Abstract
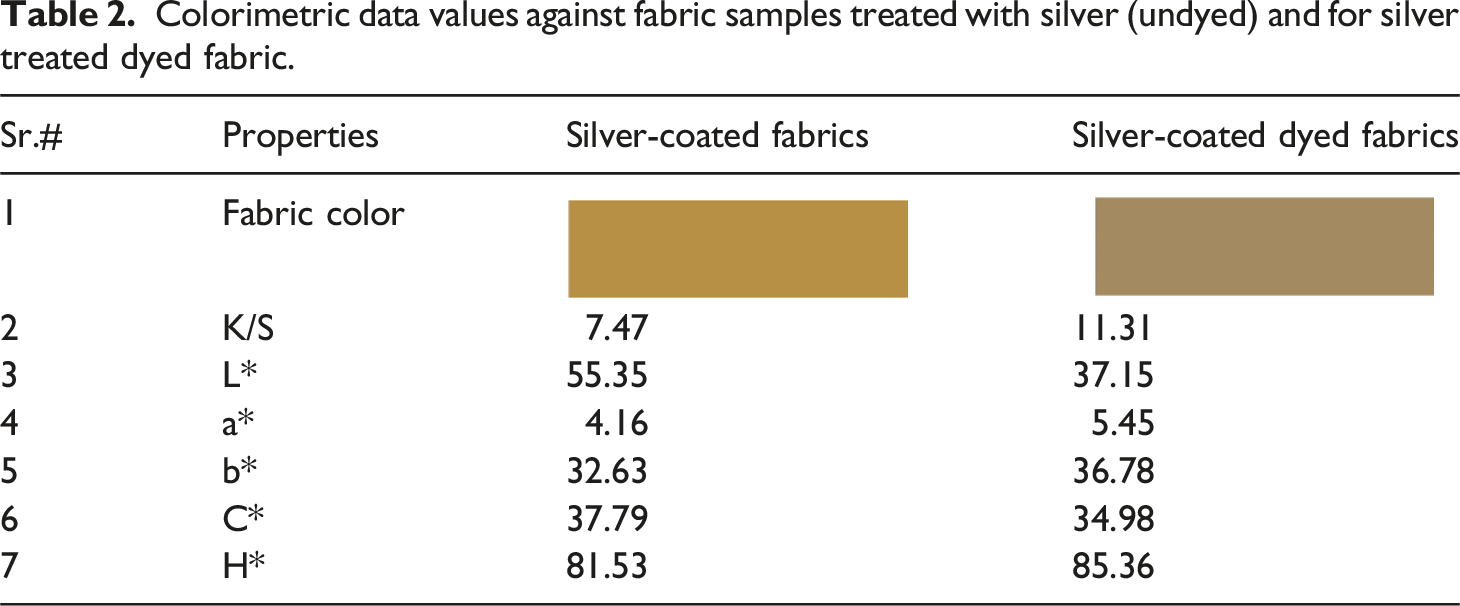
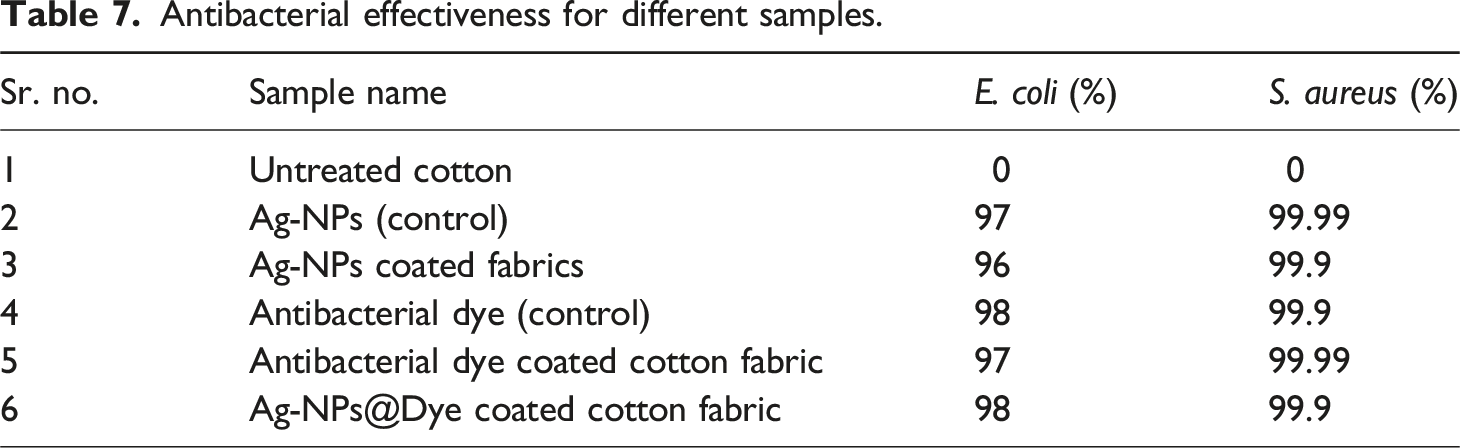
Currently the stretchable knitted fabrics are extensively used as compression bandages in clinics. In present study, we have fabricated antibacterial stretchable medicated textiles (bandages, compression bandages). The study was composed of three parts. At first, the versatile one-pot green synthesis approach was used to form a stable and concentrated solution of silver nanoparticles (Ag-NPs) through the self-assembly of tannic acid (capping agent). Tannic acid (hygienic capping agent) serving as a green dispersant and reductant, also acts as a self-assembly agent and while remaining free of hazardous chemicals. Subsequently, the synthesized Ag-NPs were coated over the cotton textile substrate. The surface morphologies of the metallic nanoparticles coated textiles were evaluated by the use of different characterization techniques such as Scanning Electron Microscopy (SEM), Dynamic Light Scattering (DLS), Fourier Transform Infrared Spectroscopy (FTIR), Energy Dispersive X-Ray Spectroscopy (EDS), and X-Ray Diffraction (XRD). In the second step, a natural antibacterial dye was extracted from pomegranate peel and exhaust dyeing process was employed over the Ag-NPs coated fabric. The color measurement parameters of the dyed substrate were evaluated using CILAB (L*, a*, b*, C, h, and K/S). The K/S values for dyed sample was relatively high (11.31) than for undyed fabric (7.47), indicating that dyeing has altered the light-toned silver coated fabric to a relatively dark-colored fabric. While, the levelness L* value of the dyed fabric was relatively low (37.15) than undyed fabric (55.35), indicating that the dyed sample has a darker shade (depth) compared to the undyed sample. Additionally, the antipathogenic performance, including antibacterial, antiviral, and antifungal effects, were determined for all coated fabrics. The silver coated fabric with post dyeing process showed extraordinary antibacterial properties and killed 99.99% of bacteria (
Introduction
Plane cotton fabrics are generally renowned for having outstanding attributes in medical textiles due to their novel properties, such as sharp absorbency, good breathability, and comfortable wear.
1
However, they may be limited when compression and bioactivity are both needed. In the case of compression bandages, the compression and antibacterial properties are compulsory for wounded skin. The knitted fabrics (stretchable) are the key component for the development of compression bandages. Textile based materials which are used in hospitals or clinics must meet high standards and essential requirements including properties such as non-toxicity, Hypoallergenic, antipathogenic, and anti-inflammatory.
2
Because of the expanding biological risks to global security, it is essential to improve antimicrobial fabrics for hygienic practices and precautionary measures to prevent cross-infections particularly in hospitals. To induce the antibacterial properties in textiles, various kinds of agents, metal particles and treatments are used.
3
Metallic nanoparticles have recently been used to functionalize textiles in an attempt to slow down the disease transmission and microbial activity. Different types of metallic nanoparticles such as Cu, ZnO, TiO2, Ag, Cu2O, CuO, and MgO and antibacterial finishing for healthcare have been widely synthesized.4,5 Among the above-mentioned nanoparticles, the most significant NPs with broad spectrum antibacterial properties are silver nanoparticles.6–11 Multiple technologies such as photochemical methods, chemical reduction, laser ablation, gamma irradiation, as well as different methods of biological synthesis, among others, have been used for the synthesis of silver nanoparticles.12,13 The green synthesis method provides less toxic and more stable dispersions of nanoparticles without the utilization of harmful chemicals. Additionally, the green synthesis approach also eliminates the need of energy input such as high temperature or pressure.
