Abstract
Jute is a natural fiber that decomposes organically and is widely used in textiles, composites, and many industrial applications. The cellulose’s crystallinity influences jute fibers’ durability, moisture resistance, and mechanical qualities. This study examines the impact of time, temperature, and peracetic acid (PAA) concentration on the crystallinity of cellulose jute fibers. Chemical concentration was varied with time and temperature variation to improve the crystallinity of jute fibers. After the experiment, the crystallinity of jute fibers improved altering chemical structure brought about by PAA’s removal of noncellulosic components from jute fibers. X-ray diffraction (XRD) analysis has been performed to find the crystallinity improvement in the fibers. After the chemical treatment which was performed at 2 M for 120 min at 50-60°C temperature, the maximum crystallinity was 75.34%. The findings suggest that longer treatment times and higher temperatures increase crystallinity because the PAA can easily reach the cellulose microfibrils. In addition, it was observed that the enhancement of crystallinity was significantly influenced by the optimal concentration of PAA, with an optimal concentration being identified.
Introduction
Bast fibers have garnered significant attention in recent times. Utilizing bast fiber materials (Jute, hemp, flax, and ramie) offers numerous benefits, including low cost, low density, favorable specified mechanical properties, and biodegradability. 1 Among their many benefits are these materials’ lighter weight, competitive specific strength and stiffness, improved energy recovery, carbon dioxide sequestration, ease of manufacturing, and environmental friendliness. 2 Besides these, more suitable and advanced applications purpose, various chemical and surface modifications of jute fibers have been attempted because the fibers have lower strength than some natural fibers, and synthesis fibers have poor thermal and dimensional stability; after the jute falls, its strength rapidly, easily rotten, etc.
Jute fiber must be able to bind and be compatible with the resin matrix well to function as an efficient reinforcing constituent which have undergone some chemical alterations to provide them with acceptable bond properties and serve as excellent candidates for reinforcement in general applications. Alkali treatment is the most cost-effective among other surface treatments. Alkali treatment may reduce the moisture absorption of fibers and enhance the interface.
3
The dissolution of cellulosic constituents in alkali treatment created voids in the fiber structure, and swelling was observed to have occurred with the increase in the crystallinity of the fibers. This resulted in a drop in linear density and made it more brittle.
4
Oxidation modification with NaClO2 (Sodium Chlorite) removed lignin but affected jute fibers’ moisture absorption, dimensional stability, moisture recovery, and microbiological deterioration can be affected.
5
The presence of waxes and pectin in the cell wall of natural fibers covered the fibers’ reactive functional groups, thus preventing the interlocking with the matrix. The inaccessibility of fibers with matrix leads to poor adhesion across the phase boundary, weak dispersion of force, and inadequate strength properties.
6
Reducing natural fiber’s hydrophilic qualities to increase its compatibility with the hydrophobic matrix is the primary goal of chemical treatment. Oxidized jute fibers may be incompatible with polymers in composite materials or surface treatments due to their altered chemical characteristics like poor adhesion, delamination, or other performance difficulties.
7
The most common bleaching agent in the textile sector is peracetic acid, which is easily broken down and is a potent oxidizing reagent which also has a unique delignification impact on the lignocellulosic biomass.
8
Peracetic acid undergoes breakdown into acetic acid and oxygen, resulting in biodegradable decomposition products that do not generate any harmful by-products.
9
The processed fibers exhibit a prominent level of whiteness while experiencing minimal damage during peracetic acid bleaching. Peracetic acid undergoes decomposition, as illustrated in Figure 1, throughout the bleaching process. The reactive oxygen molecule [O] and the peroxy radical [ Study the impact of time, temperature, and chemical concentration on the crystallinity of jute fibers treated with peracetic acid.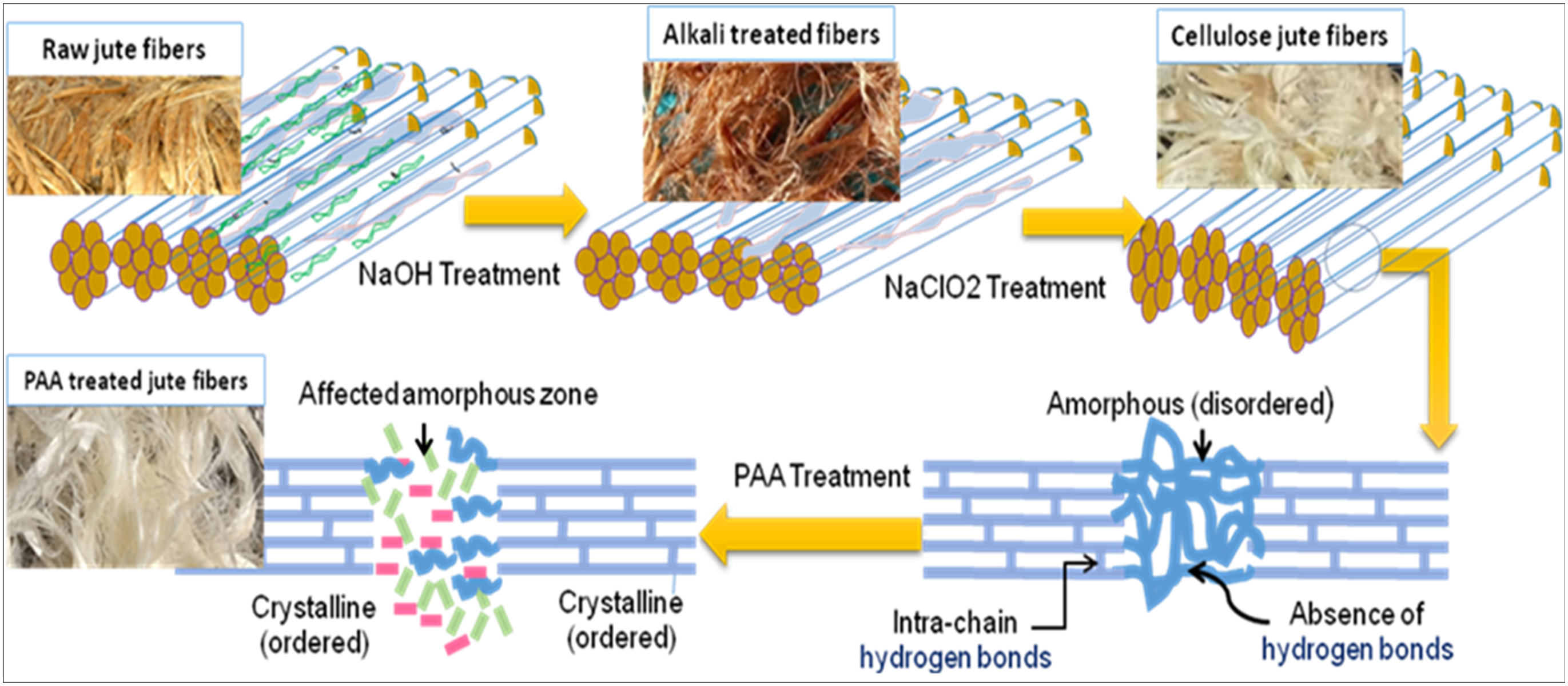
Peracetic acid treatment has recently become a promising technique for altering the surface characteristics of natural fibers, resulting in enhancements in their mechanical, thermal, and moisture absorption properties. 11 Temperature, chemical concentration, and time typically affect Most chemical oxidation processes.12,13 Therefore, it is essential to clarify the exact mechanism of thermal peracetic acid (PAA) degradation to precisely evaluate the Impact of temperature on the PAA treatment process.
