Abstract
In this study, bacterial cellulose was produced through the fermentation of a mixture of black tea, ginger, and sugar, and used to create bio-textile films on sugarcane-based media. Characterization included ribosomal RNA gene sequencing, FTIR spectroscopy, XRD, and SEM was used to examine surface morphology. The bio-textile films showed increasing UV resistance beyond 10 days of cultivation (T.UVA%; 0.13 ± 0.02, T.UVB%; 0.22 ± 0.01, UPF; 629 ± 2.12) and antimicrobial resistance was assessed by quantifying Colony-Forming Units (CFU), resulting in a 100% reduction in growth for both Escherichia coli and Staphylococcus aureus. Subsequently, after 15 days of cultivation, antimicrobial activity was evaluated using the disc agar diffusion method, yielding noteworthy outcomes. E. coli displayed a 25 mm zone of inhibition, S. aureus exhibited a 31 mm zone of inhibition, Candida albicans showed a 35 mm zone of inhibition, and Aspergillus niger presented a 22 mm zone of inhibition.
Introduction
To mitigate the environmental impact of the textile and leather industries, designers are increasingly exploring textiles created from renewable raw materials. We are well aware of the ethical and environmental challenges associated with rearing animals for leather and fur, tanning leather using hazardous methods, exploiting silkworms, and relying on petroleum-based materials to produce synthetic fibres, which are widely used in fabric and artificial leather production. 1 Consequently, the textile and garment industries rank among the world’s most polluting sectors. Therefore, bio-fabrics present an excellent alternative to non-eco-friendly textiles and leather materials.
Bio-fabrics, especially those produced through biological processes, represent the future of sustainable textiles on a global scale. These materials are cultivated by microorganisms such as bacteria, fungi, yeast, and others. Through bioengineering techniques, we can engineer these microorganisms to synthesize biopolymers that can be extruded into filaments using a spindle or molded into materials that can be retrieved without generating waste. During the biosynthesis process, bacteria or a consortium of microorganisms produce a film, known as a pellicle or membrane, consisting of a random nanofibrillar network of cellulose chains (as depicted in Figure 1).
2
This network is interspersed with regions of water, which account for 90%–98% of the material’s total volume. The structure, thickness, color, and texture of the final materials can be controlled through the microorganisms’ DNA. These microorganisms are nourished with sugar and transformed into biological fiber factories. Random nanofibrillary network of bacterial cellulose chains.
Bacterial cellulose (BC) is the most abundant biopolymer on the planet. BC is synthesized by bacteria belonging to the genus Acetobacter, with “Gluconobacter xylinus” being the most efficient producer. 3 BC consists of a network of self-assembled nanofibers, leading to a BC structure with higher crystallinity, tensile strength, and water retention capacity compared to plant cellulose. 4
Due to its unique properties, BC is emerging as a promising biopolymer for various applications. For instance, owing to its biocompatibility, 5 BC finds numerous applications in the biomedical field, including the development of artificial blood vessels for microsurgery, 6 scaffolds for cartilage tissue engineering, 7 and wound dressings. 8 It is also utilized in the manufacture of acoustic diaphragms for audio speakers and headphones, 9 electronic paper, 10 and optically transparent nanocomposites.11,12 Purified and dried BC is transformed into membranes for use in separation processes such as ultrafiltration, gas permeation, and vapor permeation, as well as in paper production.13,14 Food-related applications of BC encompass additives, emulsifiers, dietary fibers, edible preservatives, and barriers against bacterial growth.15,16
Despite the growing interest in BC, there is a limited number of publications addressing modifications in culture conditions, including strain selection, growth temperature, and carbon source. Existing studies predominantly focus on chemical modifications like acetylation,17,18 nitration, amination with diethylene-triamine, 19 and phosphorylation. 20 In this work, we present the production and investigation of bio-textile films derived from BC biosynthesis using black tea and ginger, cultivated on sugar cane media. The primary objective was the manufacturing of bio-textile films with the ability to resist microbes and ultraviolet radiation. To be considered a sustainable alternative for specific textiles and leather, especially in medical and technical applications, it should be both environmentally friendly and cost-effective. The cultivation process for obtaining biotextile films ranged from 4 to 10 days, depending on the desired thickness. It’s worth noting that the harvest time in this work is relatively shorter compared to most publications, where harvest times of 10–14 days are commonly reported.21,22
Materials and methods
Description of the biotextile films production process
The bio-textiles production is divided into four stages. - Biosynthesis for bacterial celluloses. Bacterial cellulose was biosynthesised by fermenting black tea and ginger and creating BC wet membranes (95% humidity, three-dimensional network of nano- and microfibrils) for this study. - Cultivation conditions on sugar cane media. The BC previously described has been cultured on sugar cane media. Sterilised by boiling at 110°C for 5 min, then cooling to a room temperature range of 25 to 29°C. - Production of bio-textile films. Four bio-textile films were biosynthesized according to the culture/fermentation time on sugar cane media Sterilized. - Washing and drying. The films were washed with distilled water and dried in atmospheric air.
Biosynthesis for bacteria celluloses
To biosynthesize bacterial nanocellulose (BC), we combined black tea from Lipton and 100% organic Indian ginger rhizome powder from “Imtenan- Egypt” in the first phase. This recipe involves using 1 L of distilled water, 150 g of sucrose, 10 mL of apple cider vinegar, 5 g of black tea, and 200 mL of ginger drink. These components were carefully blended before being placed in an airtight bottle, as shown in Figure 2(a). During the first phase, incubation and fermentation occurred over a 3-month period under room conditions (with temperatures ranging from 25 to 29°C and relative humidity between 45 and 50%). This process was conducted in a dark and elevated location. In the second phase, the biosynthesis of BC wet membranes required the following ingredients: 1/2 L of hot water, 4 g of black tea, 1/2 L of room temperature water, 150 g of sugar, and 1/2 L of the pre-prepared material. These ingredients were placed in a glass jar and covered securely with a breathable cloth, as illustrated in Figure 2(b). During the second phase, incubation and fermentation continued for 30 days at temperatures ranging from 25 to 29°C, with relative humidity levels maintained between 45 and 50%. This process also took place in a dark and elevated location. The BC wet membrane was formed, resulting in gel-like materials, as depicted in Figure 2(c). (a) Brewing ginger and black tea together; (b) Biosynthesis for BC; (c) BC wet membranes created by floating gel-like material.
Cultivation conditions on sugar cane media
On sugar cane media grown in Upper Egypt, the BC was cultured. The seller was “Al-Qubaisi Company of Ramses Street, Ghamra, Cairo, Egypt.” All the protocols for the use of plant material were compliant with relevant institutional, national, and international guidelines and legislation. In static culture conditions, it was Incubation for 7 days at room temperature in a dark and high place. in a temperature range of 25 to 29°C and a relative humidity range of 45 to 50%. BC film was created by floating gel-like material. The fermentation process is depicted in Figure 3(a). The findings after 5 days are shown in Figure 3(b), and the cellulose bacteria after 7 days are shown in Figure 3(c). (a) Process of fermentation on sugar cane medium, (b) after 5 days of fermentation on sugar cane medium, (c) BC wet membrane, created by floating gel-like material after 7 days of cultivation on the medium of sugar cane.
Production of bio-textile films
We made four bio-textile films with varying culture durations (4, 6, 8, and 10 days) by incubating 4 L of sugar cane juice with 50 g of bacterium cellulose generated in a 30 × 50 cm container, as shown in Figure 4(a). Then it is covered well with a breathable cloth and fermented in static cultivation conditions in a In a high place that looks like a basement. As illustrated in Figure 4(b) biotextile films were generated by floating a gel-like substance. (a) Incubating 4 L of sugar cane juice with 50 g of BC generated; (b) Bio-textile films were generated following incubation by floating a gel-like substance.
Washing and drying
The films were rinsed multiple times with distilled water and squeezed to minimise its water content, as shown in Figure 5(a) and (b). The washed film was dried for 2 days on a horizontal surface in the sun and atmospheric air, as illustrated in Figure 5(c), Figure 6 shows Scheme of bio-textile film production in sugar cane media. (a) Rinsed films multiple times with distilled water, (b) squeeze to minimise its water content, (c) dry on a horizontal surface in the sun and atmospheric air for 2 days. Scheme of bio-textile films stages production in sugar cane media.