14
The stability of nanoparticles is quite significant and plays a unique role in their antibacterial activity.15,16 Recently, Shaukat et al performed a research work to fabricate the bioactive textiles (cotton woven) by using green synthesis technique and bio dispersants to stabilize the silver nanoparticles. The dispersed silver particles showed improved performance against hospital acquired pathogens.
17
Moreover, the use of green capping agents also plays a significance role to maintain the diversity among particles. During the last few years, the researchers have been trying to prepare the tannic acid-mediated nanoparticles as well as their potential implement in the different fields of biomedical. Azam et al, successfully synthesized approximately spherical and highly concentrated (2281 ppm) Ag-NPs, capped with poly-tannic acid (AgNPs@PTA). The average particle size of AgNPs@PTA was note to be 10 nm. In normal conditions, the colloidal dispersion of the formed NPs was found to be quite stable for about more than 1 year.
18
Tannic acid consists of both catechol and galloyl groups, as they are quite well known for their metal chelation and binding properties of material surface. Therefore, they are very important in the process of green synthesis of nanoparticles in normal conditions due to their dual functionality in terms of reducing and capping agents.19–22 Several research studies have investigated the use of silver nanoparticle-coated textiles in medicated textiles.23,24 Ali et al. presented a coating of cotton fabric with silver nanoparticles to produce multifunctional textiles. The developed fabrics were highly effective towards pathogenic microbes
Thus, considering the aforementioned issues, the present study suggests a novel strategy for the fabrication of sustainable green synthesized silver-coated antimicrobial cotton fabrics (keep view on ecological and environmental factors) with an excellent aesthetic appearance. As far as we know, there are limited research works available on the development of highly antipathogenic textiles that combine aesthetic appearance with minimal toxicity. The study was composed of three parts. At first, a one-pot method was used to make a concentrated and stable colloidal solution of silver nanoparticles (Ag NPs) through the self-assembly of tannic acid, without the use of additional harmful chemicals. Tannic acid (polyphenol that is derived from plants) was utilized as a stabilizing and reducing agent for silver nitrate in a slightly alkaline state. In the second step, the natural antibacterial dye was extracted from pomegranate peel (
Experimental section
Materials
Tannic acid (C76H52O46), sodium hydroxide (NaOH), and silver nitrate (AgNO3), were obtained from Sigma-Aldrich. All these chemicals were kept in the desiccator and utilized without any additional purification. Cotton fabric with plane weave having GSM of 150 was utilized and obtained from National Textile University, Faisalabad. Deionized water was used during all synthesis procedures. Pomegranate peels were collected from a local juice corner of City Faisalabad.
