Abstract
Considering the advantages of electrospun thermoplastic polyurethane (TPU)-fibers and antibiotic (vancomycin)-containing polyvinyl alcohol (PVA)-microbeads for wound dressings, a composite membrane was developed. Electrospun-aligned TPU-fibers were collected using a high-speed drum collector. Three groups of TPU-only, double-sided PVA-microbeads composite TPU-fibers (PVA/TPU/PVA sandwich, DSV40), and simultaneous electrospray PVA-microbeads/electrospun TPU-fibers (interstitial PVA-microbeads, CV40) composite membranes were prepared and characterized. The composites were tested for morphology, infrared spectroscopy, and tensile tests along the fiber-parallel (MD) and fiber-perpendicular (CD) directions, water wettability and absorption, antibacterial activity, and biocompatibility. The results showed that the tensile strength, modulus, and strain of TPU-only in MD and CD directions were 7.31 ± 1.13 and 2.47 ± 0.43 MPa, 2.73 ± 0.36 and 0.69 ± 0.07 MPa and 285.02 ± 23.56 and 360.70 ± 34.01%, respectively. Moreover, MD-direction tensile properties of DSV40 and CV40 resulted in a significant increase in strength (9.05 ± 2.19 and 9.77 ± 1.51 MPa) and modulus (10.97 ± 1.65 and 5.47 ± 0.94 MPa), but a decrease in strain (92.42 ± 11.68 and 185.78 ± 24.85%), compared to TPU-only. However, DSV40 and CV40 showed opposite trends (respective strength of 0.99 ± 0.07 and 2.11 ± 0.33 MPa, modulus of 0.21 ± 0.01 and 0.48 ± 0.51 MPa, and strain of 460.69 ± 29.38 and 435.92 ± 29.26%) in the CD direction compared to TPU-only. In terms of wettability after 10 s observation, DSV40 obtained a hydrophilic tendency (25.91°) than CV40 (74.22°) and TPU-only (121.06°). The results of antibacterial activity showed that both DSV40 and CV40 had good antibacterial effects on Staphylococcus aureus but the inhibitory effect on Escherichia coli was poor. The biocompatibility test confirmed that neither DSV40 nor CV40 negatively affected L929 cell viability.
Keywords
Introduction
A good wound dressing can improve healing through the interaction of the dressing with the tissue to achieve good wound closure by maintaining favorable factors in the wound environment.1–7 Moreover, multifunctional wound dressings can simultaneously accelerate wound healing and reduce patient inconvenience by preventing wound dehydration and acting as a drug carrier.3–7 Electrospinning is a technology capable of producing high surface area and high porosity fibrous membranes from most polymers. Furthermore, many studies have been carried out focusing on the development of functional fibers or fiber-based composite membranes by electrospinning.6–13 Such fiber-based composite membranes have the advantages of air permeability, high specific surface area, diverse composite materials, and a three-dimensional structure similar to that of the extracellular matrix, which benefits cell adhesion. Han et al. 14 studied the directionality of aligned fibers and found that aligned fibers undergoing cell contact guidance can accelerate the migration of cells from the wound edge to its center, and the nanopores on the aligned fibers are conducive to cell adhesion and proliferation. The hierarchical structure can also improve drug-loading efficiency and sustained release. The principle of electrospray is similar to electrospinning. When a Taylor cone forms a jet during electrospray, the end of the jet splits into countless droplets, resulting in spray beads of varying sizes and shapes. Electrospray hydrogel beads are more effective for drug loading and narrower particle-size distribution than other techniques.15,16
Thermoplastic polyurethane (TPU) is a class of thermoplastic elastomers with important commercial value, which is obtained by the reaction of polyol, that is ether-, ester-, and carbonate-based diols. TPU is a multi-block copolymer with alternating hard segments composed of diisocyanates and simple diols (chain extenders) and soft segments composed of polymeric diols. The composition of TPU is a linear structure. Polyester TPU is easy to be hydrolyzed, and polyether TPU shows better hydrolysis resistance than ester TPU, but its mechanical properties are poor. The hydrogen bonds play a role in strengthening its shape. In the 1990s, the asymmetric TPU membrane prepared by Hinrichs et al. 17 has tiny interconnected pores, which can prevent rapid dehydration of the wound surface and block bacteria, have good absorption capacity, and can promote tissue regeneration. They claim that in contrast to other wound dressings, TPU is applied to excised clean wounds as a whole-skin substitute that does not need to be replaced during the healing process, but can be left on the wound until the skin fully regenerates and then spontaneously rejects. It has properties such as high elasticity, non-cytotoxicity, and blood compatibility. Accordingly, TPU is extensively used in biomedical fields, such as skin, artificial blood vessels, cartilage, ligaments, and bone scaffolds.18–23 Polyvinyl alcohol (PVA) is a typical water-soluble and biodegradable synthetic polymer with good mechanical strength, flexibility, and barrier properties. It is easy to process and is widely used in the food packaging, pharmaceutical, and biomedical fields.24–27 PVA is also a commercial material for electrospinning because of the presence of hydroxyl groups in its repeating units, which allow it to be cross-linked through its interconnected hydrogen bonds. Vancomycin (Vanco) is a glycopeptide antibiotic primarily used to prevent and treat infections caused by Gram-positive bacteria but is generally less effective against Gram-negative bacteria and cannot penetrate the outer bacterial cell wall barrier.28,29 Most wound infections caused by Staphylococcus aureus and methicillin-sensitive (MSSA) and methicillin-resistant (MRSA) species are frequently cultured, so the topical delivery of antibiotics with a broad antimicrobial spectrum such as Vanco) is attractive. The use of Vanco in MRSA-positive chronic diabetic wounds can help significantly reduce mean surface area, exudate volume, and wound coverage compared with traditional saline-impregnated dressings.
