Abstract
Heavy metal contamination is a serious concern worldwide. In order to remove heavy metal ions from wastewater efficiently, multifunctinoal biomass-based material have been utilized. Herein, hyperbranched polymer modified cellulose biomass (HBP-CB) derived from textile waste materials ramie fibers was successfully developed by construction of Fe3O4/SiO2/polymer multiple structure through an etch-fill strategy. Specifically, Fe3O4 and SiO2 particles were introduced to the oxidation etching cellulose surface, and subsequently modified by hyperbranched polymer The adsorption behavior of the adsorbent toward two classes of heavy metal ions was investigated. The results indicated that HBP-CB owned excellent adsorption capacities for chromium and Cu(II) with maximum 123.5 mg/g and 149.0 mg/g and magnetic recovery performance in aqueous medium. These are owing to the oxidization etching biomass matrix with a higher BET surface area of 4.61 m2/g and the hybrid multiple structure modified by functional hyperbranched polymer with high dense adsorptive sites on the matrix. The adsorption behavior was well described by pseudo-second-order kinetic model and Langmuir isotherm model, revealing a rapid surface adsorption and monolayer spontaneous chemical adsorption. For recycling, 81% of adsorption capacity could be retained after five recovery cycles. These demonstrate that the hybrid multiple structure based on biomass, nanomaterials and polymer could provide sustainable and high-performance adsorption property for wastewater treatment.
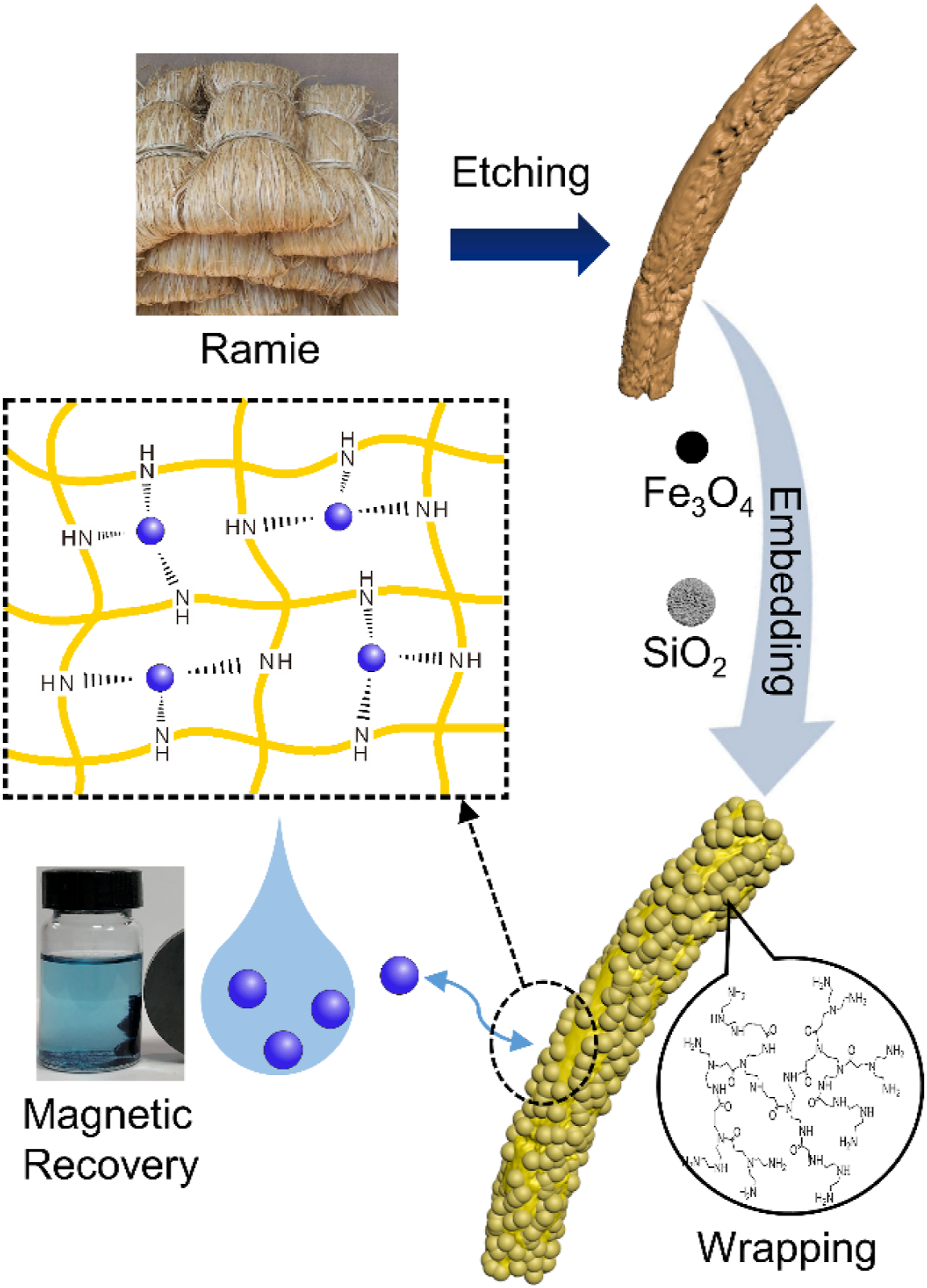
Introduction
Heavy metal contamination in the environment is a serious concern worldwide. In the event that these ions contaminate water resources, they are highly toxic to the animals, humans and the environment combined with their nonbiodegradability and ability to bioaccumulate in living organisms and the food chain.1–4 Among the heavy metals, chromium (Cr) and copper (Cu), which are categorized as environmental priority pollutants owing to the high toxicity, are considered the representatives ones and have received the most attention.5–7 In order to eliminate this risk, a great deal of research has focused on developing effective and less costly methods such as chemical precipitation, membrane separation, ion exchange, biological treatment, and adsorption.8–11 Among them, chemical precipitation methods can effectively treat wastewater with high concentrations of heavy metal ions to low concentrations, but they cannot be removed completely due to the high input of precipitating agents.12,13 Biosorption has emerged as a promising alternative to conventional methods by considering the economic benefit and removal efficiency.14–16 And plant-based biomass such as cellulose particularly have been a focus of attention owing to the outstanding uptake capacity, nontoxicity, low-cost and sustainability.17–19
Qiao et al. obtained a spherical cellulose adsorbent by preliminary chemical cross-linking strategy and unique superpores with the pore size of 0.5∼1.0 μm formed, which contributed to high specific surface area (152 m2/g). And the adsorbent displayed high adsorption capabilities toward heavy metal ions (98.86 mg/g for Cu(II) and 102.29 mg/g for Co(II)) and fast separation (equilibrium within 30 min). 20 Choi et al. prepared a thiol-functionalized cellulose nanofiber membrane by the deacetylation of electrospun cellulose acetate nanofibers and subsequent esterification of a thiol precursor molecule, and maximum adsorption capacities in the Langmuir isotherm for Cu(II), Cd(II), and Pb(II) ions were 49.0, 45.9, and 22.0 mg/g, respectively. 21 Zhou et al. designed a cellulose-based amphoteric adsorbent with a high density of amino and carboxyl groups for removal of low-level heavy metal ions. The adsorbent could completely remove Cr(VI), Cd(II), Cu(II), Zn(II) and Pb(II), which was attributed to the specialization and cooperation mechanism between amino and carboxyl groups. 22 The literature analysis revealed spherical and membrane cellulose-based biosorbents shown excellent performances owing to high surface area and enormous functional groups, and biosorbents with high density of functional groups such as amino and carboxyl groups exhibited outstanding adsorption capacities for heavy metal ions from medium media.23–26 Whereas, these fabrication processes often need multiple steps which were complicated and required strict experimental conditions.27–29 Thus, the design and development of a biosorbent with large surface area for modification and high density of adsorptive groups via simple and easy-controlled preparation process will be beneficial to improve the rapid construction of high-efficiency adsorption system for heavy metal ions.