This treatment entails oxidizing the fiber’s surface and altering its chemical makeup and structure. Prior research has shown that using peracetic acid can increase the crystalline structure of natural fibers, leading to enhanced overall performance. However, the distinctive attributes of jute fibers are intricately linked to their crystalline arrangement, which impacts mechanical robustness, moisture uptake, and other significant traits. 14 Research on natural fibers and composites has significantly increased due to growing environmental concerns regarding the risks associated with synthetic fiber-based composites. Jute-based bio and hybrid composites have been widely utilized in various applications. 15
X-ray diffraction (XRD) is a widely used analytical method for determining the crystallinity and other crystallographic parameters of materials, such as crystallite size, microstrain, dislocation density, etc. 16 Analyzing their crystallographic parameters can help better understand. 17 Crystallographic analysis can provide valuable insights into degradation mechanisms by detecting crystal structure and orientation alterations. This information can then be used to direct efforts toward enhancing the durability and lifespan of products.18,19
Chemical treatments can enhance the performance of jute fibers in many applications by modifying their crystallinity. The crystallinity of jute fibers plays a vital role in determining their mechanical strength, thermal stability, and moisture absorption properties. 20 Multiple parameters, such as treatment duration, temperature, and chemical concentration, influence the efficacy of peracetic acid therapy in augmenting fiber crystallinity. Nevertheless, complete knowledge is needed regarding these parameters’ individual and collective impacts on the crystalline structure of jute fibers.
This study examines the application of peracetic PAA treatment, a potent oxidizing agent commonly utilized in textile bleaching, as an innovative approach to enhance the crystallinity of jute fibers. PAA’s distinctive capability to eliminate lignin and other non-cellulosic constituents improves the structural integrity of cellulose, potentially mitigating the intrinsic constraints of jute fibers. The crystallinity of cellulose significantly affects the mechanical, thermal, and moisture absorption characteristics of natural fibers. Notwithstanding the possible advantages, a thorough comprehension of the effects of treatment time, temperature, and chemical concentration on jute fiber crystallinity is still inadequate.
The primary intent of this study is to systematically evaluate the impact of PAA treatment parameters on the crystallinity enhancement of jute fibers. This study contributes to the existing body of knowledge by identifying optimal treatment conditions that maximize crystallinity while preserving fiber integrity. By addressing these gaps, the findings have significant implications for advancing the use of jute fibers in sustainable industrial applications, particularly in the development of high-performance composite materials.
Materials & methodology
Materials
Jute fiber was acquired from the local market. Other analytical grade chemicals, such as peracetic acid and acetic acid, were acquired from Labtex, Dhaka-1217, while sodium hydroxide and chlorite were procured from Merck, Germany. All these chemical are graded. Deionized water was acquired from PureTech, Dhaka-1203, Bangladesh, for each investigation step.
Methodology
A pretreatment procedure is essential before treating the jute fibers to ensure thorough cleaning. Typically, this involves thoroughly cleaning and washing the fibers to remove dust, debris, or other unwanted substances. Untreated (raw) and cellulose jute fibers can significant references for comparing and demonstrating the effects of time, temperature, and chemical concentration of peracetic acid treatment on crystallinity enhancement in jute fibers.
The fibers were submerged in the solution after making the peracetic acid solution with specific concentrations. Here, time, temperature, and concentration of peracetic acid must be managed since they influence the qualities of jute fibers. After treatment, thoroughly rinse the jute fibers with clean water to eliminate any remaining PAA solution. This process can be seen in Figure 1.
The mixing ratio of acetic acid (CH3COOH) and hydrogen peroxide (H2O2) to prepare PAA in water depends on the application’s PAA concentration. Peracetic acid solutions are employed as disinfectants, sanitizers, and sterilizers in food processing, healthcare, and water treatment. 21
The jute fibers are immersed in a peracetic acid solution with the correct concentration, ensuring the time and temperature are appropriately maintained. After treatment, the jute fibers are dried to eliminate any remaining moisture before characterization. During these chemical processes, suitable chemical concentration, temperature, and time will be investigated to enhance the crystallinity of jute fibers. 22
Furthermore, it is imperative to adhere rigorously to safety measures when handling acid and other chemicals. This includes strict adherence to using personal protective equipment, adequate ventilation, and effective waste disposal techniques.
Chemical treatment
Jute fibers are coated with undesirable substances called gum. These disrupt natural functioning and hinder intended performance. Degumming is a purification technique used to eliminate contaminants. The chemical degumming process consists of successive processes: alkaline treatment, scouring, and bleaching. 23 It is necessary to trim them to approximately 10 cm lengths precisely. The NaOH solution used for alkali treatment of jute fibers has a concentration of 1%. This concentration is chosen to balance the need for effective treatment with the risk of fiber destruction that can occur with more significant concentrations. 24 Besides, this percentage can remove non-cellulose components effectively, and preserve mechanical properties.
Additionally, the duration and temperature of the alkali treatment bath impact the treatment’s efficacy. This experiment uses the temperature and time of 30 min and 30°C. Elevated temperatures and ten times have the process but may also heighten the danger of fiber injury.25,26 The fibers should be neutralized with an acid solution (acetic acid) following the alkali treatment to halt the alkaline reaction. Further rinsing with demineralized water is required to eliminate any remaining alkali in the fibers.
This experiment utilized oxidative Modification using a 0.7% NaClO2 solution, with a duration of 80 min and a temperature of 80°C, as a chemical treatment method to alter jute fibers. The technique can introduce oxygen-containing functional groups onto the surface of the jute fibers. Oxidation can alter the chemical makeup of fibers, leading to changes in their characteristics, as shown in cellulose jute fibers. 27
An acid hydrolysis process was employed to eliminate the amorphous region where peracetic acid was used, producing a highly crystalline substance known as microcrystalline cellulose.
