Abstract
Polyethylene terephthalate (PET) polyester fabric has been treated with different concentrations of sodium lignin sulphonate (SLS), a lignin based sustainable flame retarding agent. Treated PET fabric has exhibited different chemical loading on its surface varying from 15 to 60% depending on the concentration used. Treated fabric has shown 40–70% more limiting oxygen index (LOI) value as compared to the control PET fabric. 200 g/L SLS treated PET fabric (with 35% chemical loading) has shown LOI value of 27. In addition to it, melt dripping property of all the treated PET fabrics have been found to be stopped with lower shrinkage during combustion, as observed from the UL94 test results. TG analysis of the said fabric has shown 20–25% more char mass retention and lower rate of weight loss as compared to the control one. Char morphology of the control and the treated fabric has also been examined in different magnifications and analyzed in detail. Forced combustion test of the sodium ligno-sulphonate treated PET fabric has revealed 82% lower peak heat release rate (PHRR) as compared to the control PET fabric with less amount of carbon monoxide liberation during burning. Besides, a possible mechanism lies behind the flame retardancy of the SLS treated PET fabric also has been expressed and discussed scientifically.
Keywords
Introduction
Polyethylene terephthalate (PET) fibre is one of the important synthetic fibres commonly used all over the world. Polyester fibre and its blends have versatile areas of end uses in the domain of home textiles, upholsteries, curtains, apparel, automobile industries and various other technical textile applications. This fibre has many positive aspects like lusture, flexibility, affordable cost, strength. These properties have made it popular among other synthetic fibres. However, it has some serious disadvantages like non bio-degradability, lower comfort property, high flammability, shrinkage, dripping during flaming, lower dye ability (due to lack of functional groups present in its structure) and so on. Different research works have already been accomplished to compensate those negative aspects of PET fibre.1–4 Most of the researches are based on the addition of some useful functional groups as copolymer during polymerization stage of the PET and subsequently melt-spinning of the same whereas others have tried to improve the properties of it by external functionalized coating on the polyester surface. As far as the flammability property of PET is concerned, it shows limiting oxygen index (LOI) value of around 19 and during flaming fibre gets shrunk, melt dripping phenomenon starts occurring with the generation of lot of toxic smoke. Because of these critical flammability aspects, lots of fire accidents and physical damages are the common challenging issues in the field of the usage of PET polymer. Different researches already have been performed to address these critical issues of flammability.1–6 As mentioned, besides copolymerization, useful coating of a suitable fire retardant agent on the PET fibre or fabric surface is one of the common approaches to address the flammability challenges. However, high add-on%, durability of flame retardancy, toxicity of the chemicals used, cross-link of chemical with the fibre surface are the challenging issues. Researchers have reported copolymerization of polyester with bifunctional organo-phosphorous compound based on phosphonic derivative and the end product is commercialized as trade name Trevira CS. 1 Horrocks et al. also have reported that halogen based chemicals and combination of halogen/phosphorous based compound can be a worthy flame resistant for PET fabric as chemical loading required for restricting the flame and melt dripping is lower. However, the approach is not environmentally friendly. 2 From last two decades, char forming flame retardant intervention are getting more attention of the researchers as compared to the oxygen blocking halogen based alternatives. Chen et al. have demonstrated that aryl polyphosphonate is an effective flame resistant material as copolymer for polyester fibre as compared to the aliphatic and aromatic polyphosphonate due to its higher hydrolytic stability and melt flow property during polymer processing. 3 Didane et al. have registered flame retardant action of aluminium phosphinates as copolymer and also as a back coating material for PET fibre. They have concluded that in both the cases heat release from the treated fabric gets reduced (50–60% lower as compared to the control PET) but back coated material has shown more smoke generation as compared to the copolymerized PET fibre. 4 Carosio et al. have reported a new method of making fire retardant polyester fabric with silica architectures by following layer by layer technique. 5 Fabric treated by horizontal spray technique (3% silica add-on) has shown more tendency of char formation as compared to the control polyester fabric. Younis have reported the process of making flame resistant polyester fabric by using mixed formulation of ammonium phosphate, urea and boric acid. 6 Treated fabric has shown LOI value of 25–26 and the sample also gets passed in UL94 test. Gawlowski et al. have reported that PET fibre can be made flame resistant by using combination of silicon and phosphorous based chemical. Treated fabric has shown LOI value of 29.8 at 16% add-on and attains char forming behaviour. 