In this study, low-cost waste ramie fabric was adopted, for it could provide cellulose materials which own good mechanical strength for recyclable carrier. Thus, we unveiled ramie-based mesoporous silica/polymer hybrid magnetic adsorbent (HBP-CB) by etch-fill strategy as a novel magnetically separable biosorbent, and systematically assessed its adsorption performance with two hazardous heavy metal ions, Cu(II) and Cr(III). The novelty of this study is the formation of a multiple functional structure via an etch-fill strategy which could optimize the function structure and largely improve the adsorptive performance. The mesoporous silica particles were adopted embedded onto the oxidized etching cellulose surface to provide larger surface area for modification and adsorption, and the hyperbranched polymer was utilized to enhance the adsorption performance for heavy metal ions, constructing a magnetic adsorptive structure with high density of functional groups. The morphology and chemical structure of adsorbents were studied, and the corresponding kinetic and isothermal analysis were carried out. The separability and reusability of as-prepared adsorbent were also studied. Based on the results, we discussed the applicable feasibility of as-prepared biosorbent in heavy metal-polluted wastewater remediation.
Material and methods
Materials and reagents
The ramie-based biomass employed was derived from waste garments provided by Testing Center of Textile Industry (Changshu). Ferrous chloride tetrahydrate (FeCl2·4H2O), ferric chloride hexahydrate (FeCl3·6H2O), cetyltrimethylammonium bromide (CTAB), triethanolamine (TEA), tetraethoxysilane (TEOS), 3-aminopropyl triethoxysilane (APTES), sodium hydroxide (NaOH), hydrochloric acid (HCl), ammonium hydroxide (NH3·H2O), sodium periodate (NaIO4), copper nitrate (H10Cu2N4O17) and chromic nitrate nonahydrate (Cr(NO3)3·9H2O) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). All reagents used were analytically pure and without any treatment.
Synthesis of mesoporous silica particles
Amino-functionalized mesoporous silica particles (MSPs) were prepared by copolycondensation, as shown in Figure 1. 1.0 g of template agent CTAB, 100 mL of deionized water and 70 mL of ammonia were sequentially added to the three-necked flask, and stirred in an oil bath until CTAB was completely dissolved. Then 5 g TEOS was added dropwise. After 1 h, 1.67 g APTES was added dropwise, and stirring was continued for 6 h. The crystallization was carried out by standing at room temperature for 12 h, followed by filtration, washing and drying. The template agent was removed by hydrochloric acid-ethanol solution with a mass fraction of 2.5 wt%, and the product was washed for several times and dried to constant weight to obtain amino-functionalized mesoporous silica particles (MSPs).
30
Schematic illustration of the preparation of HBP-CB and adsorption for heavy metal ions.
Pre-treatment and oxidized etching of fibers
The pure ramie fabric was cut and scattered into short pieces at a length of about 10 mm, and then 12 g of the pieces were added to the NaOH solution with mass fraction of 5 wt% and boiled (100°C) for 3 h, followed by 20 wt% NaOH boiling for 1 h. NaOH was used to fully weaken the hydrogen bonding on the ramie surface in order to activate the cellulose matrix for subsequent treatment. After cooling to room temperature (25°C), the product was filtrated, washed and dried to obtain activated ramie fibers.
The oxidized ramie fibers were prepared by selective oxidation process with sodium periodate. Briefly, 10 g of ramie fibers were dispersed in 800 mL of acetic acid buffer (pH 4.0) containing 70 g of sodium periodate. The reaction mixture was stirred magnetically in the dark for 6 h at room temperature. During the oxidation etching process, large amouts of grooves and porous formed on the fiber surface, achieving an etching cellulose biomass (E-CB) with large surface area.
Preparation of magnetic, mesoporous silica particles- and hyperbranched polymer-functionalized fibers
Fe3O4 magnetic nanoparticles were prepared by co-precipitation method. The specific experimental process is as follows: 3.98 g of ferrous salt (FeCl2·4H2O) and 5.4 g of ferric salt (FeCl3·6H2O) were placed in a beaker and dissolved in 100 mL of distilled water, and stirred for 30 min at 50°C to make FeCl2·4H2O and FeCl3·6H2O fully mixed evenly. Furthermore, 25 mL NH3·H2O was added to the mixture under stirring at 50°C, and the ramie fibers were added to the mixture at 80°C for 2 h. Thus, magnetic cellulose biomass (M-CB) was obtained.
100 mg MSPs were dispersed in 100 mL anhydrous ethanol, stirred at 50°C for 30 min. After they were fully dispersed, 1 g M-CB was added and mixed well, stirred at 50°C for 2 h. The product was suction filtered, washed, and vacuum dried at 80°C to obtain mesoporous silica particles-functionalized cellulose biomass (MSP-CB).
Required amount of MSP-CB was added in 2 wt% terminal amino hyperbranched polymer (HBP) ethanol solution, and then the mixture was stirred at 60°C for 3 h under nitrogen atmosphere. The HBP, possessing a quasi-spherical dentritic macromolecular structure with low viscosity and high density of imino and amino groups, was prepared by a methodology of polymerization between AB2 type and AB type monomers according to the method by the previous study of our research team (shown in Figure 2).
30
The obtained HBP-functionalized cellulose biomass (HBP-CB) was suction filtered, washed with sufficient amount of deionized water, and dried in a vacuum oven overnight. Schematic illustration of the preparation of HBP.
Measurement and characterization
The apparent morphologies of adsorbents were studied by field emission scanning electron microscopy (FESEM; ZEISS SIGMA). Fourier transform infrared (FT-IR) spectroscopy was made by Nicolet 5700 (Thermo Nicolet) to identify the structures and chemical compositions of materials. The further X-ray photoelectron spectroscopy (XPS) measurements were conducted with an Axis Ultra HAS system. The distribution for element compositions of adsorbents surface was determined by a desktop scanning electron microscope and its supporting energy dispersive spectrometer (EDS, Hitachi, TM3030). The crystal structures of adsorbents were determined by X-ray diffractometer (XRD; Brucker AXS D8 Advance). The particle size distribution measurement was conducted by a laser particle sizer (Malvern Panalytical, Mastersizer 3000). The N2 adsorption-desorption isotherms for materials was measured at 77.34 K using Micromeritics ASAP 2020 analyzer (Micromeritics). The magnetic performance was measured by a magnetometer (VSM, Lake Shore 7404). The concentrations of heavy metal ions were estimated by inductively coupled plasma atomic emission spectroscopy (ICP; Thermo Scientific ICAP 6000 DUO).
