Abstract
Composites of chitosan chloride and bacterial cellulose were successfully prepared by in situ method. Composites of bacterial cellulose/chitosan and pristine bacterial cellulose were investigated by means of scanning electron microscope, atomic force microscope, Fourier transform infrared spectroscopy, thermogravimetric analysis, X-ray diffraction, and bacteriostatic test. The crystallization of bacterial cellulose was interfered and weakened by the chitosan chloride included in the growth media, resulting in lower crystallinity index and thermal stability. And interaction between two polymers is verified by the thermal gravimetric analysis. The ultrafine nanofibril network structure of bacterial cellulose was retained by the composites, while the diameters were larger and the aperture inside were smaller than those of pristine bacterial cellulose, as shown through scanning electron microscope and atomic force microscope figures. The antimicrobial effects were enhanced by the increasing concentration of chitosan in composites. All the characteristics of the composites provide evidence for the miscibility of chitosan and cellulose. Their biocompatibility is proved through our published data. It is strongly indicated that bacterial cellulose–chitosan nanocomposites have great potential in tissue engineering or pharmaceutical applications in the near future.
Introduction
Cellulose is the most abundant polymer in nature and has been widely used in paper, paint, textile, food, pharmaceutical industries, and so on. Cellulose can be classified by its origins, including vegetal, animal, and microbiological. In our study, bacterial cellulose (BC) is used for preparing nanocomposites with chitosan. BC is produced by the gram-negative bacteria, typically Acetobacter xylinum (or Gluconacetobacter xylinum), which is chemically identical to cellulose of vegetable origin, 1 while it is of high purity, free of hemicellulose and lignin. The BC pellicle comprises a random assembly of microfibrils of less than 100 nm diameter, which are much finer than plant cellulose. 2,3 High purity and tremendous water holding capability add to its superior biocompatibility, which favors the use of BC in the pharmaceutical, food, and cosmetic industries. BC has also recently attracted a great deal of attentions for biomedical applications. 4,5 Furthermore, A. xylinum can be readily adjusted when it is biosynthesized. For example, we can synthesize different high-order (or senior) structures through dynamic and static culture modes. Meanwhile, BC can also be modified in situ by some additives to G. xylinum growth media, such as water soluble polysaccharides, 6 carbon nanotubes, 7 lignosulfonate, 8 polyaniline, 9 and so on, in terms of its morphology, crystalline index, water uptake capability, 10 and electroconductivity. 9 Additionally, BC does not contain any components of animal origin which may cause allergic reactions. 11
Chitosan (poly(1,4-β-
Since the chemical structure of chitosan backbone is very similar to that of cellulose, it was expected that chitosan could be miscible with cellulose and the resulting mix might gain the beneficial properties of both BC and chitosan. Several methods were adopted to prepare different forms of BC and chitosan composites. Biocomposite films were produced by dissolving cellulose and chitosan in 1-butyl-3-methylimidazolium acetate (BMIMAc) and then casting the films on flat surfaces. 21 Similarly, cellulose and chitosan were dissolved in a binary system consisting of acidic ionic liquid glycine hydrochloride (Gly·HCl) and neutral ionic liquid 1-butyl-3-methylimidazolium chloride ([Bmim]Cl); chitosan/cellulose composite fibers were fabricated by spinning the solute. 22 Chitosan/cellulose fiber foam composites can be produced by freeze-drying cellulose fiber/chitosan suspension. Silver ions were then loaded by in situ reactions. 23 Bacteria cellulose films were modified by adding chitosan into the culture medium to produce BC/chitosan films. 24 Low-molecular-weight (MW) chitosan acetate and chitosan lactate were used to obtain BC/chitosan composite materials for medical applications. 25
Water soluble polymers have been included in the growth media of cellulose producing bacteria to get in situ composites, such as starch, 26 carboxymethylcellulose (CMC), methylcellulose (MC), and poly(vinyl alcohol) (PVA). 27 It was found that the additives were incorporated into the growing cellulose fibrils, resulting in composites. However, some researchers claim that the inclusion of water soluble polysaccharides, such as CMC, MC, agar, and sodium alginate, bring about altered cellulose structure and other modifications, higher water retention ability, greater thermal stability, and so on. 28
Besides water soluble polymers, water insoluble PHB (poly-3-hydroxybutyrate) has been included in the medium to produce in situ BC/PHB composites. 29 PHB was able to be incorporated into the BC fibrils during fermentation.
