Abstract
In this work, polyamide 6 (PA6) nanofiber-supported thin-film composite membranes have been studied for saline water remediation. Nanofiber-based support layers are critical in enhancing the membrane surface area and facilitating the formation of high-performance polyamide (PA) thin films. Herein, microporous nanofiber support membranes were fabricated using a lamination technique at varying temperatures, and the effects of lamination temperature on fiber diameter, pore size, and air permeability were investigated to optimize the support layer for PA thin-film membrane fabrication. Results indicate that increasing the lamination temperature from 110°C to 135°C decreases air permeability and pore size while increasing fiber diameter. Using the optimized support laminated at 110°C, ultrafiltration PA thin-film membranes were developed, and the effects of monomer type and feed pH on membrane performance were evaluated. M-phenylenediamine (MPD)-based membranes achieved a maximum salt rejection of 74% but exhibited a relatively low water flux of 0.42 L·m−2·h−1 at 400 kPa. In contrast, piperazine (PIP)-based membranes demonstrated a low sodium chloride (NaCl) rejection of 25% but a high magnesium sulfate (MgSO4) rejection of 97% at the same benzenetricarbonyl trichloride (TMC) concentration, with a significantly higher flux of 8.6 L·m−2·h−1. Acidic conditions improved the flux of MPD-based membranes up to 5-fold but significantly reduced salt rejection, whereas alkaline conditions enhanced salt rejection for PIP-based membranes while decreasing flux. This work is the first attempt in the literature to systematically investigate the influence of lamination temperature on nanofiber support for further PA thin-film membrane formation, providing new insights into optimizing support layer properties for desalination.
Introduction
Water is vital for all life on Earth, acting as a medium for nearly every chemical reaction in our bodies. It functions as a solvent, transporting nutrients, hormones, enzymes, and gases throughout our system. In green plants and algae, water is necessary for photosynthesis, which produces the oxygen we breathe. Despite two-thirds of the Earth’s surface being covered by water, only a tiny portion is potable. The limited availability of drinkable water significantly impacts economics, geopolitics, and migration. The water cycle operates relatively predictably within the hydrosphere, but climate changes—whether due to human activity or natural causes—make these predictions less reliable. Some regions experience droughts, forest fires, dust storms, and desertification, while others face excessive rainfall, flooding, and soil degradation. Combined with pollution from human activities, these challenges underscore the need for sustainable and effective methods of purifying drinking and utility water.
One of the most significant water sources is oceans and sea, which consists of 97% saline water. 1 The second source is brackish water, less salty than seawater but still has a considerable amount of dissolved solids. Its salt content ranges from about 0.05% to 3% (500–30,000 ppm), varying by location and conditions. 2 The discharge of saline wastewater causes a serious threat to the environment. Saline wastewater can be treated to meet discharge standards and provide opportunities for water reuse and nutrient recovery. Salts in wastewater can harm aquatic species, destroy ecosystems, suppress plant germination, and hinder seed growth, reducing species diversity and ecosystem disruption.3,4 Moreover, the saline water in the land will leave salt residue in soils after evaporation, cause damage to microorganisms, change the mineral balance, and damage agricultural areas. 5
The main challenges of recovery of saline water are high energy demand and low efficiency. Several techniques, such as biological, chemical, and physical, have been used to treat saline water, including their hybrid systems. The biological techniques include membrane bioreactor (MBR), wetland, bio-filter, biofilm reactor, sequencing batch reactor (SBR), anaerobic baffled membrane bioreactor, algal-bacterial granular sludge (ABGS-CFR), etc.6–8 Besides the cost-effectiveness and sustainability, the biological treatment suffers from the high salt concentration, which poses a significant problem by interfering with microbial metabolic processes. A hybrid system is needed for the reduction of salt concentration. 9 The chemical remediation of the saline water includes advanced oxidation and electrochemical techniques.10–12 However, this method is expensive, energy-intensive, inefficient at high salt concentrations, and may lead to secondary pollution. The physical methods include absorbents or membrane technology (physicochemical technology). 4 Among all technologies, membrane technology offers promising solutions for effective saline wastewater treatment and resource recovery with more cost-effective and environmentally friendly technology.13,14 Despite membrane-based desalination achieving excellent results so far, research in membrane technology continues to grow due to the increasing global population and the demand for more freshwater through cost-effective methods. Optimizing membranes for high flux and selectivity is crucial. Thin-film composite (TFC) membranes are gaining prominence for their selectivity and permeability. Incorporating a nanofibrous layer as a substrate, backed by nonwoven material, to create thin-film nanofibrous composite (TFNC) membranes shows promising potential for future desalination needs.
Recent advancements have been achieved in creating nanofiber-supported thin-film composite membranes for desalination and water treatment.15–18 However, attaining an ideal equilibrium between improved water permeability and effective salt rejection continues to pose a substantial problem. The complex relationship among membrane selectivity, structural integrity, and permeability is further complicated by factors including the morphology of the nanofiber support layer, the chemical composition of the thin-film polyamide layer, and the environmental conditions in which the membranes function. The use of a nanofiber web as a support layer for thin-film composite membranes presents numerous benefits. The high surface area and interlinked porous structure of nanofibers improve water permeability while preserving mechanical strength and flexibility. The nanoscale pores serve as an ideal substrate for depositing a thin selective polyamide layer, enhancing overall membrane performance. The customizable shape of nanofiber webs, including variable fiber diameter and pore size, enhances control over membrane characteristics, resulting in increased flow and effective salt rejection.