Natural dyeing
Bacterial cellulose can be dyed not only after it is produced (ex situ), but also while it is being produced by adding dye during cultivation (in situ). Both dyeing methods produced smooth surfaces and kept their inherent nanostructures during dye penetration, but the in situ method produced a smoother surface and more even colour than the ex-situ method.
23
Based on this, the bio-textile film was dyed by adding dye during cultivation (in situ)14 mL of food colouring was added to the carbon source (sugarcane juice). using blue food dye from Surya International, Ahmedabad, India. Figure 7(a) shows the dyed bio-textile film during washing, Figure 7(b) shows the dyed bio-textile film after drying. (a) The dyed bio-textile film is rinsed multiple times with distilled water, (b) the dyed bio-textile film after drying.
Figure 8 depicts bio-textile films created at various cultivation times. Figure 8(a) after 4 days of cultivation, Figure 8(b) after 6 days of cultivation, Figure 8(c) after 8 days of cultivation, and Figure 8(d) after 10 days of cultivation. (a) Bio-textile film after 4 days of culture time; (b) bio-textile film after 6 days of culture time; (c) bio-textile film after 8 days of culture time; (d) bio-textile film after 10 days of culture time.
Characterization
Isolation and characterization of microbes the cultivation on sugar cane media
Nutrient Agar (NA) and Potato Dextrose Agar (PDA) obtained from Sigma-Aldrich were utilized. Both NA and PDA were prepared following the manufacturer’s recommended procedures. For Hestrin-Schramm (HS) agar, a mixture was created by combining 20 g per liter of glucose, 5 g per liter of peptone, 5 g per liter of yeast extract, 2.7 g per liter of Na2HPO4, and 1.15 g per liter of citric acid. To prepare plates for enumerating microorganisms in BC fermentations, a solution containing 0.5 mg per milliliter of cycloheximide in ethanol was added to the NA and HS agar while they were in their liquid state and had cooled down. For yeast enumeration in BC fermentations, plates were prepared differently. A solution containing 25 mg per milliliter of chloramphenicol in ethanol was added to the cooled, molten PDA agar.
Isolation of dominant microbes (bacteria and yeast) culture adapted in sugar cane media
A fermentation broth was subjected to culture on three different agar types: PDA, NA, and HS. To begin, a 10 mL sample of the fermentation broth was serially diluted in sterile phosphate-buffered saline (PBS). The bacteria were isolated on NA media with the addition of cycloheximide to hinder yeast growth. This mixture was then incubated at room temperature for a duration of 24–48 h. In contrast, yeast isolation took place on PDA agar, which was supplemented with chloramphenicol to prevent bacterial growth. The yeast samples were incubated at room temperature for a period of 72 h. Additionally, microbial samples were grown on HS agar media at room temperature for 72 h. The dominant isolates from each petri dish were chosen based on their colony morphology and the results of Gram staining.
Characterization and identification of bacteria and yeast isolates using sequence analysis of ribosomal RNA genes
Bacterial isolates were identified using a PCR method along with gene sequencing of the 16S rRNA gene, employing universal primers (27F/1492R) with the following sequence:
(5′-AGAGTTTGATCCTGGCTCAG-3′ and 5′-GGTTACCTTGTTACGACTT-3′).24,25 In parallel, yeast isolates underwent identification through a PCR method, focusing on the D1/D2 region of the 26S rRNA gene, using NL-1 and NL-4 primers with the following sequence:
(5′-GCATATCAATAAGCGGAGGAAAAG-3′ and 5′-GGTCCGTGTTTCAAGACGG-3′). 26 The resulting PCR products were then subjected to electrophoresis on a 1% agarose gel with Tris-borate-EDTA. Subsequently, the agarose gel was treated with ethidium bromide and visualized under UV light. Following this, the pure PCR results obtained from the amplification of the bacterial 16S rRNA gene and yeast 26S rRNA gene were sent for sequencing.
Scanning electron microscopy (SEM)
SEM analysis was performed using scanning electron microscope. was carried out using a high-resolution scanning electron microscopy (SEM) EDAX AMETEK Quanta FEG 250 with field emission gun attached with an accelerating voltage 30 kV, FEI company - Netherlands in the central lab, National Research Centre, Egypt. the samples cut from the dried bio-textile film alized were coated with gold film before analysis.
X-ray diffraction (XRD)
The X-ray diffraction (XRD) was used to study the crystallinity and microstructure of the sample bio-textile film. The XRD pattern was captured over a 2-h period with an angle range of 0–40°. The Cu/Ka radiation source employed had a wavelength of 0.154 nm and was created at an acceleration voltage of 40 kV and a filament emission of 40 mA.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared spectroscopy (FTIR) Attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) was performed using a Thermo Scientific Spectrometer equipped with a Smart Orbit ATR accessory. The reflectance element was a dia-mond crystal. Spectra were collected in the 4000–650 cm−1 range with 16 scans and at 16 resolution.
Contact angle test
Contact angle analysis was performed in the Desert Research Centre of the Egyptian Ministry of Agriculture and Land Reclamation using the Dataphysics model tension metre and the OCA-15EC. The sample was subjected to one drop of deionized water (3 μL) at room temperature. The evaluations were conducted at 0, 1, 3, 4, and 5 s.
Percentage of water retention (PWR)
All four wet bio-textile film were weighed and dried in atmospheric air in order to completely remove the water and maintain a constant weight. Then the PWR was obtained using equation (1).
27
Yield and thickness
To calculate the yield of the biosynthesis process, we used equation (2)
where M is the dry/wet weight of BC (g), and C is the weight of the carbon source (L) used in the production medium (sugar cane juice).
The thickness test was carried out according to DIN EN ISO 5084 by using a digital thickness gauge with a roller insert measuring tool type (0.01 mm–12.5 mm).
Ultraviolet resistance analysis (UVA)
The ultraviolet resistance properties of bio-textile films were determined using the UV/Visible Spectrophotometer JASCO V-750 (from 280 to 400 nm) in accordance with the Australian/New Zealand standard method AATCC Test Method 183:2010.
Antimicrobial activity studies
Antimicrobial activity was determined in two ways. 1- Determination of antibacterial activity by measuring colony forming units (CFU) 2- antimicrobial activity the disc agar diffusion method (Zone of inhibition)
Where the antimicrobial activities of microbes were investigated Staphylococcus aureus ATCC 6538-P (G+ve) and Escherichia coli ATCC 25933 (G−ve), Candida albicans ATCC 10231 (yeast), and Aspergillus niger NRRL-A326 (fungus).
Staphylococcus aureus (bacteria G+ve)