Synthesis of silver nanocomposites
To conduct the manufacturing experiment for silver nanocomposites, 0.2 M NaOH, 1 mM tannic acid, and 0.147 M AgNO3 salt solution were prepared in distilled water and kept at room temperature. The homogeneous mixture of tannic acid (35 mL) and AgNO3 salt (10 mL) was prepared by mixing both solutions on a magnetic stirrer (500 rpm) for about 5 minutes at room temperature. The solution pH was maintained between 8 and 9 by slowly adding NaOH (1.35 mL) solution. Then, the reaction mass was shifted into the beaker followed by magnetic stirring at room temperature for half an hour. After that, the product was achieved by centrifugal separation (10–15 minutes at 10,000 rpm) and washed for the removal of impurities. The synthesized particles were initially subjected to two sequential washes with deionized water, followed by a single wash using a methanol solution. The general schematic showing the complete process involved in the fabrication of core@shell silver particles is given in Figure 1. Subsequently, the above-formed particles with different concentrations (1, 2, and 3 mg) in 100 mL of deionized water were applied on cotton fabric (5 × 5 cm2) by dip-coating method. The treated fabric was then subjected to drying in a vacuum oven for about 1 h at 60°C. Schematic illustration for green synthesized Ag-NPs.
Extraction of natural dye
At first, the pomegranate peels were cut into flakes and washed along with water to remove dirt and impurities. The washed peel flakes were then dried under shade and ground to prepare a fine powder of pomegranate peels. In the next step, the dye was extracted from the as-prepared peel powder. For this, 4 g of peel powder was added in a round bottom flask previously containing 100 mL of distilled water. The reaction mass was refluxed for 30 minutes at 80°C. After 30 minutes, the refluxed reaction mass was filtered using cheesecloth. The filtrate i.e., dye extract was prepared to use for the process of dyeing of fabric.
Dyeing the fabric with extracted dye
The extracted antimicrobial dye was applied on silver particles coated fabrics by exhaust dyeing procedure. The dyeing was done in H-T (high temperature) dyeing unit. The material-to-liquor (M: L) ratio was adjusted at 1:40. Dyeing of the textile was initiated at a standard room temperature of 20 ± 2°C, which was slowly increased to 60°C. Alkali (1 g/L NaOH) and Electrolyte (Na2SO4, 40 g/L) were added step by step in the dye bath at varying intervals of periods. Hence the further dyeing of the textile was carried out for 70 minutes. Subsequently, the dyed textile was rinsed with tap water. The deposition of silver particles followed by dyeing on the surface of cotton fabric is illustrated in Figure 2. The schematic diagram illustrating the process of silver nanoparticles coating over the knitted cotton structure and subsequently dying process.
DOI for the dyed and undyed antipathogenic textile samples.

Characterization of silver particles coated dyed fabrics
Surface analysis
The surface morphology of the silver nanoparticle (Ag-NPs)-treated cotton fabrics, and dyed samples, was analyzed using a scanning electron microscope (SEM). The examination was done with an FEI Quanta 50 SEM instrument operating at an accelerated voltage of 10 kV. X-ray diffraction (XRD) analysis was performed by utilizing a diffractometer equipped with a conventional Cu Kα₁ radiation source (λ = 1.54 Å) under operating conditions of 40 kV and 30 mA. The diffraction patterns were recorded within the 2θ range of 10° to 80°. Additionally, the dynamic light scattering (DLS) measurements were performed by the use of a Malvern Zetasizer, based on the principles of DLS theory, to determine particle size distribution.
Dye exhaustion, fixation, and total fixation measurement
The method provided in ISO 105-C06 was used to calculate the rates of exhaustion, fixation, and total fixation for the modified dye. The following equations were used:


Fastness properties
The rubbing, washing, and light fastness properties for the developed fabric samples were also assessed. The examination of rubbing fastness, washing fastness, and lightfastness was conducted in accordance with ISO 105-X12, ISO 105-C06, and ISO 105-B02 standards, respectively.
Colorimetric data measurement
CIELAB (a*, b*, h*, L*, C*) data and color strength (K/S) for all the developed fabrics were calculated using a reflectance spectrophotometer ISO 105-A05, (equipped with D65 light). Whereas negative values of a* show the extent of the dye’s greenness and positive values its redness, positive and negative values of b* reflect the degree of the blueness and yellowness shade, respectively. C* stands for chroma, L* for brightness (numbers between 0 and 100, where 100 indicates the colour white and 0 for pure black), and h* for hue angle (00–3600). K/S values) of all developed samples were evaluate using the Kubelka-Munk method (equation).