This paper focuses on the parametric study of the prepared polyamide 6 (PA6) nanofiber membranes, optimized by controlled lamination temperatures and various monomer combinations, and evaluates their characteristics as a support for the thin film-membrane preparation. Previous studies have established that the membrane lamination process significantly impacts the tensile properties, pore structure, and overall performance of the membrane, thereby influencing its permeability and selectivity.19–21 A low lamination temperature results in easily delaminated layers, whereas a high lamination temperature melts the adhesive webs, filling the porous structure and reducing membrane porosity. The membrane’s low performance is the biggest challenge in membrane desalination, which is associated with low flux and rejection of salt ions. Here, we tackled these challenges by optimizing the synthesis of polyamide (PA) thin films through the interfacial polymerization method, utilizing a variety of monomers. The first step includes membrane material optimization, and the second stage is to improve membrane permeability and rejection by changing the pH of the feed. Previous reports have documented such attempts, yet further improvement remains necessary.17,22,23 The other novelty of this work lies in employing a nanofiber web as the support layer for the thin-film membrane. The use of the nanofiber layer enhances both the surface area and porous structure of the composite membrane, thereby improving membrane performance and structural stability. Herein, the preparation of microporous nanofiber membrane support has been investigated.
Our study contributes to the literature by offering a novel approach to PA6 nanofiber support membrane preparation for thin-film composite membrane and PA thin-film membrane optimization through the use of diverse monomers, potentially enhancing both flux and salt rejection. Moreover, our investigation into the impact of feed pH on membrane performance provides new insights that could inform future design strategies for more efficient desalination processes.
Materials and methods
Preparation and characterization of nanofiber web and lamination process
PA6 nanofibers were used as the support layer for the PA thin-film membrane. The PA6 nanofibers were produced using a Nanospider (NS 1WS500U, Elmarco s.r.o, Czech Republic) under the following conditions: a 12% (w/v) PA6 solution dissolved in a mixture of acetic acid and formic acid (2:1) was fed into the electrospinning device. The humidity and temperature were maintained at 35% RH and 23°C, respectively. The electrode distance and applied total voltage were set to 180 mm and 65 kV, respectively. The density of the nanofiber layer is set as 1.5 g/m2 with approx. 20 µm thickness. The final morphology of the nanofibers was analyzed using scanning electron microscopy (SEM).
The mechanical properties of PA6 nanofibers are not inherently suitable for membrane technology, so a nanofiber layer was laminated onto a nonwoven support to enhance the structural stability of the composite membrane. An adhesive web was used to bond the nanofibers onto a spunbond nonwoven surface (120 g/m2) during the lamination process. A co-polyester adhesive web with a melting range of 90°C to 120°C facilitated the bonding, achieved by applying heat and pressure to form a layered, sandwich-like structure, as shown in Figure 1. Three specific temperatures (110°C, 125°C, and 135°C) were applied to investigate how lamination conditions influence the support layer properties. The minimum temperature was 110°C based on the adhesive’s melting point range. Overheating can damage the material, while insufficient heat may lead to layer separation. Moreover, excessive adhesive melting could reduce pore size by filling pores with molten adhesive. Proper lamination is essential to reinforce the nanofiber layer’s mechanical strength without compromising its permeability. Lamination process of nanofiber layer on a nonwoven substrate.
Air permeability testing (ATLAS128 Air Permeability Tester, SDL ATLAS, Rock Hill, SC, USA) was conducted according to ASTM D737 to ensure the adhesive web did not completely block the membrane pores. Pore size was measured using a pore size analyzer (Porometer 3G, Anton Paar GmbH) following ASTM F316-03, the standard for assessing pore size characteristics in membrane filters using bubble point and mean flow pore size. This analyzer can measure microporous structures but is unsuitable for ultra- and nano-porous structures.
Thin-film membrane preparation
Solution A: Piperazine (PIP) and m-phenylenediamine (MPD) monomers were dissolved in distilled water for the aqueous solution. The PIP solution was prepared by mixing 2% (w/v) PIP, 4% (w/v) triethylamine (TEA), and 1% (w/v) sodium hydroxide (NaOH) in distilled water, and the mixture was stirred using a magnetic stirrer until fully dissolved and clear. The MPD solution was prepared by dissolving 2% (w/v) MPD, 2% (w/v) TEA, and 0.2% (w/v) sodium dodecyl sulfate (SDS) in distilled water, followed by stirring with a magnetic stirrer until the solution was homogeneous and visibly transparent. Solution B: An organic solution was prepared by adding 1,3,5-benzenetricarbonyl trichloride (TMC, 98%) to preheated hexane at 50°C. Various concentrations of TMC were prepared in the organic solution for the optimization process.
