Abstract
Self-healing fabrics have garnered a lot of attention due to their recovering functionality upon damage. This work describes a facile technique for developing a novel self-healing coating with the goal of producing autonomous intrinsic self-healing fabrics that can recover from damage at room temperature without the use of external stimuli. The coating was developed using natural rubber latex (NRL) and consisted of a dynamic reversible metal thiolate ionic network. The formation of the reversible ionic network was assessed by Differential Scanning Calorimetry (DSC), Ultraviolet-visible spectroscopy (UV-vis), Fourier Transform Infrared (FTIR) and zeta potential analysis. Scanning electron microscope (SEM) images revealed that the coating impregnated the fibres of the fabric and improved their structural integrity. The morphology of the punctured area revealed that intermolecular diffusion had occurred during the recovery and the sample had completely healed. The results also showed that the tensile strength, tear strength and puncture strength of the fabric achieved 100% healing efficiency when the damaged fabrics were brought into contact with each other and allowed to be healed at room temperature. This technology is expected to open up a new avenue in the textile industry.

Keywords
Introduction
Projections prior to the outbreak of COVID-19 forecasted that rising levels of disposable income, population growth and rapid urbanisation would lead to the expansion of the global textile and apparel industry. Recently, both the scientific and industrial segments have been increasingly using innovative fabrication and production methods of smart textiles such as superhydrophobic,1,2 radiation-shielding,3,4 flame-retardant5,6 and conductive fabrics.7,8 One of the most intriguing techniques in the recent development of smart, sustainable fabrics is the emergence of the self-healing idea, which involves applying an innovative coating technology to fabrics. This idea enables fabrics to recover their functionality and durability upon damage without needing to be identify or repair by any kind of manual intervention.
To establish the smart coating idea and improvise fabric durability, researchers have investigated the extrinsic self-healing approach, in accordance with incorporating encapsulated self-healing agents and self-healing superhydrophobicity in the fabric. 9 David et al. discovered the enzyme encapsulation of a squid ring teeth (SRT) proteins – polystyrene sulfonate (PSS) base as a self-healing agent for fabrics. They introduced layer-by-layer (LbL) methods to fabricate SRT-containing multilayer films, with the results showing that this was a feasible method of introducing healing capability to conventional textiles. 9 In both dry and wet environments, the SRTs containing structural proteins had a high elastic modulus (>2 GPa), leading to their excellent mechanical properties. The principles behind SRT protein’s self-healing ability are the deformation and softening of its structure in the water above the glass transition temperature while it reversibly maintains its hydrogen bonds in the amorphous region. 10 The major drawback of the extrinsic self-healing mechanism is the non-recurring healing action, as the impairment of a specific location will only be restored once. Due to the capsule size, the amount of available healing agent is also limited. 11 Prior to the limitation, the aim of this study is to create repairable healing action by producing autonomous intrinsic self-healing fabrics that can recover from damage at room temperature without the use of external stimuli.
Li et al. utilized fluroalkylsilane on the fabric surface at ambient temperature to develop a self-healing superhydrophobic coating. They found the superhydrophobic coating could autonomously recover numerous times without reducing the superhydrophobicity. 12 Chen et al 5 demonstrated a simple solution-dipping process on cotton fabric that included flame-retardant and self-healing superhydrophobic coatings. They explained that the durability and healing efficiency were recovered when the fabric had been exposed to repeated abrasion. Meanwhile, Wu et al. investigated radiation-induced graft polymerisation approaches to create a self-healing superhydrophobic cotton fabric. Upon sustaining mechanical damage, the superhydrophobicity could be recovered by steam ironing. 13
Moreover, Sun et al. utilised the chemical vapour deposition (CVD) of fluoroalkylsilane in porous materials to create a self-healing superhydrophobic coating. After being disrupted, the superhydrophobicity was restored spontaneously as a result of the fluorinated material migrating to the surface. 12 While numerous ways for creating self-healing superhydrophobic materials have been effectively investigated, significant drawbacks still limit their practical implementation. The dispersion of the hydrophobic substances used to restore damaged surfaces is frequently hazardous, pricey and irritating to the skin. Furthermore, long-chain perfluorinated compounds have a well-documented capacity to bioaccumulate, as well as the potential to impact negatively on human health and the environment.14–16 Furthermore, their commercial applications have been limited due to their high costs and time-consuming processes, while a long-term self-healing process is also required. As a result, developing a low-cost, environmentally friendly, and time-saving approach for producing superhydrophobic materials that are easily repairable and have rapid self-healing properties is crucial. In creating an environment with zero pollution is impossible but reducing it is a must. Therefore, the materials employed in this research are carbon-neutral and sustainable, that could lead to new developments in the field of self-healing materials.
In this work, the authors explore a latest method for the facile fabrication of self-healing fabrics using a natural rubber latex(NRL) coating. Numerous existing works have reported the usage of NRL as a coating material, especially for fabric applications.17–19 NRL is an aqueous suspension of colloidal rubber particles with an excellent rigidity-to-elasticity ratio. 20 The NRL has piqued the attention of several researchers as a coating component for coated fabric. For environment issue and health issues, the self-healing natural rubber coating on fabric has minimal risk of allergic reaction or sensitisation on skin and upon contact with water the pH will not change as the materials is fully cured. Rupayan et al. 21 reported that fabrics coated with NRL exhibited higher tensile strength and modulus than a neat fabric. In their work, various concentrations of NRL solutions (20% and 30%) were incorporated into p-aramid (Kevlar 129) and ultra-high-molecular weight polyethylene (UHMWPE) fabrics. The result showed that the yarn pull-out force of the NR-coated p-aramid fabric was significantly higher than that of the neat p-aramid fabric. The single-layer NR-coated p-aramid fabric performed poorly in terms of peak force generated and energy absorbed when compared to neat p-aramid. This could have happened because the metal-to-yarn friction was reduced.
