Abstract
In this study, a polybutylene terephthalate (PBT) antimicrobial masterbatch containing silver, zinc, and copper antimicrobial agents was added to polyethylene terephthalate (PET) and spun into an antimicrobial partially oriented yarn which was then texturized into a draw-textured yarn (DTY). Filtration pressure rise and spinning tests indicated that the silver antimicrobial masterbatch had the best long-term spinnability. In addition, the strength and elongation of the antimicrobial DTY were reported to match practical standards, indicating that PET containing a silver antimicrobial agent can contribute to a smooth spinning process. In an antibacterial test of gram-negative and gram-positive bacteria before and after finishing, only the silver fabric maintained over 99.99% antibacterial effectiveness. Thereafter, the silver textile was tested for strains of influenza A virus H1N1 and H3N2 to evaluate its antiviral activity before and after dyeing; the textile containing 2% silver antimicrobial agent exhibited optimal antiviral effectiveness. In conclusion, the silver-containing fabrics exhibited long-lasting antibacterial and antiviral effects.
Keywords
Introduction
The nonbiodegradable polyesters polyethylene terephthalate (PET) and polybutylene terephthalate (PBT) are the most well-studied and widely utilized polymers worldwide. They are employed as raw materials in a variety of goods, such as textiles, beverage bottles, and packaging.1–3 Semicrystalline thermoplastic polymers, including PET and PBT, are best known for their outstanding chemical resistance, moisture resistance, electrical insulation, and flexibility.4,5 Since the solubility and chemical structures of these two polyesters are very similar, they have good mixing compatibilities in specific quantities. PBT is structurally identical to PET, but has a longer alkyl chain between the two parabens, allowing it to be more flexible. 6
Antimicrobial fabric treatment prevents microbial contamination, lowers the risk of cross-contamination in public spaces, and improves safety and comfort. The antimicrobial processing agents for textiles can be divided into organic and inorganic agents. The advantages of using inorganic antimicrobial agents over organic agents in textiles are their high resistance, safety, and thermal stability. However, the compatibility of inorganic antimicrobial agents and organic materials (such as polyester and polyamide) is poor, and is one of the spinning technologies investigated in this study.
Precious metals, such as silver, zinc, and copper, are employed to achieve the antimicrobial properties of inorganic antibacterial agents that are often used in textile products. Several studies7–9 have suggested the substantial antimicrobial action of metal ions on both gram-negative and gram-positive bacteria. However, the antimicrobial finishing used in the past was post-processing (such as the resin-finishing process); therefore, its antimicrobial durability was inferior, and resulted in a product with a rough feel. To overcome the shortcomings of post-processing, an antimicrobial masterbatch was added to the fiber for an additional amount of antimicrobial agent, which led to a better antimicrobial effect that lasts longer than that of post-processing techniques. Furthermore, the fabric is not affected by the contamination of the processing agent (such as resin), and is comfortable to touch.
In response to the COVID-19 pandemic, the development of functional fabrics, with antibacterial and antiviral effects, has become an urgent demand. It is challenging to evenly mix the inorganic antimicrobial agents with organic substances (such as polyesters and polyamines). Uneven mixing leads to a decrease in fiber strength and elongation. 10 Further, there are problems of yellowing and failure of antimicrobial effectiveness during manufacturing and processing.11,12 The literature reports that certain commercial antimicrobial products have been launched; however, only a few practical studies have been published. In fact, it is considerably difficult to manufacture antimicrobial products because they are either too expensive or not washable.13,14 Therefore, in this study, we aimed to explore the feasibility of using inorganic antimicrobial agents in polyester fiber spinning and the effects of the types and contents of antimicrobial agents on spinnability, fiber properties, dyeing hue, and antibacterial and antiviral durabilities before and after the finishing process.
Experimental
Materials
The PBT chip, with a melting point of 226°C and density of 1.26–1.36 g/cm3, was purchased from SHINKONG Synthetic Fibers Corporation Ltd., Taiwan. The PET chip (TiO2: 0.3 wt%, intrinsic viscosity: 0.62 dl/g), with a melting point of 253°C, was purchased from Far Eastern New Century Corporation Ltd., Taiwan. The silver-magnesium-aluminum-phosphate-borate complex (with a silver content of 1.8%) was purchased from ISHIZUKA Glass Corporation Ltd., Japan, with ZAF-HS specified as the silver antimicrobial agent (AgA). Zinc oxide (with a zinc content of 5 wt%) was purchased from Jia Fu Technology Corporation Ltd., Taiwan, with JF-002-P as the zinc antimicrobial agent (ZnA). Copper iodide (CuI) (with a copper content of 5 wt%) was purchased from Jia Fu Technology Corporation Ltd., Taiwan, with JF-003-P as the copper antimicrobial agent (CuA).