7 Ureyen et al. have reported the effect of zinc borate and melamine coating on the flammability properties of UV treated PET fabric and the treated fabric has rendered LOI value of 28–31 depending on the concentration of zinc borate. 8 Younis et al. have reported the treatment of UV irradiated polyester fabric by sol of pomegranate rind and siloxane and it has been observed that the treated one can show LOI value of 26 with more char formation instead of melt dripping. 9 In addition, treated fabric also exhibits water repellency effect due to lowering the surface energy of the fabric with the presence of silicon. Polyester fabric also has been made flame retardant by using new phosphorous nitrogen based compound. The concerned chemical was synthesized by the mixture of polyvinyl alcohol, polyester resin, phosphoric acid and dicyandimide. 10 Recently a nano coating of chitosan and ammonium polyphosphate has been developed for making char forming polyester fabric. Researchers claimed that the mixture containing nitrogen and phosphorus forms thin coating on the fabric surface. 11 Chitosan sodium polyborate polyelectrolyte has been explored by another research group for making flame retardant polyester fabric. 12 As sustainability is a big challenging issue in this field, researchers are very fascinated to find worthy alternatives of synthetic based flame retardant chemicals. To this direction, research works have been carried out to explore different plant and protein based bio-macromolecules for making fire resistant natural and synthetic fibre products.1,13–22 Recently, phosphorous based bio-moleucles like DNA, casein have been used for making fire retardant polyester fabrics .23,24 Lignin and different derivative of lignin, a polyphenolic compound, have been well explored by the researchers as flame retarding agent for the cellulosic and ligno-cellulosic natural fibre based textiles.25–27 Sodium lignin sulphonate (SLS) (a lignin derivative) has been applied on the cotton fabric and it has shown promising effect on the flame resistant property of the said cotton fabric at 30% add-on level 28 and its property has been further improved when it has been mixed with pomegranate rind extract. 19 Researchers have postulated that it enhances char formation and restricts the flammable gas formation during pyrolysis of the natural fibre, reflects condensed phase mechanism of fire restricting action.19,28 Very recently, one review paper has been published which shows systematic applications of lignin bio-macromolecule on different polymeric materials to impart flame retardant effect. 29
Flame retardancy of synthetic polymer made fabric by using natural bio-macromolecule is a challenging aspect of research as dripping of polymer needs to be addressed. Very few bio-macromolecules have been explored as coating material for synthetic fabric.5,30 However, till date no research work has been documented on the application of lignin based derivatives on synthetic textiles as best of our knowledge. On the other hand, searching of alternative sustainable material is still going on, and in this direction, in the present article SLS has been explored for making flame resistant PET fabric. This research work aimed to develop flame resistant PET fabric for making curtains, sofa cover and other home textile products for which comfort properties are not much important.
Materials and methods
Materials
Fabric
Woven PET fabric (140 g/m2) having 55 ends/inch and 30 picks/inch was purchased from the local market of New Delhi, India. Purchased fabric was ready for dyeing, heat set polyester fabric.
Chemicals
Sodium lignin sulphonate used for the experiment and other chemical ingredients like, non-ionic detergent, acetic acid required for the experimental purpose were purchased from HI MEDIA Chemicals. Sodium lignin sulphonate used for the treatment was brown color powder having 99.9% purity and density of 0.5 g/cc.
Treatment of PET fabric with SLS
Fabric was primarily treated with water at pH range of 4.5–5 (adjusted by glacial acetic acid) and thoroughly washed with cold water before using for experimental purpose. Acid treatment of the fabric was performed for cleaning action, for removing contamination, vegetable matters present in it. Polyethylene terephthalate fabric could be stored for long days after mild acid treatment.
Before SLS treatment, PET fabric was washed with the 1 g/L detergent in water at 60°C for 30 min and dried. It helped to remove the presence of any unwanted contaminants present on the fabric surface and also rendered to reduce the surface tension of the fabric surface. Dried PET fabric was coated with four different concentrations of (50, 100, 200 and 400 g/L) SLS by padding method (following 2 dip-2 nip process) at lower mangle pressure (0.4 kg/cm2) and was dried at 80°C for 5–7 min. Dried fabric was conditioned at 27°C, and 65% RH before undergoing any scientific testing. Acetic acid was added into the formulation for adjusting the pH of the treatment bath at 5.5.