Ahmad et al. 22 reported that the shear behaviour and yarn pull-out strength of Kevlar textiles covered with natural rubber latex demonstrated larger shear angles than uncoated fabrics. This improvement in mechanical properties was attributed to the capability of NRL to restrict yarn movement and group the yarns together to withstand the external work force of yarn tugging or shearing before they entirely failed at a specific point. The NRL layer’s presence on the fabric surface also contributed with increasing force loads and yarn–yarn and yarn–NRL friction. However, NRL is non-polar and has no accessible functional groups that can be converted into reversible physically or chemically interactions, hence developing self-healing NRL is a challenging task. 23 In addition, fresh NR latex spontaneously coagulates and becomes purified soon after rubber tapping; high ammonia (HA) levels are required for preservation. This is a general practice for latex industry to prepare the NRL suspension. Ammonia is the primary preservation in both field and concentrated latex concentrates. The high ammonia is used to inhibit bacteria growth in the latex and to prevent natural coagulation in latex. The ammonia was in liquid state in the rubber. The high ammonia natural rubber latex (HANRL) is not expected to influence self-healing capability of the material but more into maintaining latex stability. 24
Herein, the authors propose a rapid and simple route to fabricating self-healing fabric that can be easily repaired at room temperature upon damage. This technique was stimulated by combining the current vulcanizing system and the rubber tree. NRL consists of organelles called ‘lutoids’. When a rubber tree is damaged, the lutiods burst, releasing the protein Hevein, which then forms hevein-dimers under the influence of Ca2+ ions. The hevein-dimer then bonds to the rubber particles onto damaged surface, prohibiting the tree from producing any more latex. Zn2+, which can form a reversible network provides self-healing powers to natural rubber and sulphide molecules, which, it has been claimed, attach strongly to the rubber molecule chains, was used in a recent study, in a similar way to the role of Ca2+ in Hevein25,26 In the current work, the authors synthesised and characterised self-healing high ammonia natural rubber latex with variable zinc thiolate content to assess the feasibility of impregnating synthetic polypropylene (PP) fabric with the self-healing latex. This self-healing fabric autonomously repaired itself at room temperature, giving it long-term reliability and making it ideal for developing sustainable fabric technology.
Methodology
Materials
Synthetic polypropylene (PP) fabric was used in this research. The fabric morphology is shown in Figure 1. The PP fibers are randomly oriented. High-ammonia natural rubber (NR) latex was provided by Zarm Scientific and Supplies Sdn. Bhd. (Penang, Malaysia). Zinc oxide (ZnO), zinc thiolate (ZT), oleic acid (OA), potassium oleate (PO) and potassium hydroxide (KOH) were supplied by Bendeson Laboratory (Johor, Malaysia) and discumyl peroxide (DCP) was acquired from Sigma-Aldrich Ptd. Ltd. (Malaysia). The ingredients used to fabricate the latex coating in this work is similar to the formulation and ingredients to fabricate latex gloves and commercial latex coating for fabrication of stretchable fabric. The different is composition of the ingredients which enables reversible self-healing crosslinks. Morphological properties of non-woven PP fabrics.
The raw materials used which is NRL is tree-derived rubber and it is eco-friendly. Harvesting and using the product itself has less impact on the environment. The Rubber tree is a sustainable crop and helps maintain the global carbon balance in the atmosphere. The natural rubber resource is safe for children and other users since it is proven to be hygienic, non-toxic and antibacterial as well as anti-dust mite. The material is widely use worldwide to manufacture rubber glove, medical products, mattress and others.
According to the European Commission Health and Consumer Protection Directorate, the use of natural rubber is safe although there are few reports highlighted that some proteins in latex can cause mild skin allergic reactions. The chemicals present in the latex compound for manufacturing of the commercial latex products which is similar to the chemicals used in this work is not toxic and have minimal risk on human body. 27
Preparation of self-healing high ammonia natural rubber latex and fabric-coating method
Formulation of self-healing latex with various ZT content.

aAmount of ingredients is in parts per hundred rubber (phr).
Table 1 shows the formulation to prepare and compound self-healing HANRL with varying amount of ZT contents. Based on the formulation, the compounding ingredients were calculated and measured with a total mass of 500 g. The compounding ingredients are then added in sequence of pH modifier (KOH), stabilizer (PO), accelerator (ZT) dispersion, activator (ZnO) dispersion and DCP emulsion into HANRL. After all ingredients have been added, the self-healing HANRL is stirred homogenously up to 30 min at ambient temperature by mechanical stirrer. After being compounded, the latex is matured for 24 h at room temperature to allow the diffusion of all ingredients. Maturing method use is similar with previous work reported elsewhere.28,29 It would also be expected that maturing the latex compound at room temperature would prevent deionization of Zn2+ ions in zinc thiolate molecules for better formation of metal thiolate ionic network during curing process of self-healing HANRL.
Next, the coating method of PP fabric started with coagulant dipping. The PP fabric was firstly coated with coagulant solution. Calcium nitrate is used for forming a coagulant layer so that the self-healing HANRL able to achieve and impregnated into the fabric. The coagulant dipping process of PP fabric in calcium nitrate solution was carried out for 30 s. Then the PP fabric was continued to air dry in room temperature for 10 min. Subsequently, the PP fabric was dipped into the self-healing HANRL for 30 s. The coated PP fabric was placed inside oven for post-curing at 120°C for 40 min then cooled at room temperature for 24 h. The sample was tested with different numbers of dip-coating layers (single, double, triple and quadruple) to study the effects of different coating layers’ thickness on the self-healing fabrics. Then, the synthetic PP fabrics were characterized and evaluated based on performance and healing efficiency.