Experimental equipment
Spinning machine
(1) Barmag pilot machine, Germany, Type: PILOT. (2) Feature: Ready for production with a few kilograms of materials and the parameters can be changed rapidly.
Draw-Texturing machine
(1) Taiwan exc-fiber company, Type: TTRI-001. (2) Feature: Take-up speed: 2000–5000 m/min.
Experimental procedure
In this experiment, PBT and 20 wt% of the three inorganic antimicrobial agents (silver, zinc, and copper) were mixed in a twin-screw mixer to produce three types of PBT antimicrobial batches: silver antimicrobial masterbatch (AgM), zinc antimicrobial masterbatch (ZnM), and copper antimicrobial masterbatch (CuM). First, 95 wt% PET was mixed with a 5 wt% antimicrobial masterbatch in a spinning machine to produce antimicrobial partially oriented yarn (POY). Next, an antimicrobial draw-textured yarn (DTY) was manufactured using a draw texturizing machine and knitted into a fabric for evaluation. Three different proportions of AgM (2.5, 5.0, and 10.0 wt%) were added to the PET polymer for spin tests. The experimental process is explained using the flowchart in Figure 1. Flowchart detailing the various steps of the experiment.
Detection of antimicrobial fabrics
Scanning electron microscopy
The surface morphologies of the antimicrobial agents and their dispersion in PBT were observed using a 15 kV electron beam with a resolution of 0.7 nm. Scanning electron microscopy (SEM) was performed using a JEOL FESEM-7900F field-emission SEM.
Thermogravimetric analysis
Thermogravimetric analyses (TGAs) of the three masterbatches were performed (TA Instruments, type: Q-5000) in an air environment at a heating rate of 10°C/min and a temperature range of 25–800°C to observe the variations in weight loss of the antimicrobial agents, antimicrobial masterbatches, and antimicrobial fibers.
Color difference
The CIE L*a*b* system is perceived as a combination of red and yellow, red and blue, green and yellow, and green and blue. In this color space, the color coordinates in the rectangular coordinate system are as follows: L* represents the lightness coordinate, a* represents the red/green coordinate (with +a* and −a* representing red and green, respectively), and b* represents the yellow/blue coordinate (with +b* and −b* representing yellow and blue, respectively). The CIELAB color difference is typically expressed as ΔE*, defined as an equally weighted combination of the coordinate differences (L*, a*, b*), and calculated as the distance between two points in a three-dimensional space, that is, the total color difference ∆E* = (∆L*2 + ∆a*2 + ∆b*2)1/2, where ΔL*, Δa*, and Δb* represent the differences in lightness, red/green difference, and yellow/blue difference, respectively. 15 The values of L*, a*, and b* were measured using a Nippon Denshoku color meter NE 4000.
Antibacterial test method
The JIS L1902:2015 is a standard for the determination of the antibacterial activity and efficacy of textiles. 16 The bacterial liquid absorption method was applied using bacterial species, such as gram-negative bacteria (Pneumococcus pneumoniae) and gram-positive bacteria (Escherichia coli and Staphylococcus aureus). First, to prepare the test pieces, six test and six control samples were collected and cut into appropriate sizes (18 mm2) to obtain a mass of approximately 40 ± 0.05 g. Three test and three control samples were measured immediately after inoculation, and the remaining three samples of each type were measured immediately after inoculation.
Next, the test was performed using a 0.2 mL sterile pipette to accurately inoculate the test piece with the prepared test inoculation solution at multiple locations. Immediately after inoculation, 20 mL saline was added to each vial of the three test and three control samples, the caps of the vials were then closed and they were rinsed. Thereafter, 0.2 mL of the inoculated bacterial solution was added to a 30 mL sample bottle together with an 18 mm2 standard cloth for measurement after 24 h of incubation. Thus, the test sample adsorbed approximately 40,000 bacteria and nutrients. After culturing at 37°C for 24 h, the sample was removed and the bacteria were washed out using 20 mL of physiological saline. Thereafter, 1 mL of bacteria and 9 mL of physiological saline were added to the test tubes and mixed homogeneously. Measurements were performed after incubating the mixture at 37°C for 24 h.