28
However, it is essential to maintain a chemical concentration of 1.5 morality, a 50°C temperature, and a time of 120 min for this process. The alkali treatment degraded the hydroxyl groups on the fiber’s surface, disposing of excess lignin, hemicellulose, pectin, wax, and other contaminants. Besides, the NaOH arrangement invaded the shapeless range, causing cellulose swelling and the extension of the cell divider. This upgraded the surface roughness, encouraging a more open interface for plant fiber-resin interaction and cultivating strong adherence with the lattice utilizing mechanical interlocking. Moreover, the alkali treatment debased sensitive hydroxyl bonds in fibers, making hydroxyl bonds between cellulose molecules. The chemical treatment process can be seen in Figure 2. Chemical treatment process for achieved raw jute fiber to peracetic acid treated jute fibers.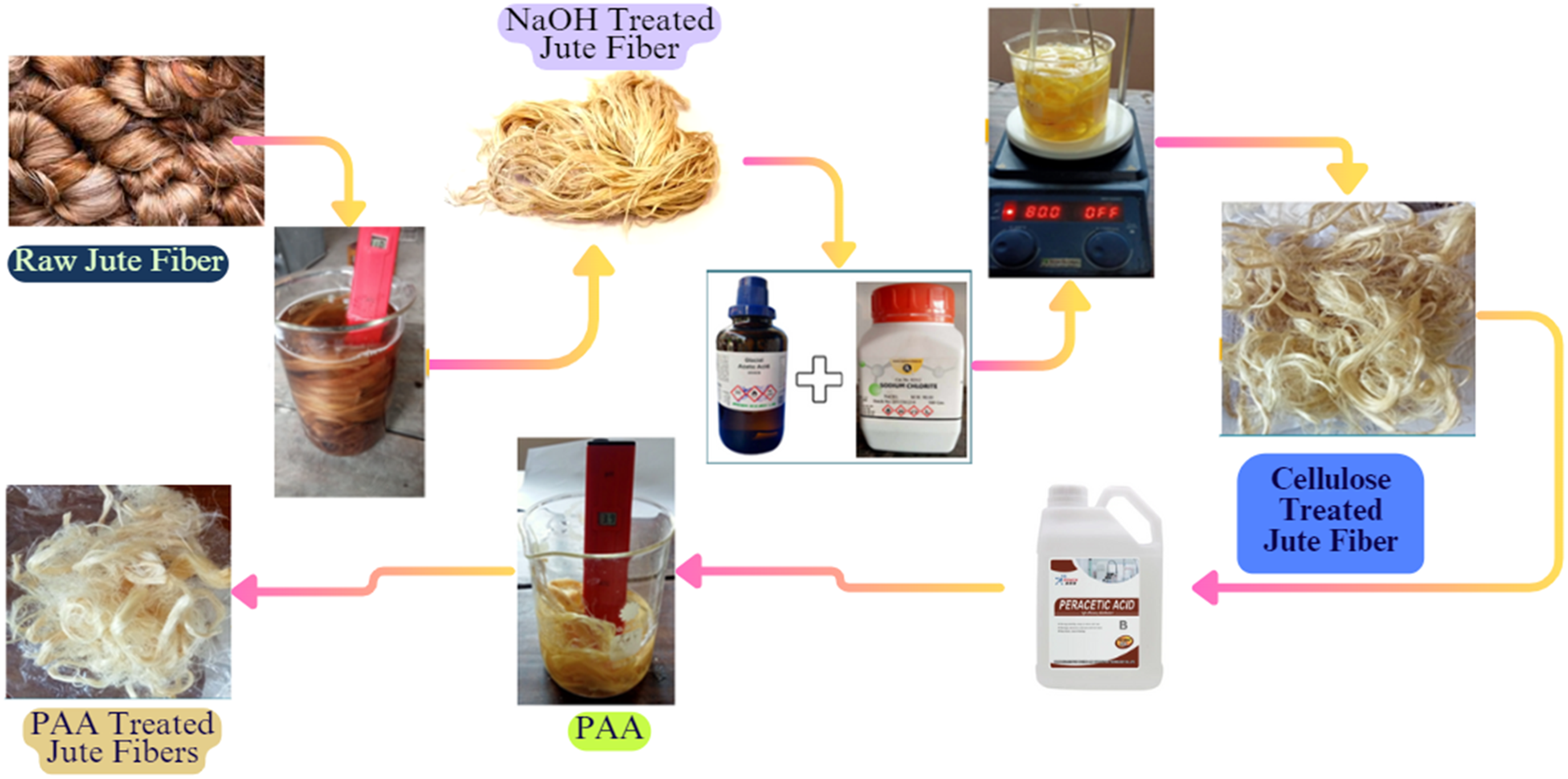
An acid hydrolysis process was employed to eliminate the amorphous region where peracetic acid was used, producing a highly crystalline substance known as microcrystalline cellulose. 28
Measurement technique of crystallinity
Raw, cellulose and peracetic acid treatment sixteen samples were generated using to assess crystallinity, with changes in time, temperature, and chemical concentration. XRD data require several steps to determine crystallinity. Raw, cellulose and sixteen selectively peracetic acid-treated samples are exposed to X-rays and measured for diffracted intensity at 5–40-degree angles from Atomic Energy Research and Establishment, Bangladesh.
When conducting XRD investigations to determine crystallinity, it is customary to compare the diffraction peak intensities of the sample with those of an amorphous baseline or reference standard. It is possible to decide on a sample’s crystallinity using the following equation no. 1 (one):
XRD data may also reveal the orientation of crystallites (texture) in a material, which can reveal its crystalinity. Determine the crystalline area of the sample PAA-JF-X1 where three sharp peaks by integrating the area under its. This can be accomplished with XRD analysis-origin software, which typically includes peak fitting and integration tools which shown as Figure 3(a) where find out 3 sharp peak area A1, A2 and A3 are 28367.06, 67193.36 and 1998.2 respectively and summation of those area is 97558.67. Measurement techniques of (a) selection crystalline area and value with summation (b) and from XRD data.
The Peak Analyzer, a typical tool gadget, can be used in Origin software to measure the amorphous and crystalline areas from X-ray diffraction (XRD) data. This allows the software to automatically calculate the identified 5-40 degree and which area are 144097.02 shown in Figure 3(b).
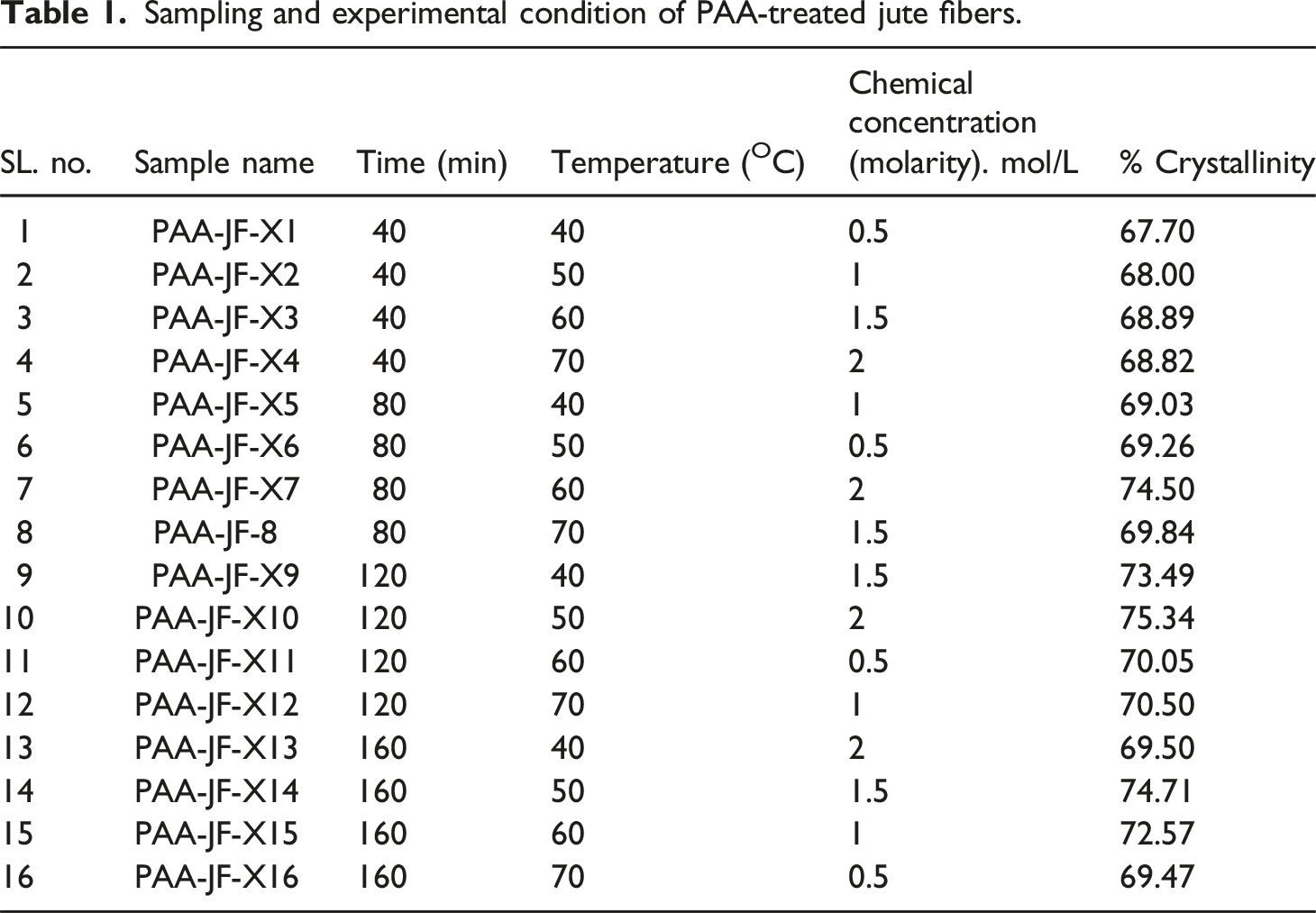
Sampling and experimental condition of PAA-treated jute fibers.