Characterisation of self-healing performance
Differential scanning characterisation
Differential Scanning Calorimetry (DSC) was used to assess the exothermic reaction during the vulcanisation of self-healing latex compounds using the Perkin Elmer Pyris model. The tests were carried out in a nitrogen atmosphere at a range of temperature of 26°C–220°C with a heating rate of 10°C/min. The controlled sample and different of zinc thiolate content of self-healing latex samples were used throughout the experiment.
UV-visible
Using a Cary 50 Conc UV-Visible Spectrophotometer in the range of 200–800 nm, the optical characteristics of the produced samples were studied using UV-visible absorption spectroscopy. The experiment was carried out on all of the self-healing latex samples with varying zinc thiolate contents.
Fourier transform infrared spectroscopy
Under attenuated total reflectance with a resolution of 4 cm−1, FTIR spectra were acquired using a Perkin Elmer model Fourier Transform Infrared (FTIR) Spectrometer. The latex film was reduced to 3 cm by 3 cm in size for FTIR. The self-healing samples' FTIR spectra were collected between 550 cm−1 and 4000 cm−1. All of the self-healing latex films were used in the experiment, each with a varied zinc thiolate content.
Zeta potential
The Malvern Analytical Zetasizer Nano ZS was used to determine the zeta potential. In this research, zeta potential was used to study the colloidal stability of the NRL with different zinc thiolate dispersion. One drop of HANRL was added to 200 mL of buffer solution to make the samples. The rubber latex was soaked in deionized water before being fed into the sample handling process for re-flushing and measurement.
Tensile testing
For self-healing HANRL coating fabric, tensile testing was conducted using dumbbell-shaped samples. At room temperature, the fabrics are tested at a crosshead speed of 10 mm/min. After failure, the two fracture surfaces were brought into contact with one another with a mild press and allowed to recover for 24 h. The fabrics subsequently retested under the same conditions after healing process had been accomplished at room temperature. The pre- and post-healing test findings were then compared and analysed. The experiment was repeated with single, double, triple and quadruple layers of self-healing HANRL fabric. The healing efficiency of self-healing HANRL fabrics were calculated using equation (1); where 
Tear test
For the tear strength of self-healing HANRL fabrics were evaluated with Instron 3366. The fabrics were cut in trouser shape for further testing. The fabrics were tested at room temperature and the crosshead speed was adjusted to 10 mm/min. After failure, the fragmented fabric surfaces were pushed together and left to heal at room temperature for 24 h. The experiment was repeated with single, double, triple and quadruple layers of self-healing HANRL fabric. The healing efficiency of self-healing HANRL fabrics were calculated using equation (1).
Puncture test
The puncture resistance of the fabric is determined by measuring the force required to penetrate each sample in a puncture test. The puncture properties of the samples are evaluated at 10 mm/min crosshead speed. After failure, the fractured samples were placed together and gently pressed before undergoing the healing process for 24 h at room temperature. After the samples had healed, they were retested using the same method. The pre- and post-healing results were then analysed. The experiment was repeated with single, double, triple and quadruple layers of self-healing HANRL fabric. The healing efficiency of self-healing HANRL fabrics were calculated using equation (1).
Scanning electron microscopy
At a 15 kV accelerating voltage, scanning electron microscopy (Hitachi TM 3000) was utilised to investigate the morphology of the fractured and recovered surfaces of the self-healing fabric. The samples were positioned on aluminium stub and sputter-coated using gold-palladium in a vacuum prior to inspection. The surface morphology of the fractured areas before and after healing were observed to determine the relationship between healing efficiency and the morphological structure.
Results and discussion
Characterisation of self-healing HANRL coating
In this work, the core feature of preparing the self-healing HANRL was the introduction of a reversible ionic crosslink network into the natural rubber particles suspended in the latex dispersion. The strategy involved the vulcanisation of zinc thiolate into the rubber molecular chains through controlled DCP-induced vulcanisation. Peroxide free radicals preceded the primary reaction between the rubber chains and persulphenyl radicals derived from zinc thiolate decomposition to generate C-C bonding in between rubber chains and C-S crosslinks between the rubber chains, hence the zinc thiolate at the start of the vulcanisation process. Both of these connections, however, were irreversible covalent bonds. The self-healing ionic networks were thought to be formed by reversible ion couples between the Zn2+ and two S− on the vulcanised zinc thiolate on the rubber chain. Because of the strong electrostatic contact between neighbouring Zn2+ ions, this ion pair of Zn2+ and two S− resulted in the production of ion multiplets. Subsequently, aggregation occurred, forming ion clusters and larger ionic networks. The reversible ionic networks promoted the dynamic crosslink system and induced self-healing behaviour in the materials. In our previous research, the self-healing reaction mechanism between Zn2+ and S−, healing efficiency and the recovery mechanism has been investigated and reported elsewhere. 23 Scratches and cracks on the coating surface would damage both the ionic and covalent bonding; however, as the fractured area were being contacted, the ionic networks rearranged and reformed, leading to full material recovery at room temperature.
