Abstract
This study aimed to evaluate the biological effectiveness of cancer therapy with tumor treating fields using a fractionated treatment scheme that was originally designed for radiotherapy. Discontinuous fractional tumor treating fields of an intensity of 0.9 to 1.2 V/cm and a frequency of 150 KHz were applied to U373 cancer cells and IEC6 normal cells for 3 days, with durations of 3, 6, 12, or 24 h/d. As the treatment duration of the tumor treating fields increased from 3 to 24 h/d, the relative tumor cell (U373) number (% of control) reduced in proportion to the treatment duration. Compared to a 25% cell number reduction (75% of control) for the group of 6 h/d treatment at 1.2 V/cm, only 5% (70% of control) and 8% (67% of control) of additional reductions were observed for the group of 12 and 24 h/d treatment, respectively. This experimental result indicates that the dependence on treatment duration in tumor cell inhibition was weakened distinctly at treatment duration over 6 h/d. For normal cells (IEC6), the relative cell number corresponding to the treatment time of the tumor treating fields at 1.2 V/cm of electric field strength was not decreased much for the treatment times of 3, 6, and 12 h/d, revealing 93.3%, 90.0%, and 89.3% relative cell numbers, respectively, but it suddenly decreased to ∼73% for the 24 h/d treatment. Our results showed that the effects of tumor treating fields on tumor cells were higher than on normal cells for treatment duration of 3 to 12 h/d, but the difference became minimal for treatment duration of 24 h/d. The fractionated scheme, using tumor treating fields, reduced the treatment time while maintaining efficacy, suggesting that this method may be clinically applicable for cancer treatment.
Introduction
Some cancers have been shown to be intractable to current treatment options, including glioblastoma multiforme (GBM), an aggressive cancer that accounts for more than 60% of all brain tumors in adults. 1 –3 The 2018 National Comprehensive Cancer Network (NCCN) guidelines recommend postoperative radiotherapy and chemotherapy for patients with GBM. 4 –6 Despite significant therapeutic improvements from the combination of surgery, chemotherapy, and radiotherapy; however, the median overall survival of patients with GBM is less than 1 year, with most patients dying less than 2 years after diagnosis. 7 Therefore, it is essential to develop new types of therapies that can effectively prolong patient survival.
One novel treatment for GBM consists of using alternating electric fields (tumor treating fields [TTFields]), with an intensity of 1 to 3 V/cm and a frequency of 100 to 300 kHz. When cells divide, charged microtubules align to form the spindle apparatus. Previous studies show that TTFields disrupt mitotic spindle assembly, resulting in the inhibition of cancer cell growth. 8,9 With this method, electric fields are administered through electrodes attached to the patient’s body, noninvasively damaging tumors without causing severe adverse effects to normal tissues. 9,10 A recent randomized phase III clinical trial compared a treatment using TTFields plus temozolomide, an oral alkylating agent, with temozolomide alone in patients newly diagnosed with glioblastoma, finding that the median survival for the combined therapy group was 20.5 months, which was 5.1 months longer than in the group treated with temozolomide alone. 3 For patients with recurrent GBM, a randomized phase III trial also showed that median survival was 6.6 versus 6.0 months, progression-free survival rate at 6 months was 21.4% and 15.1%, severe adverse events occurred in 6% and 16%, respectively, in TTFields and chemotherapy patients. 11 Based on clinical evidence, the current NCCN guidelines recommend TTFields for the treatment of GBM. 4 –6 These promising results show that TTFields have the potential to become a new cancer treatment modality.
The clinical studies have indicated that the addition of TTFields to chemotherapy for patients with GBM results in improved survival without a negative influence on health-related quality of life. Only mild to moderate skin toxicity such as itchy skin was observed as a consequence from the electrode arrays. 3,12 Although TTFields therapy has been shown to enhance the efficacy and sensitivity of chemotherapy, 13 it has been recommended that patients undergoing TTFields treatment apply the electrodes to their skin for more than 18 hours per day, except while showering or changing their clothes. 14 This treatment regimen, however, can lead to an uncomfortable feeling in daily life of a patient, and it is also possible to deepen skin toxicity due to prolonged adhesion of the electrode. 12,15 Therefore, if possible, the treatment regime should be optimized to improve the patients’ quality of life. In radiotherapy, the total dose of radiation is divided into several, smaller doses over a period of several days to reduce toxic effects on healthy cells. This fractionation scheme can maximize the effect of radiation on cancer with minimized side effects and a typical fractionation scheme divides the dose into 20 to 30 units delivered every weekday over 4 to 6 weeks. In this study, using the concept of fractionation scheme to optimize the radiotherapy, we evaluated the effectiveness and safety of a fractionated scheme in TTFields treatment.