The thin-film layer was prepared: PA6 nanofiber membranes were first immersed in Solution A for a specified duration, then removed and air-dried. Subsequently, the membranes were immersed in Solution B after another specified period. Finally, the samples were placed in an oven at 100°C for 10 min to facilitate the crosslinking of the PA thin film (Figure 2). Schematic diagram of PA thin-film membrane preparation.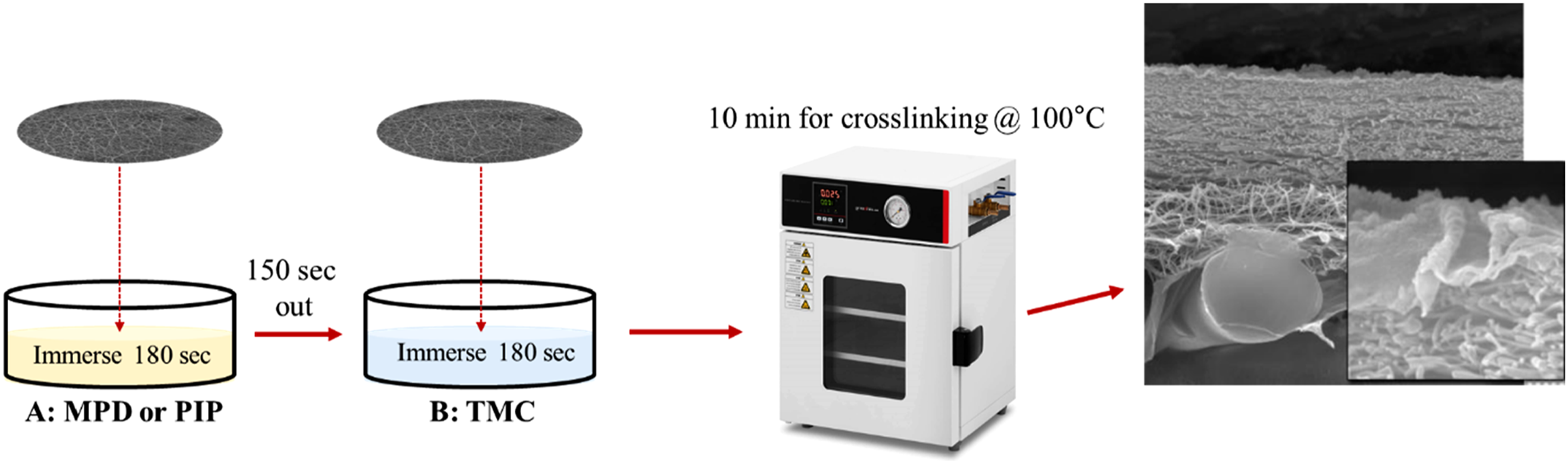
Composition and preparation details of the prepared membranes, including abbreviations, monomer concentrations, constant drying time (150 s), and constant immersion durations (180 s).

Membrane characterization
The surface morphology and defects of the prepared active thin-film layer on the composite material were analyzed using scanning electron microscopy (SEM) with a TESCAN VEGA instrument (Brno, Czech Republic). The fiber diameter of the PA6 nanofiber web was measured using the ImageJ software (free version: ImageJ bundled with 64-bit Java 8). The pore size of the nanofiber web was measured using the bubble point method26,27 with a Porometer 3G pore size analyzer from Anton Paar GmbH. The bubble point technique is often employed with a Porometer to conduct these measurements, providing precise data on the size and distribution of pores in materials. Water contact angle (WCA) measurements were performed using a Krüss Drop Shape Analyser (DS4) with deionized (DI) water and feed solutions under neutral, pH 2, pH 6, and pH 9.5 conditions. The 2, 6, and 9.5 pH values were selected to fabricate membranes under acidic, near-neutral, and alkaline conditions, respectively. These pH levels allow systematic investigation of fabrication conditions and are relevant to practical membrane cleaning operations, where acidic or alkaline environments are commonly encountered depending on the cleaning agent used.28,29 The chemical composition of the thin film was analyzed using Fourier transform infrared spectroscopy (FT-IR) with a Thermo Scientific™ Nicolet iZ10 spectrometer.
Filtration and rejection of membranes
The filtration test was conducted using a dead-end filtration unit with an active filtration area of 15 cm2 (Millipore, XFUF04701) with 50 mL filter capacity at 4 bar. A salt feed solution containing 2000 ppm of NaCl and MgSO4 was prepared by dissolving the salts in distilled water. Before introducing the feed solution, the membranes were stabilized by distilled water filtration. The membrane flux (f) and permeability (k) was calculated using equations (1) and (2), where A represents the filtration area, V is the volume of permeate collected, p is the applied pressure, and t is the time taken to collect the permeate.

The rejection rate (R) was evaluated using a conductometer calculated using equation (3), where Cf is the conductivity of the feed solution, and Cp is the conductivity of the permeate solution.
Results and discussion
Membrane characterization
Characterization of nanofiber web
The surface of the nanofiber web after the lamination process has been given in Figure 3. The red-marked area in Figure 3 highlights the adhesive point on the web, where the glue (adhesive web) has filled the pores of the nanofibers. The results indicated that the lowest applied temperature reduced the fiber web’s contact area. The elevated temperature increases the viscosity of the molten glue. The glue melted easily, penetrating the composite layers and covering a larger pore area (Figure 3(b) and (c)). As a result, the final membrane exhibits low permeability due to clogged pores. Moreover, it was found that the low porosity of the support causes more significant resistance to solute diffusion than does the interconnected pore and fiber network within the eTFC.
30
SEM image of the nanofiber membrane laminated at (a) 110°C, (b) 125°C, and (c) 135°C with an adhesive point on the web, highlighted in the red-marked area.
Nanofiber membrane properties.

Reducing the nanofiber diameter decreases the web’s pore size, as smaller fibers can form a denser and more interconnected network. This increased packing density results in narrower voids or inter-fiber spaces, thereby minimizing the overall pore size. The enhanced surface area of finer fibers contributes to a more compact and cohesive fiber structure, further restricting pore dimensions. It has been proven that there is a positive correlation between fiber diameter and pore size.31–33 Reduced pore size in TFNC membranes leads to lower permeability but enhances rejection performance, improving their effectiveness in filtration and separation applications.