Calculation of the add-on% of the treated fabric
Before testing, all the samples were conditioned in a standard atmosphere of 65% RH for 24 h, so that they are identically acclimatized. The percent add-on of the PET fabric after the treatment (i.e. the increase in the sample weight relative to the original weight) was determined by the gravimetric method as follows:
Sample prepared for the flammability test
The burning behaviour of the control and the treated PET samples was evaluated as per the standard methods. For determination of the LOI, an ignition time of 30 s was maintained as per the Indian standard IS 13501 [law resource.org1992]. UL94 test was performed by placing the fabric samples at vertical position. Size of the fabric samples was maintained 125 mm × 13 mm and this fabric was tested with 20 mm blue flame of burner for 10 s. Top of the burner was at a distance of 10 mm from the lower end of the specimen. After flame time (t1 and t2), after glow time (t3) was noted for each sample.
Characterization of the treated PET fabrics
Depolymerisation behaviour and the nature of weight degradation of the control and the treated PET materials were drawn on a Thermo Gravimetric Analyser (NETZSCH STA 44953 STA4493A-1100-M) in nitrogen atmosphere at 2 mL/min flow rate and with the heating rate of 10°C/min. Differential Scanning Calorimetry (DSC) of control and all the treated PET samples were performed in the same machine at nitrogen atmosphere in the temperature range of 50 to 450°C.
Changes in the surface morphology of the materials were studied with the help of a high resolution (up to 3 nm) Scanning Electron Microscope (SEM, ZEISS EVO 50) using SE detector. The samples were coated with a thin layer (100 angstroms) of conducting material (Gold/palladium) by using sputter coater and examined under SEM with an accelerating voltage of 20 KV.
Cone calorimeter analysis
Cone calorimeter (manufactured by DnG Technologies Pvt. Ltd., India) was used to test ‘100 × 100’ mm2 control and the SLS treated PET textile in accordance with the procedure of ISO 5660-1 standard. Specimens were tested at horizontal orientation with heat flux of 35 kW/m2 generated by cone calorimeter and to the direct application of the propane flame. Before testing all the samples were conditioned at 65% R.H and 27°C. Samples were in horizontal condition during the combustion process. The parameters measured were total heat release (THR) (MJ/m2), heat release rate (HRR) (kW/m2), time to ignition (TTI), the maximum average rate of heat emission (MARHE) and the peak heat release rate (PHRR). Three replicates of the samples were tested in each case, and the CV% values are represented in the data table.
Results and discussion
Flammability test of PET fabric
Burning behaviour of the control and the treated PET.
