Abstract
Aim:
Current diagnostics of bone metastatic disease is not satisfactory for early detection or regular process monitoring. The combination of biomarkers and the multiparametric approach was described as effective in other oncology diagnoses. The aim of the study was to improve the difference diagnostics between bone-metastatic disease and solid tumors using mutivariate logistic regression model.
Methods:
We assessed the group of 131 patients with the following diagnoses: prostate cancer, breast cancer, lung cancer, and colorectal cancer. According to the results of scintigraphy, the cohort was divided into 2 groups based on the occurrence of bone metastases. Group 0 was a control group of 75 patients with no signs of bone metastases and group 1 included 56 patients with bone metastases.
Results:
We used stepwise selection multivariate logistic regression for choosing the multimarker formula for calculation of risk score for bone metastases diagnostics. For detection of bone metastasis, it was shown to be most effective measurement of 3 biomarkers: procollagen type 1 N-terminal propeptide, growth differentiation factor-15, and osteonectin and combining with calculation of risk score by designating measured concentrations in mathematical formula: bone risk score = procollagen type 1 N-terminal propeptide × 0.0500 + growth differentiation factor-15 × 1.4179 + osteonectin × 0.00555.
Conclusion:
We identified growth differentiation factor-15 as the best individual marker for bone metastasis diagnostics. The best formula for risk score includes levels of 3 biomarkers—procollagen type 1 N-terminal propeptide, growth differentiation factor-15, and osteonectin. The new score has better performance described by higher area under the curve than individual biomarkers. A further study is necessary to confirm these findings incorporating a larger number of patients.
Introduction
Current diagnostics of bone metastatic disease are not sufficient to facilitate early detection or regular process monitoring major milestones for the improvement of treatment management. Early detection of bone metastatic disease is required to prevent severe complications such as pathological fractures and hypercalcemia. Novel diagnostic methods are needed because current diagnostics, based on imaging methods, cause patients harm as a result of doses of radiation, not to mention the high costs involved in their use. New serum biomarkers should provide additional value, and that independently of bone imaging diagnostics, while helping to improve strategies for managing bone-metastatic disease in the future.
Improvements in biotechnology along with a better comprehension of the bone metastatic microenvironment are expected to improve the reliability and predictive capacity of bone marker assessment. 1 Bone metastatic disease is a complex process incorporating both genes that are only overexpressed if the cancer has a predilection to spread into bone and genes common to metastases at all locations. The vicious circle causing bone metastatic spread arises when substances secreted by or expressed on cancer cells (eg, parathyroid hormone-related peptide) change the bone microenvironment and so activate osteoblasts and osteoclasts to produce cytokines, bone remodeling and osteolysis, hence the activation of growth promoters which increase tumor cell motility and growth with yet further release of growth factors. 2,3 The new generation of biomarkers is mostly under development using proteomic approaches to primary research of cancer pathophysiology and so could bring biomarkers with better sensitivity and specificity into clinical practice. Unfortunately, there is a lack of direct comparisons of biomarkers. Published studies of multimarker comparison are mostly “out of date,” in that they do not incorporate the novel candidate biomarkers of tumor biological activity. Over recent decades large scale studies of biomarkers have been enabled by modern analytical technologies such as the multiplex immunoanalysis used in our presented study and should improve the transition of new biomarker approaches from basic research into clinical application. Furthermore, the combination of biomarkers and a multiparametric approach has been described as being effective in other oncological procedures. 4 –7
The aim of the study was to compare the performance of a panel of 6 routine biomarkers of bone-turnover and 10 novel biomarkers in distinguishing bone-metastatic disease occurrence and the absence of bone metastasis. As a further step, we have suggested a novel mathematical model for the assessment of the risk of bone metastatic disease occurrence—the Bone risk score (BRS).
Materials and Methods
Study Design
In our study, 16 circulating biomarkers were measured and their performance compared in the distinguishing of bone-metastatic disease occurrence in a cohort of 131 oncological patients with solid tumors who underwent whole-body skeletal scintigraphy using technetium. We have compared 6 routine markers of bone-turnover and metabolism: osteocalcin (OSTEO), carboxy-terminal telopeptide (CTx), procollagen type 1 N-terminal propeptide (PINP), N-terminal propeptide of procolagen type 3 (P3NP), and bone isoenzyme of alkaline phosphatase (known as ostease - OSTA) in sera as well as parathyroid hormone (PTH) in EDTA plasma samples and the following 10 novel serum biomarkers: receptor activator of nuclear factor kappa-β ligand (RANKL), leptin, osteoprotegerin (OPG), osteopontin (OPN), sclerostin (SOST), growth differentiation factor-15 (GDF15), periostin, tartrate-resistant acid phosphatase (TRAP5), chitinase-3-like protein 1 (YKL40), and osteonectin measured using multiplex immunoanalytic methods.
The study was conducted during years: 2012 to 2015 in The University Hospital in Pilsen, Czech Republic and the data analysis was performed during years 2016 to 2017.