Finally, the bacterial cells on the test piece were measured using the mixed dilution plate method and the antibacterial value R was calculated. The bacteriostatic value R = log ((number of bacteria in the control group after 24 h−number of bacteria in the control group at 0 h)−(number of bacteria in the sample group after 24 h−number of bacteria in the sample group at 0 h)).
Antiviral test method
ISO 18184:2019 is a standard for evaluating the antiviral properties of textiles. 17 The strains of the Influenza A virus H1N1 (ATCC VR-1469) and Influenza A virus H3N2 (ATCC VR-1679) were used for the antiviral test. First, to prepare the test specimens, several 20 × 20 mm2 pieces were cut to obtain a mass of 0.40 ± 0.05 g. The yarns were prepared in bundles and cut to approximately 20 mm to obtain the same mass of 0.40 ± 0.05 g. Nine and 12 specimens were obtained for the reference cloth and antiviral test samples, respectively. Thereafter, the specimens were placed in an autoclave for 15 min at 121°C and 103 kPa for sterilization.
Following sterilization, all vials containing the specimens were removed, placed in a safety cabinet, and left to cool down for 60 min. The vial containers were checked for the absence of dew condensation, then capped and closed. Thereafter, three reference specimens and three antiviral test specimens were sterilized and placed in vials, and 20 mL of wash-out solution (SCDLP medium) was added to all vials. The vials were capped and agitated in a vortex mixer five times for 5 s each time. Thereafter, the 5 mL samples of the wash-out solution were placed in fresh tubes. Next, the specimen was inoculated at multiple locations with exactly 0.2 mL of the prepared virus suspension in vials using a pipette, and the vials were capped and closed.
Subsequently, the vials were placed in an incubator for a standard duration of 2 h at 25°C. Immediately after inoculation, the virus was washed out from three of the reference specimens, 20 mL of SCDLP medium was added to the vials, and the vials were capped, closed, and agitated in a vortex mixer five times for 5 s each time to wash out the virus from the specimens. Finally, to wash out the virus after 2 h of contact, 20 mL of the SCDLP medium was added to vials containing three reference and three antiviral test specimens. The containers were capped, closed, and agitated in a vortex mixer five times for 5 s each time to remove the virus from the specimens.
The infectivity titer was determined using a plaque assay. The plaque method was used to measure the infection value based on the extent to which infected virus cells were stained. Measurements of the unstained plaques were used to determine the number of viruses. Antiviral activity (M
v
) was calculated as follows:
Results and discussion
Filtration pressure rise test of antimicrobial masterbatch
Figure 2 shows the SEM images of the silver, zinc, and copper antimicrobial powders. Parts (a) and (b) indicate that the AgA powder particles have particle sizes of approximately 0.5–3 μm; parts (c) and (d) indicate that the ZnA powder particles are flower-like bundles of between 1–5 μm; and parts (e) and (f) indicate that the CuA powders have particle sizes of approximately 0.8–3 μm. SEM images exhibiting the surfaces of the antimicrobial agents. (a), (d) AgA; (b), (e) ZnA; and (c), (f) CuA.
Thereafter, an antimicrobial masterbatch containing 20% of the antimicrobial agents was examined. The surface images are depicted in Figure 3, where AgA was uniformly dispersed in AgM with no agglomeration, whereas an agglomeration of approximately 4 μm was observed for ZnA in ZnM and CuA in CuM. SEM images exhibiting the surfaces of the antimicrobial masterbatches. (a) AgM, (b) ZnM, and (c) CuM.
To determine whether the added inorganic antimicrobial masterbatch had too many particles agglomerated during the spinning process and whether the inorganic antibacterial agents were separated out by the filter, a filtration pressure rise test was designed to test the dispersion of powder in the masterbatch. The antimicrobial masterbatch and PET particles were mixed in an extruder, and thereafter, the mixed particles were melted and pressed into the filter screen group (pore size 10 μm; five layers of filter screen). Finally, the pressure generated by the filter screen was tested with respect to time.