In this article, a novel BC/chitosan bio-hydrogel was prepared in situ by supplementing chitosan chloride in the culture medium. The relative content of chitosan chloride and BC was determined. Its morphology, structure, and crystallinity were characterized by Fourier transform infrared (FTIR) spectroscopy, scanning electron microscope (SEM), atomic force microscope (AFM), X-ray diffraction (XRD), thermogravimetric analysis (TGA), and so on. Also, its bacteriostasis was tested against Escherichia coli. In addition, to assess further application as a tissue engineering scaffold, the bionanocomposite hydrogel was evaluated on in vivo and in vitro biocompatibility, including hematolysis, systemic allergy, pyrogen test, conjuctiva stimulation, and acute toxicity, as well as the effects on rat fibroblast (FB) and keratinocytes (KC) cells for cell compatibility. 11 Excellent biocompatibility was verified.
Materials and methods
Materials
G. xylinus (CGMCC NO. 2955) 29–30 used in this study, as described previously, was screened from a traditional Chinese drink by our group and stored in the China General Microbiological Culture Collection Center with the registered number, no. 2955.
E. coli was obtained from the Biochemical Engineering Laboratory of Tianjin University of Science and Technology, Tianjin, China.
The chitosan chloride was purchased from the Golden-Shell Biochemical Co. Ltd, China, with MW 19.8 kDa and deacetylation degree 90%.
The cellulose medium contains 20 g L−1 anhydrous dextrose, 5 g L−1 yeast extract, 5 g L−1 peptone, 5 g L−1 disodium phosphate, and 10 g L−1 citric acid, and the pH was adjusted to 6.0 by acetic acid. Then, it was sterilized at 120°C, 0.1 MPa for 20 min.
Bacterial Luria-Bertani medium (LB) medium contains 5 g L−1 yeast extract, 10 g L−1 peptone, and 10 g L−1 sodium chloride, and the pH was 7.2–7.4. It was then sterilized for 20 min at 0.1 MPa, 121°C. Solid culture medium was obtained by adding 2% agar in LB medium.
Preparation of BC/chitosan composites
G. xylinus was activated in cellulose medium at 30°C for 24 h. 31 Then, G. xylinus suspension was inoculated at 5% (v/v) in the same medium; the medium was supplemented with chitosan chloride at concentrations of 5, 8, and 10 g L−1, respectively. BC pellicles and BC/chitosan composite pellicles were formed after 10 days. They are named as BC, CBC5, CBC8, and CBC10, respectively. The harvested gel-like membranes were purified by washing with deionized water and then by boiling in 0.1 M NaOH to remove remaining cells. Finally, they were washed till neutral (pH = 7).
Scanning electron microscope
SEM (JSW-5500LV; JEOL Ltd, Japan) was used to examine the surface morphology of the materials. The freeze-dried samples were sputter coated with a thin layer of evaporated gold. All micrographs were taken at a magnification of 5000 K and an accelerating voltage of 25 kV.
Atomic force microscope
The morphology of freeze-dried BC and BC-chitosan membranes was observed with an atomic force microscope (JSPM-5200; Japanese Electronics Co., Japan). Ambient atmosphere measurements were made in tapping mode.
Fourier transform infrared spectroscopy
Samples of 0.5–2 mg were mixed with 100–200 mg potassium bromide; then, the mixtures were pressed into tablets. The spectra were recorded using FTIR spectroscopy (Vector 22; Bruker Instrument Corporation, Germany), at wave numbers ranging from 4000 to 400 cm−1.
X-ray diffraction analysis
Measurements were performed using XRD (RIGAKU D/MAX-2500; Shimadzu, Japan). The intensity of a Cu–Pd filter, 36 kV × 20 mA, was measured in a 2θ range between 5° and 70°. The crystalline index was analyzed with the help of an MDI/JADE6 software package attached to the Rigaku XRD instrument (Japan). It is calculated by the following equation 32
where A t is the total area of crystal and amorphous diffraction and A am is the area of the amorphous diffraction. Crystallite size was calculated using the software Jade 5.0. The full width at half maximum height for the two major peaks was used for this calculation. All collected data were normalized to the same baseline and shifted along the intensity axis for a better comparison of final results.