Patients
The assessed group included patients with the following diagnoses: prostate cancer (12.21%), breast cancer (59.54%), lung cancer (9.92%), colorectal cancer (6.87%), and other solid tumors (11.45%). According to the results of scintigraphy, the cohort was divided into 2 groups based on the occurrence of bone metastases. In our study, group 0 was a control group of 75 patients with no signs of bone metastases (age: median = 65 years, minimum = 39 years, maximum = 85 years), and a further group (group 1) included patients with bone metastases represented by 56 patients with no previous treatment with bone modifying agents (age: median = 67.5 years, minimum = 35 years, maximum = 87 years). The Wilcoxon test showed the groups of patients did not differ significantly in age. There were 99 women and 32 men enrolled into the study, all individuals were Caucasians.
Plasma Samples
Peripheral blood was drawn before bone scintigraphy using VACUETTE Z Serum Sep tubes and VACUETTE EDTA tubes (Greiner Bio-One, Kremsmünster, Austria) and sera were allowed to clot. Sera and plasma were separated by centrifugation at 1700×
Analytical Methods
Serum levels of multiplex biomarkers were analyzed using a MagPix instrument (Luminex corp., Austin, Texas). Growth differentiation factor-15, osteonectin, periostin, TRAP5, and YKL40 were assayed using a Human Cancer Metastasis Biomarker Magnetic Bead Panel (Merck Millipore, Darmstadt, Germany), RANKL using MILLIPLEX MAP Human RANKL Magnetic Bead and Leptin, OPG, OPN, SOST using MILLIPLEX MAP Human Bone Bead Panel. Multiplex measurements were assayed in duplicates using 2 levels of control samples for internal quality assessment.
Routine markers of bone-turnover and metabolism were assayed in duplicates using routinely implemented well-established immunochemistry methods, controlled by internal and external quality program. Carboxy-terminal telopeptide levels were measured using the electrochemiluminiscent immunoassay ß-CrossLaps Roche (F. Hoffmann–La Roche, Basel, Switzerland), Cobas e411 instrument. Osteocalcin and osteasa by chemiluminiscent assays LIAISON Osteokalcin a LIAISON BAP Ostase (DiaSorin S.p.A., Saluggia, Italy), LiaisonXL instrument. Parathyroid hormone plasma level was measured using Intact PTH kit (Beckman-Coulter Inc., Brea, California) and UniCel DxI 800 instrument. Procollagen type 1 N-terminal propeptide and P3NP levels were measured using the radioimmunoassays: UniQ PINP RIA and P3NP RIA kits (both Orion Diagnostica, Espoo, Finland) and Stratec SR300 instrument.
Statistics
Descriptive statistics, median, 25th and 75th percentiles, were calculated for all biomarkers. The Wilcoxon test (Mann-Whitney test for independent samples) was used to compare biomarker levels between groups. Significance was set up for
Results
The results of comparisons made between group 1 (bone metastasis) and the control group 0 are summarized in Table 1. Eleven of the followed biomarkers showed significantly higher levels in group 1 compared to the control group 0 (
Results of Comparison Between Group 1 (Bone Metastasis) and Control Group 0.a

Abbreviations: CTx, carboxy-terminal telopeptide; GDF15, growth differentiation factor-15; NS, nonsignificant; OPG, osteoprotegerin; OPN, osteopontin; OSTA, bone isoenzyme of alkaline phosphatase; OSTEO, osteocalcin; PINP, procollagen type 1 N-terminal propeptide; P3NP, N-terminal propeptide of procolagen type 3; PTH, parathyroid hormone; RANKL, receptor activator of nuclear factor kappa-β ligand; SOST, sklerostin; TRAP5, tartrate-resistant acid phosphatase; YKL40, chitinase-3-like protein 1.
a Descriptive statistics and results of comparison between group 1 (bone metastasis, 56 patients) and control group 0 (no signs of bone metastasis, 75 patients) using the Wilcoxon test, biomarkers with significant differences between groups are underlined.
For showing clinical applicability, all studied biomarkers can be ordered according to their decreasing ability to distinguish the occurrence of bone metastasis based upon decreasing AUC as follows: GDF15 (0.87), OPN (0.86), PINP (0.80), YKL40 (0.76), OPG (0.76), P3NP (0.73), osteonectin (0.72), periostin (0.72), CTx (0.72), TRAP5 (0.72), OSTA (0.68), leptin (0.67), SOST (0.60), OSTEO (0.58), RANKL (0.56), and PTH (0.5). Area under the curve values and 95% confidence interval values, proposed cut-off and related specificity, sensitivity, positive and negative PV, and RR are listed in Table 2 for all biomarkers.