Electrospun nanofiber-based composites have opened up a new era of fiber-based materials, showing great potential in many biomedical devices, such as biosensing, regenerative medicine, tissue engineering, drug delivery, wound healing, and more. 30 Aligned fiber-elastic polymers have developed the feasibility of designing to mimic the mechanical properties of skin tissue as wound dressing materials. For reference, the tensile strength and ultimate strain of human skin are 1–32 MPa and 35%–115%, respectively. 31 Electrospinning with drum collector technology allows TPU membranes to be oriented along the fiber axis, combined with electrospray, to produce unidirectional fiber-reinforced composites for a variety of applications. In the present study, electrospun TPU-fibers were used to prepare fibrous membranes, which had potential as a matrix for skin-wound dressings owing to their good elasticity, unidirectionally aligned fibers, and high biocompatibility. The electrospray PVA-microbeads were dispersed onto the inside or surface of the TPU membranes to prepare a composite with antibacterial, hydrophilic, and high elastic resilience. This study aimed to characterize the combined electrospinning and electrospray fabrication of PVA-microbeads as antibiotic carrier composite TPU fibrous membranes and their mechanical properties, wettability, antibacterial ability, and biocompatibility. This developed multifunctional composite membrane can be expected to have applications in chronic wounds and wound-repair acceleration.
Materials and methods
Materials
Polyether-based thermoplastic polyurethane beads (TPU, Mw: 20,000–150,000 g/mole, Kin Join Co., Ltd., Taichung, Taiwan) with a density of 1.11 g/cm3, Shore A 80, and melting temperature around 160°C was used. The other materials used in this study include polyvinyl alcohol powders (PVA, Mw: 20,000–130,000 g/mole; Beijing Guoren Yikang Technology Co., Ltd., Beijing, China), a glycopeptide antibiotic of vancomycin (Vanco, Gentle Pharma Co., Ltd., Yunlin, Taiwan), and dimethylformamide (DMF, Sigma–Aldrich Co., St Louis, MO, USA).
Preparation of electrospinning/electrospray solutions
The electrospinning solution was prepared by dissolving 3.6 g of TPU beads in 20 mL of DMF solvent to form an 18 w/v% TPU solution for electrospinning. The electrospray solution was 0.4 g of PVA powder added to 10 mL of DMF/double-distilled water (dd-H2O) (6:4) mixed solvent, and then 40 mg of Vanco powder was added. The two solutions were uniformly stirred for 12 h at 60°C and 300 r/min.
Composite-membrane manufacturing
Figure 1 shows the preparations of different composite methods used in this experiment. The equipment setup in this study was a rack-mounted high-voltage power supply (AU-60R1-LC, Matsusada Precision, Shiga, Japan), a syringe pump (YSP-101, YMC Co., Ltd., Kyoto, Japan), and a drum collector (ES-02, Falco Tech Enterprise Co., Ltd., New Taipei City, Taiwan). The TPU solution was placed in a syringe, the flow rate was 2 mL/h, the voltage used was 8 kV, and the collection speed was the linear speed of the roller collector of 9 m/s. TPU unidirectionally oriented fiber membrane was collected after 6 h (TPU-only group). Then, the TPU membrane was placed on the collector plate, and the PVA solution containing Vanco was added. The flow rate was set to 0.6 mL/h, and the voltage was 8 kV. The electrospray PVA on each surface of the electrospun TPU fibers was collected for 1.5 h to obtain the Vanco-containing double-sided electrospray PVA-microbeads composite electrospun TPU-fibers membrane (DSV40 group of PVA/TPU/PVA sandwich, PVA/TPU was 40 wt.%; Figure 1(b)). The electrospun TPU-fibers and electrospray PVA-microbeads composite membranes were prepared under the solutions for co-electrospinning/-electrospray and were set up in the pusher simultaneously, the flow rate of the TPU solution was 2 mL/h, and the flow rate of the PVA solution was 0.3 mL/h, the Vanco-containing co-electrospun TPU-fibers/-electrospray PVA-microbeads composite membrane (CV40 group, PVA/TPU was also 40 wt.%; Figure 1(c)) was prepared after collecting for 6 h. Simulation schematic and changing parameters of different composites used in this experiment: (a) electrospinning TPU and electrospray PVA solutions (40 wt.% ratios of PVA-microbeads to TPU-fibers in the composite membrane), (b) preparation of DSV40 composite membrane by double-sided electrospray PVA-microbead layer composite electrospun TPU-fiber interlayer film, and (c) CV40 composite membrane was prepared by simultaneous co-electrospun TPU-fiber composite electrospray PVA-microbeads.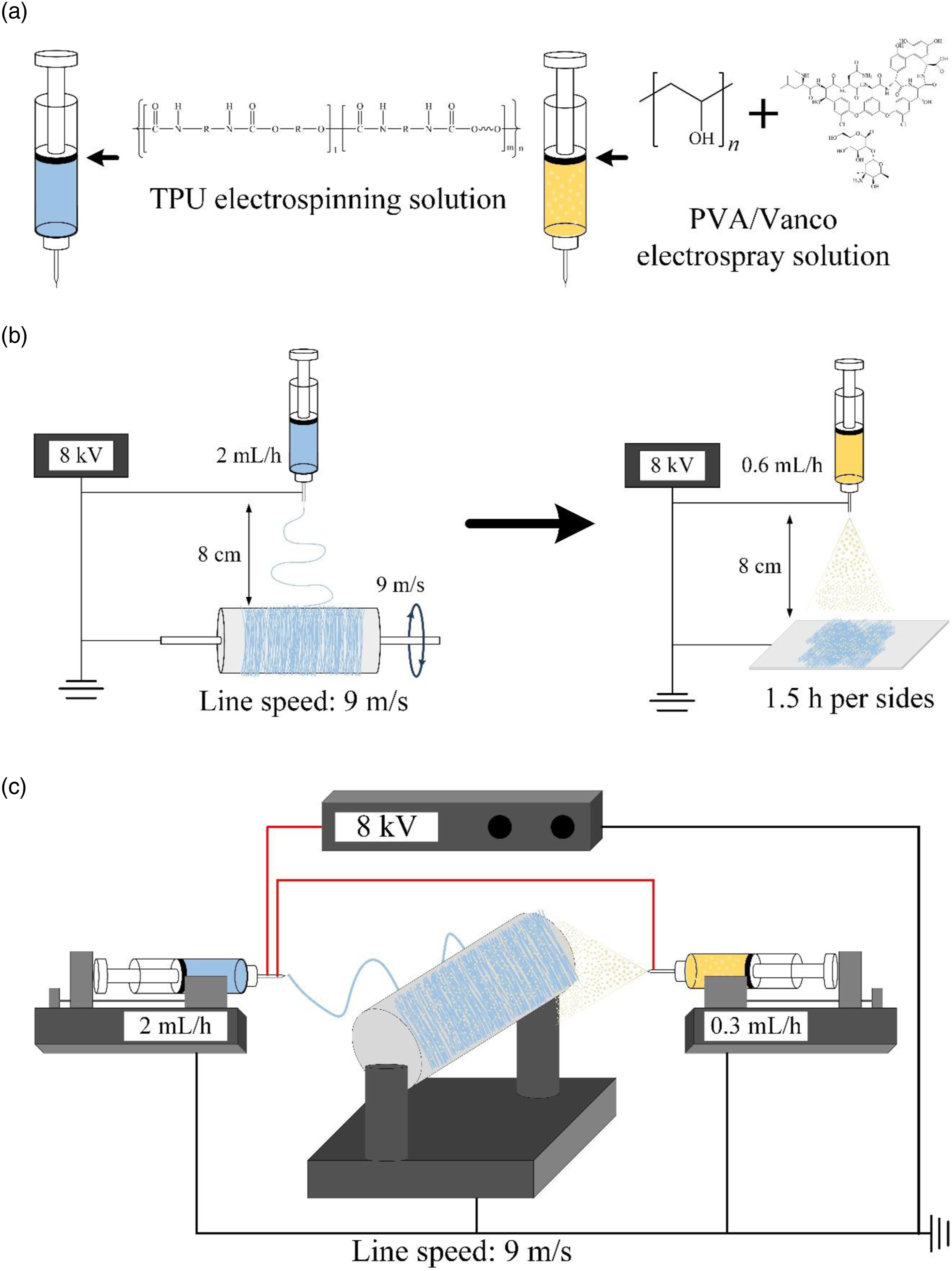