Measurement of other crystallographic parameters
One extremely useful technique for determining the crystallographic properties of materials is X-ray diffraction (XRD). 29 This approach can reveal other factors, such as crystallite size, microstrain, dislocation density, and stress, which can offer valuable insights. Extracting these features from XRD data can be achieved in the following way.
The Scherrer formula was employed to ascertain the crystallite size (D) along the prominent peak
30
:
The symbol λ represents the wavelength of X-ray diffraction, β represents the whole width at half maximum, and θ represents the diffraction angle.
The equation was used to determine the strain values (ε) of the jute fibers treated with PAA:
The subsequent equation was utilized to calculate the dislocation density (δ)
31
:
The following equation was utilized to compute the stress (σ) generated in the treated jute fibers
32
:
Results and discussion
Crystallographic analysis of raw, cellulose and PAA-treated jute fibers
The X-ray diffraction curves of raw, cellulose andvarious time, temperature, and chemical concentration conditions of PAA-treated jute fibers are shown in Figures 3–6. X-ray diffraction curve of (a) raw (b) cellulose jute fibers. X-ray diffraction curve (a) 40oC and 0.5 molarity (b) 50oC and one molarity (c) 60oC and 1.5 molarity (d) 70oC and two moralitiesat 40 min PAA treated jute fibers. X-ray diffraction curve (a) 40oC and one molarity, (b) 50oC and 0.5 molarity, (c) 60 and two molarity, (d) 70oC and 1.5 molarity at 80 min of PAA treated jute fibers.

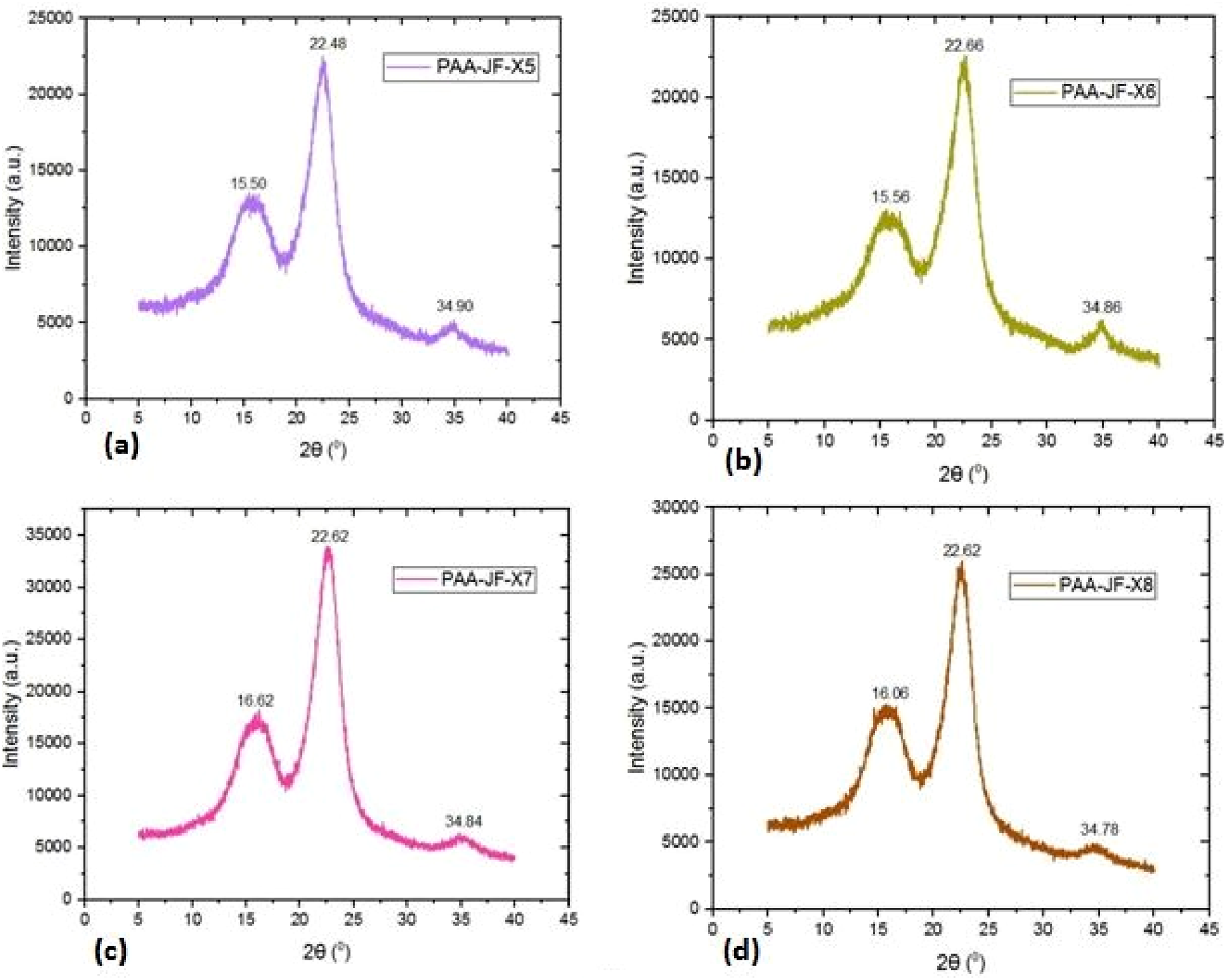
Figure 4 shows the results of the X-ray diffraction (XRD) study of raw and cellulose jute fibers. Equation (1) provides the percentage crystallinity, which is 60.30% and 65.95%, respectively. Figures 5–8 show notable variations in crystallinity for PAA-treated jute fibers (PAA-JF) under 16 experimental circumstances (designated as X1-X16). Using the crystallinity measuring technique with equation (1), the PAA-JF-X10 sample has the highest % of crystallinity among these. Samples PAA-JF-X9 and PAA-JF-X15 also exhibit significant crystallinity, albeit at a smaller level, while PAA-JF-X7 and PAA-JF-X14 follow suit with equal crystallinity values, occupying the second-highest rank. On the other hand, samples PAA-JF-X2, PAA-JF-X3, PAA-JF-X4, and PAA-JF-X5 show similar low values, while the PAA-JF-X1 sample has the least one. The experimental setup information for PAA-treated fibers is summarized in Table 1. X-ray diffraction curve (a) 40oC and 1.5 molarity, (b) 50oC and two molarity, (c) 60oC and 0.5 molarity, (d) 70oC and one molarity at 120 min PAA treated jute fibers. X-ray diffraction curve (a) 40oC and two molarity (b) 50oC and 1.5 molarity (c) 60oC and one molarity (d) 70oC and 0.5 molarity at 160 min PAA treated jute fibers.