A relationship has been established between fiber diameter, surface area-to-volume ratio, and the pore size of nanofibers. Specifically, a reduction in fiber diameter results in an increased surface area and a concomitant decrease in pore size.34,35 Can-Herrera et al. 36 demonstrated that an increase in the diameter of polycaprolactone nanofibers, achieved by modifying the applied voltage, reduces the surface area of the fibers. A finer nanofiber diameter yields a higher surface-to-volume ratio, which enhances the surface area of the coated PA thin film. This increased surface area improves the uniformity and adhesion of the coating, leading to a thinner, more selective separation layer. As a result, the membrane exhibits enhanced water permeability and filtration efficiency while maintaining high rejection performance.
Increasing the heat-press lamination temperature decreases air permeability in the nanofiber web, mainly due to substantial structural changes in the fiber matrix. At high temperatures, partial melting and fiber fusion occur, resulting in a denser and more compact configuration that reduces the size and number of inter-fiber holes, blocking airflow across the web. The adhesive web used in the lamination process melts and fills the remaining pores, reducing porosity and air permeability. The WCA did not show any significant changes by changing the lamination temperature.
Based on the SEM images in Figure 3, the highest air permeability, the lowest fiber diameter, and the lowest pore size (Table 2), the nanofiber membrane laminated at 110°C was selected as the support layer for the thin-film composite membrane.
Characterization of thin-film composite membrane
The surface structure of the PA6 nanofiber and the PA thin-film composite membranes was observed using SEM imaging (Figure 4). The average diameter of the selected PA6 nanofibers laminated at 110°C was 90 ± 19 nm. No bead structure or damage on the nanofiber surface has been observed. The avr. pore size of the PA6 nanofiber membrane was measured as 0.33 µm. SEM images of (a) PA6 nanofiber support layer for thin film composite membrane, (b) MDP-1, (c) MDP-2, (d) MDP-3, (e) MDP-4, (f) PIP-1, (g) PIP-2, and (h) PIP-3 thin-film composite membranes.
The surface morphology of the MPD-based membranes revealed that the thin film formed effectively on the nanofiber layer without any cracks or damage. As the concentration of TMC in the organic phase increased from 0.2 to 0.6 wt.%, the bead-like structures became less prominent. This can be explained by the denser structure of MPD-based membranes synthesized with a high concentration of TMC. Nulens et al. 37 observed that higher concentrations of TMC led to a denser structure in the thin film, reducing membrane permeability. Additionally, they noted that when combining a high concentration of MPD (0.185 M) with a low concentration of TMC (0.0038 M), the membrane surface exhibited the typical ridge and valley morphology characteristic of hexane. They also suggested that a minimum threshold concentration of TMC is necessary to achieve a rejection rate of 80% or higher.
On the other hand, PIP-based membranes showed nodular ridge structures on the surface. The increased amount of TMC in organic faces increased the size of the ridge structure. Compared to MPD, the high nodular-ridge structure of PIP-based membranes could be due to their ability to form a three-dimensional cross-linked structure with TMC. Similar structures have been reported before.38–40
WCA of MPD and PIP-based membranes.

The WCA results of MPD- and PIP-based membranes show a clear dependence on the TMC concentration and fabrication conditions. For MPD-based membranes, the WCA increased progressively with TMC concentration, ranging from 63.66° for MPD-1 (0.2% TMC) to 71.20° for MPD-4 (0.6% TMC) similarly reported in literature.41,42 This trend can be attributed to changes in surface morphology, as observed in SEM images (Figure 4), where increasing TMC concentrations reduced the size of bead-like structures, resulting in smoother surfaces with reduced hydrophilicity. The pH variations in MPD-3 samples affected WCA significantly, with the highest WCA of 76.72° observed at pH 6, suggesting an optimized interaction between MPD and TMC at this pH level. The variation in WCA of MPD-based membranes prepared under different pH conditions can be attributed to changes in membrane surface chemistry and cross-linking density during interfacial polymerization. Under acidic conditions (pH 2), reduced cross-linking leads to a more hydrophilic and looser polyamide structure, lowering the contact angle (69.87°), while moderate pH (pH 6) favors denser cross-linking, resulting in a slightly more hydrophobic surface (76.72°). 43 At higher pH (pH 9.5), excessive deprotonation increases surface roughness and free carboxyl groups, enhancing hydrophilicity again and thus reducing the contact angle to 65.29°.
The hydrophilic characteristic of the thin film is related to amino groups on the surface. 44 For this reason, monomers can show different water contact angles. The higher contact angle of the MPD-based membranes is attributed to the abundant hydrophobic benzene ring in their structure. 45 In contrast to MPD-based membranes, PIP-based membranes exhibited much lower WCA values, indicating higher hydrophilicity. The WCA ranged from 8.54° for PIP-1 (0.2% TMC) to 5.90° for PIP-2 (0.45% TMC) and 5.97° for PIP-3, which was cured at room temperature. The SEM analysis (Figure 4) showed that PIP-based membranes formed nodular-ridge structures, with increasing TMC concentration enhancing the size of these ridges, further contributing to their hydrophilic nature. The marked difference in surface characteristics between MPD- and PIP-based membranes can be attributed to the higher degree of three-dimensional cross-linking in PIP-TMC interactions, which promotes hydrophilicity and suppresses the formation of bead-like structures. Li et al. 44 reported that the PIP/TMC membrane exhibited a relatively low contact angle of 39.6° ± 1.6°, which they attributed to the grainy structure or increased porosity (pore size and pore density) on the membrane surface, leading to reduced wettability. Our research findings also demonstrate that the PIP-based membrane exhibits a lower contact angle, aligning with the results reported by Li et al., who attributed this phenomenon to the membrane’s grainy structure and increased porosity. It can be summarized that these findings emphasize the impact of TMC concentration, pH, and polymer type on the morphological and wetting characteristics of thin-film composite membranes.