Adsorption experiments
Typically, the adsorbent at a dosage of 1 g/L was added to Cu(II) and Cr(III) solutions (each for 100 mL) at a concentration of 200 mg/L, respectively. Specifically, control variable method was adopted to conduct the adsorption experiment. Adsorption took place during agitation of mixture solutions at 30°C for a set time segments (for 5–720 min, studied by adsorption kinetics experiments). Then the mixture was centrifuged and small amount of supernatant was taken for analysis using ICP. The adsorption capacity Q
e
(mg/g) is calculated according to the
Results and discussion
Characterizations
Morphological study for CB, E-CB, MSP-CB, and HBP-CB are displayed by SEM images. In comparison with CB containing a smooth surface shown in Figure 3(a), the E-CB fiber became rougher and flexible, and obvious irregular grooves with abundant micropores and nanopores are observed on the surface (shown in Figure 3(b)), indicating the drastic morphological changes induced by periodate oxidation and part of cellulose was converted into loosen oxidized long chains which was dissolved in solvent. The oxidation etching helps largely raise the specific surface area and chemical activity of cellulose matrix, giving rise to an efficiency enhancement of subsequent modification. After introduction of Fe3O4 magnetic nanoparticles and mesoporous silica particles (MSPs), large amounts of functional particles were adsorbed and embedded by self-assembly with a high density in the grooves and pores of fiber surface as shown in Figure 3(c). Notably, MSPs were introduced after Fe3O4 nanoparticle to prevent the pore channels from being adhered and blocked. Apparently, MSPs with an average size of 2 μm piled on Fe3O4 nanoparticles with an average size of 149 nm (the particle size was measured by particle sizer), distributing evenly on the material surface, further increase the surface area and active sites. As displayed in Figure 3(d), with the further treatment of HBP, the grooves and pores declined due to the coverage of the amorphous HBP block layer, suggesting that Fe3O4 nanoparticles and MSPs on the HBP-CB surface were wrapped by polymer, forming an etch-fill functional system. Based on the structure, HBP-CB obtained an unique 3D multiple structure of Fe3O4 nanoparticles, MSPs and HBP with large surface area and high dense adsorptive sites, providing a high-efficiency adsorptive property as illustrated in Scheme 1. SEM photographs of CB (a), E-CB (b), MSP-CB (c), and HBP-CB (d).
The TEM images of MSPs indicated their structure and size, which confirmed that the monodisperse MSPs possessed an uniform morphology of egg-like shape at a size of about 300 and 500 nm for width and length (Figure 4(a)), and the internal structure was highly ordered with parallel mesoporous channels at a diameter of 2.39 nm (Figure 4(c)). Besides, textual property for ASPs was investigated and analyzed and the BET surface area reached as high as 693.3 m2/g, which could provide the functional biomass with substantial adsorptive sites and high adsorptive performance. TEM images of MSPs (a) and the corresponding enlarged views (b, c).
The elemental mapping for HBP-CB were conducted to determine the distribution of the characteristic elements from the functional components. As shown in Figure 5, Fe and Si exhibited uniform and dispersed distribution with a high density, indicating Fe3O4 nanoparticles and MSPs were assembled and embedded evenly on the material surface impacted by steric hindrance and electrostatic action between the functional particles and the oxidized cellulose surface. Thus, a pepper-sprinkling-like functional matrix with multiple functional particles was constructed for further modification and adsorption. After graft of HBP, it was observed that the material surface was covered evenly by N element from HBP, indicating the multiple-particles cellulose matrix was fully wrapped by the hyperbranched polymer. And it is accordant with SEM results. Elemental mapping of Fe (b), Si (c) and N (d) for HBP-CB.
For better explanation, the comparison among raw cellulose biomass (CB) and modified ones was tested by FT-IR analysis as shown in Figure 6. The FT-IR spectra for CB, E-CB, M-CB, MSP-CB and HBP-CB display the characteristic band of the cellulose structure at 2918 cm−1 is ascribed to the symmetric and asymmetric stretching vibrations of the C-H groups from cellulose, and the bands at 1237, 1091 and 895 cm−1 are associated with asymmetric out-of-phase ring stretching vibration for cellulose,31,32 revealing the main cellulose structure was preserved and oxidation reaction took place only on the surface of cellulose materials. During the modification process, the characteristic bands at 2918, 1237, 1091 and 895 cm−1 shown a decline tendency in intensity compared with that of CB, demonstrating that the cellulose rings opened and the ring structure has been influenced. Meanwhile, the adsorption at 1409 cm−1 became weaker, revealing the reduction in inter- or intramolecular hydrogen bonded hydroxyl groups of cellulose. These phenomena indicate the dramatic influence of periodate oxidation etching on cellulose structure, which is consistent with the SEM results. After modification, a characteristic peak at 1735 cm−1 corresponding to the C = O stretching vibration was generated and observed in the oxidized cellulose samples (E-CB, M-CB, MSP-CB and HBP-CB), and the peak at 3366 cm−1 declined after oxidization modification, which is associated to the conversion of −OH (3366 cm−1) on C2, C3 positions into −CHO groups by the oxidation reaction.
33
And the related process was illustrated in Figure 1. In addition, new adsorption bands at 1646 cm−1 and 1554 cm−1 were observed in the spectrum of HBP-CB, which are assigned to the amide I (νC=O) and amide II (δN-H), respectively. This confirms that HBP with abundant amide groups was introduced successfully onto the cellulose surface. FT-IR spectra of CB, E-CB, M-CB, MSP-CB and HBP-CB.
The XPS studies were performed to further investigate about the chemical state of elements during the etch-fill modification. As shown in Figure 7(a), the wide scan XPS spectra for CB, E-CB, M-CB, MSP-CB and HBP-CB exhibit the apparent peaks at binding energy around 103, 154, 284, 400, 533 and 710 eV which are ascribed to Si2p, Si2s, C1s, N1s, O1s, and Fe2p, respectively.34,35 Among them, the peak for Fe2p is ascribed to the magnetic Fe3O4 nanoparticles, and the peaks for Si2p and Si2s are considered derived from mesoporous silica particles (MSP), which indicates the successful introduction and functionalization. The graft of HBP is also confirmed by the obvious N1s peak for HBP-CB spectra, which is attributed to the amino groups, and the smaller N1s peak for MSP-CB spectra could be ascribed to the amino-functionalized MSPs. Furthermore, in comparison with that of CB (Figure 7(b)), the high resolution C1s peak of E-CB could be deconvoluted into three peaks at 284.6, 286.2, and 287.5 eV corresponding to the C-C, C-O, and C = O bonds, respectively (shown in Figure 7(c)), suggesting the conversion of −OH on C2, C3 positions into −CHO groups by the oxidation reaction.36,37 And for C1s of HBP-CB (shown in Figure 7(d)), C-N species associated with the peak at 285.6 eV was deconvoluted, furtherly confirming the successful graft of HBP and MSPs. Meanwhile, new C = N species with the peak at 287.0 eV was deconvoluted which was considered from the Schiff-Base reaction between amino groups and aldehyde groups during the chemical graft of HBP and MSPs onto the oxidized cellulose surface (illustrated in Figure 1). Notably, the intensity of C-O and C = O for HBP-CB increased in a comparison with that of E-CB, suggesting the presence of MSPs and HBP with oxysilane and amide groups, Therefore, the XPS results proved the achieved modifications, which are consisted with the results of SEM, EDS and FT-IR. XPS wide scan spectra of CB, E-CB, M-CB, MSP-CB, and HBP-CB (a); and C1s spectra of CB (b), E-CB (c), and HBP-CB (d).