A peptidoglycan layer and teichoic acids compose the cell wall of gramme-positive bacteria. 28 Peptidoglycans, or mureins, are composed of linear glycans that are cross-linked by short peptides. Glycans are disaccharides comprised of N-acetylglucosamine and N-acetylmuramic acids linked β (1,4) glycosidic linkages.28–30 Teichoic acids are anionic glycopolymers that give gramme-positive cells a negative charge [80]. Two types of teichoic acids exist: those that covalently attach to the N-acetylmuramic acid of peptidoglycans and those that anchor to cytoplasmic membranes. 28
Escherichia coli (bacteria G-ve)
A Gramme-negative bacteria’s cell wall consists of a thin layer of 2–7-nm-thick peptidoglycan and an outer membrane, with lipoproteins binding these two together.31,28 The outer membrane, on the other hand, is composed of an asymmetrical lipid bilayer with an inner side comparable to a cytoplasmic membrane but an outside side distinct due to the presence of lipopolysaccharides.32,31 There are proteins in this outer membrane that tend to be organised into trimers called “porins,” and these porins contain a hydrophilic channel that allows low-molecular-weight molecules from the external environment to enter the bacterial cell.32,31
Candida albicans (yeast)
C. albicans is a common pathogen that causes mucosal infections. It also causes life-threatening bloodstream infections that spread to internal organs in certain types of immunocompromised patients. It is a polymorphic fungus that can grow in yeast, hyphal, or pseudohyphal forms. The hyphal form enters the bloodstream by penetrating epithelia and endothelia, causing tissue injury and gaining access to the bloodstream. 33
Aspergillus niger (fungus)
Niger is a widespread contaminant of food, soil, and the indoor environment due to its propensity to thrive on a wide range of substances. Despite the fact that its spores are widespread, the fungus has been considered a less likely source of human disease than other Aspergillus species. 34 A. niger typically infects tissues that have already been rendered vulnerable by bacterial infections, physical damage, or cerumen deposition in the external auditory canal. A. niger, like other Aspergillus species, is a causal agent of otomycosis, a subacute or chronic fungal infection of the ear, throat, or nose. Immune deficient individuals, like most filamentous fungal species, are prone to A. niger infections, which cause invasive pulmonary aspergillosis.35,34 characterised by a chronic productive cough with blood coughing.
1- Determination of antibacterial activity by measuring colony forming units (CFU)
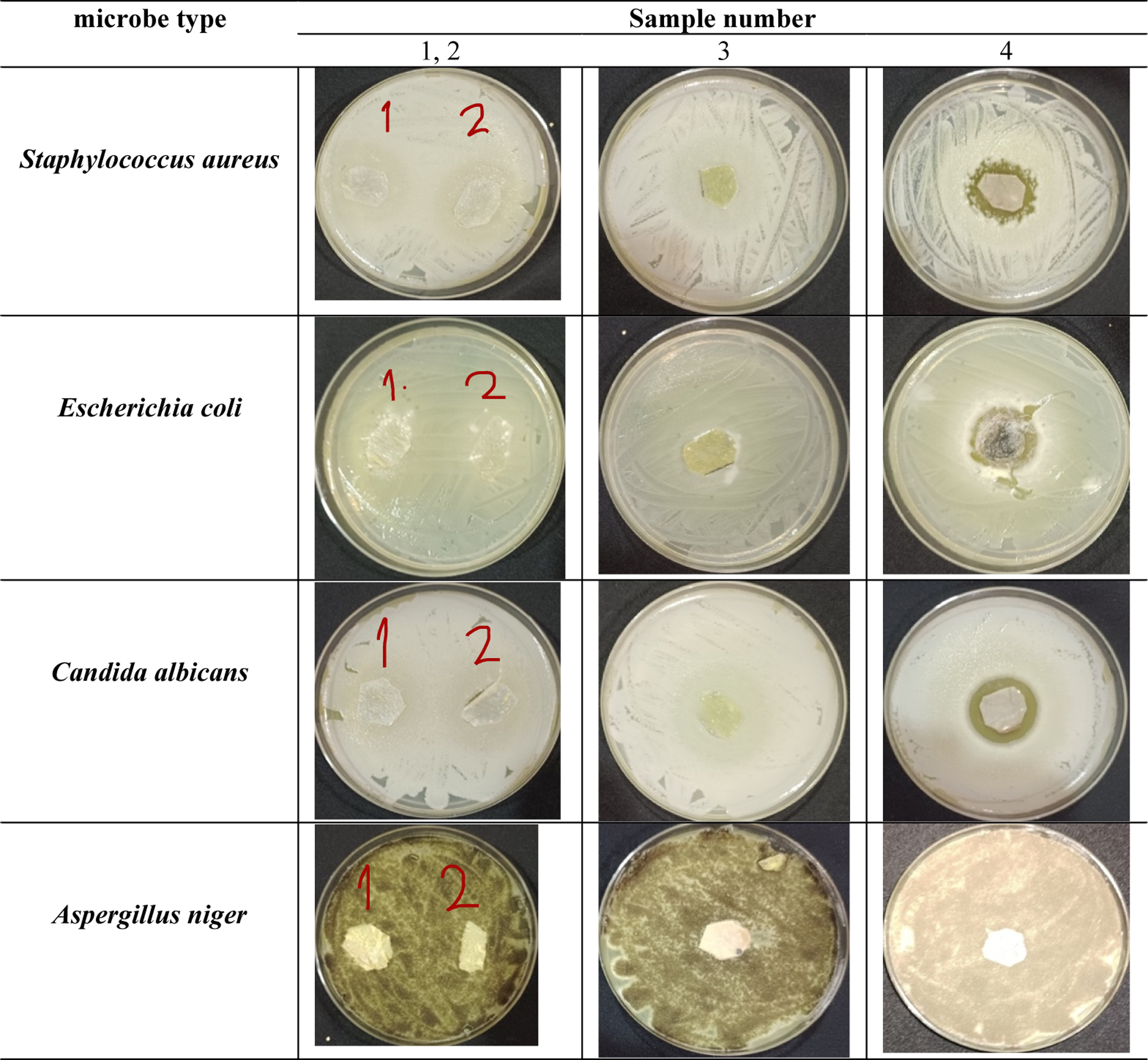
The antimicrobial activities of bio-textile films against S. aureus and E. coli were investigated using the colony forming unit technique (CFU). Bacterial stocks, totaling (100 µl) in volume, contained an estimated CFU count of around 108. were inoculated into a 20-ml freshly prepared liquid nutrient broth containing 5 g/L peptone and 3 g/L beef extract at pH 6.8 in 100 mL Erlenmeyer flasks and incubated for 24 h. Bio-textile films (250 mg) were added to the inoculated flasks (with 20 µl of inoculums), leaving the control inoculated flasks without samples) The reduction (R%) in growth was measured after 24 h of incubation at 37°C by detecting the reduction in absorbance of the inoculated flasks compared to the untreated controls. Equation (3) was used to calculate the growth reduction.
where A represents the number of colonies in the culture control and B represents the number of colonies in the bio-textile films sample. 36
2- Determination of antimicrobial activity the disc agar diffusion method (zone of inhibition)
The disc agar diffusion method was used to investigate the antimicrobial activities of the four representative test microbes used: S. aureus ATCC 6538-P (G +ve) and E. coli ATCC 25933 (G−ve), C. albicans ATCC 10231 (yeast), and A. niger NRRL-A326 (fungus). Bacteria and yeast were regularly introduced to NA plates through heavy inoculation, with each plate receiving 0.1 mL of cells per milliliter in the range of 105–106 cells/ml. PDA plates seeded with 0.1 mL (106 cells/mL) of fungal inoculum were used to evaluate the antifungal activities. The inoculated plates were covered with bio-textile film discs (15 mm in diameter). After that, the plates were kept at 4°C for 2–4 h to allow for maximum diffusion. The plates were then incubated in an upright position for 24 h at 37°C for bacteria and 48 h at 30°C for organism growth. The test agent’s antimicrobial activity was determined by measuring the diameter of the zone of inhibition in millimetres (mm). The experiment was repeated several times, and the mean of the readings was recorded.
Statistical analysis
We conducted statistical analysis using Microsoft Excel 365 from Microsoft, USA. The data were analyzed using trend lines, their equations and values of the correlation coefficient (R2) determination coefficient values, and one-way analysis of variance (ONE WAY-ANOVA).
Results and discussion
BC material formation occurred by enveloping native cellulose polyglucosan chains with water molecules from the growth medium. Subsequent drying of the BC material led to the establishment of hydrogen bonds among cellulose hydroxyl groups, transforming the gel material into a film. 24 In the static cultivation process, a BC film formed on the surface of the sugar cane medium. Within 3 days of fermentation initiation, the BC film exhibited an increase in both weight and thickness, gradually covering the entire liquid surface. Consequently, harvesting was conducted at intervals of 4, 6, 8, and 10 days to investigate the influence of culture time on various film properties, such as antimicrobial resistance, ultraviolet resistance, water retention percentage, yield, and thickness. Additionally, the bio-textile films underwent thorough characterization using diverse analytical techniques, including the sequencing of ribosomal RNA genes, FTIR spectroscopy, XRD, and SEM.
Bacterial cellulose-producing isolates screening
Bacteria that had been isolated and preserved from a previously adapted culture were selected for screening based on their distinctive colony characteristics. Before the screening process, six different colony types underwent microscopic examination, revealing that all of them exhibited uniform visual features, appearing as rod-shaped and pink when subjected to a Gram staining procedure Figure 9. These characteristics indicated that the bacteria belonged to the acetic acid bacteria group,
37
which is a common producer of BC. Following a 14-day period of static incubation, the most productive bacterium was chosen based on its notably thick appearance, determined through visual assessment. This selected isolate became the sole bacterial inoculum used for subsequent experiments. Light microscope at 1000× magnification of gramme-stained bacteria.
Sequence analysis of ribosomal RNA genes
Names of bacteria and yeast isolated.