Characterizations
Morphology and characterization of fibers and beads
The morphology of electrospun fibers and electrospray beads was observed under scanning electron microscopy (SEM; S3400N, Hitachi, Tokyo, Japan). The membranes were gold plated before SEM observation, and average electrospun fiber diameters and electrospray bead sizes were measured through SEM images (500x, 2000x, and 5000x) from randomly selected fibers (n = 100) and beads (n = 50) using ImageJ software (version 1.53e).
Attenuated total-reflection Fourier infrared spectrometry
Infrared spectra obtained through FTIR (Nicolet 6700, Thermo Fisher Scientific, Waltham, MA, USA) were used to confirm the chemical composition and the existence of chemical reactions between TPU-fibers and PVA-microbeads. The wavenumber scan range of FTIR is 600–4000 cm−1.
Membrane tensile-strength measurement
According to the ASTM D882 specification, a composite membrane of uniform thickness was selected, and the membrane was cut into dumb-bell-shaped specimens along the mechanical direction/unidirectional aligned fiber-parallel (MD) of the membrane. Another test group was in the cross-sectional direction/fiber-perpendicular (CD) of the membrane. Thickness was measured and then tested with a universal testing machine (HT-2402, Hung Ta, Taichung, Taiwan) at a tensile rate of 10 mm/min. Tensile testing was performed from 0 N load to specimen fracture, and tensile strength and strain changes were recorded.
Wettability
Using a contact-angle measuring instrument (CAM121, Creating Nano Technologies Inc., Tainan, Taiwan), the test piece was placed on the measuring platform, and a fixed amount of dd-H2O was dropped onto the surface. The shape of the water droplets was observed with a charge-coupled device, and the wettability of the sample surface was analyzed with software to confirm the hydrophilicity and hydrophobicity of the composite membrane before and after the composite membrane containing Vanco microbeads.
Immersed weight changes
We first measured and recorded the unabsorbed weight (W0) of the composite membrane. Then, the sample was placed in a 15 mL centrifuge tube, 5 mL of dd-H2O was added, and the temperature was maintained at 37°C. After soaking for different times, the tubes were taken out and excess water on the surface was wiped off with a saturated sponge. The tubes were weighed to obtain the residual weight (Wt) of water absorbed by the sample (n = 10).
Antibacterial activity
The membranes were sterilized by UV irradiation using a UV lamp at 365 nm with an incident powder intensity of 700 mW/cm2 at a lamp-to-sample distance of 20 cm for 8 h. The antibacterial activity of Vanco-containing composite membranes against two pathogenic bacteria (Gram-positive and negative) was investigated using the disc-diffusion method of the American Institute of Clinical and Laboratory Standards. 32 S. aureus (ATCC number: 25923) and Escherichia coli (ATCC number: 10798) were cultured in Luria-Bertani broth. Liquid broth (1.5%) was sterilized in a high-temperature autoclave, equilibrated in a water bath at 50°C for 30 min, poured into a 10 cm dish, and incubated on a cured trypsin soy broth (TSB) agar. Each sample of TPU, DSV40, and CV40 was made to adhere to an agar plate, which was incubated at 37°C for 24 h. After measuring the diameter of the inhibition zone around the sample (n = 3), each specimen was placed in 2 mL of TSB medium, and the changes in bacterial growth were observed during incubation at 37°C for 1, 4, 8, 24, and 48 h. A 100 μL medium suspension was prepared, and bacterial counts were determined by optical density at 595 nm (OD595) using an ELISA reader (EZ Read 400, Biochrom, Cambridge, UK) (n = 3).