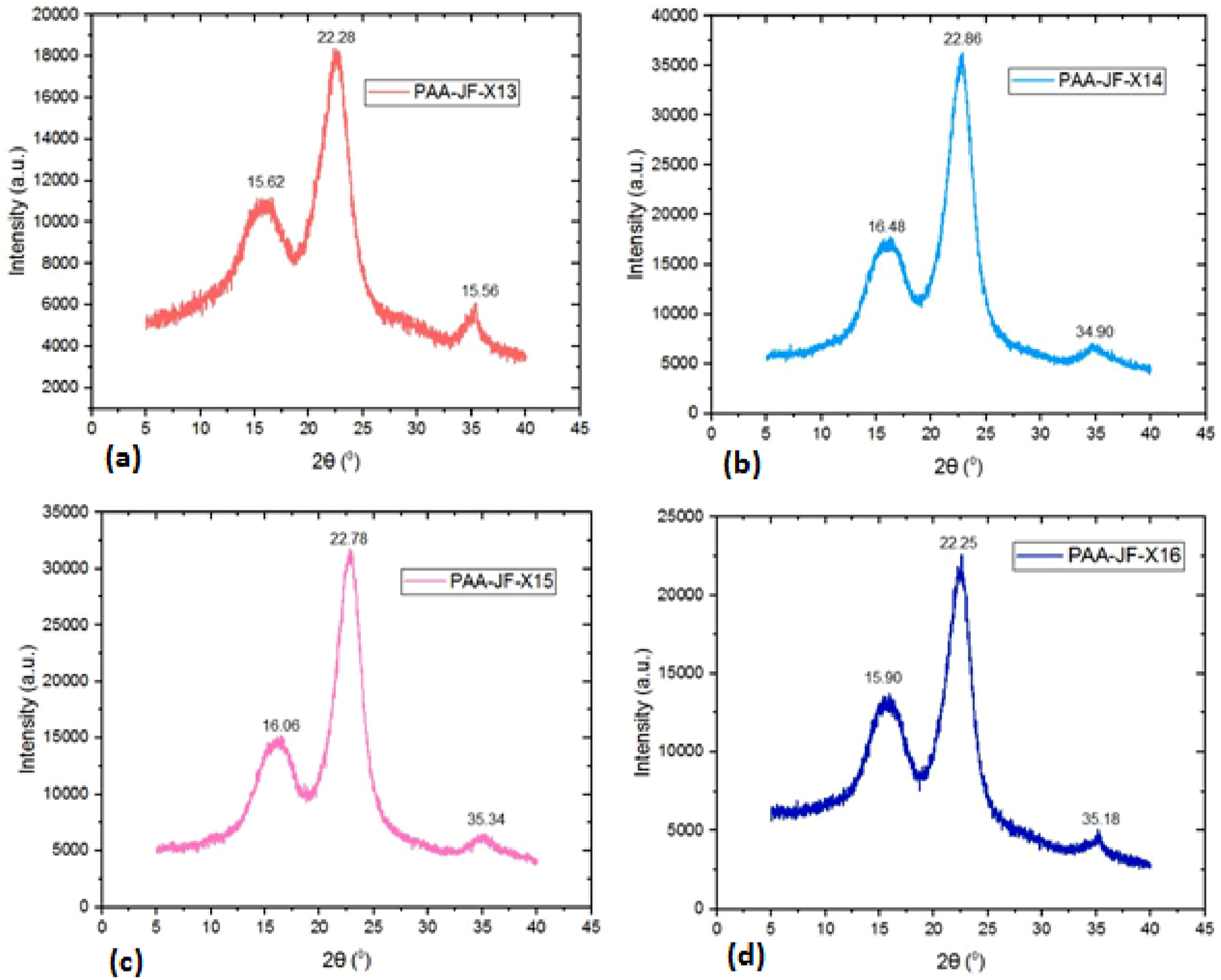
Crystallographic data of PAA-treated jute fibers.

The Tables 1 and 2 provide measured crystallinity and crystallographic value ofPAA-treated jute fibers respectively. A high crystallinity in jute fiber signifies that the fiber has highly organized and densely packed molecular structures. A high degree of crystallinity often enhances strength, stiffness, and thermal stability, although may diminish flexibility and water absorption. The pretreatment of cellulose using chemical methods to diminish its crystallinity influences its solubility and reactivity. 33 The elevated crystallinity of jute fiber mainly results from the inherent cellulose molecules that create highly organized, densely packed structures. The ratio of cellulose to other constituents, such as hemicellulose and lignin, affects the fiber’s overall crystallinity and mechanical characteristics.
Research often adjusts molarity to optimize reaction conditions and control variables and enhance experimental accuracy. Molarity is a concept that relates three components concerning solutions: two independent extensive variables, n and V, and one dependent intensive variable, M.The predominant unit for molarity is moles per liter, denoted by the symbols mol/L.Understanding molarity requires the precise relationship among these three variables, necessitating reasoning that involves the regulation and proportionality of the variables within a chemical context foreign to the students. The mathematical expression is M = n/V. 34 Furthermore, in this study, we modify the molarity to improve reaction efficiency, ensure the solubility or stability of compounds, or mitigate environmental factors such as temperature. These modifications ensure experimental reliability and validity.
FTIR analysis
Cellulose, hemicellulose, and lignin are the primary components of jute fiber. The FTIR analysis of raw, cellulose and PAA treated jute fiber are shown in Figure 9. In general, FTIR spectra of jute fibers can be separated into two characteristic regions: The vibrational modes in the 3500 to 2800 cm−1 range are usually connected with the O-H and C-H stretching modes. In the 2000 to 800 cm−1 region, cellulose, hemicellulose, and lignin compounds exhibit many peaks.
35
In bothtreated and untreated jute, the following wave numbers and corresponding functional groups were found. The strong and broad peak at 3319 cm−1 corresponds to –OH stretching vibration. This extensive peak signifies the hydroxyl (–OH) groups present in cellulose and hemicellulose.
36
The peak at 1634 cm⁻1 in the FTIR spectrum of jute fiber is typically associated with the H-O-H stretching vibration of absorbed water in carbohydrate, as well as the C=O stretching vibrations of carbonyl groups present in lignin or hemicelluloses.
37
FTIR analysis curve of raw, cellulose and 120 min, 50oC and two molarity (PAA-JF-X10) peracetic acid treated jute fibers.
