Abstract
Objective:
Tumor-treating fields are currently used to successfully treat various cancers; however, the specific pathways associated with its efficacy remain unknown in the immune responses. Here, we evaluated tumor-treating fields–mediated initiation of the macrophage-specific immune response.
Materials and Methods:
We subjected RAW 264.7 mouse macrophages to clinically relevant levels of tumor-treating fields (0.9 V/cm, 150 kHz) and evaluated alterations in cytokine expression and release, as well as cell viability. Additionally, we investigated the status of immunomodulatory pathways to determine their roles in tumor-treating fields–mediated immune activation.
Results and Discussion:
Our results indicated that tumor-treating fields treatment at 0.9 V/cm decreased cell viability and increased cytokine messenger RNA/protein levels, as well as levels of nitric oxide and reactive oxygen species, relative to controls. The levels of tumor necrosis factor α, interleukin 1β, and interleukin 6 were markedly increased in tumor-treating fields–treated RAW 264.7 cells cocultured with 4T1 murine mammary carcinoma cells compared with those in 4T1 or RAW 264.7 cells with or without tumor-treating fields treatment. Moreover, the viability of 4T1 cells treated with the conditioned medium of tumor-treating fields–stimulated RAW 264.7 cells decreased, indicating that macrophage activation by tumor-treating fields effectively killed the tumor cells. Moreover, tumor-treating fields treatment activated the nuclear factor κB and mitogen-activated protein kinase pathways involved in immunomodulatory signaling.
Conclusion:
These results provide critical insights into the mechanisms through which tumor-treating fields affect macrophage-specific immune responses and the efficacy of this method for cancer treatment.
Introduction
Tumor-treating fields (TTFs) therapy, in which small electric fields are pulsed through the skin to disturb mitosis, represents a noninvasive, regional treatment modality that was recently approved for the therapy of recurrent glioblastoma (GBM) by the US Food and Drug Administration and gained a CE mark in Europe. 1 –3 The clinical concept is that TTFs therapy delivers low-intensity (0.9-3 V/cm), intermediate-frequency (100-300 kHz), and alternating electric fields to the tumor using transducer arrays placed on the skin around the tumor site. 4 The proposed mechanisms of action involve disruption of the metaphase of tumor cells by interfering with mitotic spindle formation and disruption of anaphase by the dielectrophoretic dislocation of intracellular constituents, resulting in an overall effect of apoptosis. 3 The antimitotic effect of TTFs treatment has been showed in various cell lines as well as in GBM by tuning the appropriate frequency to specific cancer cell types. 5 –7 A phase III trial of TTFs combined with temozolomide established that this treatment protocol led to minimal toxicity, increased the overall survival rate, and improved patient quality of life with equivalent therapeutic efficacy to chemotherapy. Moreover, in addition to its combination with chemotherapy, synergistic enhancement of the cellular response was reported when TTFs were administered in combination with radiation therapy. Thus, with these merits, ongoing and future trials will evaluate TTFs to treat various cancer types, including newly diagnosed GBM, solid tumor brain metastases, nonsmall cell lung cancer, ovarian cancer, and pancreatic cancer.
Emerging evidence has demonstrated that the tumor microenvironment may greatly contribute to the final outcome of cancer treatment modalities. Several types of cells including leukocytes, fibroblasts, and vascular endothelial cells form the tumor microenvironment, which has immune cells as its major component. 8 These immune cells interact with tumor cells to influence the initiation, proliferation, elimination, invasion, and metastasis of tumors. 9 In particular, tumor-associated macrophages are immune cells that orchestrate various factors in the tumor microenvironment through the polarization of M1 or M2 macrophages. 8,10,11 Tumor-associated macrophages play a crucial role in the link between inflammation and cancer to produce inflammatory cytokines, such as interleukin (IL) 1β, interferon γ, IL-12, and IL-23, along with anti-inflammatory mediators (IL-4, IL-10, and IL-13). 12 –15
Despite increasing studies of the effect of TTFs on cancers, the influence of TTFs on immune cells has not yet been determined. Therefore, to further understand the mechanism underlying the beneficial effects of TTFs therapy and further improve this treatment modality, we investigated the influence of TTFs exposure on the function of murine macrophage RAW 264.7 cells, a frequently used in vitro model to investigate the immune response and inflammation.