Primers, Identities, and the Accession.
Scanning electron microscopy (SEM) for bio-textile film
Bacterial cellulose’s unique properties are determined by the ultrastructure and size of its fibrils. The surface microstructure of BC pellicles is revealed by SEM images Figure 10(a) to be layered nets of varying densities made of randomly oriented cellulose microfibrils. This is typical BC morphology, as described by Yamanaka and Sugiyama (2000).
38
BC nanofibrils can be seen on the surface in a multilayered structure, as shown in Figure 10(b), confirming the structure of a well-organized three-dimensional network. The crystallization of cellulose in a single direction confirms that the molecules are linear glucose linked by a β (1 → 4) glycosidic connection. When glycosidic chains join, microfibrils are formed that are oriented by intramolecular hydrogen bonds.
39
Because of the presence of binding agents (mainly sucrose from the growth medium), the structure of the BC sample was bulky, making determining cellulose fibers challenging. However, using SEM, it was possible to determine the cellulose fibers and their diameter. As shown in Figure 10(b) the diameter of microfibrils in the BC specimens varied between 20 and 95 nm. (a) SEM image of the bio-textile film; (b) SEM of the diameter of cellulose fibres.
X-ray diffraction (XRD) analysis
Here are four crystallographic polymorphisms in cellulose (I, II, III, and IV). Cellulose I is the crystalline cellulose produced naturally by a wide range of species, including trees, plants, tunicates, algae, and bacteria. Cellulose I has a thermodynamically metastable structure and can be changed to cellulose II or III. All of the cellulose strands are arranged in a highly organised parallel pattern.
40
Regeneration (solubilization and recrystallization) and mercerization (alkaline solution) can be used to convert cellulose I to cellulose II. The structure of cellulose II is monoclinic. Cellulose III is created by combining liquid ammonia and thermal treatments with Cellulose I and II. Cellulose IV is created by combining cellulose III.41,42 Diffraction patterns obtained for the bio-textile film obtained are shown in Figure 11 three distinguished peaks at 2h (2θ) = 14.35, 22.4, and 35.2, corresponding to the (101), (200), and (040) crystallographic plane. These peaks are typical reflections of cellulose I.43,44 Cellulose I is the crystal structure with the highest axial elastic modulus.
45
The peaks also appear narrow and pointed, which shows that they have a high degree of crystallinity.
46
Other diffraction peaks appear at 2h = 12.5, and 19.9, corresponding to the (110), (200), and (040) crystallographic planes, typical for type-II structure.
47
When compared to cellulose I particles, cellulose II particles lead to improved dispersibility in water, increased compatibility with materials such as proteins and lipids, and are beneficial as an excipient in tablet manufacturing.
48
Furthermore, cellulose II particles with a lower crystalline structure can improve ductility in rigid polymeric matrices.
49
X-ray diffractograms (XRD) of bio-textile film.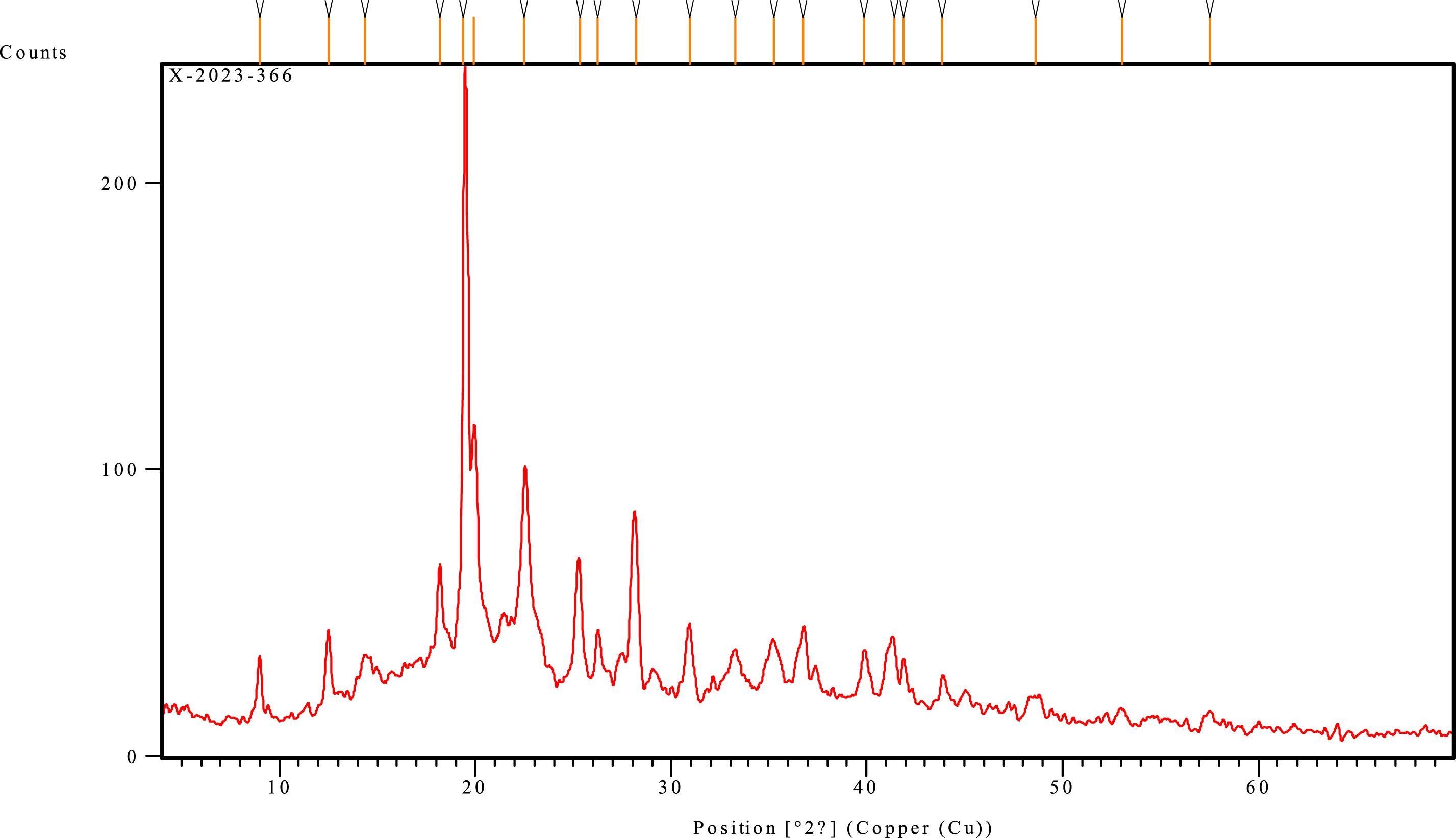
Fourier transform infrared spectroscopy (FTIR) analysis
List of band assignments for FTIR spectra.