Interestingly, the strategy used here enabled the vulcanisation of zinc thiolate onto the natural rubber chains to be controlled before crosslinking between the rubber chains occurred in the presence of DCP. The curing behaviour of HANRL was investigated using DSC, the results of which are depicted in Figure 2. It is clear that the endothermic vulcanisation of HANRL/DCP occurred at a lower temperature than the samples containing zinc thiolate. It can be regarded that an increase in zinc thiolate content shifted the vulcanisation peak to higher temperatures. This observation reveals that the vulcanisation of zinc thiolate on rubber chains could be controlled and the formation of covalent crosslinks between rubber chains could be minimised by altering the curing temperature. The endothermic peaks were further analysed to investigate the energy of the curing reactions. The energy levels required for curing to occur for HANRL/DCP, HANRL/DCP/15ZT, HANRL/DCP/30ZT and HANRL/DCP/45ZT were found to be 916.23 J/g, 892.26 J/g, 350.96 J/g and 236.69 J/g, respectively. This finding implies that forming C-C covalent crosslinks between rubber chains used more energy than forming reversible ionic crosslink networks within the material. Ionic networks dominated the crosslinks inside the material, as evidenced by the decrease in energy with increasing zinc thiolate content. DSC curves of different ZT content in self-healing HANRL.
An intuitive observation was made of the types of crosslinks that occurred during vulcanisation. These were examined using UV-Vis spectroscopy on the self-healing HANRL, which had been cured at a representative DSC endothermic curing peak. As previously reported by Shuxun et al.,
30
UV-Vis spectra can be used as an efficient method of investigating the in-situ and real-time chemical crosslinks of gels or elastomeric materials. Figure 3 shows the absorbance against the wavelength of UV-Vis spectra for HANRL/DCP and HANRL/DCP, each with different zinc thiolate content. For the HANRL/DCP sample, it can be seen that the formation of covalent crosslinks in the presence of DCP did not strongly affect the absorbance peak over the wavelength range tested. This observation was similar to those reported in other works.
31
The absence of a peak in the covalently bonded rubber could be explained by the low maximum wavelengths of C-C and C = C in the rubber chains, which are difficult to detect with UV radiation. It was apparent that the absorbance peak in the 240–260 nm range for the HANRL/DCP/ZT sample increased with higher levels of zinc thiolate content. This demonstrated the greater zinc thiolate vulcanisation on the rubber chains that formed the C-S bonds. The ultraviolet absorption spectra of C-S bonds were previously investigated in detail and reported by Marvin and Edward.
32
Moreover, it can also be seen that the HANRL/DCP/ZT absorption peak tended to shift towards larger wavelength regions as the zinc thiolate content increased. This shift in the absorption maximum to a longer wavelength is typically attributed to the Bathochromic effect,
33
which is frequently explained by the presence of auxochrome groups. The combination of NH2 and Zn-S in zinc thiolate structures would increase the maximum UV-absorption value by extending the conjugation of the molecules through resonance. UV-Visible of different ZT content in self-healing HANRL.