Materials and Methods
Experimental Setup of the Electric Fields
Tumor-treating fields were generated using a pair of insulated wires connected to a functional generator and a high-voltage amplifier, which generated sine-wave signals ranging from 0 to 800 V 12 and resulted in an applied electric field intensity and frequency of 0.9 V/cm and 150 kHz, respectively. We used 0.9 V/cm as the field intensity to match the application in clinical settings. For irradiation treatment, the cells were plated in 100-mm dishes and incubated at 37°C under humidified conditions in a 5% CO2 atmosphere until reaching 70% to 80% confluence.
Cell Culture and Preparation of Conditioned Medium
RAW 264.7 mouse macrophages and 4T1 murine mammary carcinoma cells were purchased from American Type Culture Collection (Manassas, Virginia) and cultured in Dulbecco modified Eagle medium (DMEM; Welgene, Seoul, Korea) supplemented with 10% heat-inactivated fetal bovine serum (FBS) and antibiotics (100 U/mL penicillin and 100 μg/mL streptomycin; Welgene) at 37°C in a humidified 5% CO2 incubator.
For the preparation of conditioned medium (CM), RAW 264.7 cells were treated with 1 ng/mL of lipopolysaccharide (LPS; Sigma, St Louis, Missouri) as an inflammatory stimulator control or TTFs; nontreated cells were used as the control group. After 24 and 48 hours, the supernatants were harvested and filtered through 0.45 μm membrane (Millipore, Massachusetts).
Coculture of RAW 264.7 and 4T1 Cells
4T1 cells (5 × 104 cells/mL) were plated in 12-well plates, and the 24-hour TTFs-treated RAW 264.7 cells were added to the wells for coculture at different densities: 5 × 104 cells/mL, 25 × 104 cells/mL, or 50 × 104 cells/mL. The cells were cocultured in DMEM supplemented with 10% FBS and 1% antibiotics for 48 hours at 37°C with 5% CO2.
Measurement of Cytokines
Cell-free supernatants derived from the coculture of RAW264.7 and 4T1 cells were stored at −70°C prior to measurement of cytokine production. The amounts of IL-6 (Invitrogen, Carlsbad, California), IL-1β (BD Science, San Diego, California), and tumor necrosis factor α (TNF-α; R&D System, Minneapolis, Minnesota) produced in the CM were determined with commercial enzyme-linked immunoassay kits according to the manufacturer’s protocol. The absorbance at 450 nm was obtained using a 96-well microplate reader (Thermo Scientific, Rockford, Illinois).
Cell Viability Assay
Cell viability was determined by a trypan blue exclusion assay and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Amresco Life Science, Philadeplhia, Pennsylvania) assay. An equal volume of trypan blue reagent was added to the cell suspension, and the percentage of viable cells was evaluated by microscopy. 4T1 cells were plated in 24-well plates (1 × 104 cells/well) for various times after treatment of the RAW 264.7 cell CM. Then, MTT (5 mg/mL) reagent was added to each well for 3 hours, and absorbance was measured at 540 nm using a microplate reader (Multiskan EX, Thermo LabSystems, Waltham, Massachusetts). Assays were performed in triplicate.
Detection of Intracellular Reactive Oxygen Species
Reactive oxygen species (ROS) levels in macrophages were monitored using the fluorescent ROS indicator C2′,7′-dichlorodihydrofluorescein diacetate (5 μmol/L; Molecular Probes, Eugene, Oregon). Cell-associated fluorescence was detected by fluorescence-activated cell signaling (FACS) using a FACS-Calibur flow cytometer (BD Biosciences, San Diego, California) with FlowJo software V 7.2.5 (Tree Star Inc, Ashland, Orogen). Images of fluorescent cells were acquired under a 710 confocal microscope (Carl-Zeiss, Germany).
Determination of Nitric Oxide Production
RAW 264.7 cells at a density of 1 × 105 cells/well were treated with TTFs or 1 ng/mL of LPS from Escherichia coli 055:B5 (L2880; Merck KGaA, Darmstadt, Germany) as a positive control for the activation of macrophages. After incubation, the culture supernatants of RAW 264.7 cells were collected, and each culture supernatant was mixed with the same amount of Griess reagent and incubated at room temperature for 10 minutes. The absorbance of the mixture was determined at 540 nm using a microplate reader (Multiskan EX, Thermo Fisher Scientific, Waltham, Massachusetts). All measurements were performed in triplicate. The amounts of nitrites were determined using a standard curve established with NaNO2.