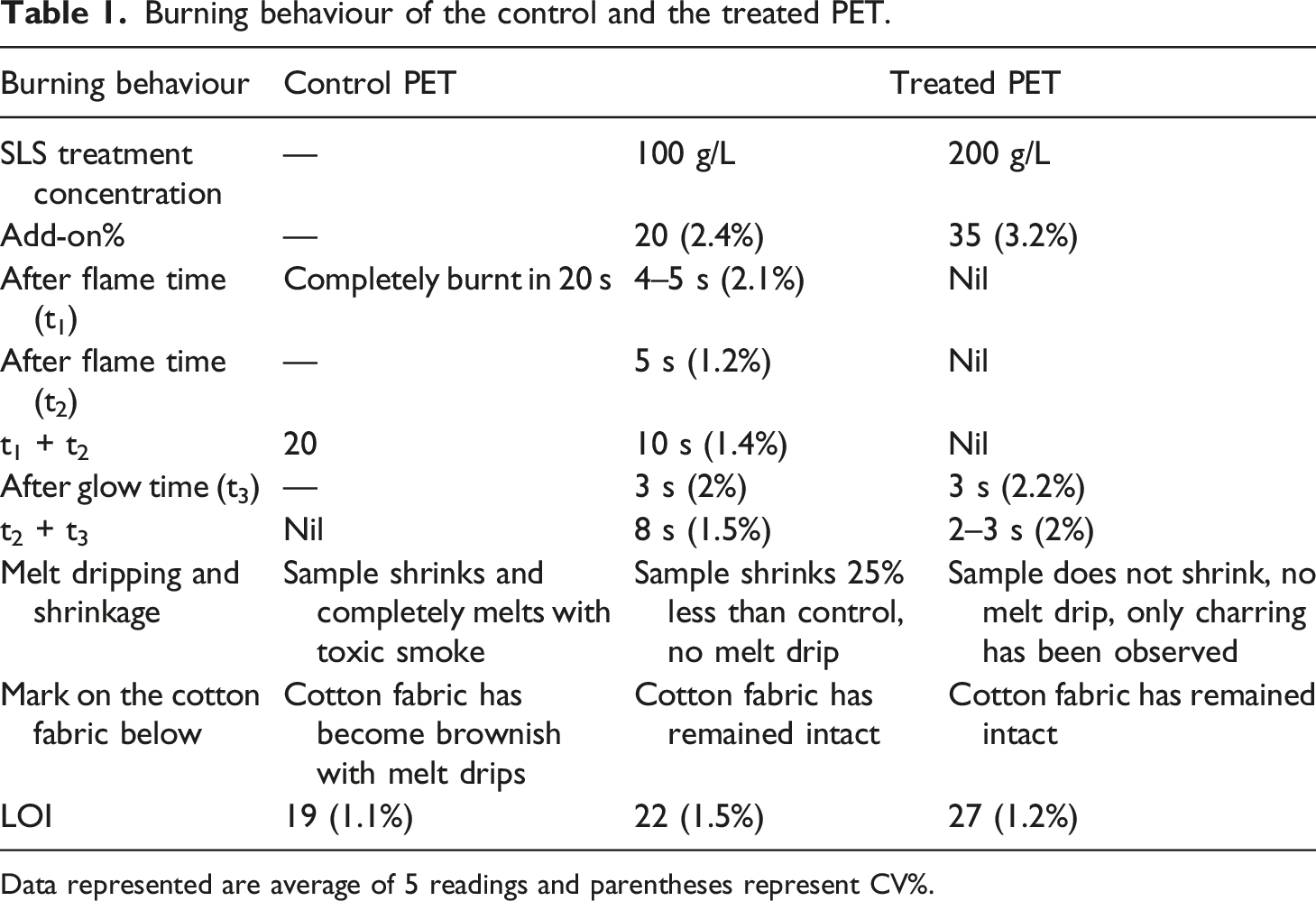
Data represented are average of 5 readings and parentheses represent CV%.

UL 94 burning behaviour of the SLS treated PET fabric at different concentrations.
In addition, melt dripping of the treated fabric has been restricted completely as the treatment acts as a char enhancer of the polymeric materials (depicted in Figures 2 and 3). Figure 2 also exhibits microscopic images showing that the treated material is uniformly deposited on the fibre surface of PET. Limiting oxygen index of the control and the treated PET fabrics has been examined for further fortification of the UL94 results. It has been observed that 200 g/L SLS treated PET fabric shows LOI value of 27 at 35% loading against the LOI value of the control PET of around 19. Increase of the LOI value of the treated fabric could be due to the deposition of polyphenolic groups of lignin on the PET fabric surface. However, only 50 g/L SLS treated PET fabric shows marginal improvement of the LOI value which is around 20 and also fortifies the shrinkage in the longitudinal direction during burning but positive finding is that in UL94 test the said sample did not show any occurrence of melt dripping phenomenon. As per standard of UL94 test, one cotton fabric was placed below the fabric sample for understanding the damaging effect of the polymer melt dripped from the control and treated PET fabric during burning. Add-on exceeded 60% for 400 g/L SLS treated PET fabric (LOI: 38) and the treated sample was very hard with such a high chemical add-on. Fire retardant results have been compared with the literature. It was registered that commercially PET fabric was treated with char forming phosphorylated sol-gel and the LOI value has been raised from 18 to 27. Trevira CS, one of the commercialized copolymer based PET fabrics has shown LOI value of 28.8,31 Table 1 represents CV% value (depends on mean value and standard deviation) of the flammability results has shown here. It is observed from the Table 1 that with increasing the loading of SLS on the PET surface, CV% values are also increasing attributed with the more variation of results. More variations may be linked with the non-uniformity of SLS thickness throughout the PET surface. However, LOI value of 200 g/L SLS treated PET fabric has shown lower CV% compared to the 50 g/L SLS treated structure. It may be linked with the flaming from total sample used for the experiment, it is a bulk property and more SLS loading could reduce the variation of LOI results. Surface morphology of the SLS (A), control (B) and the treated fabric (C) with the char pictography of the SLS (D), control and treated PET fabric (E and F) at ×100 magnification after UL94 burning test. Char surface morphology of the SLS (A), control and treated PET fabric (B and C) at X 1KX magnification.