The dispersion state of the antimicrobial powder in the masterbatch was evaluated based on the extrusion pressure value for 20 min. The lower the filtration test value, the better the dispersion of the antimicrobial powder in the masterbatch. In the filtration pressure rise test, a filtration pressure value below 15 bar indicated good long-term spinnability; if it exceeded 30 bar, the test was stopped. The filtration test values for AgM, ZnM, and CuM were 10.6, 18.8, and >30 bar, respectively, indicating that AgM possessed the best long-term spinnability, followed by ZnM. Although the filtration test value for CuM exceeded 30 bar, the pore size of the filter screen used in the PET fiber spinning factory was 16–20 μm with a five-layer structure and a base pressure of 100 bar. Therefore, CuM can still spin with short-term spinnability.
Antimicrobial partially oriented yarn spinning
Test conditions for 130D/72F POY for the various antibacterial masterbatches.

Note: ZnA powder is a flower-like bundle that causes lower friction; thus, a high tension is required.
Test conditions for 130D/72F POY for various ratios of AgM.
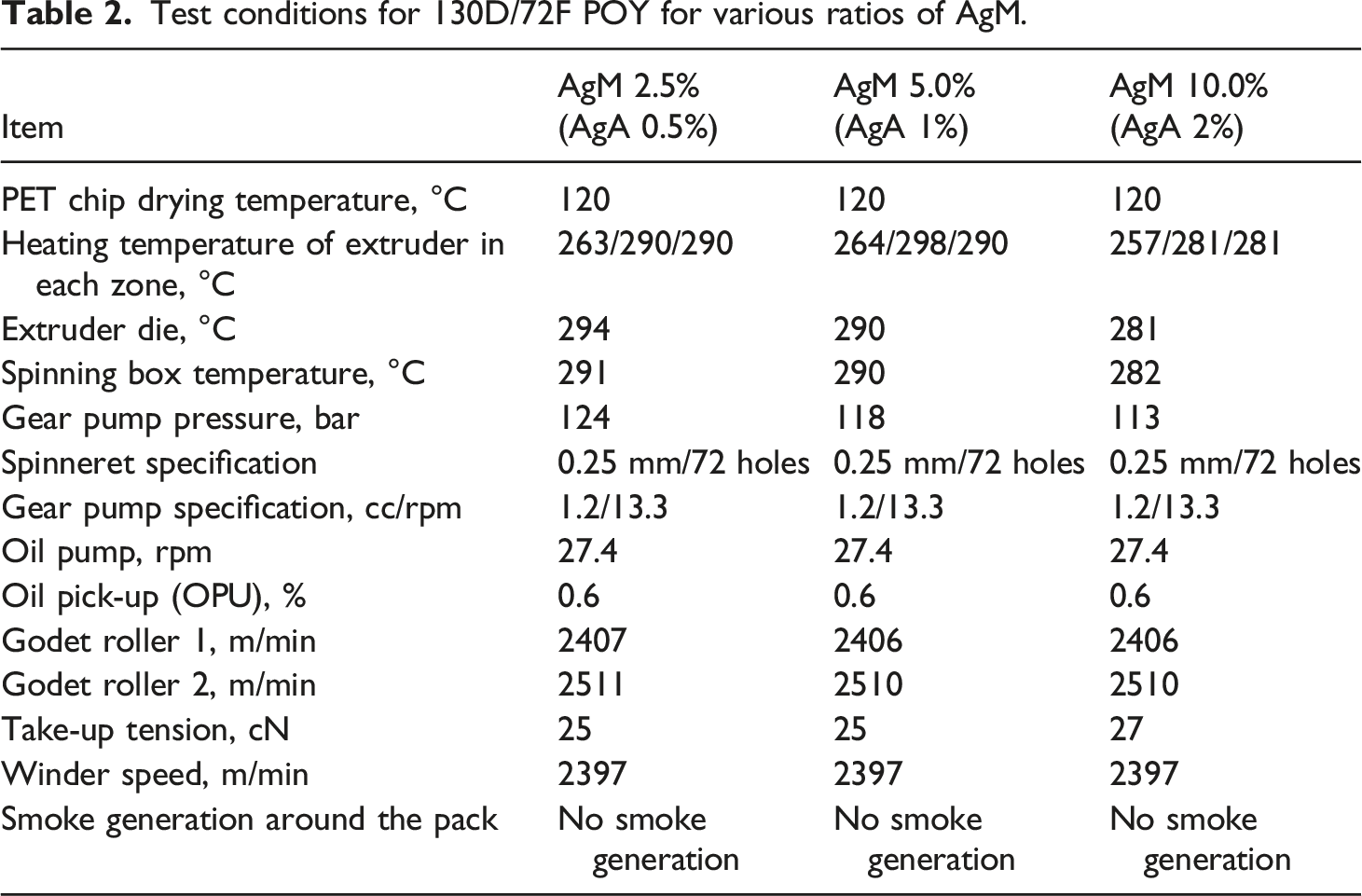
In the early stage of spinning with 10 wt% AgM, no broken filament was observed. However, feeding was unstable, and the cake had the appearance of a spider web, and the concentration of AgA (2.0%) was higher. This phenomenon also appeared during the finishing process of DTY. Based on the above inferences, the spinnability of 10 wt% AgM is barely acceptable.