Quantitative Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted from RAW 264.7 cells using TRIzol reagent (Invitrogen) and reverse transcribed using EcoDry Premix (Clontech Laboratories, Mountain View, California) according to the manufacturer’s recommendations. Quantitative reverse transcription polymerase chain reaction experiments were performed using Maxima SYBR Green qPCR Master Mix (Thermo Scientific) in LightCycler 96 Real-time PCR system (Roche Diagnostics, Mannheim, Germany). Using GAPDH as an internal reference, the relative gene expression was calculated based on the ΔΔCt method. The following primer pairs were used: inducible nitric oxide synthase (iNOS), 5′-CGA AAC GCT TCA CTT CCA A-3′ (forward) and 5′-TGA GCC TAT ATT GCT GTG GCT-3′ (reverse); IL-1β, 5′-TGA AGG GCT GCT TCC AAA CCT TTG ACC-3′ (forward) and 5′-TCT CCA TTG AGG TGG AGA GCT TTC AGC-3′ (reverse); TNF-α, 5′-ATG AGC ACA GAA AGC ATG ATC CGC-3′ (forward) and 5′-CCA AAG TAG ACC TGC CCG GAC TC-3′ (reverse); and GAPDH, 5′-ACC ACA GTC CAT GCC ATC AC-3′ (forward) and 5′-TCC ACC ACC CTG TTG CTG TA-3′ (reverse). Experiments were repeated at least 3 times.
Western Blot Analysis
Total proteins from RAW 264.7 cells were extracted in TNN buffer (50 mM Tris–Cl, pH 7.4; 1% NP-40; 150 mM NaCl, and 1 mM EDTA) supplemented with protease inhibitors (1 mmol/L phenylmethylsulfonyl fluoride [PMSF], 1 μg/mL aprotinin, 1 μg/mL leupeptin, and 1 mmol/L Na3VO4) and quantified using the Bradford method. Protein samples (15 μg) were separated by sodium dodecyl sulfate/polyacrylamide gel electrophoresis and transferred to a nitrocellulose membrane. After blocking nonspecific antibody binding sites, the membrane was incubated overnight at 4°C with mouse monoclonal antibodies against iNOS, extracellular signal-regulated kinase (ERK), phosphorylated ERK (pERK), c-Jun N-terminal kinase (JNK), pJNK, p38, p-p38, IκB-α, pIκB-α, and GAPDH. After incubation with peroxidase-conjugated secondary antibodies at 37°C for 1 hour, the protein bands were visualized using enhanced chemiluminescence reagent (GE Healthcare Biosciences, Pittsburgh, Pennsylvania) and detected using the Amersham Imager 680 (GE Healthcare Biosciences). The relative levels of protein expression were calculated with reference to the levels of GAPDH.
Statistical Analysis
All results are expressed as the mean (standard deviation). Significant differences among groups were determined by analysis of variance and Tukey post hoc comparisons using GraphPad software version 5 (GraphPad, La Jolla, California). Statistical significance was defined as P values <.05, and the individual P values in the figures denoted by asterisks (*P < .05; **P < .01; ***P < .001).
Results
Effect of TTFs on RAW 264.7 Cell Viability
To investigate the effect of TTFs on macrophages, RAW 264.7 cells were treated with TTFs (0.9 V/cm) or LPS, which is a representative activator of macrophages. 13 The morphological changes were observed under a phase contrast microscope (Figure 1A). The normal morphology of RAW 264.7 cells was round in the control group. In contrast, LPS-treated cells showed polygonal and dendritic-like morphology, indicating that LPS effectively activated RAW 264.7 cells. 14 In TTFs-treated cells, the dendritic-like shape was observed in a portion of the population, but the number of cells seemed to be decreased at 48 hours compared to that in other groups. Therefore, the cell viability was determined by a trypan blue dye exclusion assay. As shown in Figure 1B, LPS-stimulated and unstimulated control cells continuously proliferated, whereas RAW 264.7 cells treated with TTFs exhibited cell growth inhibition but without a statistically significant influence on cell viability.