Methods and Materials
Experimental Setup for the Treatment With TTFields
After amplifying the signals by connecting the function generator (AFG-2112, Good Will Instrument Co, Ltd, Xinbei City, Taiwan) and amplifier (A303, A.A. Lab Systems Ltd, Ramat-Gan, Israel), the TTFields were applied to the cells and the surrounding areas using insulated wires (Seoul Electric Wire Co Ltd, Seoul, Korea; outer diameter, 0.4 mm; polyvinyl chloride insulation thickness, 0.17 mm; dielectric breakdown, 25 kV/mm) attached to the base of the dishes (Figure 1A). We previously described the effect of the TTFields according to the frequency and showed that the most effective treatment for the U373 cancer cells with alternating electric fields was a frequency of 150 kHz. 16 The TTFields, at a frequency of 150 kHz, were applied at intensities of 0.9 V/cm and 1.2 V/cm to the cancer cells and normal cells through the attached wires, with LabVIEW software (National Instruments, Austin, Texas), which was used to control a generator for the fractionation and regulation of the TTFields. The effects of the TTFields were determined following 2 different periods of fractionation, including 20 minutes (scheme 1) and 24 hours (scheme 2), with TTFields applied for 3 days in both schemes (Table 1 and Figure 1B). For example, in scheme 1-(2), TTFields are applied for 2.5 minutes and not for 17.5 minutes; this 20-minute cycle repeats for 3 days. In scheme 2-(2), TTFields are applied for 3 hours and are not applied for 21 hours; this 24-hour cycle repeats for 3 days. The former and latter cycles are 20 minutes and 24 hours, respectively, but the total duration the TTFields are applied for both is 9 hours over the course of 3 days. The “control group” underwent the same treatment for the total experiment duration but without TTFields exposure, and the relative cell numbers were quantified for all cases at the final 72-hour time point. The number of cells remaining after 3 days was defined simply as the “cell number,” with the “relative cell number (% of control)” defined as the ratio of the cell number of the treated group to the control group.

A, A schematic diagram of the experiment. B, Fractionation scheme for the application of the tumor treating fields (TTFields) on the cells. The total treatment time for scheme 1 and scheme 2 is the same, 72 hours.
Fractionation Scheme for the Application of the TTFields on the Cells.a