Evaluation of yarn properties
Yarn strength (g/den) and elongation (%)
Properties of 130D/72F POY for the various antibacterial masterbatches.

Properties of 130D/72F POY for the various ratios of AgM.

Properties of 75D/72F DTY with various antimicrobial masterbatches.
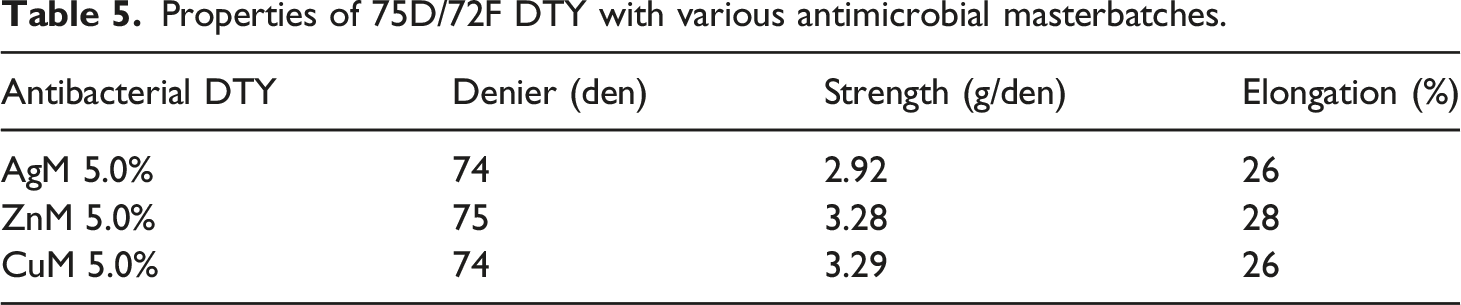
Properties of 75D/72F DTY with various ratios of AgM.
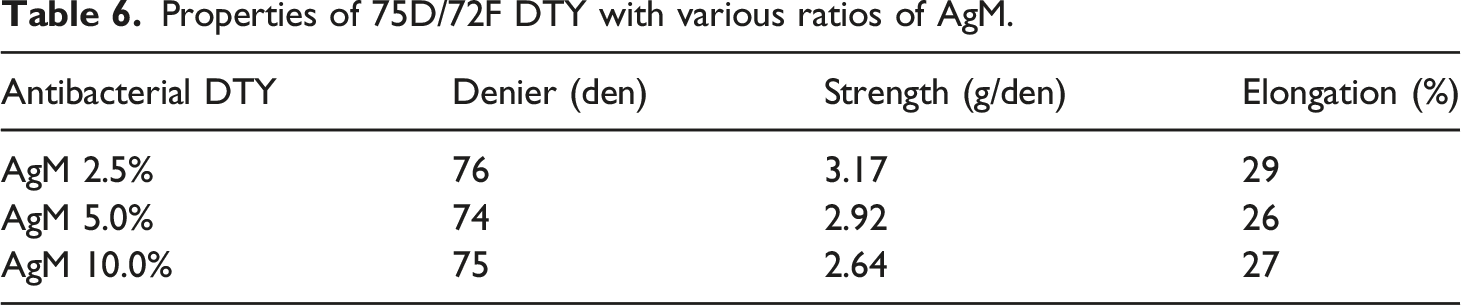
The elongation of the yarn affects the dyeing behavior and should be controlled under normal conditions. If the elongation of POY is greater than 150%, the amorphous area is too large to allow the dye to move out easily, resulting in a tendency of the cloth to fade after dyeing. In the case of DTY, the elongation was less than 10%, which indicated a smaller amorphous area; thus, the dispersed dye could easily get stuck in the pores of the fiber surface, resulting in poor dyeing speed. Based on the spinning convention, the appropriate values of the elongation for POY and DTY are 140–150% and 20–30%, respectively, which are the optimum conditions for dyeing.18–20 An analysis of physical properties, as presented in Tables 3–6, indicates that the antimicrobial yarns have achieved the required practical standard.