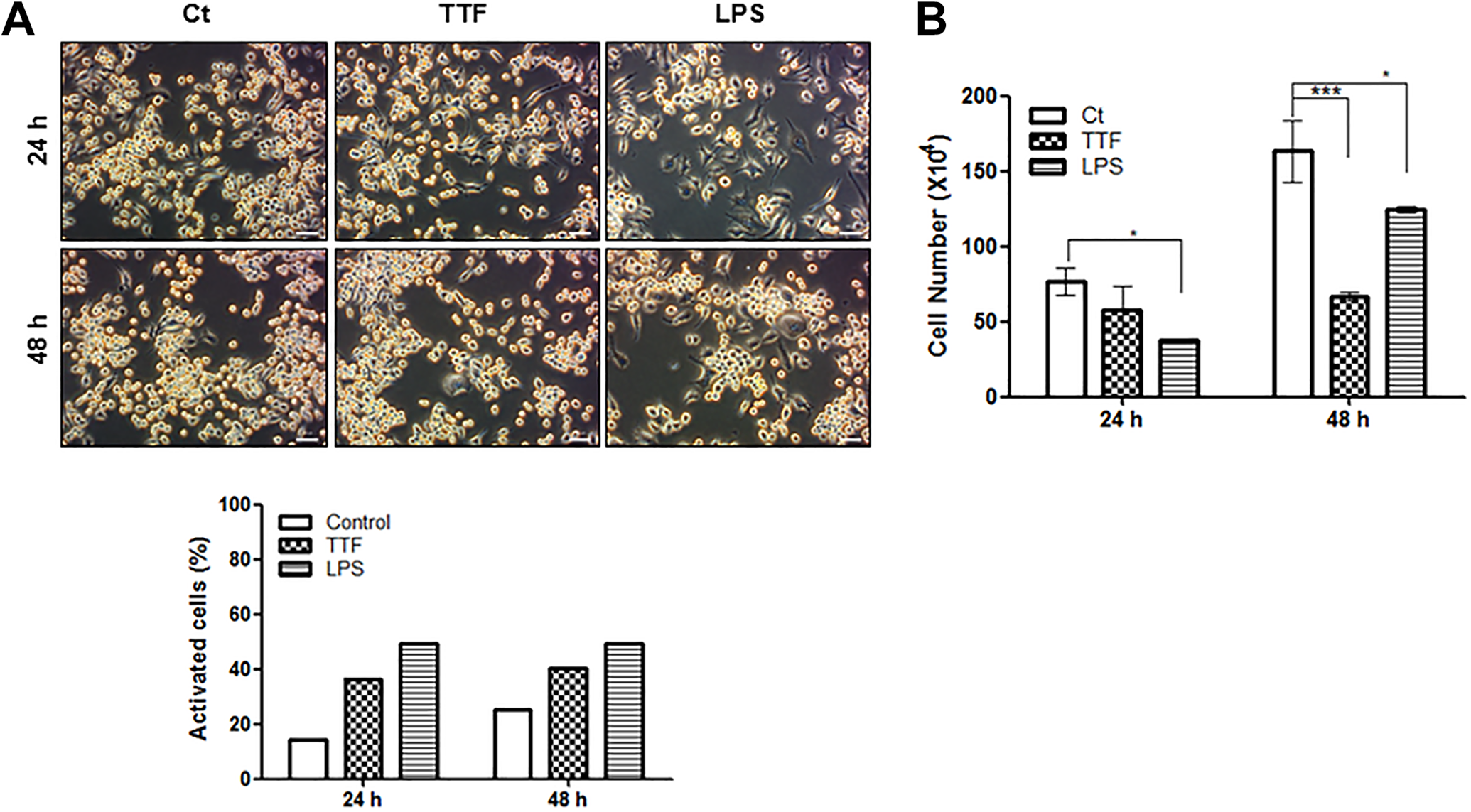
Effect of TTFs on the viability of RAW 264.7 cells. A, RAW 264.7 cells were treated with TTFs (0.9 V/cm) for the indicated times and monitored morphological changes by phase contrast microscopy (magnification ×200, scale bar = 50 μm). The bar graph represents the percentages of activated cells from a total of 500 counted cells. B, The cell viability was determined by a trypan blue exclusion assay. Data represent the mean (standard deviation) of triplicate samples. *P < .05, ***P < .001 compared with the control group. TTFs indicates tumor-treating fields.