FTIR spectra of bio-textile film.
Contact angle test
Time and contact angle for bio-textile film.


Analysis of contact angle for bio-textile film: (a) zero time; (b) 1 s; (c) 3 s; (d) 4 s; (e) 5 s.
Effect of culture time on percentage of water retention (PWR)
Water retention percentage of bio-textile film obtained.
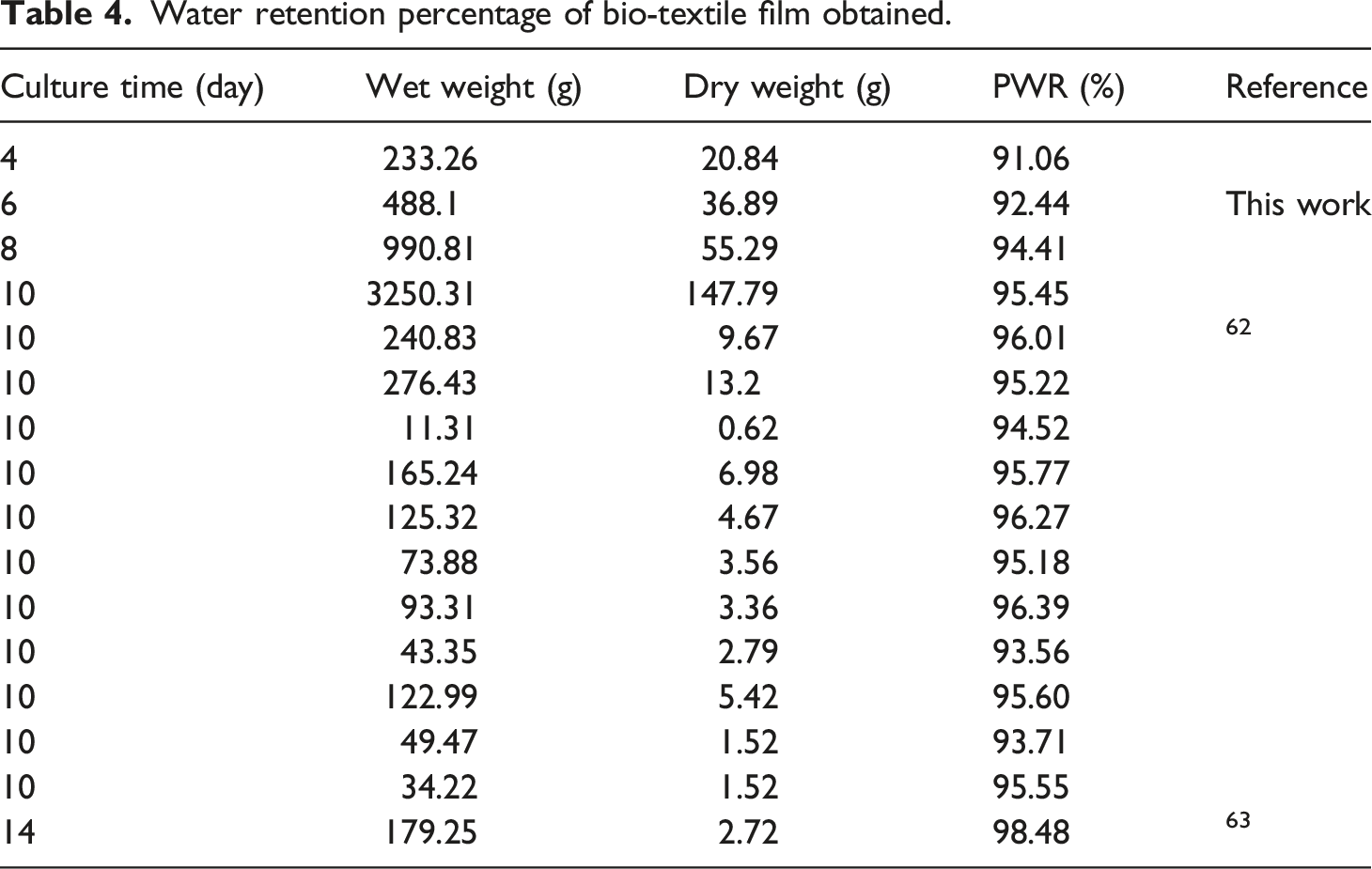

Effect of culture time on the water retention percentage of bio-textile films.
Effect of culture time on yield and thickness of bio-textile films
Effect of culture time on yield and thickness.