Thermal analysis
TGA and differential thermal analysis (DTGA) data of all samples were carried out using a SDT Instrument Q600 thermal analyzer (USA). Scan rates of 10°C min−1 over a temperature range of 20°C and 500°C were applied. All specimens were in sheet form and tested under an inert (N2) atmosphere flowing at 100 mL min−1.
Antibacterial properties of BC/chitosan composites
Testing of antibacterial activity of the chitosan membranes was performed according to the method described by Wu et al. with minor modification. 33 The samples used for the antibacterial assay were sterilized at 121°C for 30 min. In the antibacterial assay, 1-cm2 sterilized test samples were placed in test tubes. Subsequently, 10 mL of bacterial broth culture was seeded onto the BC, CBC5, CBC8, and CBC10 membranes (106 CFU mL−1), respectively. The bacterial broth without containing any material was used as control. The bacteria used for the test were E. coli. Subsequently, the membranes were put into the moisture incubator and incubated at 37°C. After 12-h incubation, the medium was diluted to a certain concentration and seeded on agar plates containing nutrient broth and incubated at 37°C for 12 h. Finally, the units of colony formation in each agar plate were calculated to examine the antibacterial ability of each BC and CBC membrane.
Results and discussion
Morphology and microstructure
Chitosan is insoluble in water, due to its intensive intermolecular forces; it can be soluble only in organic or inorganic solutions. Its hydrochloride derivative shows reliable water solubility. However, chitosan chloride did not completely dissolve in culture medium, which was attributed to the almost neutral environment (pH = 6). Flocculent precipitates appeared; the precipitates were so tiny that most of them dispersed in the medium. Very little particle settled at the bottom of the flasks. At the end of the fermentation, the pH dropped to 4.8, so that part of the solid chitosan hydrochloride became soluble. This is different from the work of Phisalaphong and Jatupaiboon 24 ; low-MW chitosan (MW 30,000 and MW 80,000) was added to culture medium at low initial pH (pH 3–4), where chitosan was soluble. In our study, gel-like BC/chitosan membranes were harvested and named as CBC5, CBC8, and CBC10, respectively, corresponding to the chitosan content of 5, 8, and 10 g L−1 in the medium. Our work was different from that of Ruka et al. 29 ; water insoluble polymer PHB resided at the bottom of the growth medium. PHB was incorporated into the BC fibrils during production, although BC pellicle was formed on the surface of the medium. The integration of chitosan and BC maybe attributed partly to the electrostatic attraction. In the culture medium, chitosan was cationic, while BC was positive charged; according to our published data, the zeta potential of BC fibril suspension was −13 mV. 34
Pristine BC membrane was translucent and soft, as shown in Figure 1; while CBC membrane showed yellowish and rigid, due to less water content than that of BC (data not shown). Typical interwoven mesh of BC fibrils network was observed in both the SEM images of Figure 2. The average fibril diameter of BC measured by Image J1.48 is 71 nm, which is a nanomaterial. The inclusion of chitosan hydrochloride, however, resulted in some changes in the morphology of CBC5 (Figure 2(b)), since chitosan was integrated into the fibrils during fermentation. Moreover, chitosan as a coating covered the fibril network of CBC membrane and filled the voids between fibrils. The average fibril diameter of CBC5 is 107 nm, due to the adhesion of chitosan. Specifically, in Figure 2(b), it is clearly shown that one single fiber had covered and uncovered parts along its length. The partially chitosan filled voids led to different density and porosity of CBC. Consequently, the final network retained less water and was, therefore, relatively rigid.

Photos of (a) pristine BC and (b) CBC5. BC: bacterial cellulose.

SEM micrographs of (a) BC and (b) CBC5. BC: bacterial cellulose; SEM: scanning electron microscope.
Through the AFM image (Figure 3(a)), the morphology of the specimens was observed. As shown in Figure 3(a), the mean diameter of BC fibers was around 50 nm, which was in line with According to Ross et al.’s data, 10,35 the morphology of CBC exhibited remarkable differences from that of BC. Figure 3(a) clearly shows a typically defined fibril network, while CBC shows aggregates of chitosan and BC and enlarged cellulose diameters. It is shown in Figure 3(b) that the network of CBC was more closely knitted, which was also reported in Huang et al.’s study. 10 It is also obvious that the surface of CBC is coated by a layer of chitosan. The film of pristine BC shows a relatively smooth roughness of 321 nm (Figure 3(a)). Along with the incorporation of chitosan, the resulting film becomes rougher, with the roughness for CBC5 of 612 nm (Figure 3(b)), which is consistent with the work of Xu et al. 36

The AFM graphs of (a) BC and (b) CBC5. AFM: atomic force microscope; BC: bacterial cellulose.