FTIR measurement revealed the production of rubber-to-rubber crosslinks as well as zinc thiolate crosslinks with rubber chains, as illustrated in Figure 4. In the presence of DCP, the covalent crosslinks between the rubber-to-rubber chain may be detected at 1380 cm−1. The reduction of rubber-to-rubber crosslinks was suggested by the decrease in absorption at 1380 cm−1 with increasing zinc thiolate content. At 1000 cm−1, crosslinks between the zinc thiolate and the rubber chain were discovered. Apparently, the absorption band intensity increased between the use of HANRL/DCP/15ZT and of HANRL/DCP/45ZT, which was evidenced in the increase in the zinc thiolate crosslinks with the rubber chain. Both the rubber-to-rubber crosslinks and rubber-to-zinc thiolate crosslinks would have affected the out-of-plane bending vibration of the C-H and C = C of the rubber chain backbones, which was seen at the 876 cm−1 absorption band. Furthermore, the absorption bands at 1200 cm−1 and 1270 cm−1 showed the characteristics of phospholipids. Asymmetric and symmetric stretching suggested a possible interaction between the persulphenyl radical of zinc thiolate and the phospholipids.
34
It should be noted here that neutral lipids, glycolipids and phospholipids, carbohydrates, ash and other substances are all found in commercially available liquid rubber. Rubber particles in the latex dispersion were also attached to long-chain fatty acids, most likely found as the phospholipids bonded to the chain ends. FTIR spectra of different ZT content in self-healing HANRL.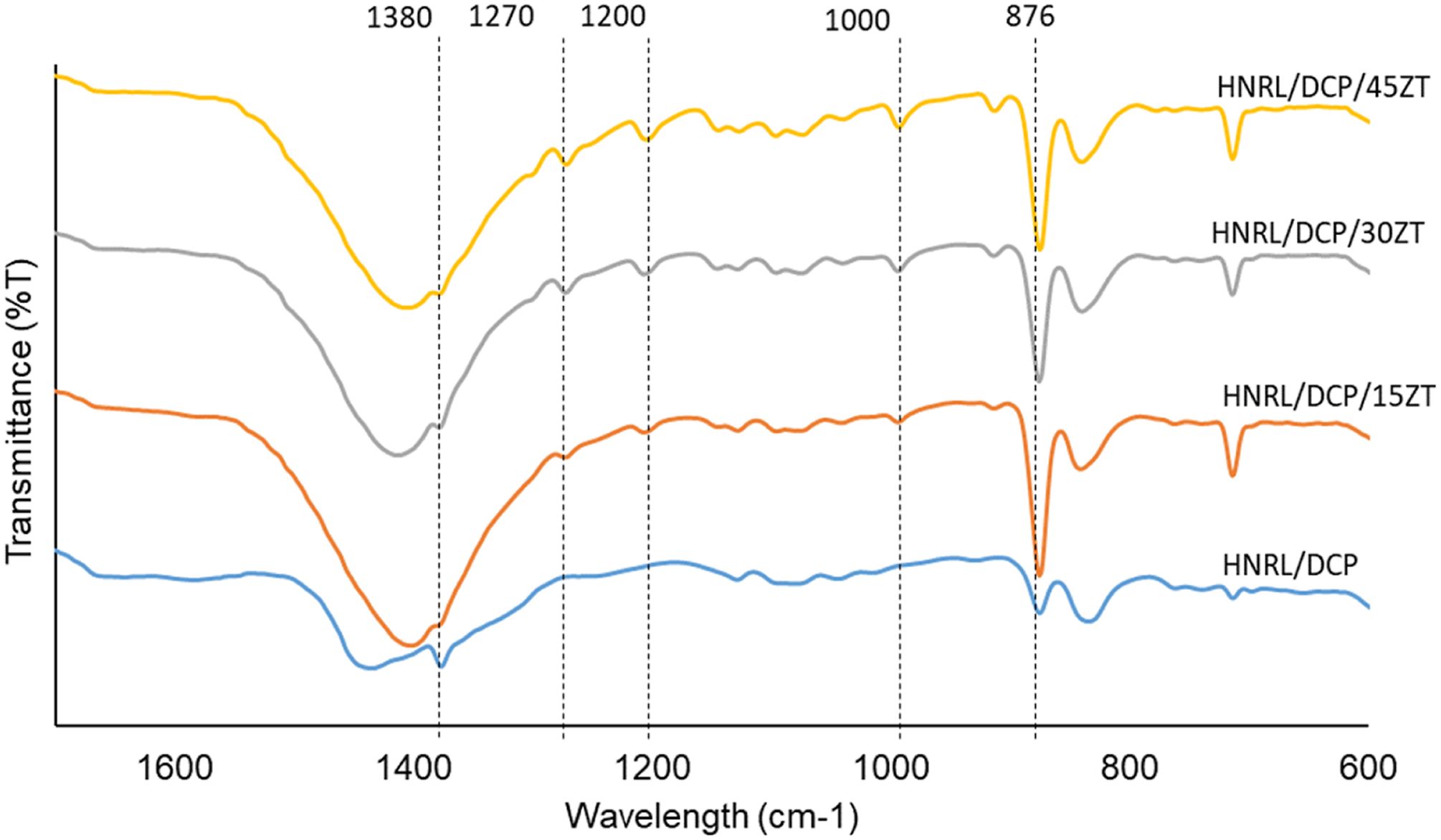
To assess the efficiency of coating and impregnating the fabric with self-healing HANRL, the colloidal stability of the dispersion was investigated using zeta potential. Figure 5 shows the zeta potential results for the HANRL latex samples with different zinc thiolate content. HANRL is colloidally stable due to having surface charges that are either −30 mV or higher than +30 mV.
35
The graph indicates that the negative zeta potential increased with the increase in ZT from 0 to 30 phr, while it decreased slightly for 45 phr of zinc thiolate content. This could be explained by the phospholipids, carbohydrates, proteins and other non-rubber ingredients contributing charge species to HANRL, resulting in significant negative zeta potential values.
36
The highest negative zeta potential for the HANRL with 30 phr zinc thiolate showed that the colloidal particles were stabilised, which thus inhibited particle aggregation, unlike with lower zeta potential values. Moreover, the presence of potassium oleate acted as a colloid stabiliser, preventing rubber particle coalescence in the controlled sample. Negative charges were generated that surrounded the latex particles, resulting in high electrostatic repulsion between them. The decrease in zeta potential for 45 phr zinc thiolate could be explained by the higher probability of latex particle aggregation at higher loading. The results revealed that the HANRL/DCP/30ZT sample showed optimum colloidal stability and thus should produce a good self-healing fabric. The mechanical performance of the self-healing fabric coated with HANRL/DCP/30ZT was investigated, as outlined in the following section. Zeta potential of different Zinc thiolate content in self-healing HANRL.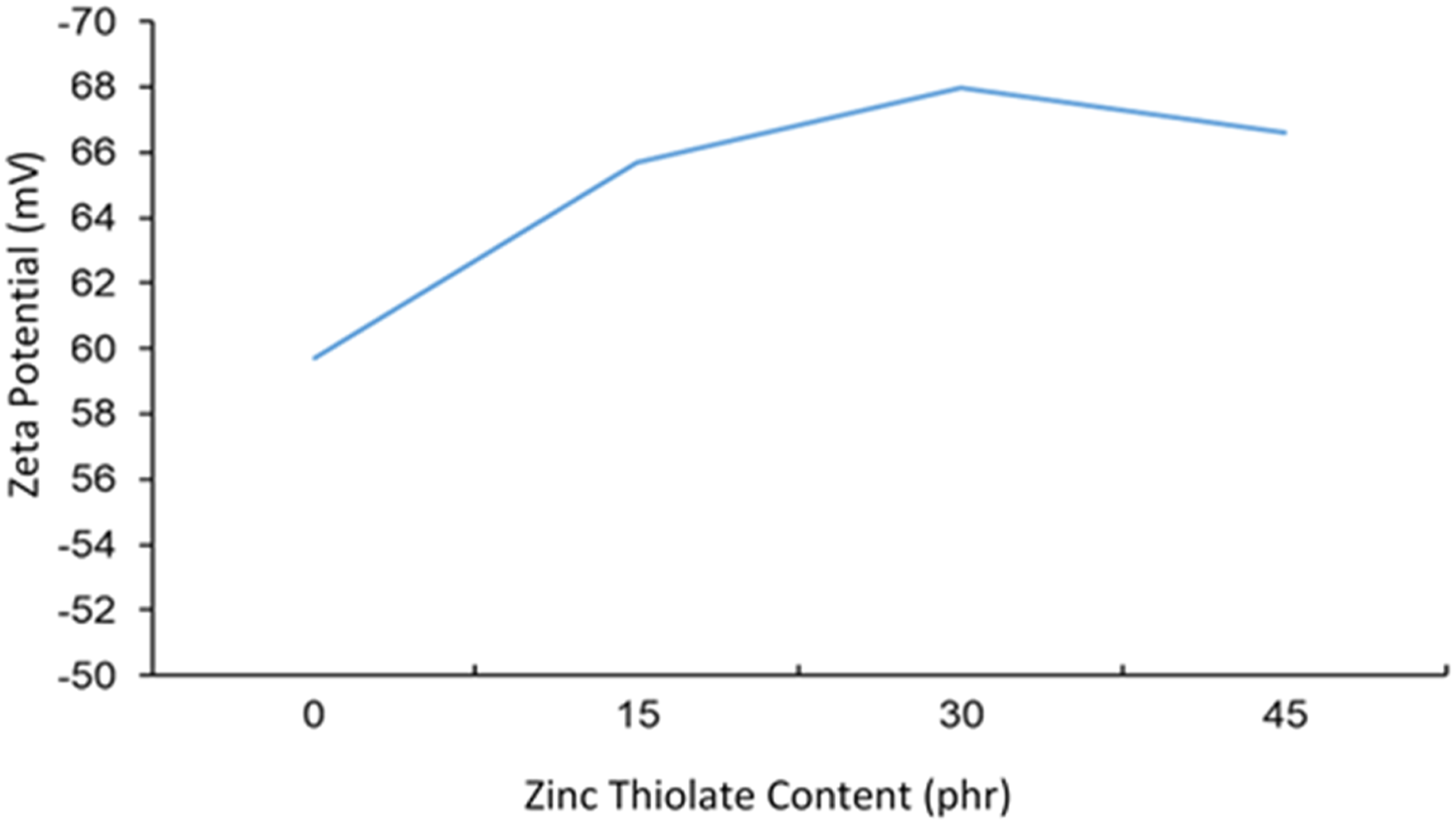
Evaluation of self-healing performance