Analyzing the levelness of applied dye
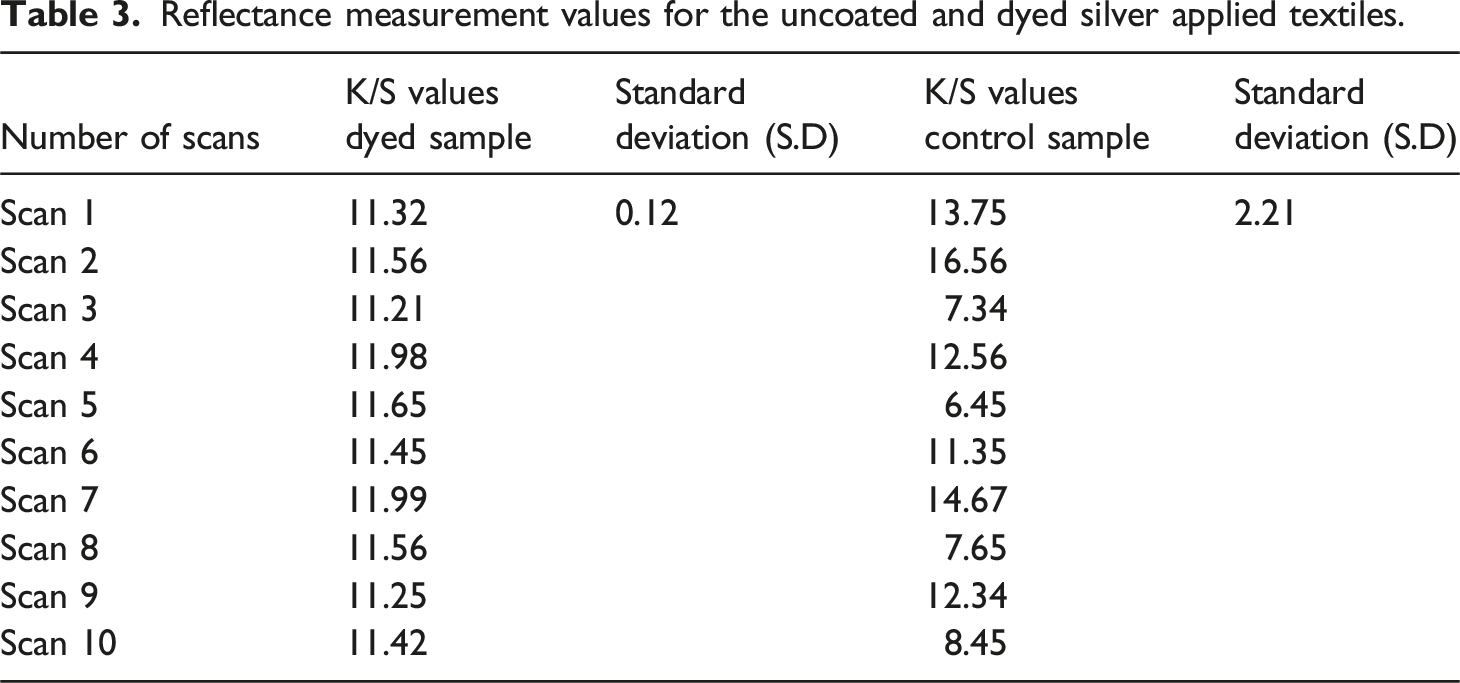
The treated dyed fabrics were assessed for dye levelness using both a sensory and quantitative techniques, following the guidelines given in ISO 105-A01. The dyed fabric was carefully examined for the visual inspection from different angles, and grading from 1 to 5 was done (5 rating is for excellent dye levelness and 1 rating is for poor levelness). For more accurate results, the degree of levelness was evaluated by an objective process. To do this, a reflectance spectrophotometer was used to identify the fabric at 12 different locations, and K/S values were computed. The estimated standard error for each K/S observation was used to evaluate the dye levelness. Excellent dye levelness is indicated by values in the range of 0.20, whereas values >1.0 represent poor dye levelness. However, A higher dye levelness is connected with lower standard deviation (SD) values. The S D for the observed K/S values was calculated by using equation (5).
Antimicrobial properties
The antibacterial activity of developed fabric samples was tested both qualitatively using AATCC 147 and quantitatively by the ISO 20743:2013.
Zone of inhibition test (qualitative measurements)
Bacterial strain preparation
Masaryk University Brno, in the Czech Republic, provided the gram-positive
Measurement of the inhibition zone
A comprehensive technique was applied to access the antimicrobial effectiveness of dyed (treated) and undyed (control) substrate samples. The samples with dimensions 3 × 3 mm squares were placed in infected (cultured) agar plates. Subsequently, the agar plates containing fabric samples were incubated for 24 hours at 37°C. The zone of inhibition (ZOI) was calculated by adding the total diameter (mm) of the fabric sample to the ring zone in which no growth of bacteria was noticed. The experiment was repeated three times and average value was calculated. 35
Reduction factor (quantitative test)
The ISO 20743:2013 transfer method was used to perform quantitative antibacterial testing. The control and dyed samples were placed on agar plates, and samples were pushed down with a 200 g weight. Each sample was separated from the agar media and positioned on separate Petri dishes, with the transferred surface facing up. Subsequently, samples were then incubated at standard incubation conditions. The other samples for both control and dyed fabrics were instantly shifted to two separate reagent vessels containing 40 mL of saline solution to determine the bacterial colony count at 0 hr. Eight serial dilutions of the saline were made and poured on agar growth media (as per ISO 20743 method). The same experiment was repeated for samples that were incubated for 24 hours to find out about the bacterial colony count. Equation (6) was employed to estimate the antimicrobial effectiveness (A) for all prepared substrates. Each sample underwent three repetitions to validate the accuracy of the results.