Cytotoxicity tests in vitro
The mouse fibroblast cell line L929 was used in the cytotoxicity test, and the procedures were conducted per ISO 10993-5:2009. L929 cells were grown with media in an incubator at 37°C and 5% CO2 and subcultured when the cell concentration was between 0.8 × 106 and 1.0 × 106 cells/mL. The medium used was a minimal essential medium alpha medium (Gibco, Thermo Fisher Scientific Inc., Waltham, MA, USA) containing 10% horse serum and was changed every 2 days of culture. The sample extract was prepared following the ISO 10993-5 standard test method of indirect cytotoxicity evaluation. The sample was immersed in a medium with a ratio of 6 cm2/mL to prepare the sample extract. Other groups in this experiment included the control group, which was cultured with a medium to simulate the culture process of normal cells; the positive control group, which was treated with 15% dimethyl sulfoxide (Sigma-Aldrich, USA); and the negative control group, which was used to confirm whether the sterilization was effective. The material used was high-density polyethylene. First, 100 μL of medium and L929 cells at a concentration of 1 × 104 cells/well were transplanted into a 96-well microplate and incubated for 24 h at 37°C in a 5% CO2 incubator to allow cell attachment. After removing the medium and adding the extract for another 24 h of incubation, the extract was aspirated and 100 μL/well was added to a new cell-culture medium along with a 50 μL/well XTT Cell-Proliferation Assay Kit (Biological Industries, Beit-Haemek Ltd., Israel). It was mixed well and incubated for an additional 4 h. Then, the absorbance of the cells was measured with an ELISA reader (SPECTROstar Nano, BMG LABTECH, Offenburg, Germany). Notably, the XTT assay absorbance was proportional to cell viability.
In the qualitative cytotoxicity test, the preparation of the extracts and the culture medium of the control group were the same as those in the quantitative test. Approximately 1000 μL of cell suspension was collected, and L929 cells were transplanted into a 48-well microliter plate at a cell concentration of 1 × 105 cells/well. The original medium was cultured in an incubator at 37°C and 5% CO2 for 1 day, and then the medium was removed. The sample extract (100 μL) was added and cultured for 24 h, and cell morphology was observed under an inverted microscope (IVM-3AFL, SAGE VISION Co., Ltd., New Taipei City, Taiwan).
Morphological observation of cell attachment
The sterilized sample was placed in a microcentrifuge tube, to which we dropped 20 μL of the cell suspension with a cell concentration of 1 × 105 cells/mL on the sample. After adding 200 μL of culture medium and incubating for 20 min in contact, the sample was moved into a 48-well plate, and 800 μL of the medium was added. After culturing for 1 h, 1 day, and 2 days, the samples were taken out and placed in a new 48-well plate, soaked in fixative, and placed at 4°C for 20 min. The samples were processed into sequential dehydration, and after drying, the samples were plated with gold and analyzed by SEM (200x and 1000x) to observe cell adhesion onto the composite membranes.
Statistical analysis
ANOVA was performed using IBM SPSS Statistics version 20 (SPSS Inc., Chicago, IL, USA) to measure the sizes of electrospun TPU-fibers and electrospray PVA microbeads. ANOVA was used to determine whether the differences between the means of multiple groups were significant. Estimates of two different variables were used when comparing differences, and significance was accepted when the p-value was less than 0.05.
Results and discussions
Morphology and characterization of fibers and beads
An ideal wound dressing comprises a fibrous membrane that should possess properties such as fine fiber diameter, small pore size, and tailored wettability and strength to achieve appreciable modulation of high air permeability, directional water transport, and cell growth.
33
As shown in Figure 2(a), a significant embedding difference existed in the number of microbeads attached to the fibers of the composite membranes of DSV40 and CV40. The TPU-fibers and PVA-microbeads of DSV40 had a sandwich structure of PVA/TPU/PVA, and the microbeads on both sides were covered on the TPU-fiber membrane in the middle. PVA-microbeads in the CV40 composite membrane were smaller than DSV40 and randomly attached to the TPU-fibers. From the internal fiber structure under the surface layer, only the internal fibers of CV40 showed obvious PVA-microbead adhesion, owing to CV40 being both co-electrospun TPU-fibers and electrospray PVA-microbeads. Thus, the microbeads can be uniformly and synchronously interstitially adhered onto the surface while the fibers were being collected. To further confirm the structural differences between DSV40 and CV40, morphologies of membrane cross-sections were observed (Figure 2(b)). In the cross-section of the PVA-TPU-PVA sandwich composite membrane of DSV40, the coexistence of microbeads between fibers cannot be seen in the middle layer of TPU-fibers, from which it can be seen that the beads on both sides of the electrospray PVA-microbeads were attached on the TPU-fibers and the aggregation even form a covering film. When the co-electrospun TPU-fibers and electrospray PVA-beads composite membrane of CV40 were observed at high magnification, the dispersed microbeads were found to be attached within the aligned fibrous structures. Therefore, we confirmed that the microbeads of the CV40 group did exist in the interfiber space under the composite surface. DSV40 and CV40 had two different structures and thus affected the performance of the composite membranes. Morphology of top and side views of aligned fibers of TPU-only and TPU-fiber/PVA-microbeads composite membranes of DSV40 (PVA/TPU/PVA sandwich) and CV40 (co-electrospun TPU/-electrospray PVA beads) at different magnifications: (a) SEM images observed on the top view of membrane surfaces (2000x and 5000x; PVA-microbeads shown in circles), and (b) side views observed on internal cross-sectional images (500x and 2000x; Red circles indicate zoomed-in sites).
Measurement of TPU-fiber diameters and PVA-microbead sizes of each group of composite membranes (mean ± standard deviation).

FTIR analysis
The TPU-fiber surface had many PVA-microbeads. To determine whether Vanco, Vanco/PVA, or PVA interacted on the surfaces of TPU-fibers, the composite membranes were analyzed by infrared spectroscopy, and the results are shown in Figure 3. After comparison especially in TPU spectrum, we found that 3323 cm−1 was the stretching vibration band of N-H in the urethane group, 2939 and 2854 cm−1 were those of C-H, and 1699 cm−1 was that of C=O stretching vibration (amide I band). The stretching vibration band at 1530 cm−1 was the bending vibration peak of N-H (amide II band), and bands at 1300–1000 cm−1 were the stretching vibration peak of C-O-C and C-N (amide III band).23,35–39 CV40 and DSV40 had similar characteristic spectra to TPU, and no new absorption was observed in each group. Additionally, the characteristic bands of Vanco and PVA were not observed in the composite membranes possibly owing to the similar functional groups in the absorption bands of Vanco, PVA, and TPU, as well as the extremely lower content of Vanco and PVA than that of TPU. Infrared spectra were obtained from raw materials and experimental groups of electrospun fibrous TPU, DSV40, and CV40 composite membranes.
