast cancer can also contribute to an increased risk of endometrial cancer, gastric cancer, colon cancer, and ovarian cancer. 26 Particularly, the risk of endometrial cancer is observed to be higher after tamoxifen treatment. However, current studies did not find a significant correlation between OS and the presence of prostate cancer or breast cancer in patients with GBC.
Another interesting phenomenon in this study is the distinct clinical features observed in GBC patients with a history of prior cancer compared to those without previous malignancies. Our investigation revealed that GBC patients who had prior cancer are older (≥65 years), a trend consistent with research concentrated on other types of cancers.27,28 The Kaplan–Meier, Multivariable Cox regression analysis and Subgroup analysis showed a decrease in the specific mortality of gallbladder cancer in patients with a history of previous cancer, aligning with the previous findings in other cancer types.18,29 These studies showed an increased risk of death from previous cancer or non-cancer causes when compared to patients without a previous cancer history. Subgroup stratified analysis further showed that the previous cancer history may act as a protective factor against specific mortality of gallbladder cancer, except in cases where the tumor size > 6, stage II and stage III. No significant correlation with specific mortality was found in other subgroup analyses, suggesting that these factors contribute to a lower risk of specific mortality. The possible explanation is that patients with a history of robust tumor biology and treatment response are more likely succumb to primary tumor-related causes. In addition, most prior cancers are in a localized or regional stage. These results may be due to the fact that these CS often live long enough to develop a second type of cancer.
Finally, in addition to considering the influence on prognosis, there are other potential causes to exclude patients with previous cancers diagnosis from clinical trials. Firstly, patients who have undergone chemotherapy and radiotherapy for previous malignancies may have compromised resistance to the current therapeutic trial, thereby disrupting the effectiveness. 17 Secondly, prior cancers can induce a range of immune reactions, damage target organs, or result in additional health conditions such as immune deficiency. These factors significantly reduce the reliability of the outcomes and the usefulness of experimental patients in the trial. However, there are alternative strategies that can be employed to address this issue. For example, some clinical trials have implemented exclusion criteria for patients with concurrent critical conditions or organ dysfunction, as well as those who have previously received cancer therapies.30,31
To comprehensively evaluate the impact of previous malignant tumors on heterogeneity, we used subgroup analysis based on specific cancer site stratification. Within our cohort, past hematological malignancies were found to have an adverse impact on the OS rate of gallbladder cancer. This finding aligns with a previous study that demonstrated poor survival outcomes among individuals with previous hematological tumors. 32 We hypothesize that the adverse prognosis could be attributed to the invasive biological behavior and high mortality rate associated with hematological tumors. We found that types of previous cancers, other than hematological tumors, did not have a significant impact on the OS prognosis of gallbladder cancer. Therefore, it is crucial to develop more precise and specific inclusion criteria for future clinical trials. These criteria should be based on factors such as treatment tolerance, organ function, and overall health status of the patient. For instance, instead of considering previous cancer diagnoses, certain treatments for past cancers such as radiation therapy, chemotherapy, biological therapy, or recurrence could be used as exclusion criteria. Like the exclusion of patients with other severe comorbidities or organ dysfunction, many clinical trials currently exclude individuals who have previously received cancer treatment. To ensure a more accurate representation of the real-life population and enable the participation of patients with a history of cancer in clinical trials, it may be beneficial to modify the exclusion criteria, especially in early-stage clinical trials. The modified criteria could exclude only those patients who currently require active anti-cancer treatment. Undeniably, this modification would increase the complexity of evaluating efficacy and progress. 33 Nonetheless, by investigating the specific effects of previous cancer stratified according to clinical features on the prognosis of patients with GBC, we aim to derive more accurate conclusions that can effectively guide clinical treatment.
Some potential restrictions should be acknowledged in this study. Firstly, this study relies on retrospective data from the SEER database, which inevitably introduces selection bias. Although the study attempts to mitigate this bias by utilizing the PSM, it is possible that there are hidden biases caused by unobservable confounding factors that cannot be completely ruled out. Secondly, the SEER database lacks comprehensive information on treatment details and relapse occurrences. Specific characteristics of previous malignancies were not available in this dataset, limiting the analysis to focus solely on type stage and interval time. The accumulated evidence illustrates that GBC is a persistent inflammatory illness fostered by various risk elements, such as gallstone disease, smoking, sedentary lifestyle, drinking, metabolic disorders, hypercholesterolemia, and high-fat diet, as well as various forms of infections. 34 Unfortunately, the SEER database does not provide information on these lifestyle elements. Thirdly, coding inaccuracies and extensive variability within large databases may have influenced the accuracy of the results. Finally, the SEER database covers only approximately 9.4% of the US population. Therefore, the generalizability of the research results to the entire population requires further confirmation.
Conclusion
In summary, the characteristics of demographics and clinical characteristics of GBC patients who have previous cancer are obviously distinct from those of patients who do not have cancer. Previous cancer didn’t result in poorer outcomes for cancer survivors. Patients who had previous cancer should be rethought for inclusion in clinical trials. Additional research is necessary to verify our discovery. This may help to appropriately increase the number of trials enrolled and draw more general conclusions to guide prospective treatment of gallbladder cancer.
Supplemental Material
sj-tif-1-tct-10.1177_15330338231183937 - Supplemental material for The Impact of a History of Different Other Cancers on the Long-Term Outcomes of Patients with Gallbladder Cancer: A Propensity Score–Adjusted, Population-Based Study
Supplemental material, sj-tif-1-tct-10.1177_15330338231183937 for The Impact of a History of Different Other Cancers on the Long-Term Outcomes of Patients with Gallbladder Cancer: A Propensity Score–Adjusted, Population-Based Study by Jing Wang, Chan Zhang and Dong Yan in Technology in Cancer Research & Treatment
Supplemental Material
sj-pdf-2-tct-10.1177_15330338231183937 - Supplemental material for The Impact of a History of Different Other Cancers on the Long-Term Outcomes of Patients with Gallbladder Cancer: A Propensity Score–Adjusted, Population-Based Study
Supplemental material, sj-pdf-2-tct-10.1177_15330338231183937 for The Impact of a History of Different Other Cancers on the Long-Term Outcomes of Patients with Gallbladder Cancer: A Propensity Score–Adjusted, Population-Based Study by Jing Wang, Chan Zhang and Dong Yan in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
We gratefully acknowledge the Surveillance, Epidemiology, and End Results (SEER) program tumor registries for the creation of the SEER-Medicare database.
Author Contributions
DY, JW conceived and designed the present study. JW, CZ analyzed the data and wrote the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
Competing Interests
The authors declare that they have no competing interests.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Beijing Municipal Education Commission Science and Technology Project KM202010025005;The capital health research and development of special (2022-2-7083); Beijing Municipal Natural Science Foundation7222100.
Ethics Statement
The authors state that this article does not contain any studies with human participants or animals so exempt from institutional review board approval. Informed consent from study participants was not required as this was a retrospective analysis of an existing database.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