From the morphology presented by SEM and AFM graphs, the intertwining of chitosan and BC led to an interpenetrating network, suggesting that the two polymers were compatible well with each other, and structurally cooperative. 37
Fourier transform infrared spectroscopy
The FTIR spectroscopy of the chitosan in Figure 4(a) shows that the absorption peaks at 1624, 1521, and 1320 cm−1 corresponding to the amide I, II, and III bands, respectively. The BC spectroscopy in Figure 4(b) shows that the absorption band at about 1430 cm−1 is attributed to the symmetric bending of CH2 and 1658 cm−1 is the glucose carbonyl of cellulose, which is in accordance with Barud et al.’ study. 38 The band at 1060 cm−1 is due to C–O symmetric stretching of primary alcohol; and the bands at 1163 and 2919 cm−1 correspond to C–O–C asymmetric stretching and C–H stretching. The peak at wave number of 3346 cm−1 is ascribed to O–H stretching frequency. 39 From the spectra in Figure 4, it is indicated that CBC is mainly dominated by its cellulose component, with the new peaks at 1600 and 1320 cm−1, which correspond to typical amide I and III bands from chitosan, respectively. This indicates that the CBC is a composite of BC and chitosan. It is noticeable that the amide I band shifts from 1624 to 1600 cm−1 in the composite CBC. This might result from the interaction between the component polymers, confirming the complete miscibility of two polymers. Earlier reports have also drawn similar conclusions. 24,40

The FTIR of chitosan chloride, BC, and CBC5. FTIR: Fourier transform infrared spectroscopy; BC: bacterial cellulose.
The broader O–H peak implies that the hydrogen-bonding strength in CBC was reduced, 10,41 since the hydrogen bonds between cellulose molecules were interfered with chitosan molecules. The weakening of these hydrogen bonds in CBC reflects the decrease of their crystalline regions, and this will be discussed later.
X-ray diffraction
The XRD patterns of BC, CBC5, CBC8, CBC10, and chitosan hydrochloride are shown in Figure 5. In the case of pristine BC, two characteristic peaks at 14.74° and 22.64° were to crystalline plane (110) and (002), respectively, 42,43 which is an attribute of cellulose IⅠ. BC is not a completely crystalline material, so that broad diffraction peaks were observed. 43 The diffractograms of composite CBC5, CBC8, and CBC10 show little difference to that of BC and slight variation in intensity accompanying angle changes. According to equation (1), the inclusion of chitosan hydrochloride causes a decrease in the crystallinity index (CrI), as shown in Table 1, which is in agreement with the results of others’ work. 10,24 Chitosan molecules were too large to enter the crystalline zone between subfibrils due to steric hindrance. They stuck discontinuously on the surface of microfibrils and interfered with the crystallization of BC by preventing the aggregation of microfibrils and the formation of ribbons during in situ biosynthesis. As a result, crystallization was weakened and reduced.

XRD patterns of BC, CBCs, and chitosan chloride. XRD: X-ray diffraction; BC: bacterial cellulose.
Crystallite size and CrI of BC and CBC composites.
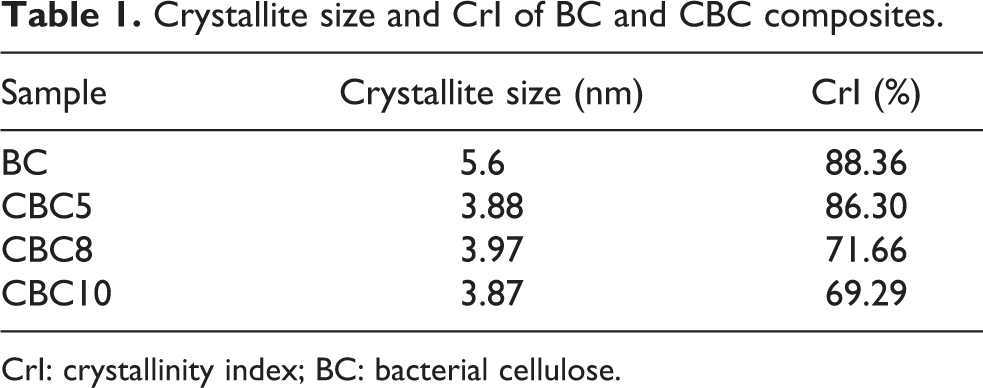
CrI: crystallinity index; BC: bacterial cellulose.