XRD analysis was carried out for clarifying the crystal phases and composition structures of synthesized materials. The XRD results of pure CB, E-CB and HBP-CB indicate that two main phases exist (shown in Figure 8). For CB, the characteristic diffraction peaks for cellulose Iβ type can be found at 14.5°, 16.6°, 22.8° and 34.3°, which reveal the characteristic polymorph of cellulose with high crystallinity. And the broad peaks around 16° and 22° are related to the amorphous region and crystalline area of natural cellulose, respectively.32,33 After oxidation modification, the width of diffraction peaks for three materials seemed nearly constant and only the peak height declined along with the oxidation reaction. This phenomenon demonstrates that the modifications including oxidation reaction took place only on the external surface of cellulose materials, which is also consistent with the results of FT-IR. Specifically, as for M-CB, the characteristic peaks for cellulose crystal form declined obviously, indicating that the crystallographic structure was destructed. And the loss of crystallinity was ascribed to the ring-opening reaction of glucose molecules and the destruction of their ordered packing. Moreover, for HBP-CB, the XRD patterns display five clear characteristic diffraction peaks at 2θ values of 30.4°, 35.8°, 43.3°, 57.3°, and 63.1°, corresponding to (220), (311), (400), (511), and (440) Bragg reflection of the face-centered cubic structure of Fe3O4, respectively.38–40 It demonstrates that the Fe3O4 nanoparticles has been introduced and embedded evenly on the material surface, which is accordant with the results of SEM and EDS. XRD patterns of CB, E-CB and HBP-CB.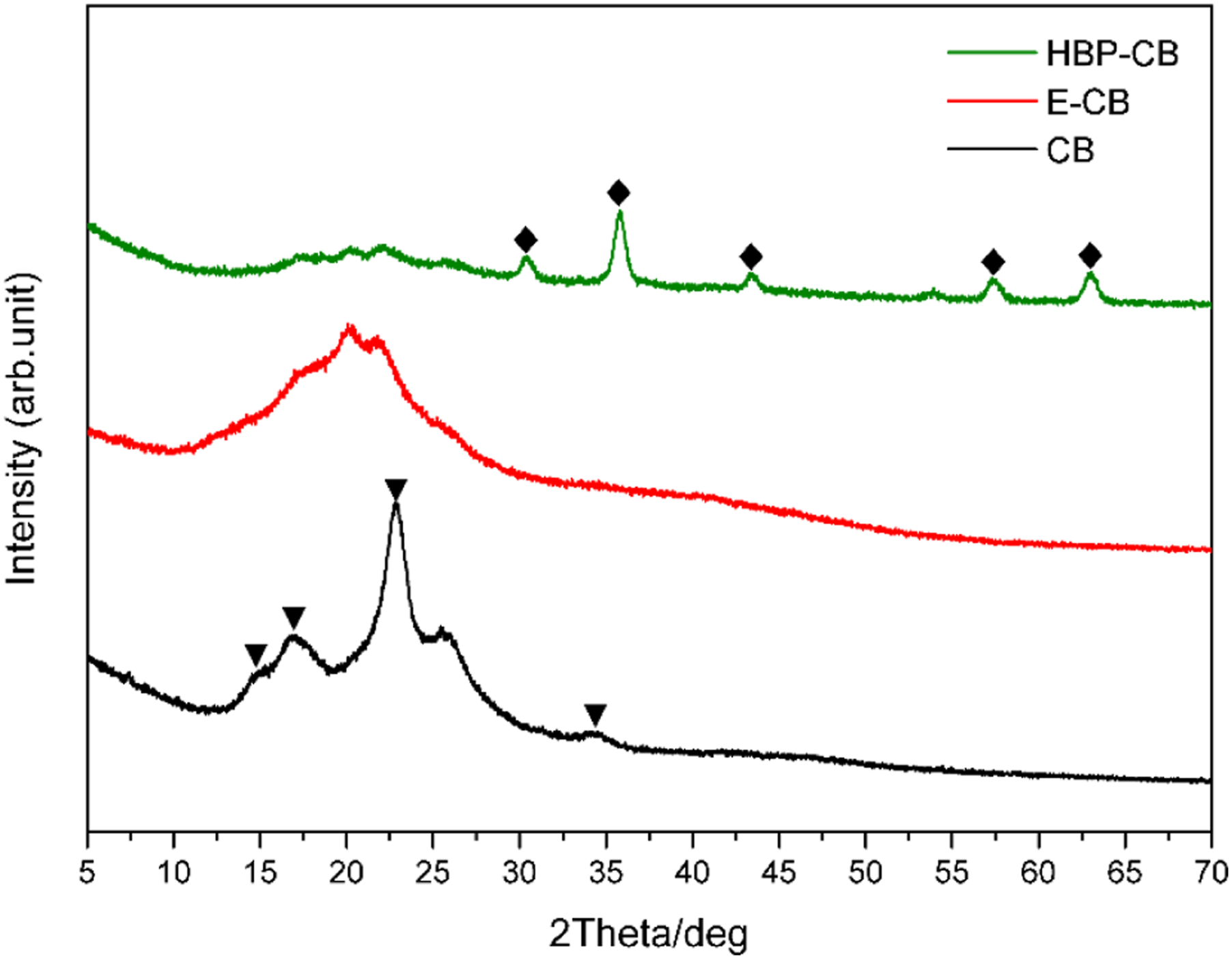
Furthermore, the specific surface area of as-synthesized MSP and cellulose materials was investigated by N2 adsorption-desorption isotherms and analyzed by using the Brunauer−Emmett−Teller (BET) method as depicted by Figure 9. And the corresponding pore volume and pore size were calculated by the Barrett-Joyner-Halenda (BJH) method. The data derived from the basis of the isotherms are summarized in Table 1. It can be observed by Figure 9(a) that MSP exhibited a quasi-type IV isotherms without an obvious adsorption-desorption hysteresis loop, revealing a complicate, irregular and uneven pore texture affected by the introduction of functional organosilane via co-condensation process.41,42 Nevertheless, BET surface area of MSP reached as high as 810.28 m2/g, and its pore volume and pore diameter were 0.43 cm3/g and 2.14 nm, indicating the low-density structure with a large surface area.
41
After oxidation modification, the main part of curves for adsorption-desorption marked by dotted lines increased to the range of 0.44–0.68 cm3/g from 0.16–0.36 cm3/g, and the curve of E-CB became fluctuating to some extent. These demonstrates the surface of E-CB became rougher and irregular, giving rise to a growth of specific surface area. Owing to the uniformly coverage of MSPs with super large surface area, the MSP-CB shown quasi type IV isotherm with an adsorption-desorption hysteresis loop at P/P0 above about 0.7, revealing the distinct capillary condensation and narrow pore size distribution.43,44 And the main part of curves for adsorption-desorption reached around 1.4–3.2 cm3/g, which was higher than that of other materials. N2 adsorption−desorption isotherms for MSP (a), CB (b), E-CB (c) and MSP-CB (d). Surface area, pore volume and pore size of MSP, CB, E-CB and MSP-CB.