Self-healing behaviour of the coated fabric is demonstrated in Figure 6. Figure 6(a) shows the appearance of the coated fabric. The fabric was cut into two and the healing was conducted by bringing both cut surfaces into contact with each other by gentle pressing at ambient temperature. After that, each sample was stretched and, as can be seen in Figure 6(c), the sample’s coherence was recovered. The morphological characteristics of the neat and coated fabrics were further investigated by SEM, the results of which are shown in Figure 7. As this figure illustrates, the neat fabric fibres were smooth and randomly oriented, with gaps between each fibre. After the fibres had been coated with self-healing HANRL using the dip-coating technique, the coating was impregnated into the fibre, also filling in the gaps between the fibres, as shown in Figure 7(b). The coated fabric images also show that the self-healing HANRL was homogeneously coated over each fibre, which is vital to ensure the material recovers fully during the healing process. In order to evidence the healing mechanism, the morphology at the fractured surface after two pieces of the broken samples were brought in contact with each other was analysed and the SEM images is shown in Figure 8. The SEM images clearly showed that the fracture surface was fully recovered without any obvious gap which suggests that the intermolecular surface diffusion occurred at the contact surfaces and reconstructed the metal ionic crosslinks to form a reversible ionic network that regain material’s original properties and healed. Visual inspection of self-healing fabric at room temperature: (a) preliminary sample, (b) cut sample, (c) healed sample under stretching, (d) damage surface of the healed sample. SEM images of the examined samples: (a) Uncoated sample, (b) Coated sample. SEM of self-healing coating: (a) broken sample and (b) healed sample.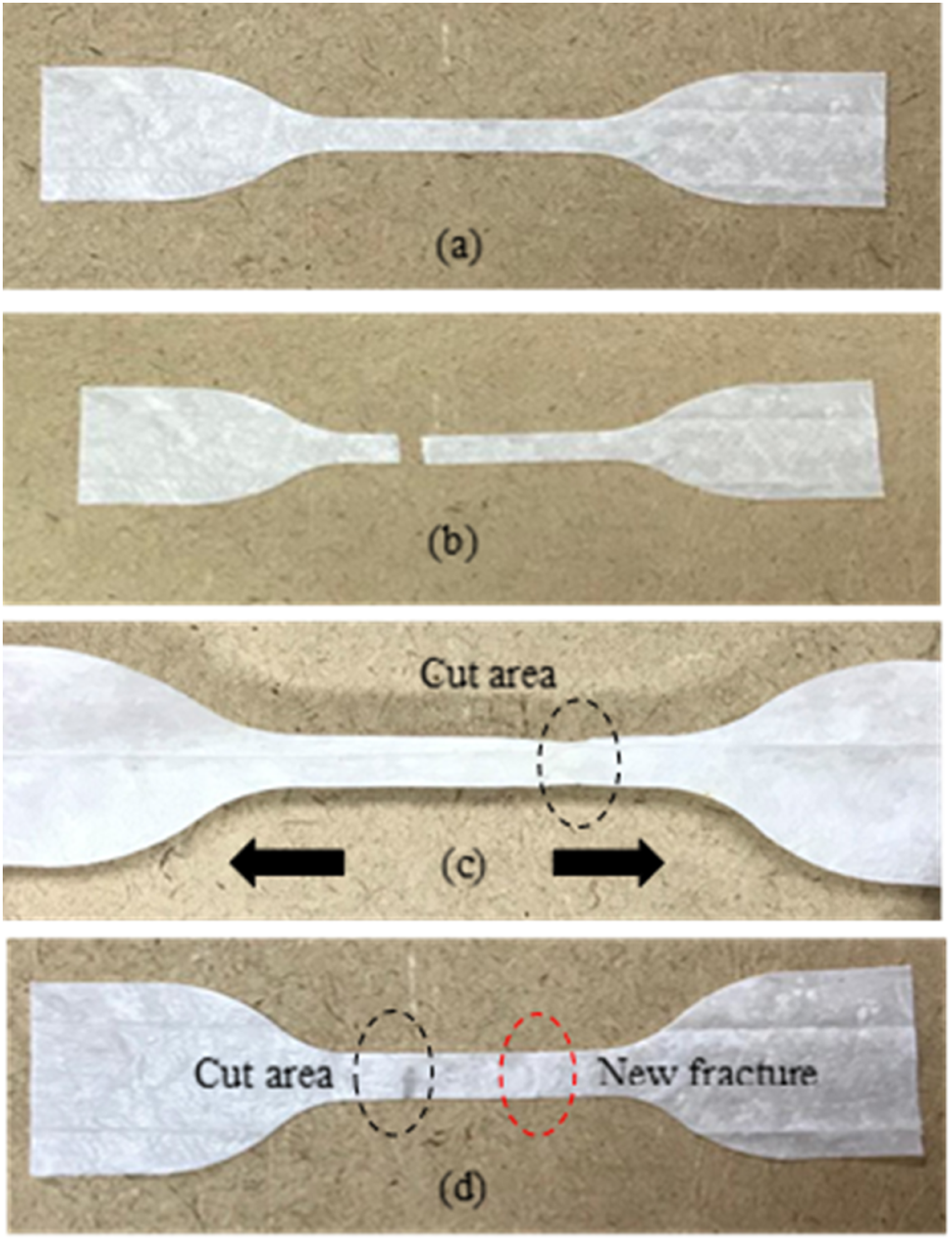


The proficiency of self-healing fabric was quantitatively evaluated in tensile experiments on the neat, coated and healed fabrics; the results of the average stress-strain curves are shown in Figure 9. As this figure illustrates, the fabric’s tensile strength and elongation at break increased substantially after it had been coated with HANRL/DCP/30ZT. The average improvements in tensile strength and elongation at break for the coated fabric were 19% and 52%, respectively. This clearly shows that the impregnation of the fabric with self-healing coating enhanced the adhesion and interaction between neighbouring fibres, which led to the formation of closed-pack fibre orientation. The closed-pack orientation of the fibres restricted the mobility and degree of freedom of the fibres, while it also improved the efficient stress transfer between the coated rubber and the fibres. Therefore, more energy was required to break down the sample, resulting in higher ultimate tensile stress. The improvement in elongation at break for the coated sample could be explained by the increase of slippage between the rubber molecular chains and the fibres during the deformation of the material. As previously observed in the SEM images, the neat fabric fibres were randomly oriented, with gaps between each fibre. The weak interaction between the neighbouring fibres limited the material’s flexibility during stretching, resulting in a lower extension of the sample before it broke. After the self-healing coating had been impregnated into the fabric, forming the closed-pack fibre orientation, the rubber molecules chain surrounding the fibre underwent coiling and uncoiling motions under stretching. This facilitated the slippage between the rubber chains and the fibres, resulting in the increased elongation at break. Stress versus strain curve of neat, coated and healed fabrics.