The crystallite size of BC was 5.6 nm (shown in Table 1), and the integration of chitosan hydrochloride resulted in smaller crystallite sizes, ranging from 3.87 to 3.97 nm, further verifying its interference in the polymerization and crystallization process.
TGA test
TGA was used to understand thermal decomposition behavior of BC and CBC composites. Their TGA and derivative thermogravimetric (DTG) curves are depicted in Figure 6, and the corresponding data are listed in Table 2; the onset temperature of thermal degradation is represented at 5% weight loss. 21 The DTG peak (Figure 6(b)) appeared at 328.36°C, due to a cellulose degradation process such as depolymerization, dehydration, and the decomposition of glucosyl units followed by the formation of a charred residue. 44 –46 Chitosan was less stable than BC and CBC composites, with the peak maximum temperature at 224.27°C. All of the CBC composites showed two peaks; degradation started at lower temperature and occurred over a broader temperature range. The initial decomposition appeared at a temperature range of 302–305°C, and the second decomposition appeared at 343–353°C, suggesting less thermal stability than BC. The reduced thermal stability of CBCs, compared with BC, might be the result of a weaker hydrogen bonding and decreased CrI, as discussed above. It implies that CBC composites decomposed slower than BC. Neither of the peak temperatures of CBC composites matches the temperatures of peak decomposition rates of neat polymers, implying certain interactions established between the two polymers. 21

TG and DTG curves for BC and CBC composites. TGA: thermogravimetric; DTG: derivative thermogravimetric; BC: bacterial cellulose.
Summary of the main degradation (onset and peak temperature) of the samples.

Bacteriostatic test
It is known that chitosan has intrinsic antimicrobial properties. 47 The antimicrobial effect relies on the interaction between polycationic chitosan and anionic groups on the microbial cell membranes, causing the leakage of intracellular constitutes. 48 –50 Besides, chitosan can be a chelating agent to bind metals to inhibit the microbial growth and toxin production. 51 Its antibacterial activity is relevant to the concentration and MW. It has been proposed that the antimicrobial model of chitosan is to attach on the surface of bacteria. 52,53 In our study, the bacteriostatic activity of CBCs against E. coli (gram negative) was determined, and the results are presented in Figure 7. CBC materials showed moderate bacteriostatic activity against E. coli; the inhibition effect of bacteria growth was significantly enhanced according to the increase of chitosan content in the materials. It is reported that at the concentration of 20 ppm, all chitosan samples of different MWs had the stimulative effect on the growth of E. coli; while the concentrations exceeded 200 ppm, all chitosan samples could kill almost all of the bacteria. 52 Therefore, in order to reliably promote the antibacterial activity of CBCs, we recommend increasing the content of chitosan in CBC materials.

Bacteriostatic tests of BC and CBC materials. BC: bacterial cellulose.
Conclusions
Composites of chitosan chloride and BC were successfully prepared by in situ method during biosynthesis. Composites of BC/chitosan and pristine BC were investigated by means of SEM, AFM, FTIR, DSC, XRD, and bacteriostatic test. Amide I and III bands appeared in FTIR spectra of the composites compared with BC, proving the integration of chitosan and BC. The composite retains the fine network structure of BC, while the diameter was enlarged and the aperture was smaller than BC in the SEM and AFM graphs. The decreased CrI and crystallite size as well as thermal stability against BC were probably due to the interfered and weakened hydrogen bonding during fermentation. Thermal degradation behavior of CBCs suggests that the interactions existed between BC and chitosan chloride. The antimicrobial activity of CBCs was enhanced by the introduction of chitosan and showed dose-dependent effect.
The composite membranes had better effect on cell growth, cell proliferation, and biocompatibility over BC. 11 The prepared CBC nanocomposite was bioactive and its potential as scaffold materials was recognized.
Footnotes
Acknowledgements
The authors are grateful for the financial support from the National Natural Science Foundation of China as well as Natural Science Foundation of Tianjin.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has received financial support from the National Natural Science Foundation of China (project no. 21106105 and 21576212) as well as Natural Science Foundation of Tianjin (15JCZDJC32600).