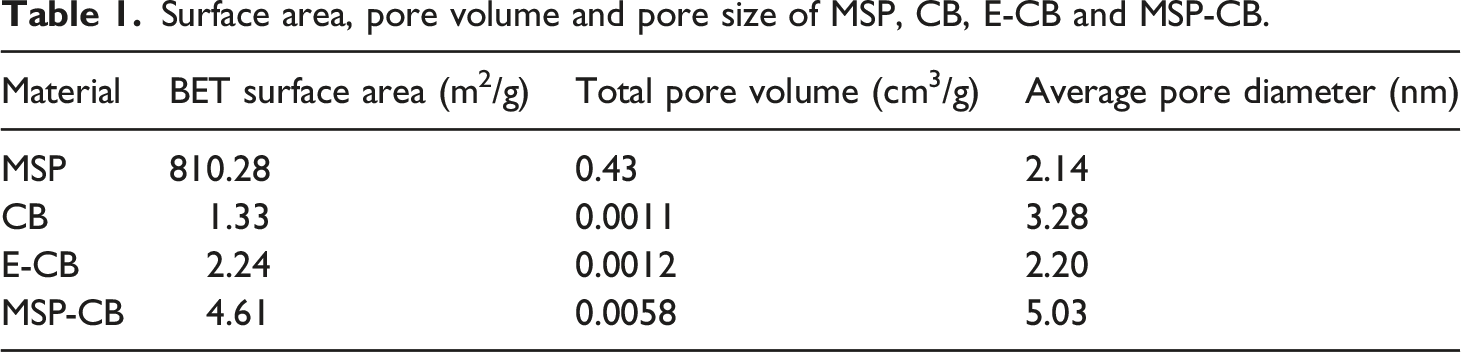
As shown in Table 1, in a comparison with CB, BET surface area of E-CB raised to 2.24 from 1.33 m2/g due to the periodate oxidation etching of material surface, while the average pore diameter decreased to 2.20 from 3.28 nm due to the elimination of macro pores and the formation of micropores and nanopores during oxidation reaction. Therefore, the larger specific surface area is ascribed to both the conversion of crystalline to amorphous state and the more porous texture of the cellulose network, which is identical to the results of SEM and XRD.45–48 As for MSP-CB, all the BET surface area, total pore volume and average pore diameter increased dramatically to 4.61 m2/g, 0.0058 cm3/g and 5.03 nm, respectively. This phenomenon is ascribed to the embedding of MSPs on the material surface with a uniform distribution besides the large surface area provided by MSPs. And this structure provides an excellent multifunctional matrix for further modification and adsorption.
Magnetism property by Fe3O4 nanoparticles can provide magnetic recovery for adsorbents, which is significant for the practical adsorption application. The magnetism performance for M-CB, MSP-CB and HBP-CB was investigated by VSM measurement (depicted in Figure 10), and the maximal saturation magnetization presents as 4.40, 4.00 and 3.82 emu/g, respectively. It suggests that Fe3O4 nanoparticles were assembled and embedded evenly and stably on the material surface, and the introduction of MSPs and HBP might slightly impact the effectiveness of magnetization. Thus, VSM results indicate the successful magnetization modification which is beneficial to rapid separation via applying an external magnetic field over adsorption and desorption processes. These are consistent with the results of SEM, EDS and XRD. Magnetization curves of M-CB, MSP-CB, and HBP-CB.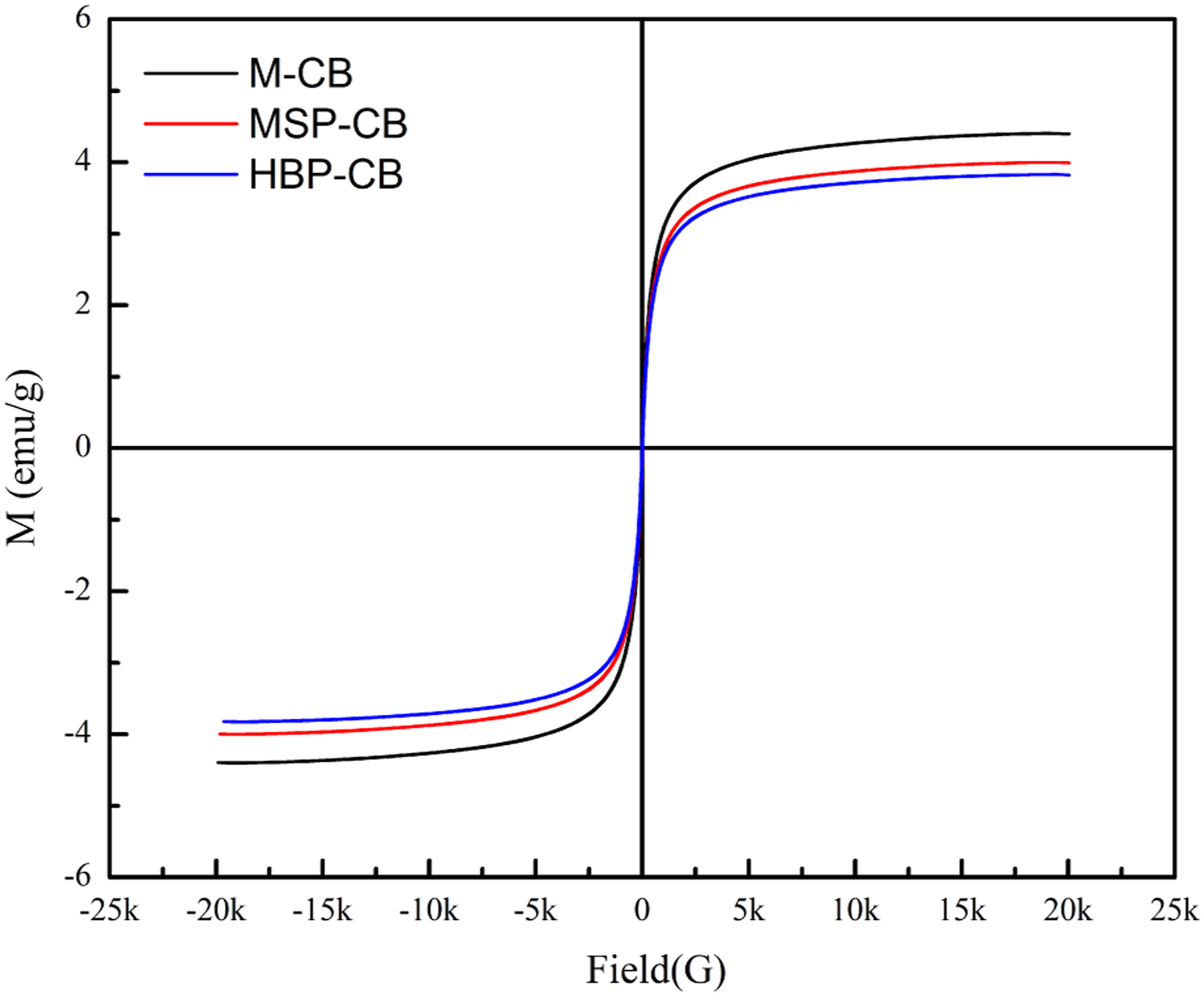
Adsorption property and kinetics