This absorption band at 1639 cm⁻1 shows a noticeable difference in intensity between raw and treated samples. This suggests that the amount of lignin and hemicellulose in the treated samples has decreased compared to the raw samples. The reduction in these components is likely due to chemical treatments that have selectively removed or altered the lignin and hemicellulose content. The wavenumber 1012
Thermo gravimetric analysis (TGA)
Thermo gravimetric analysis (TGA), which provides important information on the crystalline structure of jute fibers, is an important technique for evaluating their heat stability and decomposition behavior. The TGA results of raw, cellulose and peracetic acid (PAA) treated jute fibers are shown in Figure 10, which also highlights how chemical treatments affect thermal performance and crystallinity. Thermo gravimetric analysis curve of raw, cellulose and peracetic acid-treated jute fibers.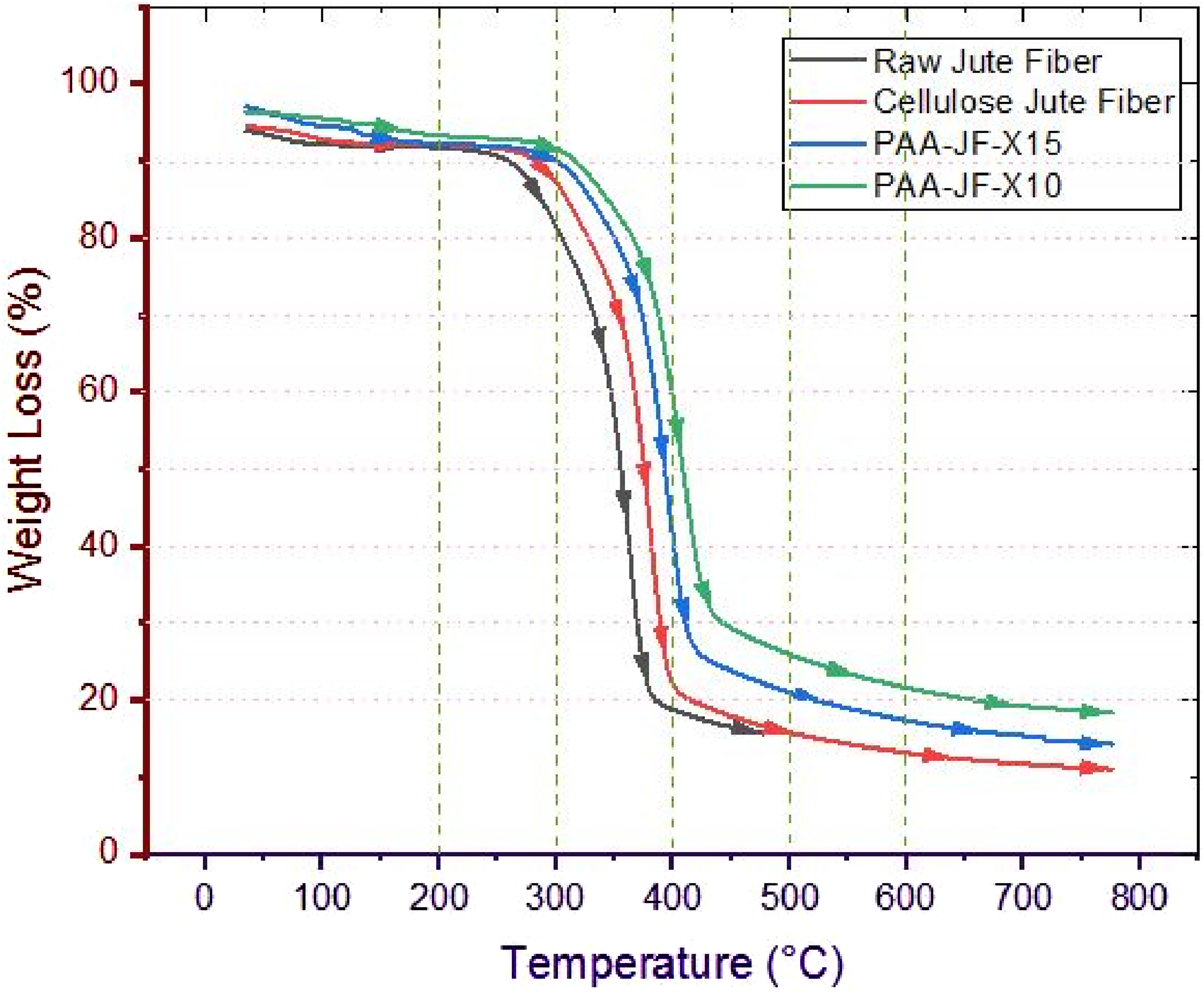
The thermo gravimetric analysis (TGA) of PAA-treated jute fibers demonstrates delayed and sharper weight loss transitions, indicating enhanced crystalinity compared to raw and cellulose treated fibers. Superior thermal stability was demonstrated by samples PAA-JF-X10 then PAA-JF-X15, with PAA-JF-X10 displaying the highest crystalinity and superior thermal resistance. The elimination of hydrophilic hemicelluloses and lignin, which lowers moisture content at low temperatures (∼100°C), and improves structural integrity are the reasons for these samples’ better thermal performance. All jute fibers lose weight first as a result of moisture evaporation, and then they significantly degrade between 200 and 350°C, which is when hemicellulose, lignin, and amorphous cellulose break down thermally. Lower thermal stability is a result of the existence of amorphous components, highlighting the importance of crystalinity in enhancing the thermal behavior of fibers treated with PAA.
According to comparative studies, fibers treated with PAA had higher levels of crystalinity, which is associated with better thermal stability. This enhancement demonstrates how well chemical changes can restructure the molecular structure of jute fibers, increasing their suitability for industrial uses where great thermal resilience is required. The interplay between crystalinity and thermal stability highlighted by TGA reinforces the potential of treated jute fibers for advanced material applications.
Morphology of jute fibers
The morphology of raw, cellulose-treated, and peracetic acid (PAA)-treated jute fibers was studied using scanning electron microscopy (SEM), as shown in Figure 11. The SEM images show that the fibers’ cross-sectional geometry is imbalanced, with dimensions that fluctuate along their length. This structural irregularity represents the inherent variability of jute fibers. The research also identifies a core lamella rich in lignin, which bonds the individual fibers and provides structural stability. Furthermore, the intermediate lamella contains different amounts of cellulose, hemicellulose, and lignin, which affects the mechanical and chemical properties of the fibers. The morphological differences between raw, cellulose-treated, and PAA-treated fibers show the impact of chemical treatments on the fiber’s structural composition and prospective applications. Cross sectional SEM image (a) raw (b) cellulose (c) PAA-JF-X1 (d) PAA-JF-X5 (e) PAA-JF-X10 and (f) PAA-JF-X15 of jute fiber.
A lesser concentration of microspores in the cross-sectional structure frequently indicates undeveloped fibers, and the surface morphology of jute fibers exhibits an uneven texture with irregular microholes. The existence of large amorphous regions in raw fibers results in poor crystallinity, whereas cellulose-treated fibers have better crystallinity because non-cellulosic components are eliminated. The PAA-JF-X10 sample outperforms samples PAA-JF-X1, PAA-JF-X5, and PAA-JF-X15 in terms of crystallinity enhancement among the peracetic acid-treated fibers. The best treatment conditions, which successfully eliminate amorphous components without sacrificing the structural integrity of cellulose, are responsible for this improvement. Even though longer treatment times increase crystallinity, they also increase the risk of cellulose breakdown, which highlights how crucial it is to precisely regulate treatment parameters.