Mechanism of flame retardancy
The molecular weight of ligno-sulphonate varies between103and 106 g/mol. It contains guaiacyl units (G units with small amount of P-hydroxyphenyl units) and hydroxyl groups, carboxyl groups with the presence of sulphonate groups. Lignin contains different kind of alcohols like sinapyl, cumaryl and colyphenyl alcohol. Single unit of these structures contain hydroxyl groups at different isomeric position and also composed of phenolic aromatic ring.30,32 These lignin based structures itself are a char enhancer and act as thermal stable material on combustion due to self-crosslinking of aromatic groups with each other and cyclidized the combined one. This material could easily be deposited on the PET fabric surface from the water medium by following padding techniques and represented in Figure 4 with chemical structure. As a result, active ingredients like phenolic groups and sulphonate groups get uniformly distributed on the wet PET fabric. The layer deposited on the fabric surface is bad conductor of heat and it gets exfoliated on burning as observed from the surface morphology images of the char mass of the treated fabric. In addition of the presence of phenolic groups, sodium present in the structure renders in dehydration and flame produced from the sample is turned to be yellowish. Sulphonate groups, hydroxyl groups present in the structure are polar in nature and attract water molecules from the environment and assist combinedly to restrict temperature increase during flaming. Therefore, cyclidization of aromatic groups of lignin, effect of sodium ions and moisture absorption of auxochromic sulphonate groups present in the SLS combinedly assist to restrict the melt dripping, high temperature generation, shrinkage phenomenon in the treated sample. Evidence of char formation has already been fortified by TG analysis and surface morphology of char mass. Low heat release from the treated sample during combustion has been fortified by forced combustion analysis and it shows that treated PET fabric generates 60–70% less heat as compared to the control PET fabric. Overall SLS treatment on the PET fabric has followed the condensed phase mechanism of flame retardancy. Chemical structure of lignin deposited on the surface of PET fabric.
Surface morphology
Surface morphology of the SLS treated PET fabric shows a uniform coating on the fabric surface whereas morphology of SLS also shows good coating ability on its own. Interstices of the PET fabric are also filled up with the SLS during the coating process. Char morphology of the SLS shows much consolidated structure with the presence of some diffused craters in its surface. It proves the extensive thermal stability of the SLS component, used for coating of the PET fabric. Control PET fabric shows left over melted damaged structure after burning whereas char mass of the SLS treated PET fabric shows many bubbles formation with the presence of small holes and pearl like very small balls throughout the surface of the char as depicted in Figure 3. This phenomenon proves the intumescent behaviour of the SLS when it is deposited on the PET fabric. Small holes may assist to release the non-flammable gases from the structure and insulation of the coating may protect the underlying material from being melted. In this way, underneath fabric surface does not get sufficient temperature for melting and before the melting phenomenon takes place, upper coated material (SLS) gets exfoliated in volume and restricts the temperature flow towards the inner surface of the coated layer. It is reported that the same kind of char behaviour also has been observed when SLS has been applied on the cotton fabric surface.
25
Cross-sectional view of the control and SLS treated PET fabric has been represented in Figure 5. It clearly shows the char forming tendency of the PET polymer after SLS treatment. Cross-sectional view of control PET fabric (A), control PET fabric after burning (B) and SLS treated PET fabric after burning (C).
Thermo-gravimetric analysis
It has been reported that TGA of SLS shows almost 50% of char mass retention at 600°C (in TG analysis at air atmosphere). It is reported that the degradation of SLS is a stepwise degradation process, depends upon the type of bonds presents in the SLS. Sodium lignin sulphonate represents polyphenolic structure and different types of bonds present in the SLS degrade at different temperatures, leads to structural breakdown of SLS. All the ether bonds, present in the SLS, are weaker and break early as compared to the other strong covalent bonds, break at higher temperature.19,28 Last residue left (50%) in SLS contains carbonized aromatic structure, which is consolidated one, as shown clearly in Figure 2.