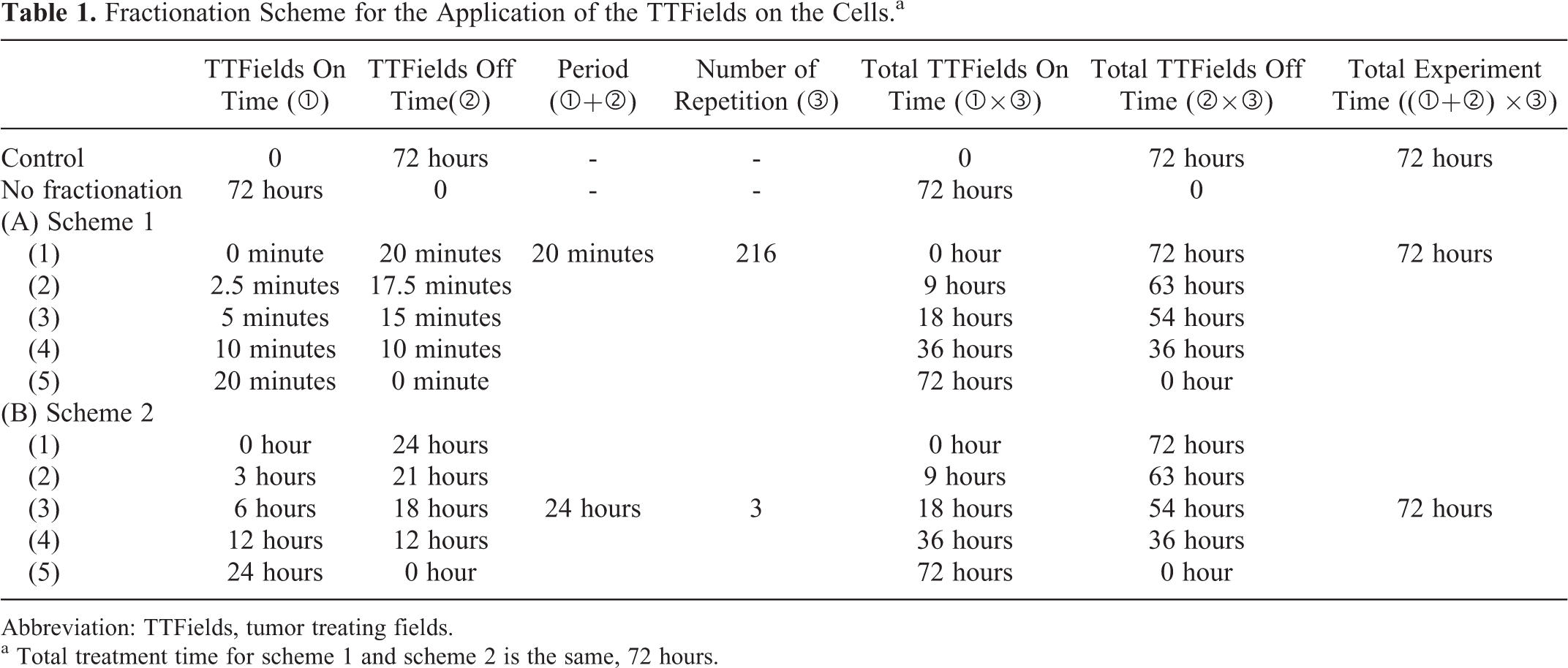
Abbreviation: TTFields, tumor treating fields.
a Total treatment time for scheme 1 and scheme 2 is the same, 72 hours.
Cell Culture
The human glioblastoma (U373) and intestinal epithelial (IEC-6) cell lines were obtained from the Korean Cell Line Bank (Seoul, South Korea). The U373 and IEC-6 cell lines were grown in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS), glutamine, (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid [HEPES]), and antibiotics at 37°C in a 5% CO2 humidified incubator. The doubling time of U373 cells and IEC-6 cells was estimated as 18 and 50 hours, respectively.
Western Blotting
Antibodies to cleaved Poly (ADP-ribose) polymerase (PARP) (9542S), cleaved caspase-3 (9661S), p-cdc2 (9111S), cyclin B (4138S), and cyclin A (4656S) were purchased from Cell Signaling Technology (Danvers, Massachusetts). Anti-phospho-Histone H2AX (05-636) was obtained from Millipore (Billerica, Massachusetts). Anti-β-actin (sc-47778) and secondary antibodies (sc-2031, sc-2004, and sc-2020) were purchased from Santa Cruz Biotechnology (Santa Cruz, California). The protein from the treated cells was extracted with radioimmunoprecipitation assay buffer, separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis, and transferred to nitrocellulose membranes. The membranes were blocked with 1% (vol/vol) nonfat dry milk in Tris-buffered saline containing 0.05% Tween-20 and were incubated overnight at 4°C with the indicated primary antibodies at 1:1000 dilution, followed by washing and an incubation for 1 hour at room temperature with secondary antibodies at 1:5000 dilution. The immunoreactive protein bands were visualized by enhanced chemiluminescence (Amersham Biosciences, Little Chalfont, UK) and were scanned.
Cell Cycle Progression
The cells were seeded in 60 mm dishes at 60% confluence. After the TTFields treatment, the cells were trypsinized, harvested, and fixed in 1 mL of 70% cold ethanol and were incubated overnight at 4°C. The cells were centrifuged at 2000 rpm for 3 minutes, and the cell pellets were resuspended in 500 μL of propidium iodide (10 μg/mL), containing 300 μg/mL of RNase (Sigma, St. Louis, Missouri). The cell cycle distribution was determined by flow cytometry using a FACSCaliber (Becton Dickinson, San Jose, California) and was calculated from 10 000 cells with CellQuest software (Becton Dickinson).