Determining the antifungal activity
The AATCC 100-2004 standard method was used to evaluate the antifungal action treated dyed sample. This test employed 
The fungal spore count for the control fabric (untreated) is denoted by A, whereas B represents the fungal spore count for dyed (treated) fabric samples
Antiviral activity
The applied method called Behrens and Karber’s was employed to calculate the reduction in virus titer from the initial viral concentration of infectivity (107) titer. The cultural medium which is normally termed as Dulbecco’s Modified Eagle Medium (DMEM) with 2% penicillin-streptomycin and 9% fetal-bovine serum (FBS) was used to keep Vero-E6 cultures alive (PSA). The coronavirus infected Vero-E6 cultures. The supernatant was filtered for 30 minutes using moderate centrifugation at 5 to 7°C and 3700 rpm. Vero-E6 cell lines were stored in 96-well plates at a concentration of 2 × 105 and cultured under standard conditions (24 hours at 37°C in 6% CO2) to evaluate virus concentration. Behrens and Kerber’s method was utilized to determine coronavirus titer in cultured cell cultures. Ensuing that, the fabric samples were placed in 20 × 20 mm fabric sample vials. 100 μL of infection rates were passed through the test samples, and the filter was then utilized to clean any extractable viral loads in vessels. The coronavirus was diluted 101 to 108 times. All serial dilutions were injected into Vero-E6 cell cultures and cultured for 3 days at 37°C with 6% CO2. Coronavirus titers in fostered cell lines were determined using the Behrens and Karber method.
Durability of bioactive fabrics
The durability of fabric samples was tested to ensure their serviceability. The fabrics were washed according to ISO 105-C01. All fabrics were washed with standard detergent and washing parameters with a liquor-to-detergent ratio was adjusted to 50:1. Following that, the coated fabric samples were washed for 45 minutes at 45°C, and washing machine adjusted at a speed of 600 rpm. After that, the fabrics were dried under normal atmospheric conditions. The durability against antimicrobial efficacy before and after washing was analyzed.
Evaluation of comfort parameters
The air permeability was assessed as per the method described in International Standards Organization (ISO) 9237 with the help of SDL air permeability tester. The pressure difference between the two surfaces of the material was noted to be 100 Pa. While the Relative water vapor permeability (%) (RWVP) (also known as moisture vapor transmission) was analyzed by using apparatus Permetest. 25
Results and discussion
Peels extract analysis and dyeing reactions with cellulose
The ATR-FTIR spectrum obtained from the pomegranate peel extract displays a broad region at 3300 cm−1 as indicated by Figure 3. This is due to the presence of water and polyhydroxylated aromatic compounds in the sample. The band at wavenumber 2981 cm−1 is obtained because of aromatic ring C–H bonds. Further, the band detected at 1719 cm−1 is accompanying to the C = O group within the 6-member ring lactone structure, which is the property of compounds that belong to ellagic acid and its derivatives. Moreover, the band observed at wavenumber 1603 cm−1 is due to the aromatic ring vibrations and those at 1176 and 1014 cm−1 indicate the presence of C–O linkages. The major absorption band of the extract of pomegranate peels was shown at 272 and 376 nm (Figure 3). It was found that the absorption bands of phenols and phenolic acids are located at 250–290 nm, however, the absorption band of flavones and flavanols are usually detected at 250–350 nm.
36
In fact, it might be possible that the spectrum obtained from pomegranate extract which has been studied in this research, is due to punicalagin, i.e. a known hydrolysable tannin.
34
Although at wavenumber 1344 cm−1, deformation of OH group and C-O stretch combination (phenols) was observed. Similarly, a C-O-C anti-symmetrical stretch was seen at 1071 cm−1, in addition to an out of plane twisted vibrations of CH at 868, 813, and 773 cm−1. This data resembles to the infrared (IR) data of ellagic acid which shows that the extract of pomegranate peel may contain ellagic acid.
34
ATR-FTIR spectrum obtained from the pomegranate peel extract.
During the process of dying color uptake occurs when different components interact in the Potential chemical interactions (a) In between the hydroxyls of cellulose and phenolic groups (present in dye structure), (b) between the carbonyl groups present dye and the hydroxyl groups of cellulose.
Colorimetric data measurement
Colorimetric data values against fabric samples treated with silver (undyed) and for silver treated dyed fabric.
Levelness of silver treated undyed and dyed fabric
Reflectance measurement values for the uncoated and dyed silver applied textiles.
Fastness properties of silver-coated dyed fabric
Exhaustion, fixation, and fastness (washing, rubbing, and light) results of dyed fabric.

Morphology of silver coated knitted dyed cotton fabrics
The average size was measured by DLS methods, which is based on the Brownian motions of the particles. Figure 5(e) demonstrate the average particle size distribution for silver nanoparticles. The average size of silver nanoparticles was noticed about 11.2 ± 1.7 nm. Figure 5(a) shows the nanometer-scale visuals of silver particles before applying to fabric. The spherical features without aggregations were observed. The claim was further justified by Zeta potential and polydispersity index (PDI) values. The recorded value for zeta potential were −51.63 ± 5.19 mV and for PDI were about 0.292. The values ensured that the particles were evenly distributed throughout the suspension and are highly polydisperse.