Mechanical properties
An important factor to control when handling electrospun fibers was their orientation as they were being collected. The two main fiber orientations obtained during electrospinning were aligned and random. Given that random fibers were randomly oriented at different angles throughout the collected sample and aligned fibers were oriented in the same direction, randomly oriented fibers were not suitable for some special applications, such as skin-tissue generation. 40 Epithelial cells in particular have better mobility and proliferative capacity on aligned electrospun structures. Aligned fiber orientation can also enhance mechanical properties by increasing tensile strength and preventing sample elongation.
Figures 4 and 5 show the tensile-test results of different groups of fiber composite membranes along with the fiber-parallel (MD) and -perpendicular (CD) directions, respectively. When testing the MD samples, the tensile strength and modulus of DSV40 and CV40 showed a clear upward trend (respective increased rates of 123.78 ± 30.01 and 133.71 ± 20.70% in strength and 401.29 ± 60.31 and 200.02 ± 34.27% in modulus) after fiber composite microbeads compared to TPU-only (Figure 4). The reason for this is presumed that because PVA itself is a viscous polymer, PVA-microbeads prevent fiber slippage by increasing the cohesion point between fibers, thereby improving the effect of membrane mechanical properties.
41
After the PVA-microbeads have adhered to fibers, they can bond and thus increase the mechanical strength and modulus of the composite membrane compared with TPU-only. In terms of strain, the strain of the DSV40 and CV40 composite membranes showed a significant downward trend, and the strain rates of DSV40 and CV40 were 32.43 ± 4.10 and 68.18 ± 7.67% of the modulus, respectively, compared with the strain of TPU-only. It was the PVA-microbeads that caused the reduction in the active gap between TPU-fibers, resulting in decreased strain. Figure 4(e) shows a fiber sample under uniaxial loading along the fiber-parallel direction, which exhibits high tensile strength and modulus, accompanied by significant elongation as the sample becomes thinner. When the TPU-fibers spin, slide, and the high-modulus region breaks beyond the load-bearing point, the sample springs back immediately after a tensile fracture. The strains of DSV40 and CV40 also significantly differed, which may be due to the structural difference. The microbeads in the DSV40 group were coated on the surface, so they cannot withstand the strain difference between the surface and the inner layer fibers after the sample was deformed. Consequently, the surface and the inner layer had uneven stress, so the deformed DSV40 had only half of the CV40 strain. Figure 5 shows the tensile results of the tested CD samples, the strains of 360.70 ± 34.01%/TPU-only, 460.69 ± 29.39%/DSV40, and 435.92 ± 29.26%/CV40 for all three groups were significantly higher than the MD tensile strains of 285.02 ± 23.56%/TPU-only, 92.42 ± 11.68%/DSV40, and 185.78 ± 21.85%/CV40. The reason is that when the fibers are stretched in the vertical direction, although there are still some asymmetric fibers interlaced instead of the CD direction to prevent fiber desorption (Figure 2), the fibers slide past each other and gradually pull apart instead of directly breaking the fibers. Finally, the gaps between the fibers were large enough to limit the fibers to slip or the PVA-microbeads to fall off the binding points between the fibers, indicating that all the binding points between the fibers were destroyed, resulting in the tensile fracture of the sample. The different mode of sample rebound after tensile fracture is shown in Figure 5(e). This finding showed the excellent deformation of the TPU-fibers/PVA-microbeads composite membranes.