The stress-strain curves of the antimicrobial yarns are shown in Figure 4. Due to the excellent molecular chain alignment in the DTY fibers, the strain increases with increasing stress, and the initial modulus in the stress-strain curve is not clear, as shown in Figure 4(a). As shown in Figure 4(b), the POY fibers reached the yield point immediately upon stressing; the stress remained unchanged and the strain increased rapidly up to a certain point. Subsequently, the molecular chain orthotropy and stress in the POY fibers increased gradually, causing the fibers to break. In comparison, antibacterial POY has a lower stress and a higher strain. The stress and strain curves for DTY (a) and POY (b), using AgA 0.5%, AgA 1.0%, AgA 2.0%, ZnA 1.0%, and CuA 1.0%.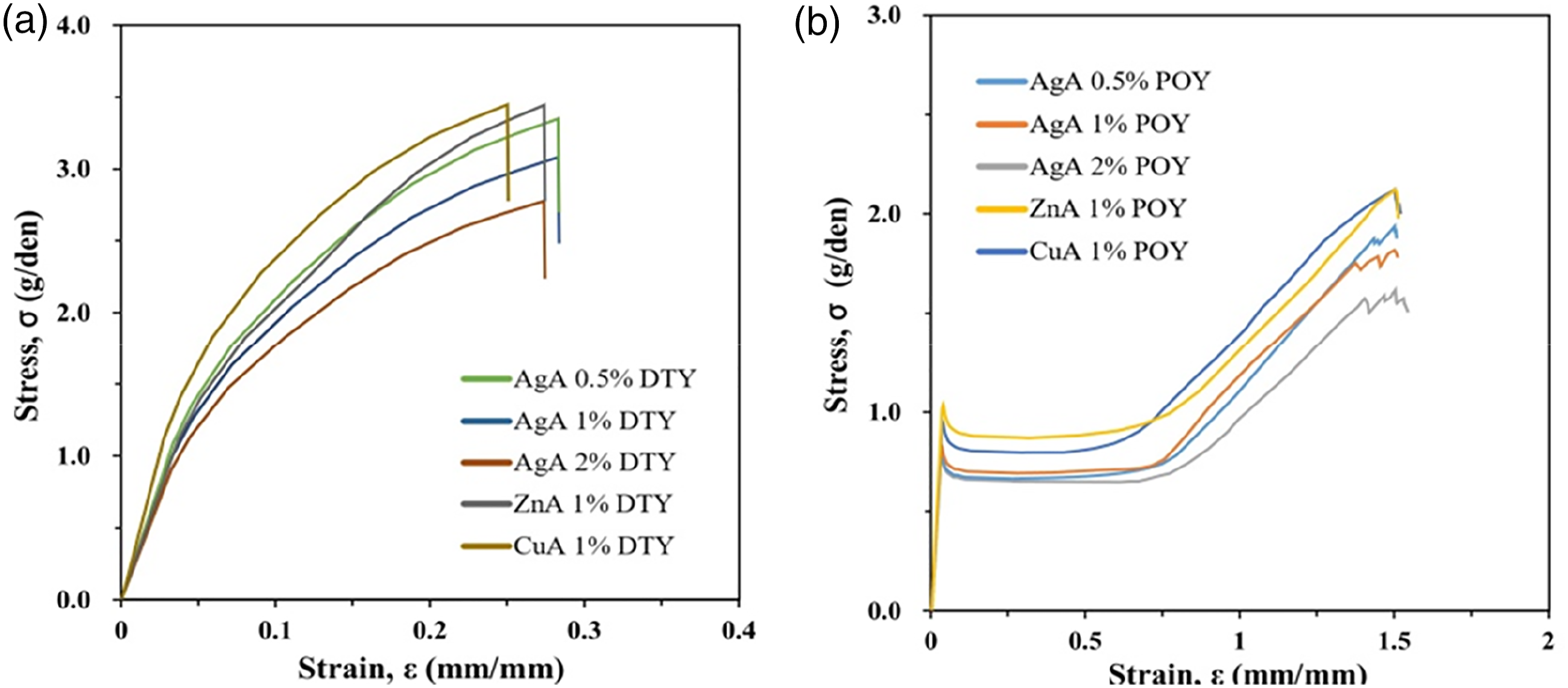
Morphology of antimicrobial fibers
The SEM images of the fiber surface and cross-sectional area illustrate the microscopic morphology when using AgA 1.0%, as shown in Figure 5. SEM images of the (a) fiber surface and (b) cross-sectional area.