40
Furthermore, the tannic acid acts as a capping agent and form a layer of poly tannic acid (PTA) during the NPs synthesis process by undergoing a self-polymerization process. Hence high aqueous stability makes them suitable for a homogeneous coating on the cotton fabric. The structural morphologies were also observed using scanning electron microscopy (SEM) on silver particles deposited fabrics before and after dyeing Figure 5(b) and (c). The coating of silver became more consistent and denser as particle concentration increased from 1 mg to 3 mg. This also indicated a greater proclivity for the formation of a percolated channel of silver nanoparticle as their concentration was increased. The dyed silver- treated fabrics demonstrated a highly uniform proportion of nanoparticles on the cotton surface. SEM images showed that silver nanoparticles synthesized under optimal conditions are roughly spherical in character and have good mono-dispersity. EDS analysis was performed to examine the accurate and consistent elemental composition of the synthesized silver nanoparticles, and the findings are shown in Figure 5(d). The major chemical constituents of the synthesized nanoparticles are oxygen (O), silver (Ag), and carbon (C). The peaks around 3 keV are attributed by the surface plasmon resonance of silver, similarly, stabilizing agent poly (tannic acid) cause some more prominent peaks at about 0.6 and 0.8 keV. As a result, it is possible to conclude that Ag is embedded by a thick layer of poly (tannic acid) in the synthesized silver nanoparticles. The comprehensive overview of elemental composition of substrate is shown in Table 5. SEM images of (a) silver particles, (b) silver particles coated cotton fibers, (c) silver particles coated and dyed cotton fibers, (d) EDX analysis, (e) size analysis and (f) XRD of silver particles. Comprehensive overview of the elemental composition of coated substrate.

The XRD analysis was utilized to determine the phase composition of the silver nanoparticles. Figure 5(f) depicts XRD spectra for the 2 range of 20 to 80° with a 0.02-degree step. The ideal indexing of diffraction pattern refers to the silver structure identify the phase purity and specific crystallographic planes of prepared silver nanoparticles. Four distinct diffraction peaks were appeared against Ag-NPs coated fabrics at 2θ values of (wide angle x-ray spectroscopy) at 38.1°, 44.3°, 64.5°, 77.5°, which were absent in case of untreated cotton fabric. The peaks were assumed to correspond with (111), (200), (220), and (311) crystal planes metal (silver); as identified and documented in the International Center for Diffraction Data (JCPDS data number 04-0783 card). Moreover, not a single peak was observed for other impurities or defects such as silver dioxide or silver sulphide etc. 41
Weight gain percentage
As mentioned in Material section, we have utilized the plain cotton woven fabric with GSM of 150. However, our final sample was made of 5 × 5 g per square meter (having calculated weight about 0.075 g (75 mg). The as-prepared particles with different concentrations (1, 2 and 3 mg) in 100 mL of deionized water were applied on cotton fabric by dip-coating method. For the purpose of dyeing, the material-to-liquor (M: L) ratio was adjusted at 1:40. The percentage weight gain against each sample is shown in Figure 6. It is clear that the Sample S1, S2 and S3, coated with a concentrated solution containing silver nanoparticles, demonstrated a maximum weight gain of approximately 20%. The percentage weight gain was more obvious (almost 43 % maximum), when additional dye was applied to fabrics previously coated with silver nanoparticles. Hence the samples S1, S2 and S3 were after applying the dye were termed as S1D, S2D, and S3D. This enhanced weight gain can be attributed to the higher concentration of dye, as well as the synergistic effect of dyeing the fabric already coated with silver particles, resulting in an increased accumulation of both dye and silver on the fabric surface. Moreover, the effect of weight gain percentage against the deposition of silver and copper nanoparticles is previously described by Azam et al. Where they claimed the maximum weight gain almost 40% of cotton fabric after the dense deposition of silver nanoparticles followed by electroless plating.
42
Weight gain percentage against silver coated undyed and silver coated dyed fabrics.
Antibacterial activity
The antibacterial efficacy of both undyed and dyed silver treated textile substrates was evaluated by qualitative and quantitative standard testing protocols.