Immunocytochemistry
Immunocytochemistry was performed to determine the nuclear distribution of γH2AX in the individual cells. The cells were grown on chambered slides for 1 day prior to the TTFields treatment. After the TTFields exposure, the cells were fixed only at the end of the 72-hour treatment. All the treatments were performed while the cells remained attached to the slides, followed by fixation with 4% (wt/vol) paraformaldehyde and permeabilization with 0.5% (vol/vol) Triton X-100 in phosphate buffered saline (PBS). Detection was performed after the slides were blocked in 10% (vol/vol) FBS/1% (vol/vol) bovine serum albumin for 1 hour with a 1:1000 dilution of Fluorescein Isothiocyanate (FITC)-labeled mouse monoclonal antibody against γ-H2AX (05-636, Millipore). The images were obtained using a Zeiss LSM-710 confocal laser scanning microscope (Carl Zeiss, Oberkochen, Germany)
Detection of Apoptotic Cells Through Annexin V Staining
After TTFields exposure for 72 hours, the cells were subsequently washed with ice-cold PBS, trypsinized, and resuspended in 1× binding buffer (10 mm HEPES/NaOH [pH 7.4], 140 mm NaCl, and 2.5 mm CaCl2) at 1 × 106 cells/mL. Aliquots (100 μL) of the cell solution were mixed with 5 μL of Annexin V-FITC (PharMingen) and 10 μL of a propidium iodide stock solution (50 μg/mL in PBS) via gentle vortexing, followed by 15 minutes of incubation at room temperature in the dark. Buffer (400 μL, ×1) was added to each sample, which was then analyzed on a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, New Jersey). A minimum of 10 000 cells was counted for each sample, and data analysis was performed using CellQuest software (BD Biosciences, San Diego, California).
Statistical Analysis
Data are presented as mean (SD; n = 3). Statistical significance was determined using a Student t test with SPSS 23.0 software (SPSS, Inc, Chicago, Illinois). Differences were considered significant at P values less than .05 and .01.
Results
Figure 2 shows the percentage of the U373 and IEC6 cells following the TTFields treatment relative to the total number of the control cells under the various experimental conditions, with percentages under 100% indicating that the cell growth was inhibited by the application of the TTFields. Although the relative cell numbers of U373 were reduced in proportion to the treatment times under 6 h/d, this dependence on treatment time was weakened significantly at treatment times over 6 h/d (Figure 2A). For example, the relative cell number of U373 cells treated for 3 h/d differed by ∼10% from that of the cells treated for 6 h/d, whereas the relative cell number of the cells treated for 12 h/d differed by only ∼7% from that of the cells treated for 24 h/d. Moreover, when the total treatment time was kept constant, the relative cell number was independent of the fractionation period. For example, the relative cell numbers of the cells treated for the 20 minutes (scheme 1) and 24 hours (scheme 2 in Table 1) fractionation periods were not significantly different (Figure 2A). The dependence of the relative cell number on the total treatment time, but not on the fractionation period, is more clearly seen in Figure 1B, which shows that at a fixed TTFields treatment time of 12 h/d, the relative cell number of the U373 cells treated for the 20 minutes (0.69 [0.08]), 6 hours (0.71 [0.03]), 12 hours (0.70 [0.02]), and 24 hours (0.71 [0.05]) fractionation periods was similar.

Relative cell number of the tumor and normal cells following the tumor treating fields (TTFields) treatment under various experimental conditions. All experiments were performed for 3 days, and TTFields were turned on/off according to the treatment scheme. The values are presented as the mean (standard deviation, SD; n = 3; *P < .05, **P < .01, relative to control). A, Effects of the TTFields treatment on the U373 cells as a function of the fractionation period. Scheme 1 is for a 20-minute fractionation period, and scheme 2 is for a 24-hour fractionation period. B, The effects of the fractionation periods on the U373 cells under the same conditions (12 h/d for 3 days, total 36 hours). C, The effects of TTFields (scheme 2 in Table 1) at an intensity of 0.9 V/cm on the relative numbers of IEC6 and U373 cells. D, The effects of the TTFields (scheme 2 in Table 1), at an intensity of 1.2 V/cm, on the relative numbers of IEC6 and U373 cells.
Figure 2C shows that the relative cell number of the tumor cells treated with the TTFields at a field intensity of 0.9 V/cm was much lower than that of the normal cells for all the fractionation schemes. These findings suggest that the TTFields treatment can effectively suppress cancer cells without significantly damaging normal cells. Moreover, the differences in the effects of the TTFields on the relative cell number of the cancer and normal cells were more pronounced for the treatment times of 3 to 12 h/d, but became minimal for the treatment times of 24 h/d. For example, the difference in the relative cell number between the cancer cells (U373) and the normal cells (IEC6) treated for 12 h/d was 17.8%, but this difference decreased to 6.2% when the TTFields treatment was applied for 24 h/d (Figure 2C). These findings indicate that to maximize the damage to the tumor cells while minimizing the damage to the normal cells, the application of the TTFields for 24 h/d should be avoided, and a fractionation scheme should be applied. For example, increasing the field intensity from 0.9 to 1.2 V/cm and decreasing the treatment time from 24 to 12 h/d resulted in the same level of inhibition of the relative tumor cell number (∼0.7% difference), while significantly reducing damage to the normal cells (∼13.3% difference; Figure 2C and D).