Since the antimicrobial agents were dispersed during the experiment, a small amount of antimicrobial agent was observed on the surface and cross-sectional area of the antimicrobial fibers.
Thermogravimetric analyzer
Based on the TGA curve presented in Figure 6(a), the weight loss of the silver antimicrobial agent before 300°C was approximately 3.47 wt%, which represents the weight loss of the impurity in the sample. This indicated that there was no visible weight loss from 300–800°C, suggesting that the silver antimicrobial agent was thermally stable and that no thermal degradation reaction occurred. Figure 6(b) indicates that the weight loss was divided into two stages. The first stage of PBT polymer cracking occurred because of the heat cracking interaction between the PBT and PET polymers in the antibacterial agent sample, the second stage occurred because of PET polymer cracking, and the last residual weight represented the inorganic content. The ZnA and CuA had final residual inorganic contents of 8.85% and 5.14%, respectively. The TGA curve demonstrated that the silver, zinc, and copper antimicrobial masterbatches had no weight loss before 300°C, whereas the silver masterbatch had a one-stage weight loss at 400°C, with a final residual inorganic content of 19.38%. Weight loss was reported in two stages in the zinc and copper masterbatches manufactured using a composite antimicrobial powder. The pyrolysis temperatures of PBT and PET polymers were 400°C and 450°C, respectively. The final residual weight was attributed to the inorganic content resulting from the thermal cracking reaction of the PBT and PET polymers in the masterbatch. The final residual inorganic content of the zinc and copper antimicrobial agents was 2.82% and 1.00%, respectively. The TGA results for the antibacterial fabric are presented in Figure 6(c) and (d). The temperature ranged from 25–800°C, and weight loss of the antimicrobial fabric was detected. Before 350°C, the TGA curve demonstrated that antimicrobial fabrics manufactured using a 5% antimicrobial masterbatch underwent no weight loss. A two-stage thermal cracking process occurs when PET is burned in the air. In the first step, residual carbon products are generated, whereas the second stage is concerned with the thermal degradation of the carbon residue. Weight loss in the first stage accelerated as the temperature increased. During combustion, PET generates carbon residues that react with oxygen at 500–600°C, causing a significant weight loss. TGA spectra of (a) antimicrobial agents, (b) antimicrobial masterbatches, (c) antimicrobial fabrics, and (d) AgA antimicrobial fabrics with various ratios.
Dyeing hue
Values of L*, a*, b*, and △E* for PET antimicrobial fabrics when dyed with various colors.

Antibacterial test results
Antibacterial activity tests were performed according to JIS L1902:2015. The antibacterial effect of the tested fabric was examined by determining the antibacterial activity of the finished fabric. Test strains of gram-negative (P. pneumoniae) and gram-positive bacteria (E. coli and S. aureus) were used to evaluate the antibacterial effect of the fabric before and after dyeing and finishing. Antibacterial durability is a significant evaluation index for antibacterial fabrics. It is important to note that antibacterial agents on the fabric surface may be lost during washing.
Antibacterial activity tests for silver, zinc, and copper fabrics.

Note. JIS L1902, gram-negative bacteria (Pneumococcus pneumoniae); # = No reduction.
Antibacterial activity tests including 50 washes of the silver fabrics with various AgA ratios.