In order to confirm the chemical structure of the PA layer, the FTIR spectra for MPD and PIP-based membranes were measured and shown in Figure 5. FTIR image of MDP- and PIP-based membranes.
The spectrum of the MPD-based membrane, represented by blue-colored bands, exhibits peaks at 1661 cm−1 and 1534 cm−1, corresponding to characteristic C=O stretching vibrations (amide I) and the N–H bending vibrations (amide II) of the amide group (–CONH–), respectively. The sharp peaks observed between 700 and 900 cm−1 are attributed to the out-of-plane bending vibrations of the aromatic C-H bonds in the phenyl ring of MPD. Additionally, the bands at 1600 and 1480 cm−1, indicating aromatic ring breathing, and at 1250 cm−1, characteristic of amide III, are specific to the polyamide (PA) structure. The absence of an acid chloride signal confirms the successful polymerization.
For the PIP-based membrane, represented by red-colored bands, the N-H bending vibration (amide II) appears at 1576 cm−1, while the C=O stretching vibration (amide I) is observed at 1660 cm−1. These peaks are typically formed during interfacial polymerization between piperazine and acid chlorides, indicating the presence of functional groups in the polyamide (PA) selective layer. The broad peak between 3200 and 3500 cm−1 corresponds to the N-H stretching vibrations from secondary amines, as piperazine contains two secondary amine groups (-NH-).
These spectral differences underscore the molecular diversity between MPD’s aromatic and PIP’s aliphatic frameworks, with MPD exhibiting intense aromatic and amine-related vibrations, while PIP displays features associated with its cyclic secondary amine composition.
Filtration results
PIP-based membranes exhibit higher water flux than MPD-based membranes due to their more open and less dense structure, which allows water molecules to pass through more easily. However, this loose structure makes them less effective for monovalent salt ion separation, such as NaCl removal, as smaller ions can easily permeate through the membrane. On the other hand, the higher permeability of PIP-based membranes makes them ideal for removing divalent salts, which are easier to reject due to their larger hydrated radius and stronger electrostatic interactions with the membrane. In contrast, MPD-based membranes form a denser and tighter network, 45 providing better rejection of monovalent salts and making them more suitable for their separation. For this reason, MPD-based membranes are used for NaCl separation, while PIP-based membranes are preferred for MgSO4 separation. The larger free volume in PIP-based membranes prevents the permeation of divalent SO42− ions, making them more suitable for divalent salt removal than monovalent salts.
MPD based membrane
Figure 6 illustrates the flux and salt rejection rates of different membrane samples, identified as MPD-1, MPD-2, MPD-3, MPD-4, MPD-3/pH2, MPD-3/pH6, and MPD-3/pH9.5. The deionized (DI) flux shows considerable variation among the samples, with MPD-1 exhibiting the highest DI water flux of 60 Lm−2h−1, followed by MPD-3 and MPD-3/pH9.5, both at 40 Lm−2h−1. MPD-2, MPD-3/pH2, and MPD-3/pH6 have slightly lower DI water flux values of 33.33, 35 and 33 Lm−2h−1, respectively, while MPD-4 has the lowest DI water flux of 16 Lm−2h−1. The reason can be associated with compactness and dense structure of the membranes. When the pH value of neutral water increased to pH9.5, no changes were observed in the flux. Influence of MPD-based preparation parameters and feed pH on NaCl rejection and the flux of deionized and saline water.
The saline water flux exhibited a similar trend to that observed with DI water, with the MPD-1 membrane demonstrating the highest flux. In contrast, the saline water flux of the MPD-4 membrane was higher compared to MPD-2 and MPD-3, as well as across their respective pH variations. Decreasing the pH of the feed resulted in a reduction of both the membrane flux and the NaCl salt rejection rate. The NaCl rejection of MPD-based membranes was generally maintained or slightly enhanced, a phenomenon likely attributable to changes in surface charge. 46 A notable decrease in NaCl rejection occurred when transitioning from neutral to acidic pH, which can explain the diminished electrostatic repulsion between the charged membrane surface and ions. This decrease in repulsion is due to the reduction of the membrane’s negative surface charge as the feed pH decreases from 7 to 2. This trend is consistent with findings reported in the literature.47,48
Increasing the concentration of TMC results in higher rejection rates while reducing water flux up to a specific threshold. This effect is likely due to forming a denser and more compact active barrier layer in MPD-based membranes, which is influenced by the aromatic amine structure. Consequently, these membranes exhibit selective rejection properties, particularly for monovalent salt ions such as NaCl. 22 On the other hand, MPD-based membranes showed low flux due to their highly dense and tightly cross-linked polyamide structure. This dense network significantly reduces the pore size, limiting water permeability and resulting in lower flux. While this tight structure enhances NaCl rejection by preventing the passage of small monovalent ions, it also restricts water flow, making the membrane less permeable and reducing overall flux.