Figure 11 shows the effect of contact time on the adsorption capacity of HBP-CB for Cu(II) and Cr(III). The saturated adsorption capacities are 123.5 and 149.0 mg/g for Cr(III) and Cu(II). Specifically, the adsorption capacity increased rapidly with the growth in contact time during initial period with more than 90% of the saturated adsorption capacity was achieved within about 100 min. It is mainly owing to the fast adsorption by high surface energy related to the multiple structure with evenly particle distribution and high surface area. After initial period, the adsorption curves became gentle and constant, which is ascribed to the slower chemical interactions which make adsorbates immobilized, such as complexation and chelation for metal ions by functional molecules cage structure of HBP and amino groups of MSPs, as illustrated in Figure 1. Thus the adsorption equilibrium was reached within about 250 min. Effect of contact time on the adsorption by HBP-CB for Cu(II) and Cr(III). Three repeat experiments were performed.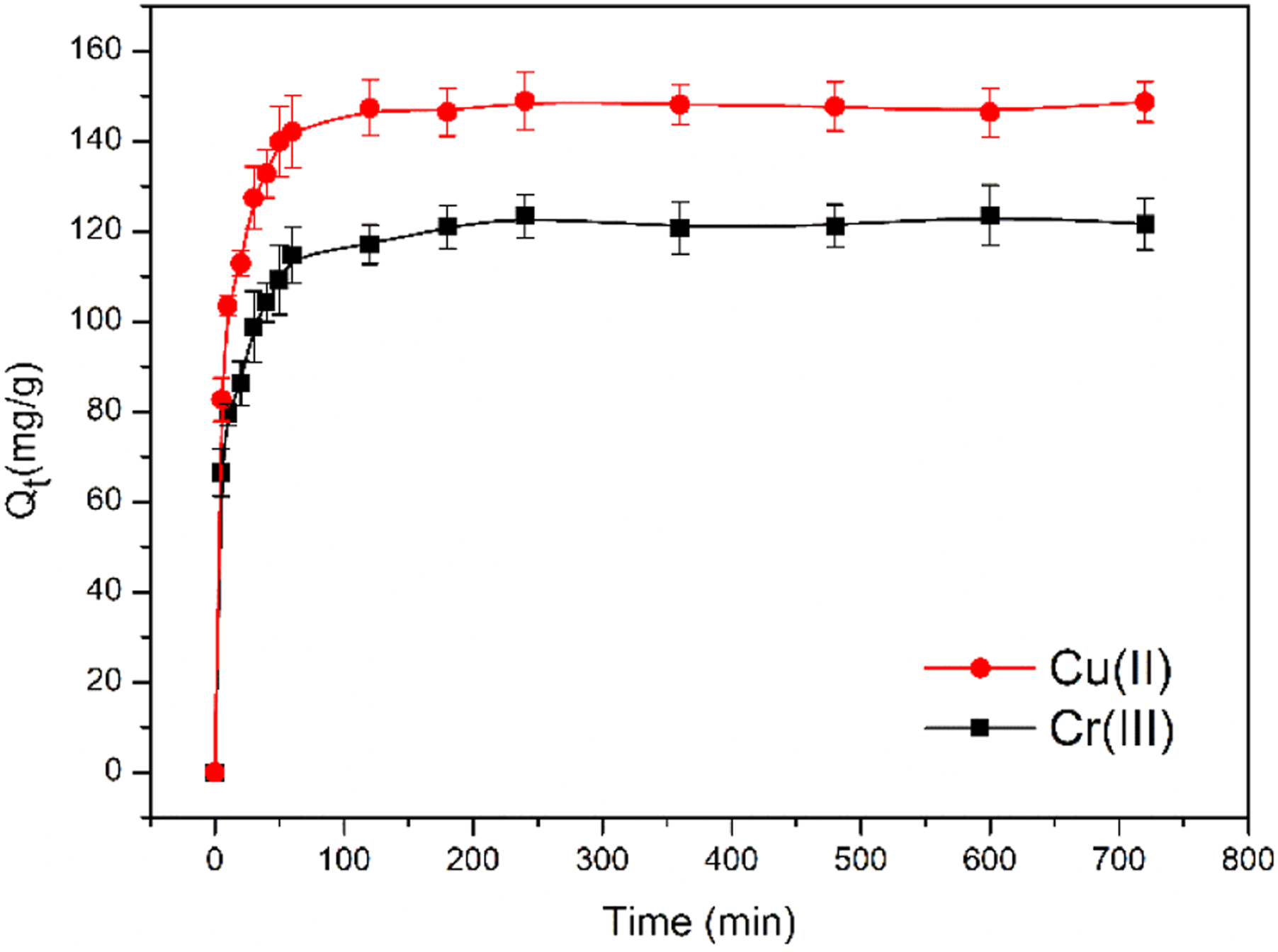
In order to investigate the kinetic mechanism, which controls the residual adsorption time during adsorption process, the Lagergren pseudo-first-order model (Figure 12(a)) and pseudo-second-order model (Figure 12(b)) are adopted to depict the adsorption process by following equations. Peudo-first-order kinetic (a) and pseudo-second-order kinetic (b) plots for HBP-CB.
The pseudo-first-order kinetic model is given as

Kinetic parameters and experimental adsorption capacities for the removal of Cr(III) and Cu(II) by HBP-CB.

Adsorption isotherm
As shown in Figure 13, the equilibrium adsorption capacity increases continuously with the initial concentration and then stabilized due to the increase in the concentration gradient driving force. In addition, the adsorption isotherm can indicate the distribution of adsorbate molecules between the liquid and solid phases as the adsorption process reached equilibrium (Figure 14). The equilibrium adsorption isotherm was described