ANOVA test results.
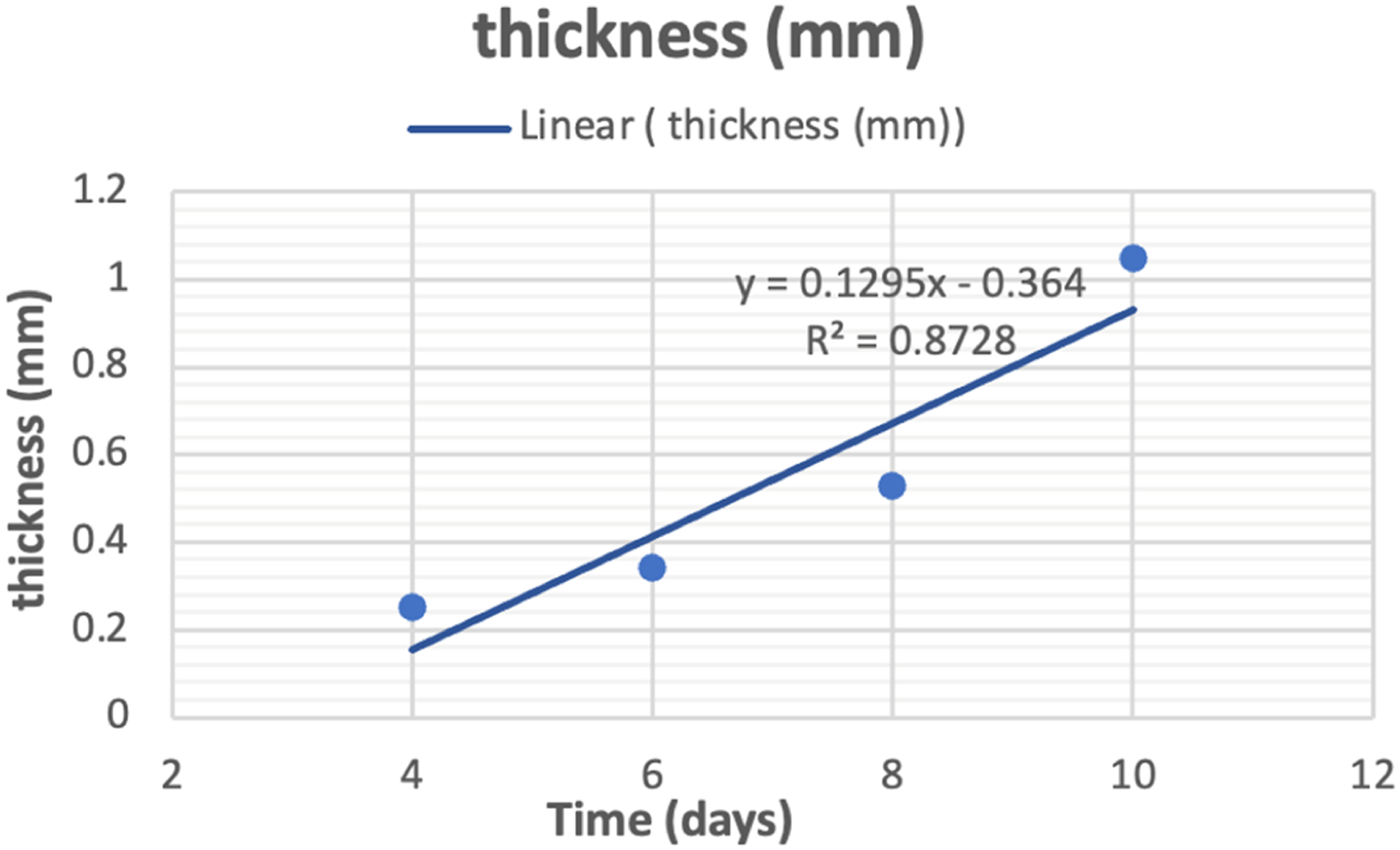
Effect of culture time on thickness.
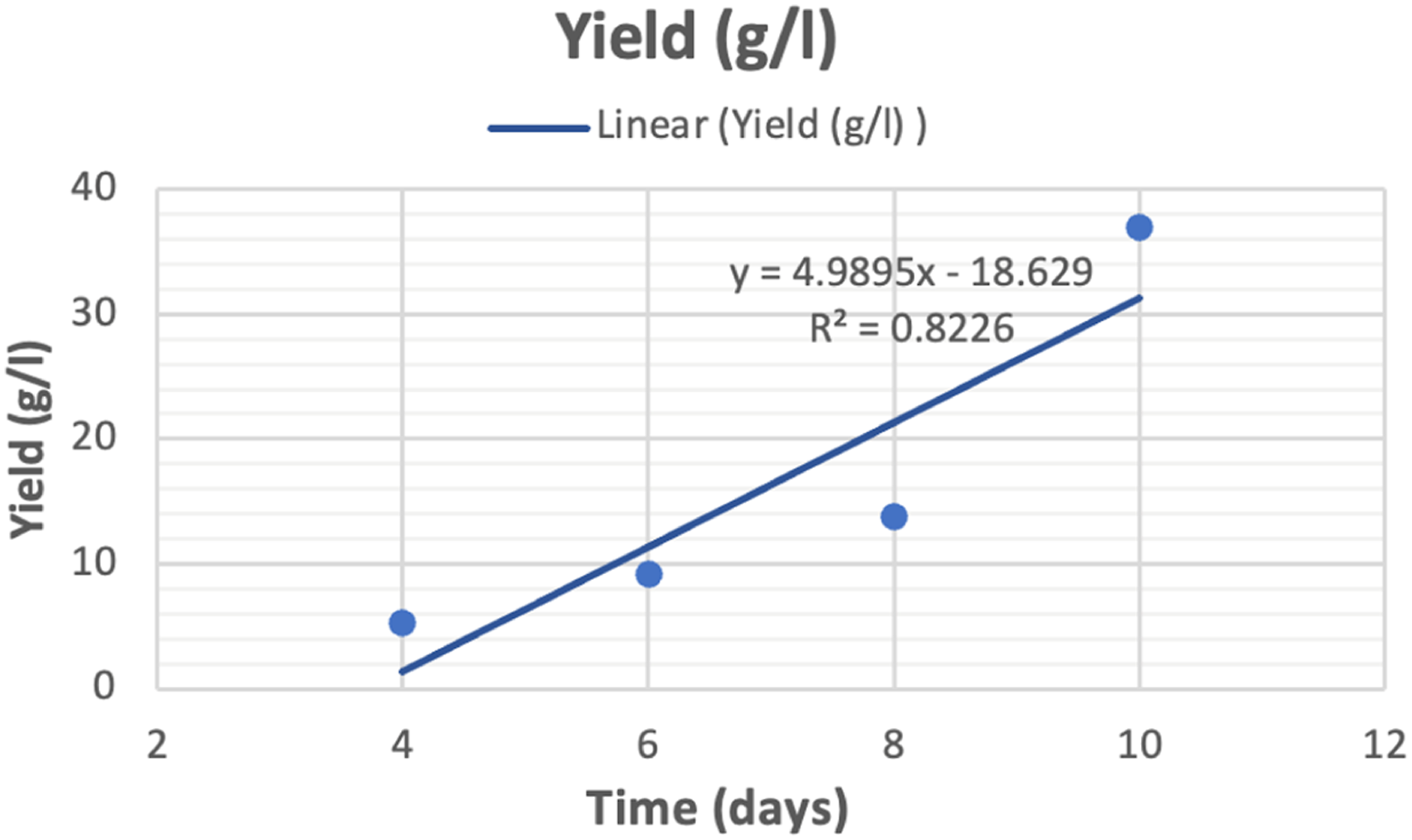
Effect of culture time on yield.
Effect of culture time, and media type on yield from previous literature.

Effect of culture time on ultraviolet resistance analysis
UPF rating and protection grades.

UV-resistance characteristics of bio-textile films.
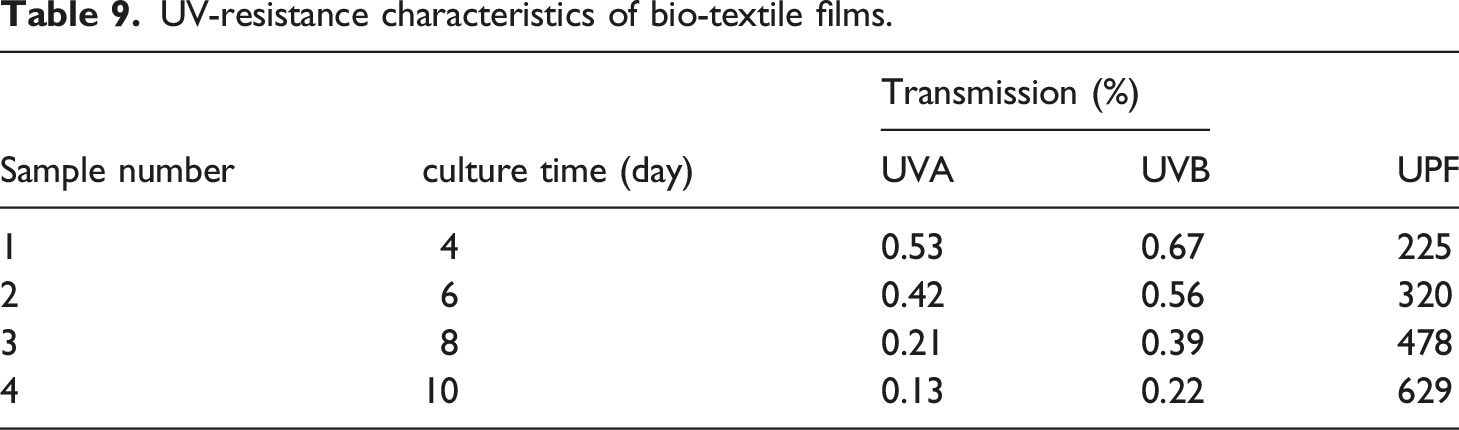

Effect of culture time on Transmission of (UVA, UVB).