PIP based membrane
The analysis of PIP-based membranes prepared under various conditions and evaluated at different pH levels shows significant variation in flux and MgSO4 salt rejection rates. The membranes exhibit distinct performance metrics for the saline water flux, as illustrated in Figure 7. PIP-1 and PIP-2 show moderate flux values of 10 and 8.6 L/m2·h, respectively, with 91% and 97% rejection rates, suggesting an inverse relationship between flux and rejection rate in these cases. Results indicated that elevated concentrations of TMC result in enhanced rejection rates, which are attributed to an increase in cross-linking density and membrane thickness. However, this improvement in rejection performance is typically associated with a reduction in water flux. Influence of PIP-based membrane preparation parameters and feed pH on NaCl rejection and the flux of deionized and saline water.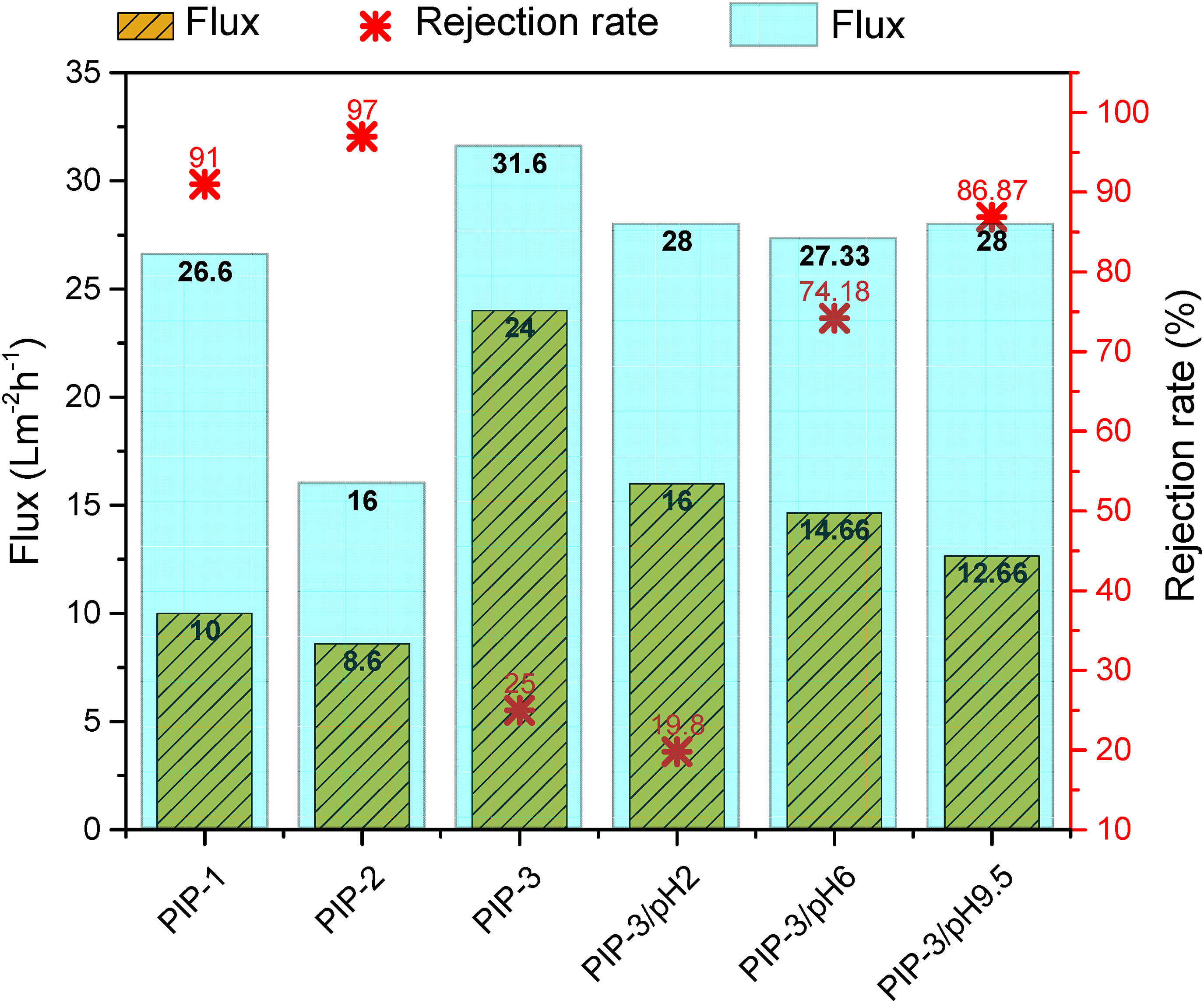
In contrast, lowering the concentration of TMC has been observed to enhance water flux, albeit at the expense of diminished rejection efficiency.22,49 Subsequently, the optimal TMC concentration (PIP-2), which yielded the highest rejection rate for divalent salts, was evaluated for performance with monovalent salts such as NaCl. The results indicated a low % rejection rate of 25%, accompanied by an increased water flux relative to the rejection performance observed for divalent salts. This behavior can be attributed to MgSO4 large space volume, which does not allow the divalent SO42− ions to permeate the membrane. 50 Moreover, forming a charged active barrier layer enriched with pendant carboxylic acid groups imparts selective rejection characteristics favoring divalent ions, such as MgSO4, in the PIP-TMC-based membrane.