When each sample was broken down, both fractured pieces were contacted to each other and gently pressed in room temperature to allow the healing process to occur between the fractured surfaces. After that, each sample was retested. The results for the healed sample showed that the fractured sample capable to restore to its original properties, with a slight tensile strength increase of approximately 9%, compared with the initial coated fabric. From a phenomenological perspective, when the fractured surfaces were brought into contact, the molecular interdiffusion and rearrangement of the Zn2+ ion intermediates formed new molecular entanglements, promoting to the recovery of the fibres’ structural integrity. Although the tensile stress had improved, the strain at break results for the healed sample decreased by 20% compared with its initial properties. This strain at break decline could be explained by an increase in stress concentration points at the broken fibre intervals, as well as the formation of gaps between neighbouring fibres after the broken pieces had been brought into contact. These factors resulted in a lower extension of the fabric before it broke.
Figure 10 shows the tear strength of the neat, coated and healed fabrics. Similar to the tensile properties, the tear strength of the coated sample was found to be higher, at 41.09 N/mm, a 10% improvement compared with the neat fabric. The increase in tear strength could be explained by the increase in the fibres’ structural integrity after being impregnated with HANRL/30ZT/DCP. As previously shown in the SEM morphology, the coated fabric closed the gap between each individual fibre and restricted the fibres’ movement and orientation during deformation. As a result, higher stress was needed to initiate a tear and the del-zone, or the tear-opening region near the crack tip, would greatly deform.
37
The fibres’ capability to self-heal after the torn pieces had been brought into contact with each other is shown by the digital image in Figure 11. After the healed sample was assessed, the crack path varied from the initial direction, indicating that the fabric was able to fully recover to its original properties at a 100% recovery rate. Furthermore, the energy required to counteract the shear stress occurring in parallel to the crack plane at the new area of the crack front is likely attributed to the minor increase in tear strength for the healed sample. The energy was projected to reach the critical volume during the tearing process, breaking both covalent bonds and ionic networks. The healing process was initiated when the torn pieces came into contact with each other. It was expected that the ionic dangling and brushes that formed during the tearing would reassociate and subsequently progress into chain interdiffusion at the crack tip. Therefore, more energy was required to initiate cracking during the sample retesting, which inhibited the growth of the initial crack, although the crack would perhaps be initiated at a different path.
38
Tear strength of neat, coated and healed fabrics. The virtual images of tear samples showing tear test before and after healing process.