Effect of culture time on ultraviolet protection factor.
Effect of culture time on antibacterial activity by measuring colony-forming units (CFU)
Effect of culture time on antibacterial activity by measuring colony forming units (CFU).

Photograph images of the Determination of antibacterial activity by measuring colony forming unit (CFU).
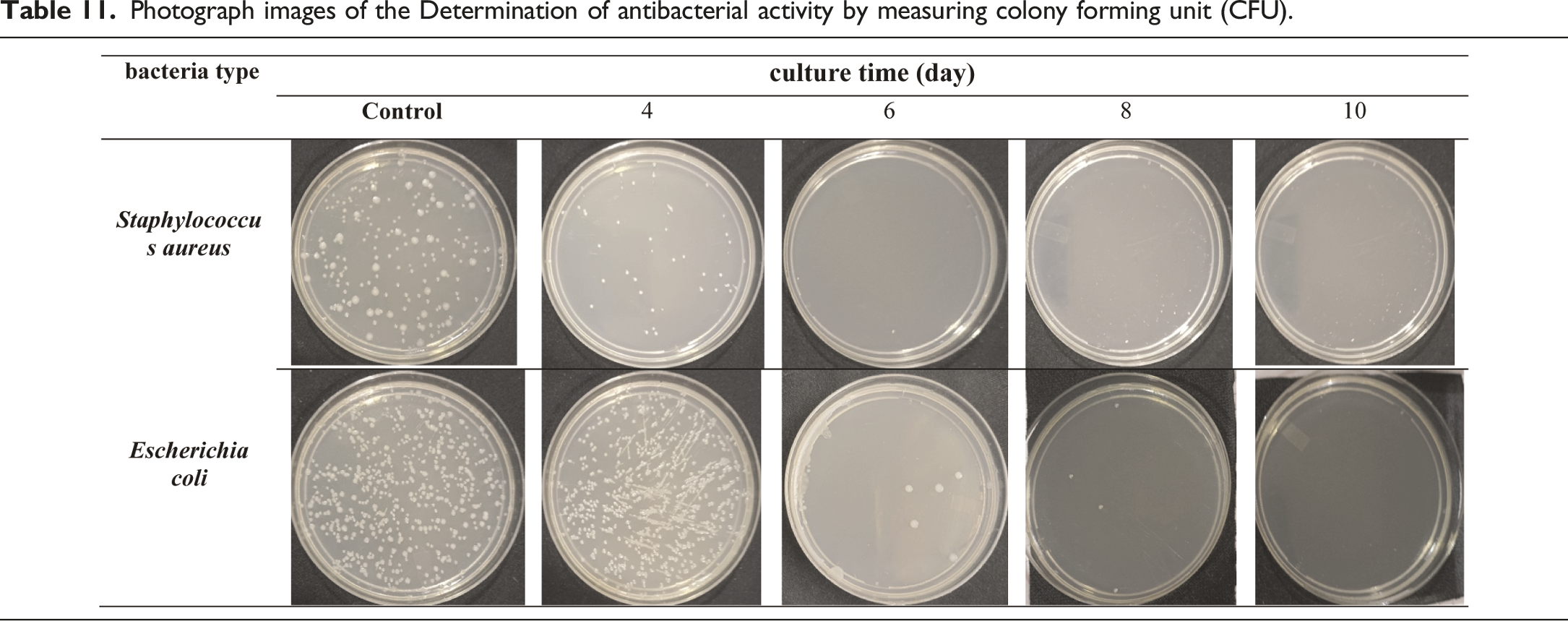
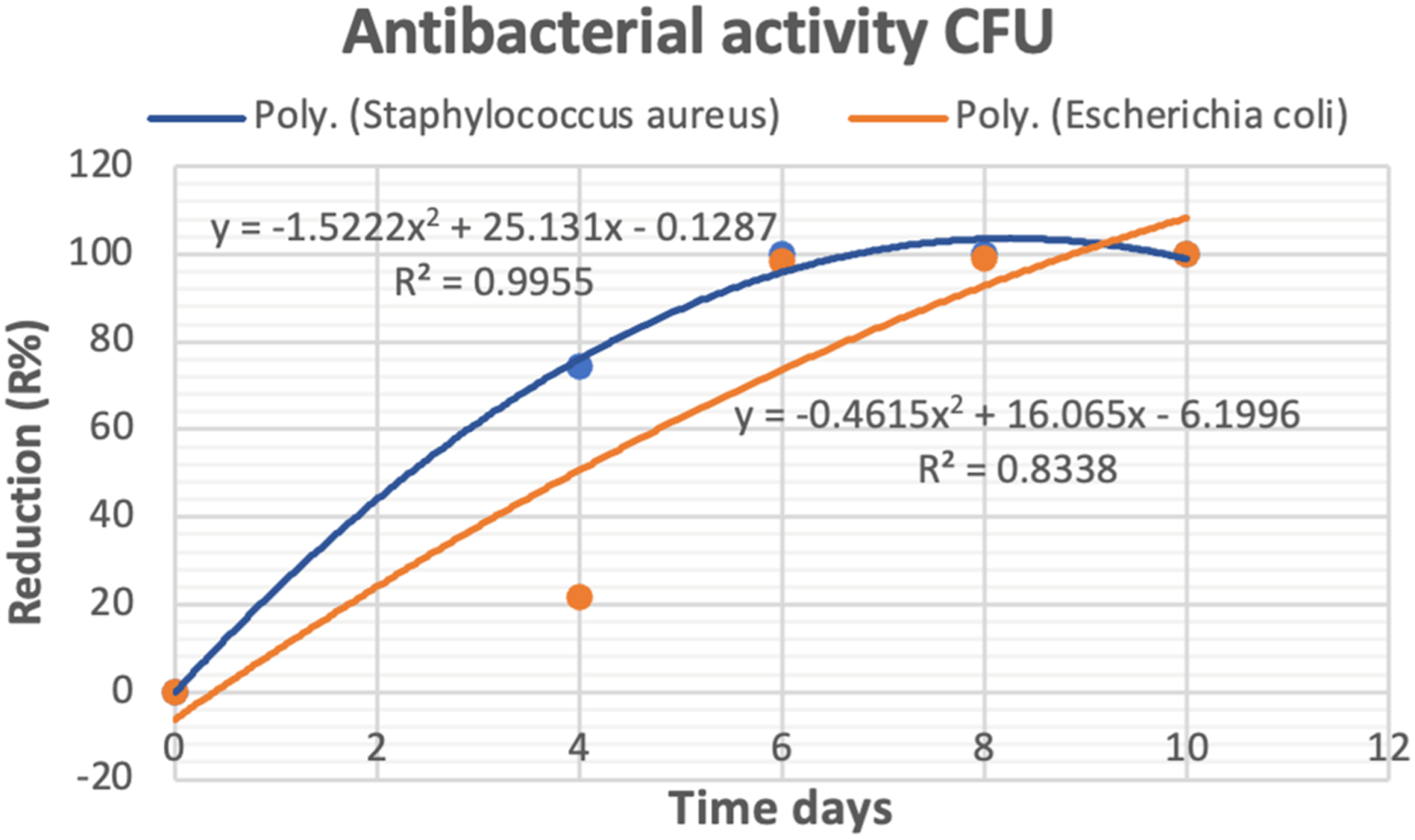
Effect of culture time on antibacterial activity by (CFU).
Effect of culture time on antimicrobial activity by the disc agar diffusion method (zone of inhibition)
Effect of culture time on the antimicrobial activity against different test microbes.