using the Langmuir and Freundlich model. According to the Langmuir isotherm model, the adsorption is based on the formation of adsorbate as monolayer coverage onto the homogeneous adsorbent surface with identical sites in terms of energy. Thus, the metal ions were supposed to be adsorbed via a monolayer spontaneous chemical reaction, and complex bond and surface attraction are the dominant adsorption mechanisms. Effect of initial concentration on the adsorption by HBP-CB for Cr(III) and Cu(II). Three repeat experiments were performed. Freundlich (a) and Langmuir (b) adsorption isotherm plots for HBP-CB.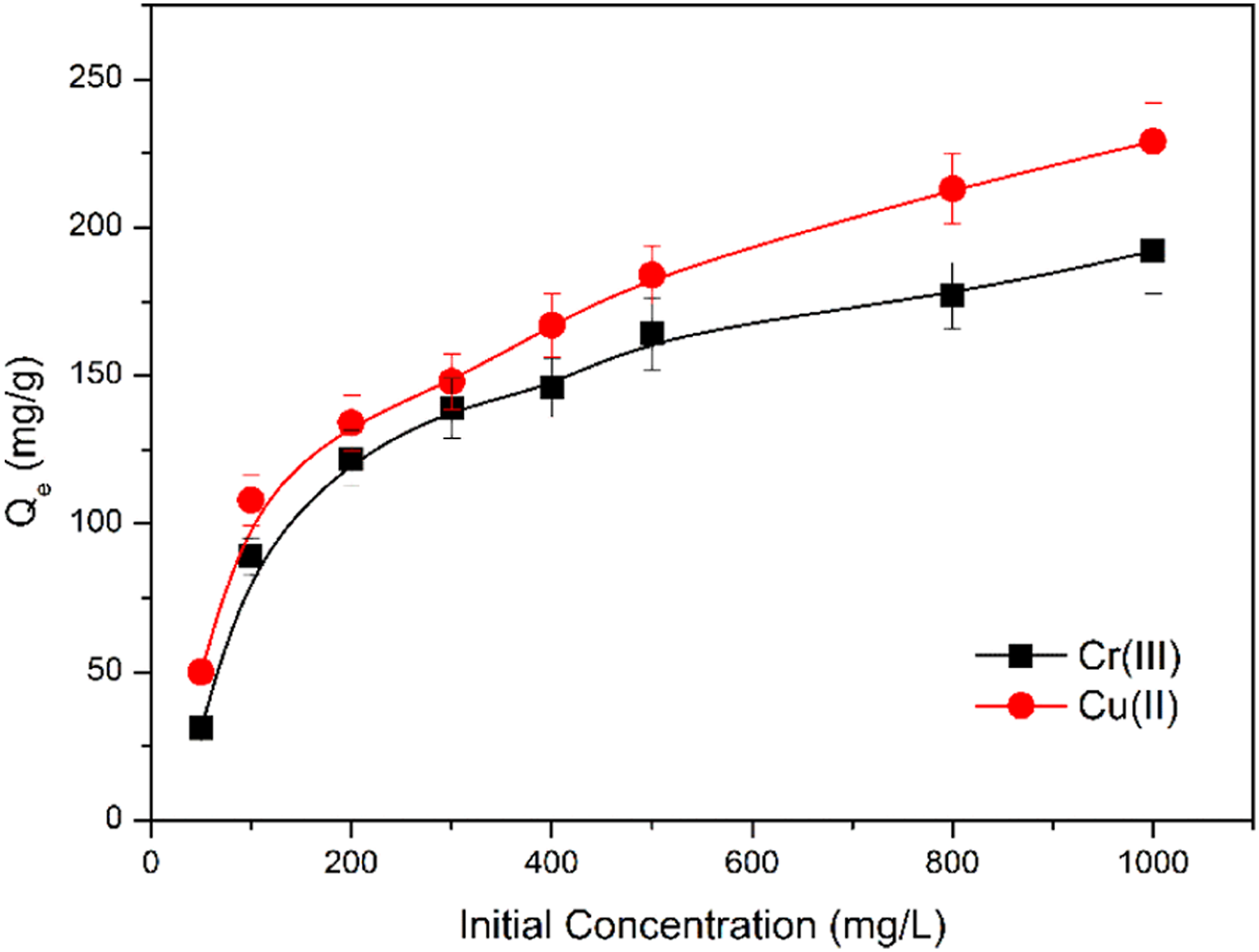
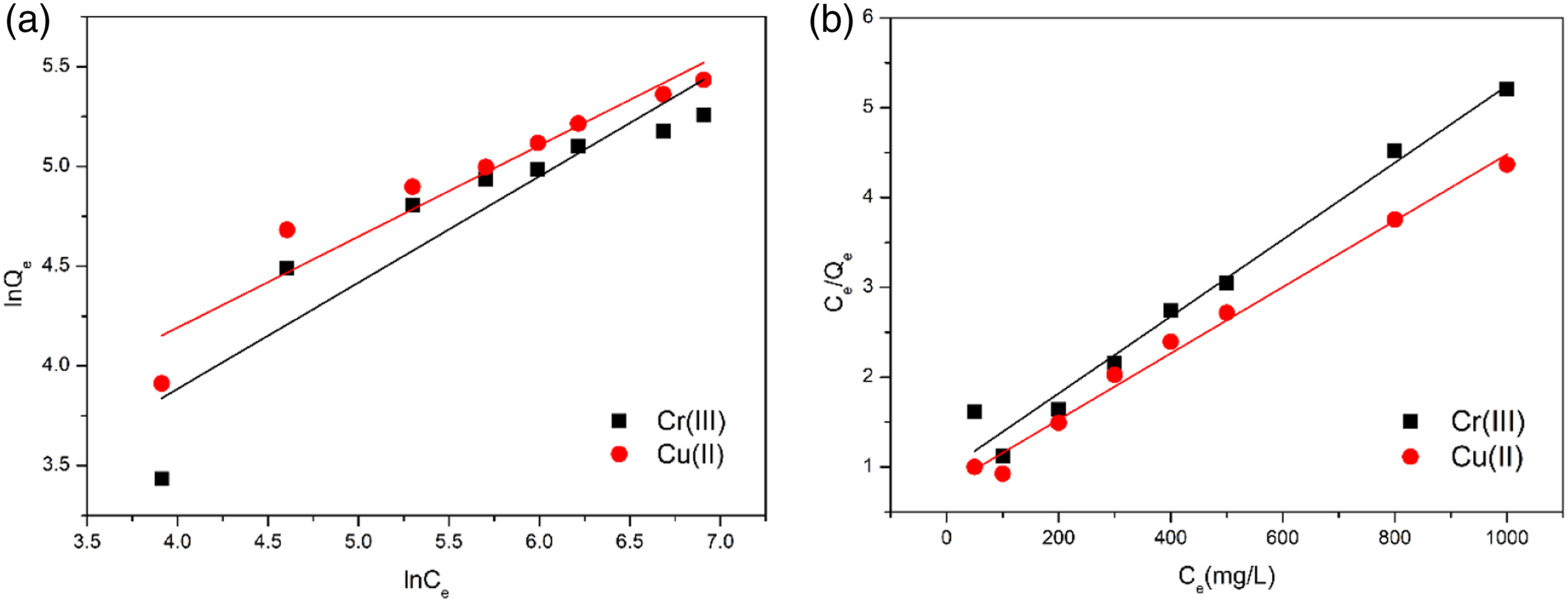
And equation (4) represents the linearized Langmuir isotherm

The empirical Freundlich isotherm model presumes that multilayer adsorption occurs on a heterogeneous surface. The equation is given as follows
Langmuir and Freundlich parameters for adsorption of HBP-CB.

Regeneration study
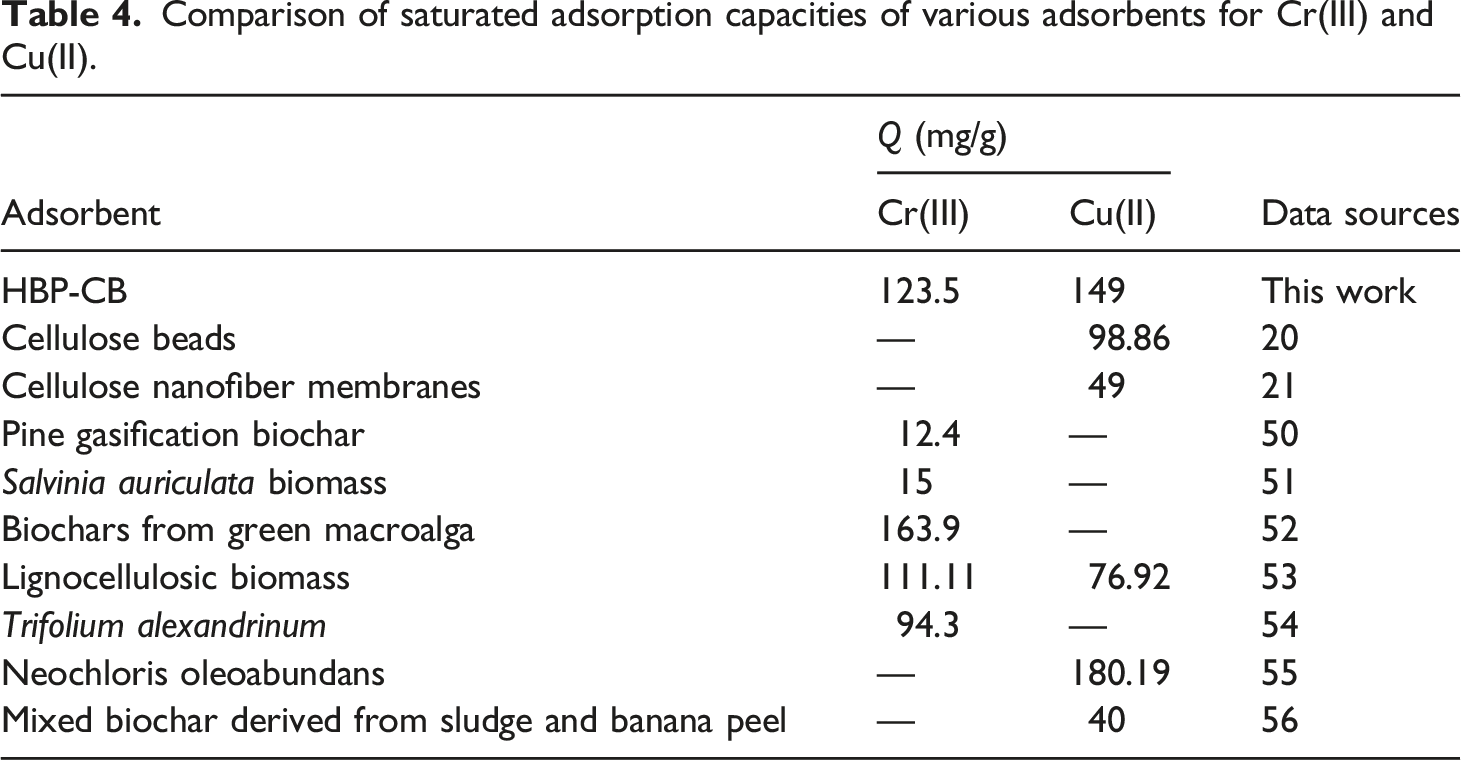
After desorption treatment, the regenerated HBP-CB was reused to adsorb heavy metal ions from the aquarium medium. As depicted in Figure 15, the saturation adsorption capacity of HBP-CB decreased continuously during the recycling process, while on average about 81% of the adsorption capacity was retained after five cycles. Thus, the results demonstrate that HBP-CB is a sustainable and excellent biosorbent for practical applications (Table 4). Saturated adsorption capacities of regenerated HBP-CB for Cr(III) and Cu(II) in different cycles. Three repeat experiments were performed. Comparison of saturated adsorption capacities of various adsorbents for Cr(III) and Cu(II).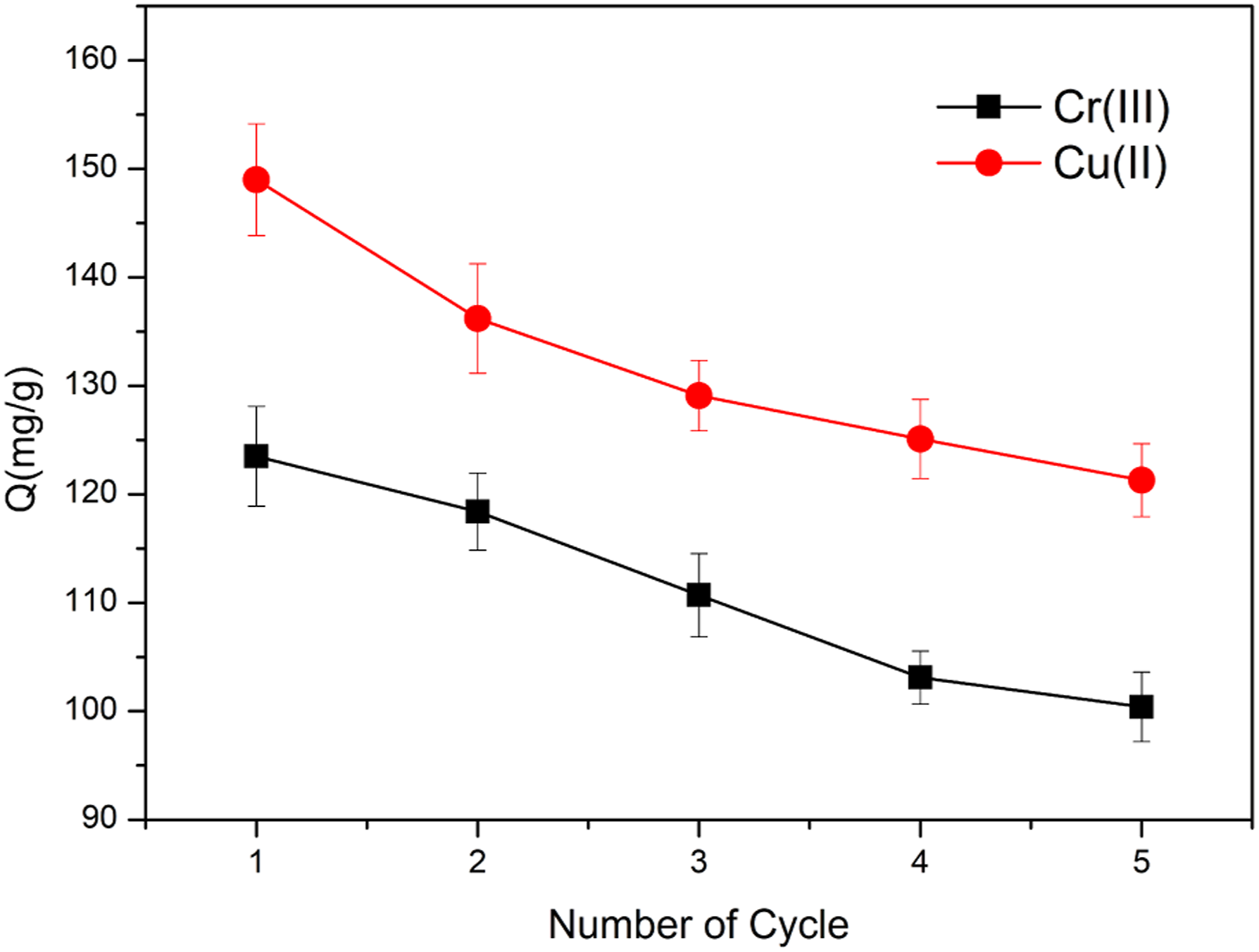
Conclusions