The increasing elimination of non-cellulosic components during treatment results in smoother fiber surfaces and better structural organization. Enhanced exposure and alignment of cellulose microfibrils help to develop a more crystalline structure, as indicated by sharper and more prominent crystalline peaks in PAA-treated fiber XRD patterns. The crystallinity index, which measures the degree of order in cellulose chains, emphasizes the importance of optimum treatment settings in increasing crystallinity. However, over-treatment can cause surface etching and cellulose degradation, resulting in a loss in overall crystallinity. This balancing emphasizes the need of regulated treatment conditions in maximizing crystalline organization while maintaining fiber integrity.
Effect of time, temperature, and chemical concentration on crystallinity of jute fibers
Figure 12 illustrates the % of crystallinity of jute fibers at sixteen experimental conditions. The figure shows that the sample PAA-JF-X10 shows the highest % of crystallinity (75.34%). Following that, the samples PAA-JF-X7 and PAA-JF-X14 occupy the second highest value and show almost similar values of % crystallinity; these are 74.5% and 74.71%, respectively. Additionally, it is seen that the % of crystallinity of samples PAA-JF-X9 and PAA-JF-X15 is also high but less than the samples mentioned previously. On the other hand, sample PAA-JF-X1 depicts the most negligible value of % of crystallinity (67.70%), and similarly, samples PAA-JF-X2, PAA-JF-X3, PAA-JF-X4, and PAA-JF-X5 show almost similar values ( around 68%). % of crystallinity various experimental conditions peracetic acid treated jute fibers.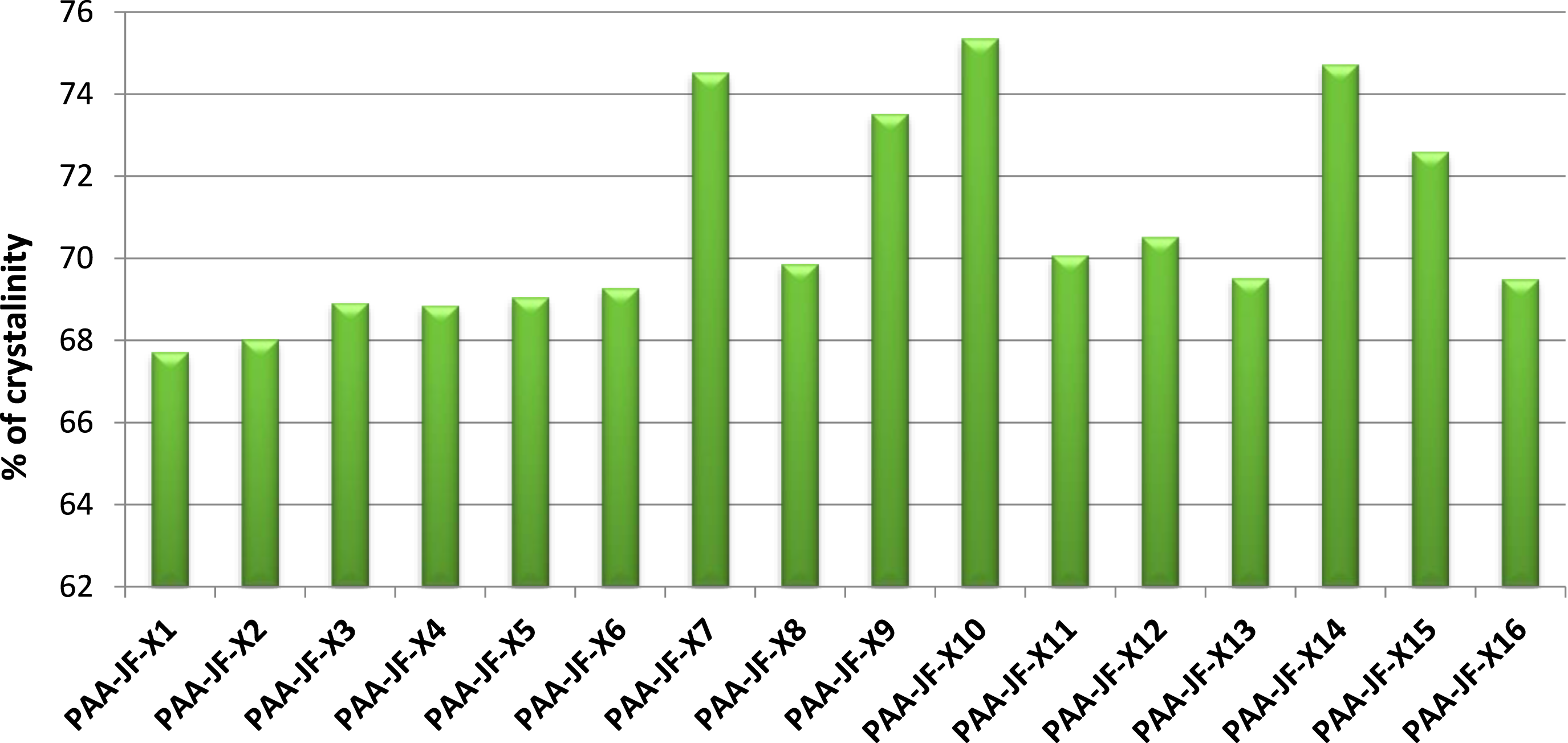
Now, from Table 1 and it is seen that the chemical concentration values of the peracetic acid solution used for samples PAA-JF-X10, PAA-JF-X7, and PAA-JF-X14 are 2M, 2M, and 1.5 M, respectively. Meanwhile, the values for samples PAA-JF-X1 and PAA-JF-X2 are 0.5 M and 1 M, respectively. Herein, it can be concluded that the crystallinity increases with increasing the concentration of peracetic acid solution. However, there are other samples, for instance, PAA-JF-X3, PAA-JF-X4, PAA-JF-X8, and PAA-JF-X13, which also have high concentration values but low % of crystallinity. Therefore, it is clear that some other factors influence the % of crystallinity. Time and temperature play a crucial role in that case. The samples PAA-JF-X10 and PAA-JF-X4 were treated as a solution of the same chemical concentration (2 M), but the temperature and time used were not the same. Sample PAA-JF-X10 was immersed in the solution at 50°C for 120 min, whereas sample PAA-JF-X10 was submerged for 40 min at 70°C. Consequently, a considerable difference becomes apparent in the % of crystallinity. In short, time, temperature, and chemical concentration significantly affect the crystallinity of jute fibers, which is described in detail in Figures 13–15. Effect of time and temperature on acid hydrolysis (0.5 moralities PAA) treated fibers. Effect of temperature and chemical concentration on acid hydrolysis (40 min) treated jute fibers. Effect of chemical concentration and time on acid hydrolysis (at 40oc) treated jute Fibers.