Photograph images of the inhibition zone of against different test microbes.

Antimicrobial activity against different test microbes after 15 days of culture.

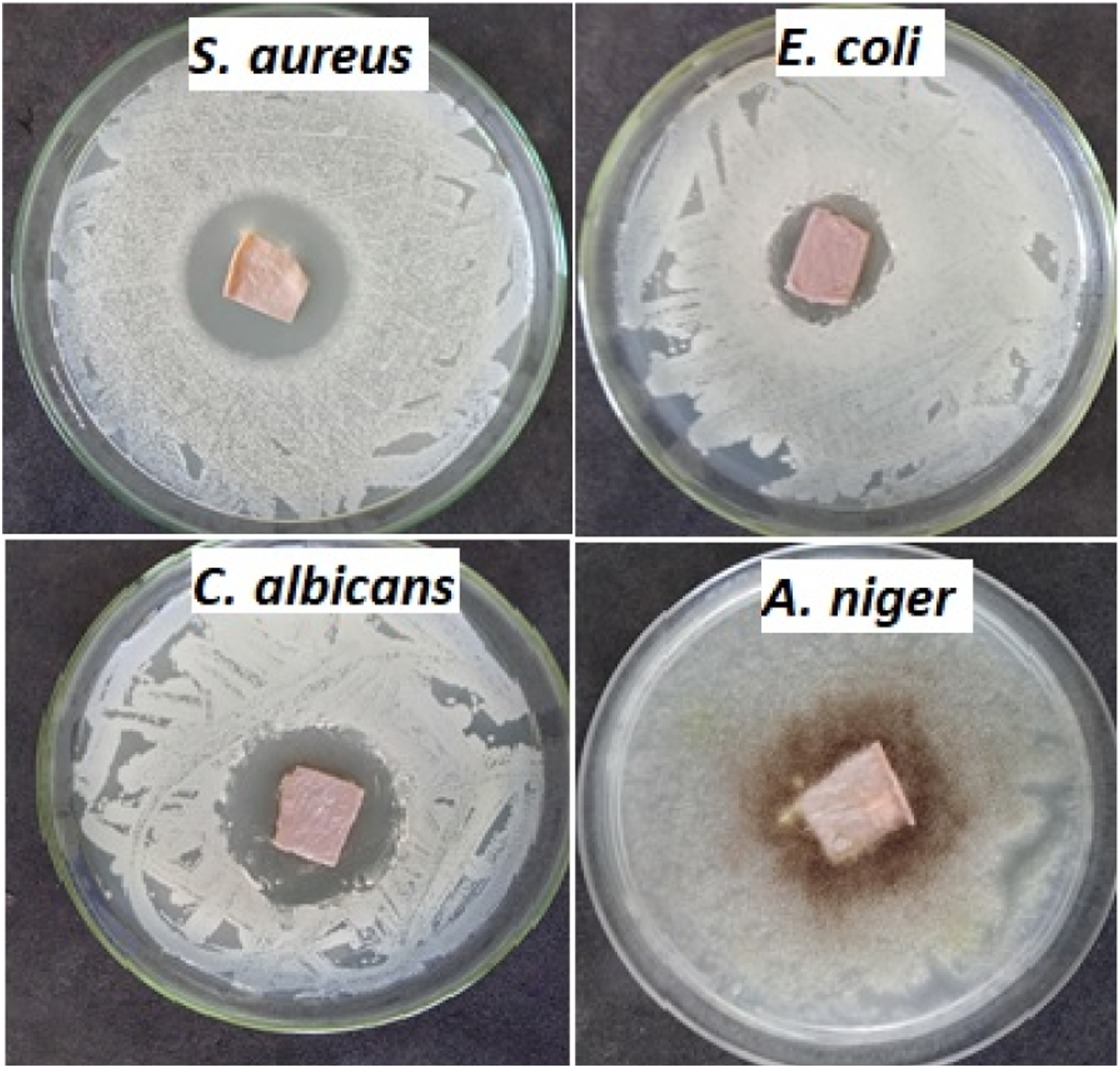
Photograph images of the inhibition zone of against different test microbes after 15 days of culture.

Effect of culture time on antifungus activity by the (zone of inhibition).

Effect of culture time on antibacterial activity by the (zone of inhibition).
Conclusions
Bacterial cellulose biosynthesis, isolated from black tea and ginger, is an exciting area of research that has the potential to revolutionise the way we think about and use cellulose. According to the results reported, this bacterial strain is capable of producing quality cellulose at high yields. Up to (650 g/L wet weight, 36 g/L dry weight) after 10 days of culture time with sugar cane as the culture medium. They also contributed to the economy of the bioprocess, as high values of productivity were obtained without the use of more expensive raw materials such as yeast extract. X-ray patterns and FTIR spectra confirmed the presence of cellulose I and II crystalline polymorphisms. The contact angle test was conducted to confirm if the bio-textile films created are extremely hydrophilic or hydrophobic. The results showed that the films are exceptionally hydrophilic. The study proved that the effect of culture time on BC production is significant. As the culture time increases, the amount of cellulose produced increases. This is because the bacteria have more time to produce cellulose, and the longer the culture time, the more cellulose. As a result for cultivation increase period, several positive changes were observed in the bio-textile film. After 10 days of cultivation, there was an increase in film thickness, a rise in the percentage of water retained by the bio-textile film, and an enhancement in its antimicrobial properties. This was evident in the antibacterial activity measured by CFU, where for E. coli and S. aureus, the reduction in growth (R%) was 100%. Subsequently, after 15 days of cultivation, the antimicrobial activity was assessed using the disc agar diffusion method, which revealed notable results with E. coli displaying a 25 mm zone of inhibition, S. aureus with a 31 mm zone of inhibition, C. albicans exhibiting a 35 mm zone of inhibition, and A. niger displaying a 22 mm zone of inhibition. and the possession of excellent UV resistance characteristics Up to (T.UVA%; 0.13 ± 0.02, T.UVB%; 0.22 ± 0.01, UPF; 629 ± 2.12). The experiment also demonstrated the feasibility of naturally dyeing the bio-textile films produced. Making it ideal for use in a wide range of applications. Bio-textile films produced in this study hold significant promise for utilisation in various domains, including: - Healthcare and Medical Applications: Bio-textile films can have applications in healthcare and medicine, such as wound dressings, tissue engineering, and drug delivery systems. These films can potentially improve patient care and medical treatments. - UV Protection: Bio-textile films with enhanced UV resistance can provide better protection against harmful UV radiation, benefiting individuals who spend time outdoors and reducing the risk of skin damage and skin cancers. - Antimicrobial Properties: Bio-textile films with antimicrobial properties can have applications in hygiene and healthcare, reducing the risk of infections and improving overall health outcomes. - Sustainable Agriculture: Bio-textile films can also have applications in agriculture, such as in crop protection and soil improvement. They can help reduce the use of synthetic chemicals and enhance agricultural sustainability. - Economic Opportunities: The production and commercialization of bio-textile films can create economic opportunities, including job creation and revenue generation, particularly in regions where raw materials like BC, tea, or ginger are readily available.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.