TG curve of PET, depicted in Figure 6 has shown two degradation stages. 1st major degradation has been occurred at around 400°C, may be due to the depolymerisation of the polymeric chain structure of the PET and another loss has been occurred at around 450°C, assigned with the char formation. Researchers have postulated that at 400°C degradation in the polymeric chain gets started through beta CH transfer reaction and it delivers vinyl and carboxyl terminated oligomers with the generation of volatile gases like carbon monoxide, carbon di oxide, methane, formaldehyde, benzaldehyde.
30
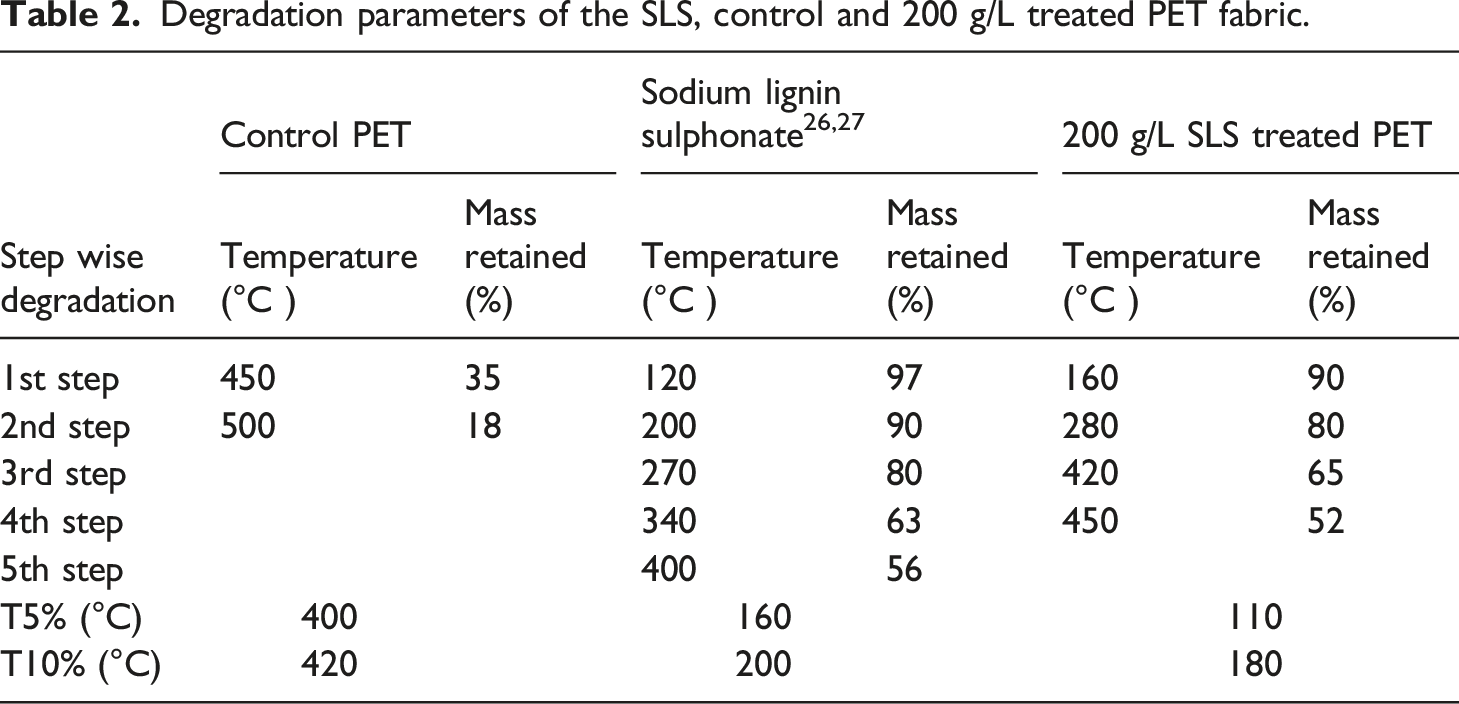
At this stage almost 70% mass loss has been observed and at final charring stage another 10–12% mass loss has been occurred. At 500°C only 18.5% char mass has been found to be left out. On the contrary, treated PET fabric has shown different mass loss stages, however, rate of weight loss is low as compared to the control PET and at 500°C almost 40% char mass has been left. Amount of char mass left for the 200 g/L SLS treated PET fabric is more as compared to the 100 g/L SLS treated fabric. In both the curves, 1st mass loss stage is in between 90–100°C and in this stage almost 5% mass loss has been occurred, due to the evaporation of moisture from the treated fabric. 2nd mass loss zone is bigger i.e., from 200 to 300°C and in this stage 13–15% total mass has lost, due to the degradation of lignin present in the composition of the deposited material. Sodium lignin sulphonate is the lignin containing large molecular weight polyphenolic compound with sulphur moieties. Only 25–30% mass loss has been occurred up to 400°C. 3rd mass loss zone is between 400°C to 450°C and only 30% mass loss is occurred in this stage and finally char formation gets started from 450°C. As of comparison, control and the treated fabric have shown retention of 18 and 39% residual mass at 500°C, fortifies enhanced thermal stability of the treated fabric. Degradation of the treated PET fabric in successive steps also clears the fact that