Receiver Operating Characteristic Analysis of the Biomarker Calculated Bone Risk Score.a

Abbreviations: CTx, carboxy-terminal telopeptide; GDF15, growth differentiation factor-15; OPG, osteoprotegerin; OPN, osteopontin; OSTA, bone isoenzyme of alkaline phosphatase; OSTEO, osteocalcin; PINP, procollagen type 1 N-terminal propeptide; P3NP, N-terminal propeptide of procolagen type 3; PTH, parathyroid hormone; RANKL, receptor activator of nuclear factor kappa-β ligand; SOST, sklerostin; TRAP5, tartrate-resistant acid phosphatase; YKL40, chitinase-3-like protein 1.
a Receiver operating characteristic (ROC) analysis of the biomarkers and calculated novel Bone Risk Score. The area under the ROC curve (AUC) was calculated. All biomarkers and Bone Risk Score (BRS) are ranked according decreasing AUC. Two cut-off values were proposed for each biomarker and BRS corresponding to approximately 90% and 95% specificity. Calculations were made for each cut-off to ascertain sensitivity, positive predictive value (PV+), negative predictive value (PV−) and risk ratio (RR).
Using stepwise selection mutivariate logistic regression analysis the optimal formula for BRS calculation was defined as follows:
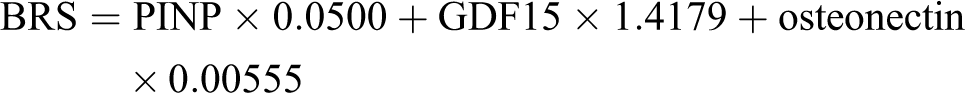
The best formula for risk score includes the levels of 3 biomarkers—PINP, GDF15, and osteonectin. When we compared ROC analyses of the best single biomarker GDF15 and calculated the new risk score BRS, the new score has a significantly better performance (
Receiver operating characteristic curves of BRS, PINP, GDF15, and osteonectin, that is, ROC of calculated BRS and best performing single markers for comparison are shown in Figure 1, which highlights the best performance of calculated BRS for bone metastatic group discrimination.

Receiver operating characteristic curves of bone risk score, GDF15, osteonectin, and PINP. GDF15 indicates growth differentiation factor-15; PINP, procollagen type 1 N-terminal propeptide.
Discussion
Bone metastatic disease causes severe complications such as pathological fractures and hypercalcemia, and lowers the quality of life of oncologic patients mainly through severe pain and immobility. Current diagnostics of bone metastatic disease, based on imaging methods, are not satisfactory. The ability of traditional bone biomarkers to contribute to the diagnostics of bone metastases is limited. Novel biomarkers which were recently introduced also failed to meet the criteria for an “ideal” individual marker. In this situation, we can use statistical methods to increase the efficiency of the diagnostic value. One of the best examples is the prostate health index which significantly improved the process of prostate cancer diagnostics. 8
In our study, we have suggested a novel mathematical model for the assessment of the risk of bone metastatic disease occurrence incorporating results of 3 circulating biomarkers selected on the basis of direct comparisons made of 16 biomarkers. The performance of individual markers and the novel risk score were compared using ROC analysis for the discrimination of bone metastatic disease. The best performance (AUC 0.94) was shown by the novel risk score BRS, calculated from circulating levels of PINP, GDF15, and osteonectin. The BRS has a superior performance to the best single biomarker in our study—GDF15. In ROC analysis, GDF15 showed a promising AUC of 0.87. The GDF15 molecule was introduced as a novel tumor biomarker in connection with various types of tumors. Increased GDF15 levels were observed in melanomas, oral squamous cell carcinomas, colorectal, pancreatic, prostate, breast, and cervical epithelial cancers. 9 Association between increased GDF15 levels and the metastatic process has been proved in several studies. 10,11 Growth differentiation factor-15 was also confirmed as a possible indicator of bone metastasis in the case of prostate cancer bone metastasis 12 and was identified as one of the proteins produced by bone metastases in patients suffering from prostate cancer. 13 In our previously published research, GDF15 was considered as a potential biomarker for bone metastatic disease and its combination with other biomarkers was recommended to increase sensitivity and diagnostic efficacy. 14
Further promising biomarkers emerging out of the ROC analysis in our study, with an AUC above 0.8, were OPN and PINP. Chitinase-3-like protein 1, OPG, P3NP, osteonectin, periostin, CTx, and TRAP5 were found to be what we might call second-line biomarkers which, according to our results, showed an AUC between 0.7 and 0.8. As routine markers of the turnover of collagen I in bone remodelation PINP and CTX are widely discussed as markers of bone metastasis. One study, performed by Lumachi
Nowadays, the role of biomarkers is discussed for proper treatment tailoring in oncology. Brown J.E.
New trends in clinical biomarker research, including for bone metastatic disease, are related to the expression of microRNA. Ell
Johansen
Several studies support the importance and efficacy of biomarkers in patients with bone metastases. The role of biomarkers in the management of bone-homing malignancies was recently precisely reviewed by D’Oronzo
Conclusions
We identified GDF15 as the best individual marker for bone metastasis diagnostics. The best formula for risk score includes the levels of three biomarkers—PINP, GDF15, and osteonectin. The new score has a superior performance described by a higher AUC than individual biomarkers. A further study is required to confirm these findings which would incorporate a larger number of patients.
Footnotes
Authors’ Note
The study was approved by the local Ethical Committee on July 13, 2011. Patients signed an informed consent before enrollment into the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the research project of Ministry of Health of The Czech Republic IGA NT 13655-4.