We also examined whether the TTFields enhanced cytotoxicity, by assessing the activation of the main enzyme involved in cell apoptosis, caspase-3, and by evaluating PARP fragmentation in the normal and cancer cells (Figure 3A). We found that treatment with the fractionated TTFields enhanced of both caspase-3 activation and PARP cleavage in the cancer and normal cells. Both caspase-3 activation and PARP cleavage in the tumor cells increased gradually as the treatment time increased from 0 to 24 hours. In the normal cells, however, caspase-3 activation and PARP cleavage were relatively low at the treatment times of 3 to 12 hours, with abrupt increases at the treatment time of 24 hours. These results were also confirmed by analyzing Annexin V, a protein used for apoptosis cell detection (Figure 3B). Treatment with the fractionated TTFields also delayed the clearance of γH2AX, suggesting that the TTFields maintained DNA damage, thereby increasing cell sensitivity. Similar results were found in the γH2AX focus analysis as well as by Western blotting (Figure 3C and D). In addition, the abnormally accumulated γH2AX foci are also observed in cytoplasm, and this cytosolic accumulation of γH2AX has been reported to be associated with tropomyosin-related kinase A-induced cell death. 17 To investigate the cell cycle phases affecting the TTField-induced cell death following fractionation, we examined the cell cycle profiles and the expression of the cell cycle regulators involved in the G2/M transitions after the fractionated TTFields treatment in normal and cancer cells (Figure 4). Although they are not directly proportional, Western blotting results showed a tendency that the longer duration of TTFields treatment per day results in the more accumulation of p-cdc2 and cyclin B1 (Figure 4A). For example, relative intensities of cyclin B1 in IEC6 (U373) were 1.00 (1.00), 1.05(1.05), 1.05(1.08), 1.27(1.14), 2.21(1.38) for 0, 3, 6, 12, and 24 hours of treatment per day, respectively. This result suggests that the longer duration of TTFields treatment leads to more cells remaining in the G2/M phase. Flow cytometry was consistent with Western blot data in terms of increased number of cells remaining in the G2/M phase as the duration of TTFields treatments increase (Figure 4B). However, there was no direct (or linear) proportionality between Western blot data and flow cytometry for the duration of TTFields treatments. For example, although it is proportional, a large increase in the cyclin B levels at 24 hours in the IEC-6 cells was not accompanied by an increase in the fraction of cells in G2/M (Figure 4). Western blot analysis shows that the cyclin A band was not significantly altered by fractionated TTFields treatment (Figure 4A).

Effects of the fractionated tumor treating fields (TTFields) treatment on apoptosis and DNA damage in normal and cancer cells. The TTFields, at a field intensity of 1.2 V/cm and a frequency of 150 kHz, using options (1) to (5) in scheme 2 of Table 1, were applied to the normal and cancer cells. The values are presented as the mean (standard deviation, SD; n = 3; *P < .05, **P < .01, relative to control). A, Equal amounts of cell lysates (30 µg) were separated by electrophoresis and were analyzed by Western blotting for the indicated antibodies. The band intensities of the target proteins were normalized to that for β-actin. B, Results of Annexin V and Propidium Iodide (PI) staining after U373 and IEC6 cells were exposed to 72 hours of TTFields, using options (1) to (5) in scheme 2 of Table 1. C, Equal amounts of cell lysates (30 µg) were separated by electrophoresis and were analyzed by Western blotting for the indicated antibodies. The band intensities of the target proteins were normalized to that for β-actin. D, Immunocytochemistry for phosphorylated H2AX, a marker of the DNA damage response, in the IEC6 and U373 cells exposed to TTFields at the indicated time points.

Effects of the fractionated tumor treating fields (TTFields) on the cell cycle for normal and cancer cells. The TTFields, at a field intensity of 1.2 V/cm and a frequency of 150 kHz, using options (1) to (5) in scheme 2 of Table 1, were applied to the normal and cancer cells. The values are presented as the mean (standard deviation, SD; n = 3; *P < .05, **P < .01, relative to control). A, Cyclin expression was analyzed by Western blotting. The total time of the TTFields treatment was 72 hours. Equal amounts of cell lysates (30 µg) were separated by electrophoresis and were analyzed by Western blotting for the indicated antibodies. The band intensities for the target proteins were normalized to that for β-actin. B, An analysis of the cell cycle by flow cytometry.