Effect of TTFs on the Production of NO and ROS by RAW 264.7 Cells
Nitric oxide (NO) is an important inflammatory mediator that can be produced by activated macrophages to kill tumors and pathogens as well as convey intracellular signals. 15 As shown in Figure 2A, LPS- or TTFs-treated cells increased the messenger RNA (mRNA) and protein expression of iNOS compared with those of the control. The production of NO in TTFs-treated RAW 264.7 cells was slightly increased, whereas LPS stimulation significantly enhanced NO production (Figure 2B). It has been demonstrated that activated macrophages exhibit increased ROS accumulation under inflammatory conditions; therefore, we evaluated whether TTFs affect the generation of ROS. As shown in Figure 2C, ROS levels were increased in RAW 264.7 cells when exposed to TTFs for 24 hours. Concomitantly, LPS-induced ROS generation was also evident. These results indicate that TTFs increased the production of NO and ROS in activated RAW 264.7 macrophages.

Oxidative molecule induction by TTFs treatment in RAW 264.7 cells. A, The mRNA and protein expression levels of iNOS were evaluated by qRT-PCR and Western blotting 24 hours after treatment, respectively. B, The total amount of nitric oxide (NO) was measured using Griess assay at the indicated times. C, ROS generation was determined by DCF-DA fluorescence 24 hours after treatment using a fluorescence microscope and FACS. Data represent the mean (standard deviation) of triplicate samples. *P < .05, ***P < .001 compared with the control group. DCF-DA indicates C2′,7′-dichlorodihydrofluorescein diacetate; FACS, fluorescence-activated cell signaling; iNOS, inducible nitric oxide synthase; mRNA, messenger RNA; qRT-PCR, quantitative reverse transcription polymerase chain reaction; ROS, reactive oxygen species; TTFs, tumor-treating fields.
Effect of TTFs on the Production of Proinflammatory Cytokines by RAW 264.7 Cells
Among the various cytokines, TNF-α can activate macrophages in an autocrine manner to induce the expression of other inflammatory and immunomodulatory mediators. 16 RAW 264.7 cells were treated with TTFs or LPS for 24 hours, and the mRNA expression levels of proinflammatory cytokines such as IL-1β and TNF-α were determined (Figure 3A). The mRNA levels of IL-1β and TNF-α were significantly upregulated by the administration of TTFs, and further increase was observed in LPS-activated RAW 264.7 cells. We further investigated the release of some key proinflammatory cytokines in the coculture medium. Compared with that in other groups, IL-1β, TNF-α, and IL-6 levels in the coculture of TTFs-treated RAW 264.7 cells with 4T1 cells were significantly increased at 48 hours in a manner dependent on the ratio of the 2 cells (Figure 3B). To evaluate the cytotoxic activity of the CM of TTFs-treated RAW 264.7 cells against 4T1 cells, an MTT assay was performed. As shown in Figure 3C, 4T1 cells exposed to the CM of unstimulated RAW 264.7 cells did not show statistically significant decrease in viability, whereas 4T1 cells exposed to CM from TTFs-treated RAW 264.7 cells exhibited decreased viability after 24 or 48 hours contact with the CM. As expected, the CM of LPS-treated RAW 264.7 cells also significantly decreased the viability of 4T1 cells. These results suggested that TTFs activate macrophages and promote the expression of proinflammatory cytokines.

Upregulation of inflammatory cytokines in TTFs-treated RAW 264.7 cells. A, Cells were treated with TTFs (0.9 V/cm) or LPS (1 ng/mL) for 24 hours, and mRNA levels of IL-1β and TNF-α were evaluated by qRT-PCR. B, RAW 264.7 cells were treated with TTFs (0.9 V/cm) for 24 hours, and the cells were cocultured with 4T1 cells at the indicated ratios (4T1:TTFs-RAW 264.7). 4T1 cells, untreated- or TTFs-treated RAW 264.7 cells were designated as 4T1, Ct, and TTF, respectively. Co-cultivations were performed for 48 hours, and the levels of proinflammatory cytokines (IL-1β, TNF-α, and IL-6) were determined by ELISA. **P < .01, ***P < .001 compared with the TTF group. C, RAW 264.7 cells were treated with TTFs (0.9 V/cm) or LPS (1 ng/mL) for 24 hours, and the culture medium (CM) of RAW 264.7 cells was used to treat 4T1 cells for 24 and 48 hours. The untreated RAW 264.7 cells were designated as Ct. The percentage of cell viability was determined by the MTT assay and calculated relative to that of untreated 4T1 cells (none). Data represent the mean (standard deviation) of triplicate samples. **P < .01, ***P < .001 compared with the 24 hours none group. †† P < .01 compared with the 48 hours none group. ELISA indicates enzyme-linked immunoassay; IL, interleukin; LPS, lipopolysaccharide; mRNA, messenger RNA; MTT, 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide; TNF, tumor necrosis factor; TTFs, tumor-treating fields; qRT-PCR, quantitative reverse transcription polymerase chain reaction.