In this study, ramie-based biosorbent with Fe3O4/SiO2/polymer multiple structure was fabricated by an etch-fill strategy. SEM, EDS, FT-IR, XPS, XRD, N2 adsorption-desorption isotherm and VSM results confirmed that the multiple particles have been evenly introduced and embedded onto the oxidation etching cellulose matrix. Subsequently the matrix was wrapped and functionalized by HBP, forming a high-efficiency and stable adsorptive structure. During the modification, the periodate oxidation reaction was confirmed occurred only on the surface of the cellulose matrix. Due to the oxidation etching and evenly coverage of MSPs with large surface area, HBP-CB exhibited a quasi-type IV isotherm with an adsorption-desorption hysteresis loop, revealing an uniform porous structure with high BET surface area of 4.61 m2/g, and surface pore volume of 0.0058 cm3/g. The magnetism from Fe3O4 could provide good magnetic recovery for HBP-CB. HBP-CB shown excellent adsorption capacity for Cr(III) and Cu(II), which is owing to the hybrid multiple structure with high dense adsorptive sites. The kinetic adsorption results accorded well with the pseudo-second-order kinetic model, and the equilibrium adsorption data fitted Langmuir more than Freundlich. These phenomena demonstrate the heavy metal ions are adsorbed via a monolayer spontaneous chemical reaction. During this process, surface attraction and chemical adsorption are the dominant adsorption mechanisms. In the future research, pH variable is supposed to added to in the adsorption study to provide valuable data for adsorption in actual aqueous medium with different pH values. And adsorption behavior for co-existed pollutants should be also studied for practical applications.
Therefore, HBP-CB possessing a multiple structure with high dense active sites is considered to provide an essential implication for fabrication of sustainable and high-performance hybrid biosorbent.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by China Postdoctoral Science Foundation (No. 2020M671594), Natural Science Foundation of Colleges in Jiangsu Province (No. 19KJB540002), National Natural Science Foundation of China (No. 51903019), and Natural Science Foundation of Colleges in Jiangsu Province (No. 19KJB430006).