the active ingredients of SLS has been coated on the fabric surface and act as hindrance in the way of depolymerisation of polymeric structure, thus, it restricts the formation of toxic volatiles during chain depolymerisation stage (350–380°C). This phenomenon has been fortified with only 10–15% mass loss occurred in this stage as observed from the Figure 6. Table 2 represents different steps of the degradation in terms of temperature and mass%. In addition of it, corroboration of every steps of degradation has been established with the structure of the polymeric material. T5 and T10% are the specific temperature (°C) at which 5 and 10% mass loss has been observed. T5 and T10% of the treated PET fabric is much lower (290 and 240°C lower) than the control PET, may be due to the reaction of PET with the different active components of SLS and it also signifies the dehydration potential of the polymeric structure with addition of SLS. TG analysis of the control and SLS treated PET fabric in air atmosphere. Degradation parameters of the SLS, control and 200 g/L treated PET fabric.
Combustion behaviour
Summary of the cone calorimeter data of the control and the treated fabric (parentheses represents CV% data).

Note: HRR: heat release rate, THR: total heat release, MARHE: maximum average rate of heat emission. Parentheses represent the CV% of data.
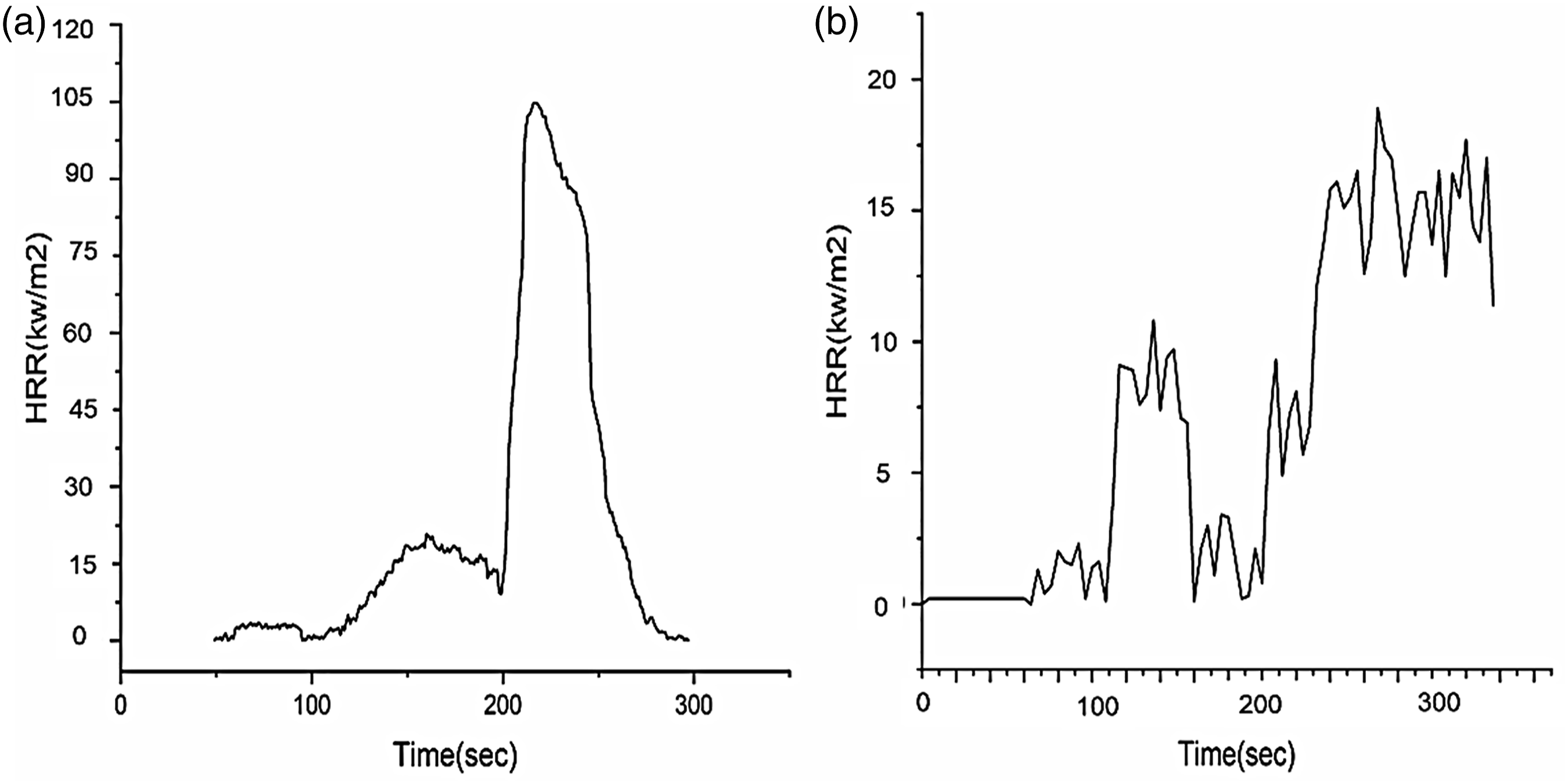
Forced combustion behaviour of (A) the control and (B) the treated PET fabric.
Durability of the treatment
Self-extinguishment behaviour of SLS coating on PET fabric surface can withstand up to 2 home launderings. After 2 home launderings with non-ionic detergent (2 g/L) at room temperature, 200 g/L SLS treated fabric has shown LOI value of 24 as compared to the LOI value of 27, before washing. However, dry-cleaning with nonpolar trichloroethylene could be very much effective and the treated fabric has maintained its LOI value of 27 even after 2 dry cleaning cycle.