Zone of inhibition test (qualitative measurements)
ZOI against

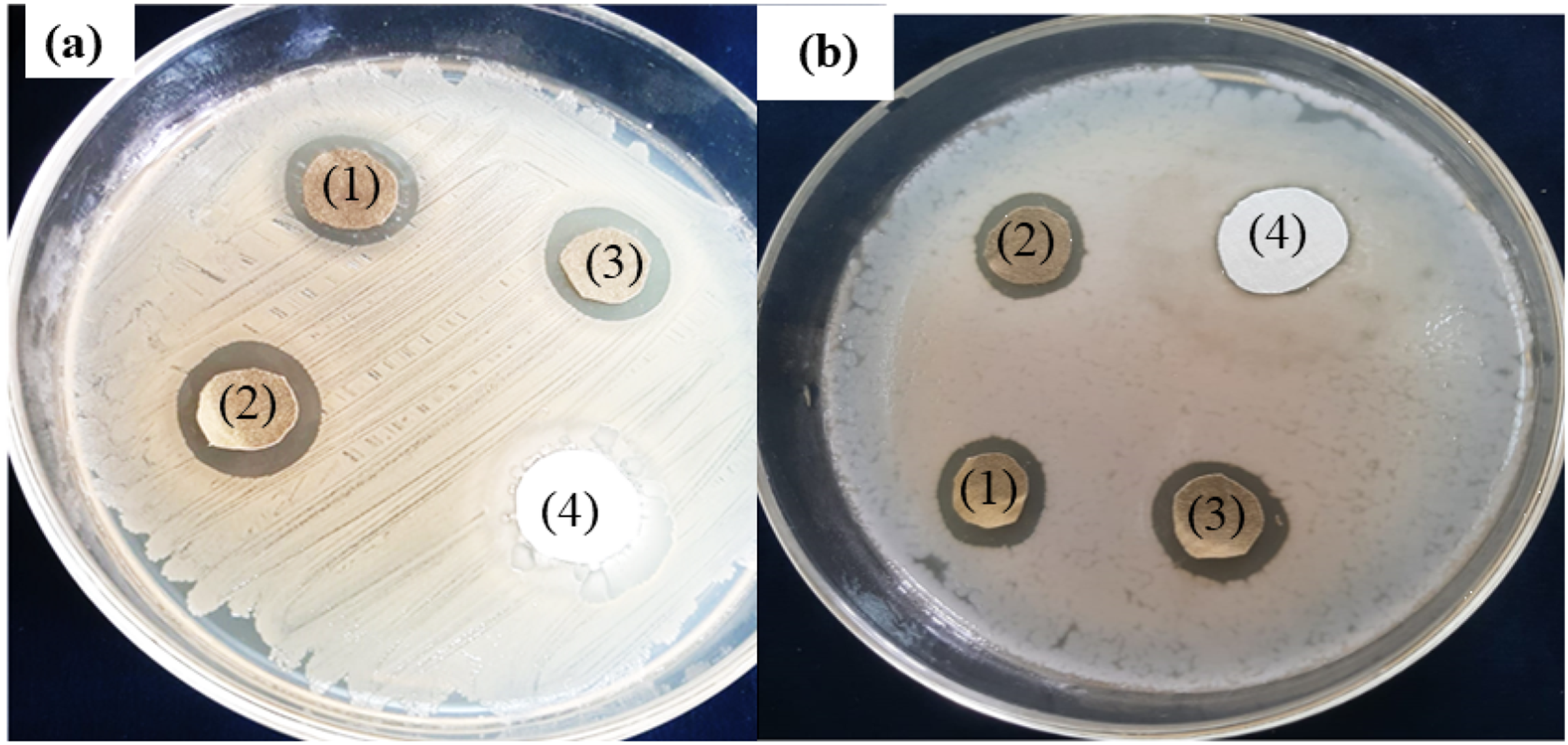
ZOI around silver treated undyed (1) dyed (2 & 3) and control (4) fabric samples against (a)
Reduction factor (quantitative test)
The ISO-20743 method was followed for the quantitative assessment of antibacterial effectiveness of all prepared fabrics towards (a) Reduction in percentage against antimicrobial quantitative analysis against 
To clarify, the decrease in CFU/ml (colony forming unites per mL) of survived bacteria was determined, and the resulting results are shown Figure 8(b). The untreated cotton fabric seemed to have a significant number of surviving bacterial colonies and higher CFUs/ml values (7.45 for
The findings were also evidenced by the images shown in Figure 9(a) and (b) for untreated and treated dyed fabrics towards Images for the bacterial colonies cultured media (at 0 h) and sustained (at 24 h) for samples termed as UT, S and S3D (a) 
Antibacterial effectiveness for different samples.

(a) Percentage reduction of antibacterial activity (b) reduction percentage of antifungal activity.
Antifungal activity of treated samples
The AATCC-100 method was followed to quantify antifungal activity towards
Antiviral effectiveness
Behrens and Karber’s method was employed to calculate the reduction in virus titer from the initial viral concentration of infectivity (108) for Corona Virus. Figure 11 shows the virus infectivity titer log at 0 minute and after 60 minutes. Figure illustrates the shift in corona virus infectivity titer for all test specimens. Antiviral activity was reported to be higher in all dyed samples, indicating that dye has considerable antiviral action. The viral infectivity titer was considerably lowered in all treated samples. The sample S3 (between all undyed fabrics) and S3D (between all dyed fabrics) demonstrated the maximum reduction, with 80% and 84% antiviral activity, respectively. The untreated fabric exhibited no antiviral action against the virus, confirming that the antiviral activity in all treated dyed and undyed fabrics was due to the use of Ag-NPs and natural antimicrobial dye. The antiviral activity of silver-treated undyed and dyed fabrics could be attributed to the binding of metallic nanoparticles and phenolic part of the polyphenols with glycoproteins at the viral surface, which acts as an inhibitory action for viruses. A recent research work was carried out to fabricate the Ag-NPs coated fabric using a photo-deposition method. The combined effect of the Ag0/Ag+ redox-active agent demonstrated a 97% reduction in viral activity, specifically against SARS-CoV-2.
44
Reduction in viral infectivity titer (left) and the corresponding percentage reduction (right).