p38 Mitogen-Activated Protein Kinase and Nuclear Factor-κB Signaling Pathways in TTFs-Administered RAW 264.7 Cells
To investigate the signaling pathways involved in the activation of RAW 264.7 cells by TTFs, mitogen-activated protein kinase (MAPK) and nuclear factor-κB (NF-κB) signaling pathways, which are vital targets for regulating inflammatory responses, were examined using Western blot analysis. The expression of 3 MAP kinase family members, namely, ERK, JNK, and p38 MAPK, was determined. The phosphorylation levels of p38 MAPK were significantly increased in TTFs-administered RAW 264.7 cells at 24 hours after treatment. However, activation of MAPK in LPS-treated RAW 264.7 cells was not observed at that time point as LPS treatment activated RAW 264.7 cells more quickly and much more strongly than TTFs treatment. Under normal cell conditions, NF-κB exists as an inactive cytoplasmic complex with its inhibitor IκB-α. 17 Tumor-treating fields–administered RAW 264.7 cells showed increased phosphorylation of IκB-α and p65, indicating that the p65 subunit of NF-κB was released from IκB-α, allowing for its translocation to the nucleus to regulate the transcription of many genes that activate macrophages (Figure 4).

Activation of RAW 264.7 cells via MAPK and NF-κB signaling pathways. A, RAW 264.7 cells were treated with TTFs (0.9 V/cm) or LPS (1 ng/mL) for 24 hours. Levels of the indicated proteins were determined with Western blotting, and GAPDH was used as the internal control. A representative result of 3 independent experiments is shown. B, Band intensities corresponding to the indicated proteins were quantified by densitometry using Image J software, normalized to the GAPDH, and expressed as the fold-change from each control. Data represent the mean (standard deviation) of triplicate samples. *P < .05 compared with the control group. LPS indicates lipopolysaccharide; MAPK, mitogen-activated protein kinase; NF-κB, nuclear factor-κB; TTFs, tumor-treating fields.
Discussion
Tumor-treating fields have recently been reported as a promising and noninvasive therapeutic approach for cancer therapy with good clinical results. The underlying mechanical mechanisms include the disruption of mitosis and selective killing of rapidly proliferating cells by delivering continuous (>18 h/d) low-intensity, intermediate-frequency, alternating electric fields to the tumor site. Tumor-treating fields ultimately lead to caspase-dependent or caspase-independent–induced apoptosis. 18 –20 Tumor-treating fields have been shown to apply its antimitotic effects in preclinical systems of various cancers, including GBM through the similar molecular mechanisms of activity. 5,6,21,22 Furthermore, TTFs have been tried in preclinical systems combined with cytotoxic, chemotherapeutic agents in order to enhance the general antitumor effects. Combined treatment of TTFs with these chemotherapeutic agents (microtubule inhibitors, nucleoside analogues, folate antimetabolites, alkylating agents, and immune checkpoint inhibitors) demonstrated an additive cytotoxic effect. Taxanes and TTFs showed a synergistic effect against cancer cells. 23 In addition, in in vitro preclinical models, TTFs were shown to expose calreticulin to the cell surface, which ultimately significantly decreased the tumor volume in vitro and to considerably decrease the tumor volume in combination with antiprogrammed T-cell death 1 to significantly increase antigen-presenting cell infiltration of the tumor. 24 These events show the potentiation of immunogenic cell death by the treatment of TTFs, 25 which needs further investigation. Although a large number of studies using TTFs as an anticancer therapy are ongoing, the influence of TTFs on normal tissues in the tumor microenvironment remains largely unknown. Thus, in the current study, we demonstrated that TTFs exert potential immunostimulatory activity via regulation of NF-κB/MAPK signaling pathways in RAW 264.7 macrophages.