No cross-linker and binder has been used in the SLS formulation to assist in crosslinking with polyester fabric as most of them hinders the self-extinguishment property of the treated PET. Moreover, nature of polyester is hydrophobic and there is no polar group present in the PET structure, therefore chance of crosslinking with the polar SLS molecule is a bit difficult even after increasing the temperature (like curing) after drying. Another scientific hurdle is that SLS is a large molecule having high relative molar mass whereas specific volume of polyester is not enough for providing space for SLS molecules inside its structure even after increasing the temperature. Therefore, high temperature process has not been followed for fixation of this SLS treatment.
Conclusion
In the present study, SLS has successfully been used for the treatment of PET polyester fabric by following padding method. It has been found that 35% add-on on the fabric surface is adequate to stop the after-flame and melt-dripping and the treated PET fabric has shown LOI value of 27. Moreover, hand feel and other physical properties of the treated fabric has almost been remained unchanged up to 35% chemical loading on the fabric surface. TG analysis of the SLS treated fabric has shown 25% more char retention at 450°C, with slow rate of weight degradation phenomena as compared to the control PET fabric. Char analysis of the treated PET has shown much consolidated structure of expanded volume with bubble formations as compared to the control PET fabric. It has been hypothesized that the cyclidized behaviour of polyphenolic groups of lignin, dehydration assistance of sodium ions of sulphonate groups and moisture absorption of the deposited structure (due to the presence of polar groups like -OH, -COOH, -SO3Na) on the fabric surface have combinely assisted in char forming ability of the synthetic polymer instead of depolymerisation and melting of the control PET fabric. Developed flame resistant PET fabric could be used for making curtains, sofa cover and other home textile products.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