In this experiment, Figure 13 depicts the time and temperature effects on the crystallinity of PAA-treated jute fibers at a constant chemical concentration (0.5 M). It is seen that the % of crystallinity increases with an increase in treatment time of up to 120 min and then decreases. Between the periods, the crystallinity increased at a specific rate to 80n min; after that, from 80 min to 120 min, the increasing rate increased slightly. A prior study demonstrated that extended treatment durations with PAA can lead to a higher breakdown of hydrogen bonding in cellulose chains. This can enhance the relative number of crystalline regions since amorphous regions are preferentially hydrolyzed or oxidized. However, if the exposure is too lengthy, even crystalline portions can begin to break down. 2
Similarly, in the case of temperature, the % of crystallinity grows as the temperature increases at a specific range (up to 60°C). Likewise, the phenomenon of crystallinity concerning temperature occurred in the same manner as before. Initially, the temperature rose to 50°C, and after that, the rate of increase intensified. For subsequent increases in temperature, it shows a decreasing trend of crystallinity. Previous studies demonstrate that elevated temperatures can promote the elimination of amorphous regions, potentially boosting the crystallinity index. However, high temperatures and prolonged treatment times could also damage crystalline areas, diminishing total crystallinity.14,39
Figure 14 depicts the correlation between crystallinity temperature and chemical concentration, with a set treatment time of 40 min. The graph shows that an increase in chemical concentration leads to a corresponding increase in the % of crystallinity. This is because higher quantities of PAA can lead to increased oxidation with a more aggressive nature. This method continues to function effectively on shapeless components; however, if it needs to be adequately controlled, it may also impact sections with a defined crystalline structure. Excessive exposure can cause degradation of cellulose chains, resulting in a decrease in overall crystallinity, even in crystalline parts.
Furthermore, the effect becomes more intense when concentrations and temperatures increase, leading to significant changes in the amorphous and crystalline areas. Nevertheless, if the temperature is not constant, the outcome of crystalinity may differ.
Figure 15 demonstrates the variation in crystallinity over time and chemical concentration while maintaining a constant temperature of 40°C. The graphic shows an initial increase in crystallinity up to a particular range, followed by a peak, and then a subsequent decline as both chemical concentration and time increase. Nevertheless, the outcome may differ if the constant temperature is altered. The figure shows that as the chemical concentration exceeds 1.5 M, there is a discernible drop in crystallinity. However, according to Table 1, sample PAA-JF-X10 exhibits the highest level of crystallinity while having the maximum chemical concentration of 2 M. The cause of this phenomenon is the temperature. The temperature for sample PAA-JF-X10 is 50°C, but in Figure 12, it is 40°C. Thus, it may be inferred that an excessive concentration of chemicals and extended treatment can destroy crystallinity.
Crystallographic parameters in relation of % crystallinity of jute fibers
The relationship between crystallinity and crystallite size is complex and can be affected by various factors, including the material’s composition, manufacturing method, and processing conditions.19,40 Figure 16 reveals that crystalinity typically increases as the size of crystallites reduces. Crystallinity is enhanced by reducing the size of crystallites, increasing the ratio of surface area to volume, and increasing surface energy. Crystallinity increases as more atoms or molecules arrange to save energy. Smaller crystallites may hinder atom or molecule mobility due to higher surface-to-volume ratios. It prevents amorphous regions and helps atoms and molecules form lattices.
41
Due to lattice structure or grain border defects, smaller crystallites can increase strain. Crystallite atoms or molecules may reorganize into a more structured crystalline structure to reduce strain and increase crystallinity. Depending on material and experimental settings, this relationship may need to be more accurate. Crystallinity and crystallite size characterization may impact the connection. % of crystallinity with (a) average crystal size and microstrain and (b) average dislocation density and average stress for PAA-treated jute fibers.
The fiber’s structure and properties complicate the proportions of crystallinity in jute fiber and microstrain rise. Microstrain increases fiber-crystalline nucleation sites. Strain regions increase crystallization and crystallinity by organizing atoms or molecules into crystalline structures. Structural disorder increases with microstrain on the fiber’s crystal lattice. Contrary to popular belief, microstrain-induced disorder can increase crystallinity by creating smaller, more frequent crystalline domains. Crystalline fibers are more energetically favorable due to higher microstrain, which may affect thermodynamic stability. Microstrain-induced stress in the fiber may rearrange atoms or molecules into a more ordered crystalline lattice, enhancing crystallinity. Crystallization grain growth is affected by microstrain. Excessive microstrain can hinder the development of grains, forming numerous smaller crystalline domains within the fiber. 40 With lower average microstrain, crystallinity may rise (Figure 16). The relationship between crystallinity and microstrain depends on the jute fiber’s quality, processing, and testing methods. More research and testing are needed to understand how these factors affect jute fiber.
Context is vital since various parameters affect jute fiber crystallinity and dislocation density. Crystallinity implies polymer chain order. Reduced dislocation density indicates a less disordered crystal lattice. Dislocations in a material’s crystal structure can disrupt crystalline growth. As dislocation density lowers, crystal lattice organization improves. Jute fibers are cellulose; a lower dislocation density may suggest better cellulose chain alignment and crystalline structure. Crystallinity can increase with polymer chain order. How manufacturing, thermal history, and additives affect crystallinity and dislocation density is complex. Fiber crystallinity and structure are assessed using X-ray diffraction and infrared spectroscopy. Jute fibers’ behavior relies on treatment and conditions. 41 Material crystal lattices have dislocations. Lower dislocation density suggests better crystal lattice organization. Organized polymer chains form crystalline regions in cellulose, an essential component of PAA-treated jute fibers. Reduced dislocation density minimizes cellulose chain alignment disturbances. Chain alignment improves fiber organization and crystallineness. Increased crystallinity indicates crystalline structure. Highly structured polymer chains promote crystallinity.21–23,41 Figure 16 shows that overall, the experimental case decreased dislocation density and organized jute fiber polymer chains, enhancing crystallinity. XRD and infrared spectroscopy measure fiber crystallinity. These approaches can show structural changes in jute fibers as dislocation density decreases, suggesting fewer dislocations increase crystallinity.
The compressive stress in jute fibers decreases because of the improved structural organization seen in the crystalline areas. 42 As the level of crystallinity increases, the polymer chains of jute fibers line with greater accuracy, leading to the formation of a well-organized and compact structure. This improved structure arrangement enables efficient dispersion and the ability to withstand the compression of the fibers. The enhanced load-bearing capability and reduced susceptibility to deformation of the crystalline areas improve stress distribution, decreasing compressive stress. Higher crystallinity in jute fibers leads to a more robust and organized internal structure, reducing compressive stress (refer to Figure 16) by improving the fibers’ capacity to endure and distribute compressive stresses.
Conclusion
This study examined the influence of PAA treatment parameters such as duration, temperature, and chemical concentration on the crystallinity of jute fibers. The best result has been obtained when the chemical concentration was 2 M, time was 120 min and temperature was 50–60°C. At this condition, the maximum crystallinity was 75.34%. Under these circumstances, the PAA treatment efficiently eliminated noncellulosic constituents while maintaining the structural integrity of the cellulose microfibrils. The improved crystallinity of jute fibers has considerable practical implications. Enhanced crystallinity augments mechanical strength, thermal stability, and moisture resistance, rendering the fibers more appropriate for advanced composite materials in industrial applications, including textiles, packaging, and sustainable composites. The experimental parameters were limited to defined limits of duration, temperature, and chemical concentration, allowing for the possibility of investigating wider or more severe situations. Moreover, the possible environmental repercussions of PAA therapy and its enduring effects on fiber characteristics necessitate additional research.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