PIP-3 achieves the highest flux of 24 L/m2·h with a reduced rejection rate of 25% due to its inherently looser and more open structure than MPD-based membranes, indicating a balance between permeability and selectivity. This balance makes PIP-based membranes more suitable for applications where high water flux is prioritized over salt rejection, such as partial desalination or softening processes. The flux performance decreased further with pH adjustment: PIP-3/pH2, PIP-3/pH6, and PIP-3/pH9 yield lower flux values of 16, 14.66, and 12.66 L/m2·h, respectively. Decreasing the feed pH to acidic reduces the MgSO4 salt rejection rate to 20.8% compared to neutral pH. On the other hand, the basic pH of the feed solution increased the MgSO4 salt rejection rates up to 86.87%. PIP-3/pH9.5 demonstrates a flux of 12.66 L/m2·h and a marginally improved rejection rate of 86.87%, reflecting the influence of alkaline pH on membrane selectivity. The increased rejection efficiency of MgSO4 by PIP-TMC membranes at higher pH levels, as compared to lower pH levels, can be attributed to several factors, including changes in surface charge, the Donnan exclusion effect, solute speciation, and the membrane’s structural properties. At elevated pH, the membrane surface acquires a more pronounced negative charge, thereby enhancing electrostatic repulsion of sulfate ions and improving rejection performance. Conversely, at lower pH levels, the membrane’s charge density diminishes, leading to reduced rejection efficiency, potentially compounded by structural modifications of the membrane. 51
The performance of MPD- and PIP-based membranes is significantly influenced by pH, as it alters membrane surface chemistry during interfacial polymerization. The charge properties of these membranes are primarily determined by the functional groups in the polyamide layer, specifically the carboxyl (–COOH) groups from TMC) and the amine groups from monomers such as PIP and MPD. At higher pH levels, deprotonation of carboxyl groups increases the membrane’s negative charge, while at lower pH, protonation of amine groups (–NH– from PIP or –NH2 from MPD) enhances its positive charge. Anions like chloride (Cl−), less hydrated than cations such as sodium (Na+), tend to adsorb onto the membrane surface, further amplifying its negative charge. 52 These pH-dependent chemical modifications directly affect membrane structure and separation efficiency. 53 Under acidic conditions, protonation of amine groups in MPD reduces their reactivity with acyl chloride, leading to a less cross-linked, more hydrated polyamide layer. This looser structure increases hydrophilicity and free volume, enhancing water permeability and resulting in higher flux. However, the reduced cross-linking lowers salt rejection efficiency. Conversely, under alkaline conditions, deprotonation of amine groups in PIP increases their nucleophilicity, promoting the formation of a highly cross-linked, dense polyamide network. This tighter structure reduces pore size, enhances salt rejection, and compromises water permeability, leading to lower flux. These structural and charge-related changes play a crucial role in determining the desalination performance of MPD- and PIP-based membranes. Optimizing pH conditions during membrane fabrication is essential for balancing permeability and selectivity, ultimately improving membrane efficiency in water treatment applications.
These findings underscore the critical role of pH in modulating PIP-based membrane performance, where acidic and neutral conditions have higher flux compared to alkaline conditions but compromise salt rejection, while alkaline conditions offer a more balanced performance. This analysis highlights the trade-offs between permeability and rejection efficiency, emphasizing the importance of optimizing preparation conditions and pH settings to tailor membrane functionality for specific desalination and separation applications.
Jahanshahi et al. 38 studied a similar concentration of PIP-based membrane as in this study for PIP-1. The membrane demonstrated average water fluxes of 53 and 101 kg/(m2h) at 5 and 10 bar pressures, respectively. The membrane achieved NaCl rejection rates of 44% at 5 bar and 35% at 10 bar, while MgSO4 rejections were 78% at 5 bar and 70% at 10 bar. Han et al. 40 reported that the PIP-based membranes exhibited the lowest permeability of 17.86 L/(m2h·bar), with Na2SO4 rejection of approximately 98% at an operating pressure of 8 bar. Adding CaCl2 to the PIP membrane increased permeability by 67% while maintaining the same rejection rate.
Comparative analysis of prior research findings and current study results.

The current research findings indicate that for the MPD-based approach, a PA6 nanofibrous membrane with 2 wt.% TEA and 0.2% SDS achieves a rejection of 74% and a flux of 0.42 Lm−2h−1 using a 2000 ppm NaCl feed under a pressure of 4 bar. This performance appears to be on the lower end compared to similar studies. For instance, polysulfone ultrafiltration membranes without specific additives demonstrate a significantly higher rejection of 99.6% under a higher pressure of 15.5 bar, albeit with a reduced flux of 0.75 Lm−2h−1. 55 Another comparable study using microporous polysulfone ultrafiltration membranes with multiple additives achieved a rejection rate of >97% and a much higher flux of 29.7 Lm−2h−1 at a similar pressure of 15 bar. 57 Similarly, polyvinylidene fluoride membranes without additives exhibited 89.64% rejection at 15 bar with flux values below 0.8 L m−2 h−1, indicating superior rejection but slightly lower permeability compared to the current study operating at a considerably lower pressure. 58 Moreover, other advanced modifications, such as polydopamine/metal-organic framework membranes, achieved 93.5% rejection with a flux of 8.48 L m−2 h−1 at 4 bar, 59 and SDS/sodium carbonate-modified PS membranes reached 97.6% rejection with a flux of 34 L m−2 h−1, 60 further highlighting the comparatively moderate performance of the present membrane. These comparisons suggest that while the current membrane demonstrates reasonable permeability under mild operating conditions, further optimization is necessary to enhance its selectivity and overall separation efficiency.