42
Compared with the tensile strength in MD direction, the strength in CD direction was significantly reduced in all three groups. The tensile strength (0.99 ± 0.07 MPa) and modulus (0.21 ± 0.01 MPa) of DSV40 were even significantly lower than CV40 (respective strength and modulus of 2.11 ± 0.33 MPa and 0.48 ± 0.05 MPa) compared to TPU-only membrane (respective strength and modulus of 2.47 ± 0.43 MPa and 0.69 ± 0.07 MPa). Fiber-parallel (MD) tensile-test results for TPU, DSV40, and CV40 membranes (n = 10; * indicates significant difference at p < 0.05): (a) schematic diagram of MD sample and typical tensile stress-strain curves, (b) measured modulus, (c) values of tensile strength, (d) failure strain, and (e) MD tensile testing, sample adjustment settings, sample morphologies before and after tensile fracture (images left to right). Fiber-perpendicular (CD) direction tensile-test results for TPU, DSV40, and CV40 membranes (n = 10; * indicates significant difference at p < 0.05): (a) schematic diagram of CD sample and typical tensile stress-strain curves, (b) measured modulus, (c) values of tensile strength, (d) failure strain, and (e) CD tensile testing, sample adjustment settings, sample morphologies before and after tensile fracture (images left to right).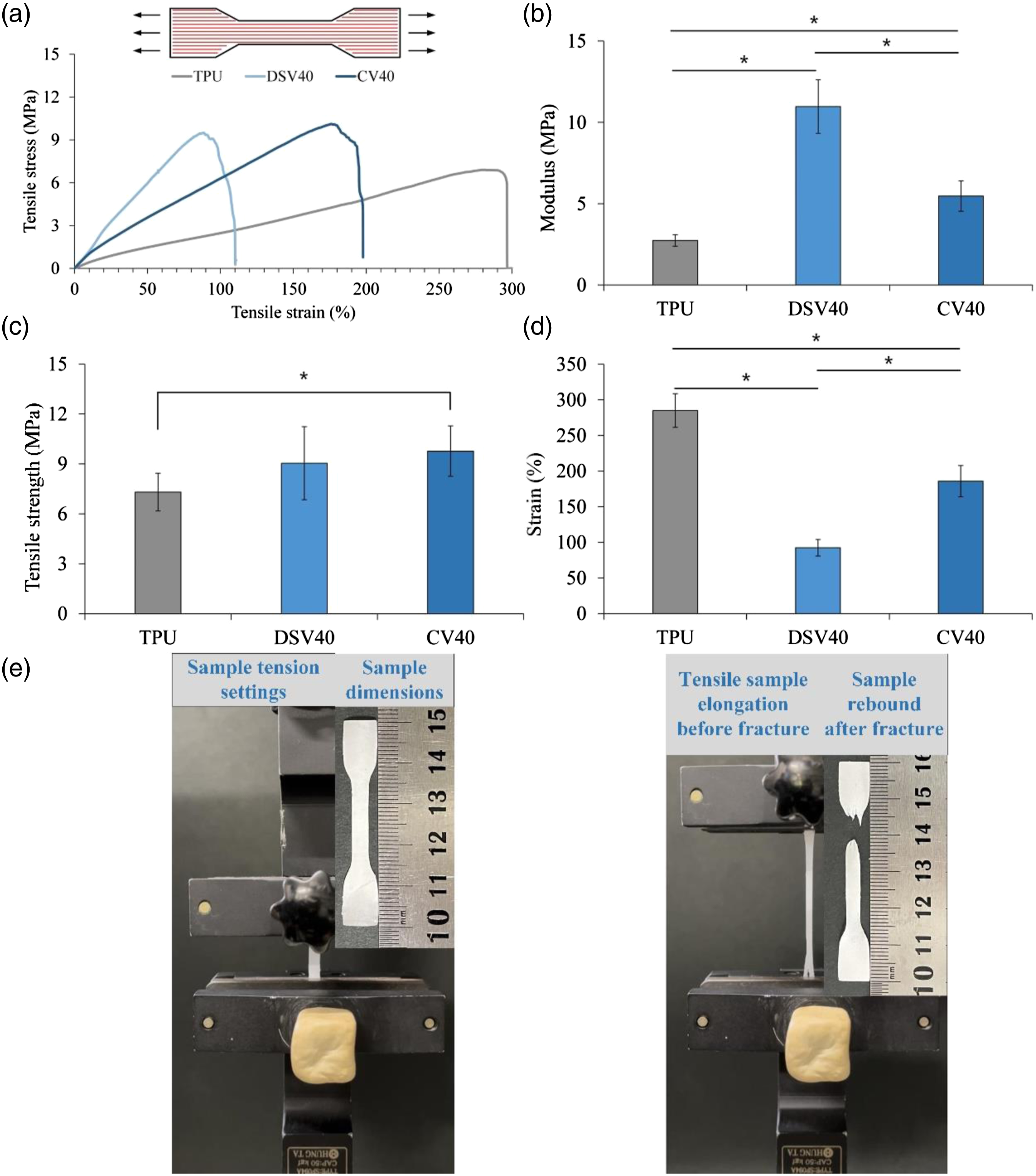

Wettability and water absorption
Wound dressings with asymmetric wettability surfaces can effectively prevent bacterial colonization and tissue dehydration, showing great potential in diabetic wound-healing applications. Wound healing is a complex pathophysiological process that requires an ideal microenvironment, of which moisture is one of the most important factors.
7
Antibiotic-containing hydrophilic degradable microbeads prevent wound infection and accelerate wound healing through spontaneous drug release in early care treatment.
43
In addition, the nanofiber surface and internal geometry (fiber diameter and membrane porosity) have been found to significantly affect the efficiency of the membrane in terms of water wettability and absorbency.
44
Figure 6 reveals the captured dynamic images of contact angles at different time points. In the TPU-only group, the membranes exhibited high hydrophobicity at time points of 0, 5, and 10 s, and the measured contact angles ranged from approximately 115° to 130°. However, the angles of the DSV40 and CV40 groups changed after 5 s of contact, and the average water contact angles of the DSV40 and CV40 groups dropped to 41° for hydrophilic and 94° for relatively hydrophilic. This finding was due to the greater amount of PVA-microbeads on the DSV40 surface than in other groups and PVA’s excellent hydrophilicity,
45
which can effectively improve the surface properties of the composite membrane. Captured dynamic images of contact angles at different time points in the TPU, DSV40, and CV40 groups (n = 3).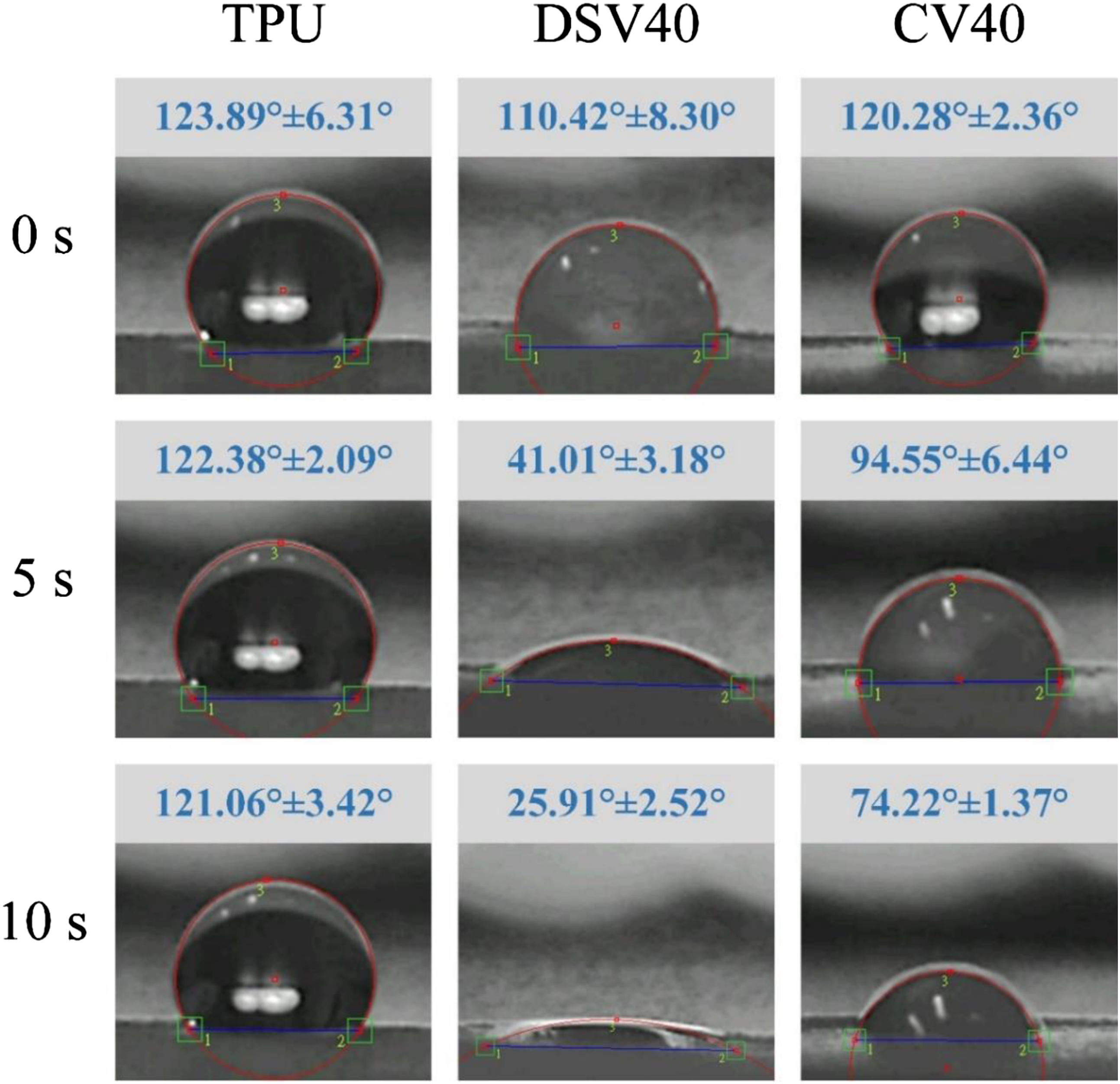
The successful transition of wounds from the acute inflammatory phase to the subsequent proliferative and remodeling phases involves many complex and subtle interactions.
46