Macrophages are crucial for host defense against infections and in inflammatory processes through the release of molecules, such as NO, TNF-α, and IL-6. 26 On the basis of our results, the study showed that TTFs increase macrophage activation in RAW 264.7 cells. Furthermore, TTFs significantly enhanced the production of NO in RAW 264.7 cells in vitro. After its release from macrophages, NO acts as an intracellular messenger to mediate the nonspecific immune response 27 and has also been suggested to be a critical factor in the immune response. 28 Nitric oxide is a gaseous, free radical and acts as a signaling molecule in biological systems, recruiting leukocytes to affected tissues. 29,30 Inducible NOS is the inducible enzyme for NO production and is responsible for increased levels of NO. Accumulating evidence indicates that intracellular ROS also serves as a second messenger in inflammatory signal transduction by modulating the release of other inflammatory mediators and stimulating MAPK activity. 31 –33 In our result, we observed that the stimulation of RAW 264.7 cells by TTFs increases iNOS expression as well as the production of NO and ROS, although the increase was much lower than that observed in LPS-treated cells. Therefore, TTFs may have immune mediating/modulating effects, including activation of macrophages and mediating their biological functions such as tumoricidal activity through NO-dependent pathways. 34
Once activated, macrophages release abundant cytokines that act as signals to control homeostasis via regulating cell differentiation, proliferation, apoptosis, and immune responses. 35 Some of these key cytokines that play critical roles in immune and inflammatory processes include TNF-α, which is also produced by T-lymphocytes and natural killer cells, and IL-1β, which facilitates the release of activated macrophages. 35,36 In the present study, TTFs markedly treated RAW 264.7 cells to release TNF-α and IL-1β, indicating that TTFs-stimulated macrophages contribute to provoking an inflammatory response. The levels of TNF-α, IL-1β, and IL-6 were markedly higher in TTFs-treated RAW 264.7 cells cocultured with 4T1 murine mammary carcinoma cells than in 4T1 or RAW 264.7 cells with or without TTFs. Moreover, the viability of 4T1 cells treated with the CM of TTFs-stimulated RAW 264.7 cells decreased, indicating that macrophage activation by TTFs effectively killed the tumor cells.
As essential pathways of inflammation, we therefore evaluated the effect of TTFs on NF-κB and MAPKs activation in RAW 264.7 cells. Nuclear factor-κB is a ubiquitous transcription factor that plays a crucial role in inflammatory processes via the induction of a wide variety of genes, including inflammatory cytokines, mediators, and chemokines. 37 The phosphorylation-induced degradation of IκBs activates and dissociates from NF-κB. As demonstrated in the present result, TTFs significantly increased the phosphorylation of IκB-α in RAW 264.7 cells, thereby inducing nuclear translocation and transcriptional activation of NF-κB in macrophages. Moreover, MAPKs regulate various inflammatory mediators, including TNF-α, IL-1β, IL-2, IL-6, cyclooxygenase 2, and iNOS, 38 –40 suggesting that the inflammatory activity of TTFs is associated with its effect on the MAPK signaling pathway, a key upstream signaling pathway in the regulation of inflammatory mediators. 41 Mitogen-activated protein kinases, including ERK, p38, and JNK, participate in the activation of NF-κB to regulate the gene expression and protein synthesis of inflammatory mediators. 41 Interleukin 1β, a major proinflammatory cytokine, can easily activate MAPK pathways. 42,43 Thus, we investigated whether the inflammatory effects of TTFs involved the MAPK regulation. Indeed, p38 MAPK signaling pathways were obviously activated in TTFs-treated macrophages. Therefore, our results indicate that TTFs may increase inflammatory responses by inducing p38 MAPK pathways in macrophages.
Administration of TTFs did not cause cell death in RAW 264.7 cells; however, cell growth inhibition was clearly observed, suggesting that highly proliferating cancer cells are selectively targeted by TTFs therapy. Lipopolysaccharide-treated cells were morphologically changed from a round-shape to dendritic-like shape, and the number of cells was decreased at the early stages but was subsequently recovered. Therefore, the effect of TTFs on various types of normal cells should be further investigated, and in vivo assays are also needed. Although this study cannot be extrapolated to in vivo conditions, it may provide a theoretical and experimental basis for immune responses specific to TTF treatments in the future.
In conclusion, this is our first result to demonstrate the role of the p38 MAPK/NF-κB pathway in the TTFs-induced inflammatory action in RAW 264.7 cells. A clearer biological understanding of the functional significance of this treatment may constitute a potential new therapeutic option. Thus, these results can provide a preclinical basis for TTFs therapy and expand research geared toward the development of novel anticancer modalities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Korea Institute of Radiological and Medical Sciences (KIRAMS), which was funded by the Ministry of Science, ICT, Republic of Korea (50531-2018).