Tests for puncture resistance and the efficiency with which the fabric healed its damage were performed to determine the maximum load required to perforate the fabric. The results of force versus displacement for the neat, coated and healed fabrics are shown in Figure 12. Generally, the puncture stress values of the coated fabric before and after healing were higher than those of the neat fabric. The puncture strength of the coated fabric before healing was 1.3 N, a puncture resistance increase of up to 63%, compared with the neat fabric. This increase in puncture resistance could be explained by the presence of HANRL/30ZT/DCP on the fabric’s outermost layer, which served to protect the fabric and the fibres in the fabric’s system from severe damage when the nail moved downward. The fibres were bound closely together, and the coating absorbed a substantial amount of energy when the nail penetrated the fabric, while the strength elongation of the coating film persisted.
19
Hence, greater force was needed to penetrate the fabric, resulting in high puncture strength. The visual inspections of the self-healing of the neat and coated fabrics are presented in Figure 13. When the nail penetrated the fabric to its maximum penetration force, the fibre broke, resulting in a hole in the middle of the sample. Interestingly, the hole was disappeared after the process of healing on the damaged fibre. As can be observed in Figure 13(c), the damaged area fully recovered and the puncture strength increased by up to 31%, with the maximum force being 1.7 N. Figure 14 compares the punctured areas of the damaged and healed fabric. This clearly reveals that the fabric’s surface morphology had fully healed, indicating that the material had been able to regain its original properties following the healing process. Again, the intermolecular interaction between the ionic intermediates and the rubber chains within the fabric could explain this, allowing the natural rubber chains and zinc thiolate to diffuse and rearrange during the healing process. The broken rubber chains re-entangled with other Zn2+ molecules, giving rise to new molecular entanglements. After some time, large-scale intermolecular diffusion developed between the natural rubber chains and the zinc thiolate, allowing each chain to restore its mechanical properties.
23
Puncture strength of neat, coated and healed fabrics. Visual inspection of self-healing NR coating at room temperature: (a) initial sample, (b) penetration area of probe before healing, (c) penetration area after healing. SEM images of damaged fabric and healed fabric during puncture test.


Effect of coating layers on self-healing performance of the fabric
The material’s potential to restore its impaired functions following damage to the system can be described as the quality of healing, generally referred to as healing efficiency.
39
The effects of coating layers and thickness were also investigated to assess the optimum healing performance of the healed fabric. The thickness of the coating layers was controlled by the number of coating layers applied to the fabrics. The coated fabrics were subjected to tensile, tear and puncture tests. After the sample failed, the broken surfaces were contacted with each other to heal the damaged area. The sample’s healing performance was then calculated by considering the average healing efficiency obtained from the tensile, elongation at break, tear strength and puncture tests from different number of coating layers as shown in equation (2). To eliminate broad outliers, a harmonic mean was employed instead of integrating the normal numerical means of each test to generate an overall healing efficiency equation. The total healing efficiency was calculated as follows:
Where n is the number of tested properties, HEp =
The influences of the coating layers and thickness on the overall fabric self-healing performance are depicted in Figure 15. Healing efficiency was found to decrease as the number of layers and thickness increased. This decrease could be attributed to poor interfacial adhesion between the layers. As previously explained in the methodology section, the dip-coating method was applied after the previous layers had been fully cured. It’s likely that the weak adhesion between the coating layers caused the fabric performance to decline, which is believed to be due to the greater surface concentration area that occurred during deformation. Furthermore, the weak adhesion between the coating layers might reduce the interaction and recombination of the ionic networks during healing, in addition to reducing the interdiffusion of the molecular chains between the broken surfaces. It is worth noting that the adhesion between the coating layers could be further improved if different treatment methods are followed. These methods might use Formic acid, Deurub K or liquid smoke, Sulphuric acid, Alum, Triple Super Phosphate (TSP) Fertiliser, Para and Alum Vinegar, some of the most commonly used coagulants34,40–42 However, more research is required to assess this. Total healing efficiency of different numbers of coating.
Conclusion
This study discovered a novel method of fabricating intrinsically self-healing NRL-coated fabric that has the ability to self-repair and regain its properties at room temperature. The strategy involved forming a reversible ionic crosslinks network through the vulcanisation of zinc thiolate on rubber molecular chains using a controlled DCP-induced vulcanisation process. The DSC results demonstrated that the crosslinks within the materials were dominated by ionic networks at higher temperatures. The FTIR analysis indicated the crosslinks between natural rubber chains with zinc thiolate were obtained at rising peaks at the 1000 cm−1 absorption band as the zinc thiolate content grew from 0 to 45 phr. The zeta potential showed negative values with good colloidal stability. In terms of the UV-visible characterisation, a strong absorption peak was obtained with increased ZT content due to the higher ion concentration. A visual inspection showed that the fractured surface healed after the broken surfaces had been brought into contact with each other, indicating that the material was able to fully recover. To support the finding, SEM images proved that the fabric was successfully impregnated by HANRL because the coating filled in the gaps between the fibres. The tensile strength, elongation at break, tear strength and puncture strength of the healed fabric were fully recovered after healing at room temperature. The effect of coating thickness was further investigated by multiple dipping layers. This revealed that a single coating produced a favourable HANRL-impregnated fabric, which fully recovered, unlike the multiple-coated fabrics. The latex coating is normally used for the manufacture of waterproof canvas fabric. The application for self-healing fabric can be expand to protective clothing, garments, leather coating for garments and accessories. This self-healing can improve the longevity and durability and especially for prolong protection. Overall, the self-healing fabrics developed in this study offer a new, smart and efficient use of natural rubber for the fabrication of smart textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the Ministry of Higher Education Malaysia for the Fundamental Research Grant Scheme with Project Code: FRGS/1/2021/TK0/USM/02/1.