Accordingly, designing and understanding an appropriate and rapid response for the fluid absorption of wound dressings is also an important issue. As shown in Figure 7, three groups of membranes all reached a water-absorption platform after soaking in water for 1 h, and the difference was not significant. This finding indicated that the main water-absorption mechanism was still the pores generated by water adhesion between fibers. The relative particle size and content of composite microbeads were lower than those of inter-fiber pores, resulting in no significant difference in water absorption among the groups. We further confirmed that the particle size of the microbeads was much smaller than the pores between the fibers (Figure 2). Additionally, the PVA-microbeads began to dissolve in contact with water, so the overall water absorption cannot be increased. Water-absorption rate of TPU, DSV40, and CV40 membranes within 24 h of immersion (n = 3).
Antibacterial activity
The basic elements of wound healing can be described in three consecutive and overlapping stages, namely, inflammation, proliferation, and remodeling. Many factors contribute to chronic wounds, including age, ischemia, and infection.
43
All wounds eventually host bacteria, typically within 48 h of contamination, although the point at which bacteria negatively impacts wound healing remains controversial. Incorporating some antimicrobial activity into wound dressings is an appropriate strategy to prevent initial infection. Figure 8 compares the difference in antibacterial ability between composite membranes with different structures. The antibacterial qualitative results showed that the S. aureus inhibition zone of DSV40 was significantly larger than that of CV40 (Figure 8(a)), which was due to the obvious difference in the number of drug-containing microbeads located on the surface. These results showed that DSV40 and CV40 had good antibacterial activity against S. aureus but a poor one against E. coli, consistent with the description of Vanco. Compared with the CV40 group, which was co-electrospun TPU-fibers/-electrospray PVA-microbeads, more drug-containing PVA-microbeads appeared on the surface of DSV40 group, so the antibacterial effect of DSV40 was significantly better than that of the CV40. Results of bacteriostatic quantification in Figure 8(b) showed that the optical density values (OD595) of the DSV40 and DV40 groups were significantly lower than those of the control group after 4 h of culture, confirming that DSV40 and CV40 had drug-release and bacteriostatic ability at least within 48 h. (a) Qualitative bacteriostasis (unit: mm) against S. aureus and E. coli. (b) Quantitative results of bacteriostasis to S. aureus of TPU, DSV40, and CV40 membranes (n = 3, OD595: optical density at 595 nm).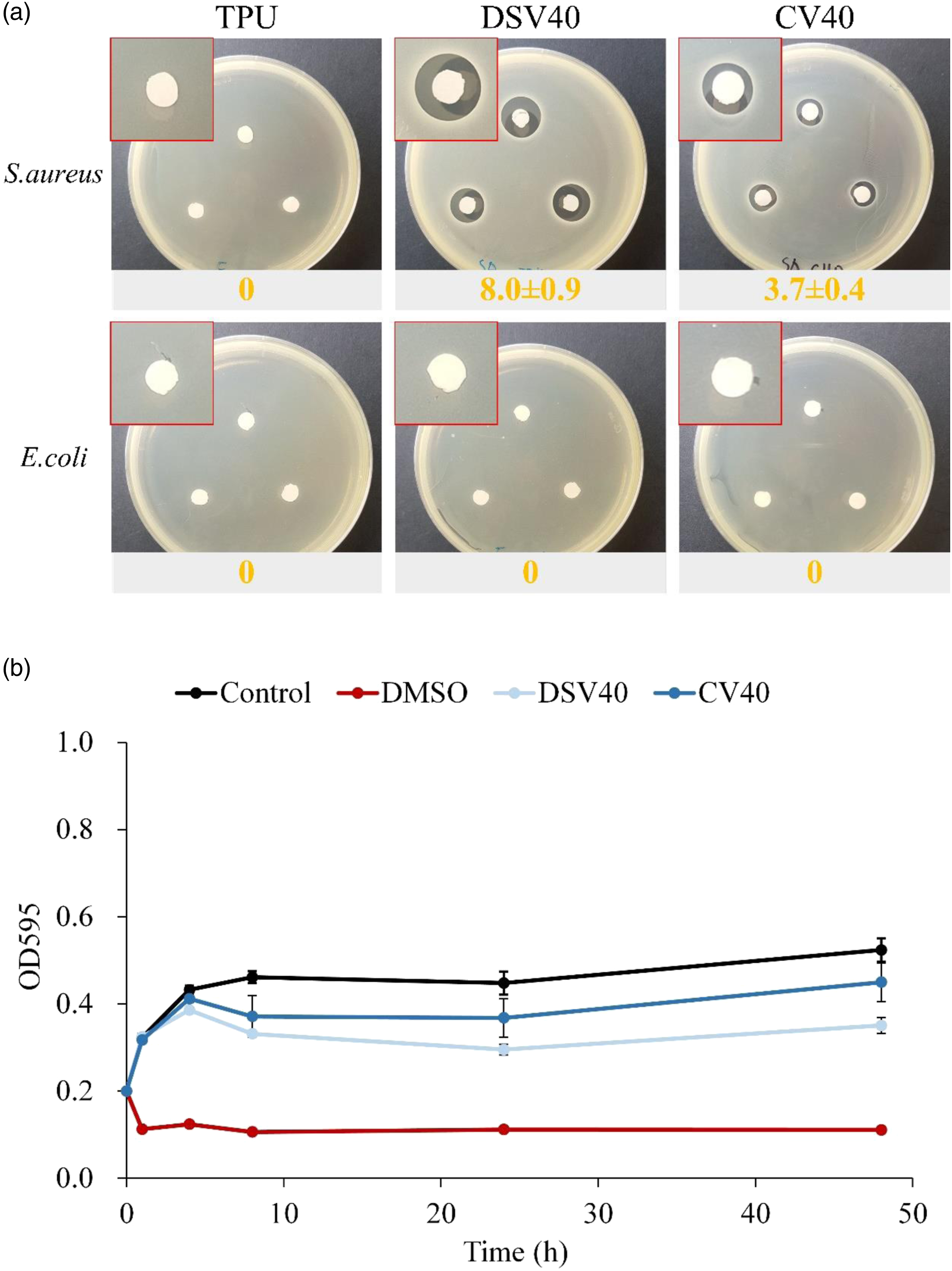
Cytotoxicity tests in vitro
Wounds heal more easily in a moist, clean, and warm environment. A moist wound dressing allows the migration of growth factors and multiple cell types including epithelial cells, thereby promoting wound-edge shrinkage. First, the absence of toxic or irritating particles is the most important criterion in all wound treatments. In terms of cytotoxicity in this study, given that high-density polyethylene (HDPE) was the negative control, the specimens were fully sterilized and no adverse effects on the experimental design were observed. Figure 9(a) shows that the cell viability of TPU, DSV40, and CV40 were all greater than 70%. This finding indicated that neither had cytotoxicity and thus proved that DSV40 and CV40 composite membranes had synergistic bacteriostatic ability without cytotoxicity. Figure 9(b) shows that the cell morphology of the three groups did not denature, indicating that the sample extract did not cause cell denaturation or death. Cytotoxicity of mouse fibroblast cell line L929 cultured in TPU, DSV40, and CV40 membrane extracts: (a) quantitative measurements (n = 3), and (b) cell-morphology observations. DMSO: 15% of dimethyl sulfoxide presented to positive control; HDPE: high-density polyethylene was designated as the negative control group according to IOS 10993-5 specification.