In the PIP-based membrane, our PA6 nanofibrous membrane achieves a rejection of 97% and a flux of 8.6 Lm−2h−1 using a 2000 ppm MgSO4 feed at 4 bar, demonstrating strong rejection performance. Compared to the literature, a PS ultrafiltration membrane without specific additives reaches only 75.4% rejection at a higher pressure of 15.5 bar. 55 Incorporating 5 wt.% sodium phosphate into a porous PS substrate improved the rejection to >81.93%, with an exceptionally high flux of >36.7 L·m−2·h−1 at 5 bar. 50 A similar approach using porous PS with 4.0% TEA and 0.01% SDS additives achieved a rejection of approximately 70% and a flux of ∼55 L·m−2·h−1 at 5 bar. 64 While these membranes show higher flux values, their rejection rates are notably lower compared to our current membrane. The membranes modified with Fe3+ ions and tannic acid on a PS ultrafiltration membrane achieved less than 70% rejection at 2 bar, 67 whereas a PA6 nanofiber with nonwoven support achieved 90.2% rejection with a higher flux of 53.1 L·m−2·h−1 at 4.8 bar. 22 Although the latter shows a higher flux, our membrane still delivers superior rejection performance. Our current PIP-based membrane shows a balanced and enhanced performance in high rejection and moderate flux at relatively low operating pressure compared to various reported PIP-based membranes, confirming its potential for effective MgSO4 removal.
The current study exhibits an outstanding equilibrium between rejection and flux, particularly at reduced operational pressures. However, although rejection rates are competitive, flux values require more adjustment to align with or exceed the higher flux values reported in the literature.
Recommendations for future research
Future research needs to concentrate on overcoming the significant challenge of achieving a balance between higher water permeability and effective salt rejection, while also guaranteeing the long-term stability and fouling resistance of nanofiber-based thin-film composite membranes employed in saline water remediation. Considering our findings about the impact of variables such as lamination temperature, monomer composition, and feed pH on membrane performance, it is crucial to explore further how these parameters influence long-term membrane behavior in actual environmental settings. Future research needs to comprehensively evaluate membrane durability and efficacy over time, especially in varied and complex water matrices characterized by varying salinity, pH variations, and the existence of organic and inorganic contaminants. Moreover, research needs to focus on understanding the mechanisms of membrane degradation and the particular effects of operational parameters on selectivity, permeability, and structural integrity. Through extensive long-term and field-based testing, researchers can clarify the trade-offs between selectivity and permeability while identifying strategies for reducing performance degradation, thereby advancing the development of more resilient and sustainable desalination technologies. Additionally, the integration of Design of Experiments (DOE) with advanced optimization algorithms, such as the Bee Algorithm, Grey Wolf Optimizer (GWO), and Particle Swarm Optimization (PSO), possibly combined with machine learning models, is suggested to systematically design, model, and optimize membrane fabrication parameters. This combined approach can significantly accelerate the development of high-performance membranes for saline water remediation by enabling efficient exploration and exploitation of the experimental design space.
Conclusion
This study enhances saline water remediation by introducing a novel technique for producing PA6 nanofiber-based thin-film composite membranes, emphasizing the complex relationship between thermal treatment and nanofiber morphology and subsequent thin-film membrane optimization. Our parametric study, which changes monomer combinations and examines the impact of feed pH, provides essential insights into the design parameters influencing flow and salt rejection. The results indicate that high lamination temperatures result in changes in morphology in the nanofibers, including increased diameter and reduced air permeability, which can directly affect the efficacy of the composite membranes. The comparative investigation of MPD- and PIP-based membranes under varying conditions highlights the necessity of systematically adjusting both chemical composition and operational pH to attain an equilibrium between permeability and selectivity. MPD-based membranes demonstrate a complex response to changes in pH influencing NaCl rejection, whereas PIP-based membranes exhibit enhanced rejection of divalent ions such as MgSO4, particularly in alkaline environments. Despite these promising findings, a key limitation of this study lies in the scalability and energy costs associated with the lamination, filtration under high pressure, and thin-film formation process, which may hinder large-scale membrane production. The requirement for precise process control and the associated energy consumption underscore the need for alternative, energy-efficient fabrication methods. Future research should explore sustainable approaches, such as solar-assisted techniques, to reduce operational costs and enhance environmental sustainability. It is also suggested to evaluate the membranes’ long-term stability and fouling resistance using continuous filtration systems with larger capacity to simulate practical desalination conditions better. The study emphasizes the importance of optimizing preparation methods and operational parameters to create effective membranes designed for particular desalination and separation requirements, providing a promising foundation for further advancements in membrane technology.
Footnotes
Author contributions
F.Y: Writing – original draft, Supervision, Resources, Project administration, Methodology, Visualization, Investigation, Funding acquisition, Formal analysis, Conceptualization. V.K.V.: Methodology, Investigation, Formal analysis. B.Y: Writing – original draft, Methodology, Investigation, Formal analysis, Visualization, Conceptualization. A.S.: Writing – original draft, Methodology, Investigation, Visualization, Conceptualization. M.K.: Writing – original draft, Supervision, Methodology, Investigation, Conceptualization. J.C: Writing, Investigation, Formal analysis. P.I.: Writing – original draft, Supervision, Resources, Project administration, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the support provided by the Research Infrastructure NanoEnviCz, funded by the Ministry of Education, Youth and Sports of the Czech Republic under Project No. LM2023066. Also, this work was kindly supported by the European Union under the REFRESH – Research Excellence for Region Sustainability and High-tech Industries [project number CZ.10.03.01/00/22_003/0000048] via the Operational Programme Just Transition. The study was funded by the Czech Science Foundation (GAČR), project number 25-15195S.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