Durability of silver coated dyed fabrics
Antimicrobial properties before and after washing.

Comfort properties
Effect of air permeability
Air permeability is a significant factor in the comfort of bandages. When perspiration and heat are produced by the body, it aids in the exchange of air. Figure 12(a) illustrates the air permeability results for all fabric samples. The results showed insignificant effect of silver- nanoparticles coating against air permeability. The air permeability of untreated fabric is approximately 956 mm/s, whereas the air permeability of silver nanoparticles coated dyed fabric samples at various concentrations was 941, 938, and 935 mm/s. Deposition of nanoparticles and dyeing reduces air permeability slightly. This phenomenon can be attributed to two factors. First, the fabric structure may undergo relaxation-induced shrinkage as a result of multiple dips in solution, potentially cause the yarns to become close and obstruct the flow of air. Second, nanoparticles have been incorporated into interstitial spaces of fabric during the application process, which ultimately reduces the fabric pore size. In fact, the fabric pore size is proportional to its air permeability. As a result, the fabric’s air permeability has decreased due to pore size reduction. However, the developed cotton bandages have no effect on air permeability. (a) Air permeability and (b) Water vapour permeability through Ag-NPs coated dyed fabric untreated fabric sample.
Effect of water vapor permeability
Water vapor permeability (WVP) is the ability to diffuse vapor from wound exudates in the body. The flow rate of water vapor through a fabric is vital for the determination of comfort because it provides the human feelings about cool or being warmth. Figure 12(b) shows that, there is no significant change in the relative WVP of cotton bandages. The untreated fabric showed WVP of 91%, while the silver nanoparticles coated fabric samples have a relative water vapor permeability of 88, 86, and 83 mm/s at different concentrations. The relative water vapor permeability decreases slightly after the deposition of nanoparticles. The reason is that (as previously described in the case of air permeability), the number of dips in a solution relaxes the fabric chains, bringing them closer together, and nanoparticles might well be deposited in the yarn interspaces. 25 In another related research, the deposition of TiO2 and ZnO nanoparticles via printing pastes on fabric led to a considerable increase in air permeability. However, its noticeable that the overall effect of the presence of nanoparticles on fabrics can have both positive and negative effects on air permeability, depending on the specific treatment and nanoparticles used 45 The nanoparticle network formed on the fiber surface has a minor impact on water vapor permeability because it has partially covered the pores in the fabric, resulting in reduced porosity. As a result, the most crucial comfort parameters for textile air and water vapor permeability are unaffected. The passing of adequate air and vapor through developed bandages provides an indication of permeability and breathability (body fluids such as perspiration, sweat, and moisture) for fabricated samples. Sweat on the skin easily passes through bandages. As a result, skin itchiness, skin irritation, and the maceration effects can be avoided when bandages are applied.
Ali et al, analysed the comfort parameters of air and water vapour transmission of silver nanoparticles coated textile woven substrates. Result showed very low effect of finely formed Ag-NPs on air permeability. The values of air permeability of the simple uncoated fabric sample were about 790 mm/s and while for Ag-NPs coated substrate it was around 782 mm/s. The same trend they analyzed for the relative water vapor permeability values, which were about 78.8% (for plain fabric), while 73.9% for silver particles coated textile. Water vapor permeability is slightly affected by the silver coating on the surface of fibre, which is quite porous, resulting in the impaired porosity. The passage of proper air and vapour through conductive fabrics provide an indication of the breathability, and permeability (of body fluid like perspiration, sweat and moisture) of developed samples. 46
Conclusion
The study was consisted of three phases. Initially, a stable and concentrated dispersion of green-synthesized Ag-NPs was obtained. Where, hygienic tannic acid, serving as a green dispersant and reductant, was used, which also acts as a self-assembly agent and free from any hazardous chemicals. Subsequently, the synthesized Ag-NPs were coated over the cotton textile substrate. The surface morphologies of the formed metallic nanoparticles coated textiles were analyzed by different characterization techniques such as SEM, FTIR, EDX and XRD. During second phase, natural antibacterial dye was extracted from pomegranate peel and applied on silver coated fabric. Hence, a total of 6 hygienic samples (S1, S2 and S3 undyed and S1D, S2D and S3D dyed) were made. Amount of silver deposition and dye uptake was analyzed by the percentage weight gain. The Sample S1, S2 and S3, coated with a concentrated solution containing silver nanoparticles, demonstrated a maximum weight gain of approximately 20%. The percentage weight gain was more obvious (almost 43 % maximum), when additional dye was applied to fabrics previously coated with silver nanoparticles. The antibacterial activity of dyed (silver nanoparticles applied and dyed) and control fabric (undyed fabrics) were evaluated by both qualitative and quantitative measures. The zone of inhibition was found to be high for dyed silver coated fabrics than undyed where dyed fabric sample S3D exhibits the highest antibacterial effect. During quantitative analysis, the highest bacterial colonies reduction was found in S3 and S3D samples, with CFU values dropping to 0 from 7.45 for
Footnotes
Acknowledgement
The authors of this study extend their appreciation to the Researchers Supporting Project, King Saud University, Riyadh, Saudi Arabia, for supporting this study (RSP2025R378) and for funding this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a researchers supporting project (RSP2025R378), King Saud University, Riyadh, Saudi Arabia.