Morphological observation of cell attachment
As shown in Figure 10, compared with the three groups, the microscopic images after 1 h of cell culture clearly showed that DSV40 adhered the best, whereas, for CV40, some cells started to adhere. When cells were cultured on TPU-only, the majority of cells were found to be lightly attached and maintained a spherical phenotype. This result suggested that PVA-microbeads can improve wettability, thereby promoting the adhesion of L929 cells. Cell adhesion was the best owing to the large number of PVA-microbeads on the surface of DSV40, even though PVA-microbeads on the surface of CV40 were fewer. Cell adhesion also significantly improved compared with cell adhesion in the TPU-only group. Images of fibroblast L929 cells attached to TPU-only, DSV40, and CV40 composite membranes at different time points: (a) low (200x, cells shown in circles) and (b) high magnifications (1000x; Zoom in on the images in circles, arrows show cell attachment morphology on the composite membranes).
Conclusion
In this study, antibiotic-loaded PVA-microbeads were successfully prepared by electrospray and then composited on the surface or in the structural gap of the electrospun TPU-fiber membrane. The strategy of compositing PVA-microbeads with TPU-fiber has a significant effect on mechanical properties. The PVA-microbeads composite membranes of DSV40 and CV40 both showed a positive increase in tensile strength and modulus in the fiber-parallel MD direction, while the tensile strain showed a decreasing trend compared to the TPU-only membrane. However, the trends in strength, modulus, and strain were reversed when tensile tests were performed in the fiber-perpendicular CD direction rather than the MD direction. The tensile strength and modulus in the CD direction are much lower than those in the MD direction, and only the strain increases. Unlike the reduced values in the MD tensile test, the elongation before failure was significantly higher for DSV40 and CV40 with strain in the CD direction than for TPU-only. The wettability of the DSV40 and CV40 composite membranes was also greatly improved compared to the hydrophobic membrane using TPU-only. The water absorption showed that the composite membrane could reach the peak water absorption after 5 min immersion. According to antibacterial activity, the two groups of DSV40 and CV40 had a better bacteriostatic effect on S. aureus but the less bacteriostatic effect on E. coli. As for cytotoxicity, all groups showed good biocompatibility. Wound dressings with different mechanical properties and antibacterial functions can be fabricated using different composite strategies of electrospun TPU-fibers with electrospray PVA-microbeads. We expect these composite membranes to be applied to wound dressings in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support provided by the Ministry of Science and Technology, Taiwan (MOST 110-2314-B-037-089-MY3, 110-2221-E-035-013, 109-2221-E-035-043, 109-2622-E-035-014-CC2).