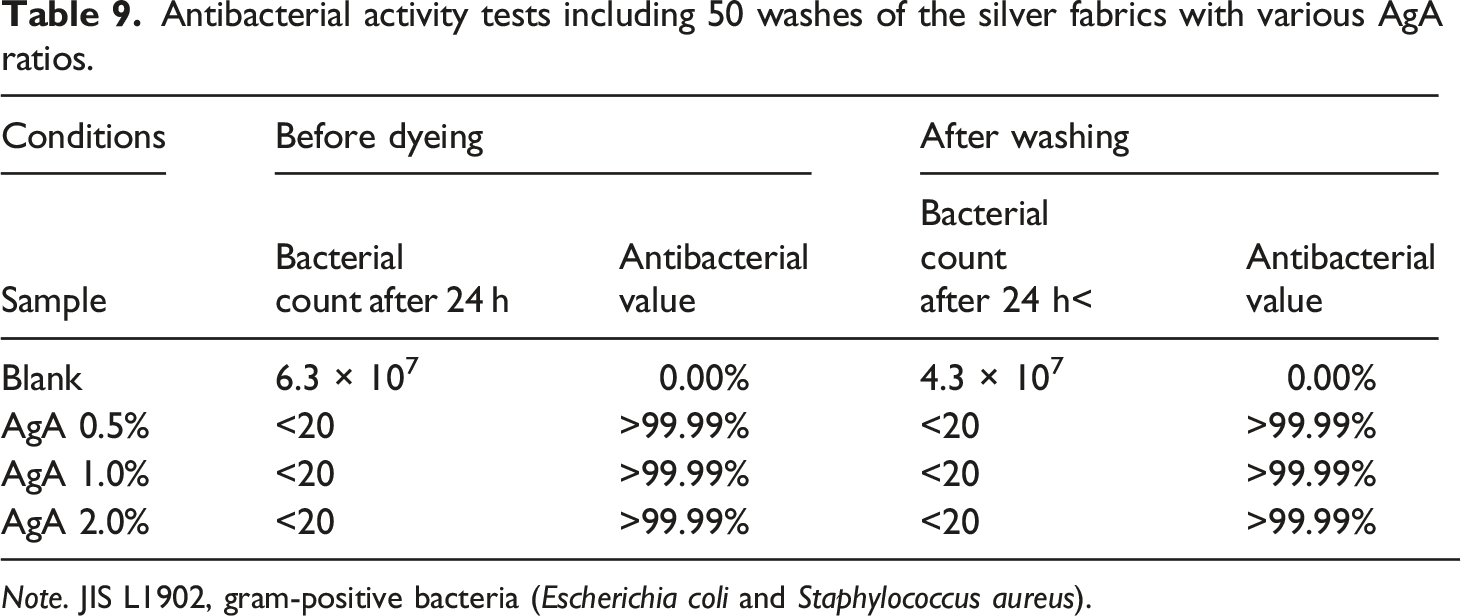
Note. JIS L1902, gram-positive bacteria (Escherichia coli and Staphylococcus aureus).
Antiviral test results
Antiviral efficacy tests for the various textiles.

Remarks: Efficacy of antiviral activity value (informative); Good effect: Antiviral activity value ≧2.0; Excellent effect: Antiviral activity value ≧3.0.
Conclusion
In this study, three types of antimicrobial agents, AgA, ZnA, and CuA, were added and spun into an antimicrobial DTY. The SEM images showed that AgA was uniformly dispersed, whereas an agglomeration of 4 μm was observed for ZnA and CuA. The results of the filtration pressure rise test indicated that AgM had the best long-term spinnability, followed by ZnM, while CuM showed the worst results. Thus, PET containing AgA was able to efficiently complete the spinning process. ZnA and CuA agglomerated in ZnM and CuM during the spinning process, and the antimicrobial agents were partially exposed on the fiber surface. Therefore, some of the ZnA and CuA particles were washed away during the dyeing and post-finishing processes, which reduced the content of antimicrobial agents in the fiber, resulting in reduced antibacterial activity. AgA does not have the disadvantage of particle loss, and permanently retains its antibacterial ability. Therefore, the silver fabric displays long-lasting antibacterial and antiviral effects.
Footnotes
Acknowledgements
The authors are grateful to Far East New Century Corporation, AUSPRING CO., LTD. and Taiwan Textile Research Institute for providing manufacturing equipment to accomplish this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