Discussion
Our results showed that as the duration of treatment increases, the antimitotic effects of TTFields on cells increases. Therefore, to optimize the treatment results, TTFields on time per day should be applied as long as possible if the strength of electric field is fixed. Current treatment scheme (TTFields on time for 18 h/d, ie, all day long) follows this method since the output voltage (or electric field applied to patient skin) coming from device is fixed. But, as seen in Figure 2, the antimitotic effects of TTFields on tumor cells increase rapidly as TTFields on time increases from 0 to 6 h/d, but the slope (or the proportionality) decreases as TTFields on time increases from 6 to 24 h/d. This result suggests that the effects of TTFields are not linearly (or directly) proportional to the duration of treatment per day. Therefore, to optimize the efficacy of TTFields based on our results, one might decrease TTFields on time per day with an increased strength of electric field. Although the reduced duration of TTFields with an increased treatment intensity is a possible way of optimization for TTFields treatment, there is a limitation of this optimization since the increased intensity is associated with the heating problem. In general, the heat generation is dependent on the intensity of treatment field as well as the duration of TTFields on time. Although an increase in intensity will increase the heat under skin, the decrease in duration of TTFields treatment will decrease the heat under skin. Therefore, the optimal treatment scheme with maximum possible intensity of TTFields with reduced TTFields on time per day should be chosen on the condition that this treatment scheme can provide the same biological efficacy of original treatment without heat side effect.
The experimental results suggest that the effects of TTFields might persist not only during the time of the treatment but also during the intervals between the treatments. In general, the effects of radiation appear several hours (as DNA damage), several days (as DNA mutation and cell death), and even several years (as cancer) after the cessation of the treatment. Our findings suggest that, like conventional radiation, TTFields affect the cells after the cessation of the treatment. Moreover, the TTFields had greater effects on the cancer cells than on the normal cells, suggesting that the TTFields can selectively damage the cancer cells. Although this study tested fractionated therapy in GBM cells only, this investigation should be extended by conducting experiments involving reproductive cell death, and an in vivo study using tissue from several cancer types can be targeted to provide a scientific rationale to treat TTFields.
Our data provide a molecular biological basis by which a fractionated TTFields treatment has therapeutic efficacy against cancer cells. The biomarkers associated with apoptosis, such as cleaved PARP, caspase-3, and Annexin V, were observed in the normal cells that received continuous treatment but were not observed in the fractionated TTFields treatment group. In contrast, the effects of the TTFields on cancer cells were proportional to the treatment time. Thus, the application of a fractionation scheme resulted in a greater degree of apoptosis in the cancer cells than in normal cells. The TTFields therapy seems to induce apoptosis in cancer cells as well as slows cell cycle progression. However, since the effect on cell cycle progression is not dramatic, the mechanism by which TTFields therapy has a relatively selective effect on cancer cells is presumed to be by DNA damage-induced cell death. 18 A relatively selective effect on cancer cells seems to come from the shorter doubling time of U373 cells (18 hours) compared to that of IEC-6 cells (50 hours) and it is well matched with previous study, 8 which showed that treatment with TTFields have a higher therapeutic effect with shorter cell doubling times. Because this study only assessed one normal and one cancer cell line, there is a need to assess the effects of TTFields in other cell lines, as well as to evaluate the long-term outcomes in cells treated with the TTFields. Moreover, this study was performed in an in vitro system, so in vivo research using animal models should be performed to investigate the effects of fractionated TTFields prior to clinical trials and to minimize possible complications in clinical applications. Further studies are needed to optimize the field intensity, treatment duration, fractionation period, and frequency for tailored therapy.
In summary, our results demonstrate, for the first time, that fractionated TTFields treatment is associated with the inhibition of tumor cell survival, DNA double-strand breaks, and the cell cycle in cancer cells, suggesting that fractionated TTFields could be exploited as a novel strategy to treat cancer cells using alternating electric fields. Treatment efficacy may be optimized by shorter treatment times at a higher intensity on the condition that the optimized treatment scheme can provide the same biological efficacy of original (continuous) treatment without heat side effect.
Footnotes
Authors’ Note
Our study did not require an ethical board approval because it did not contain human or animal trials
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Nuclear R&D Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science and Technology (NRF-2013M2A2A7067089, NRF-2017R1D1A1B03028923, NRF-2018R1D1A1B07047770) and the Korea Institute of Radiological and Medical Sciences (KIRAMS), which was funded by the Ministry of Science, ICT, and Future Planning, Republic of Korea (50531-2018).
